Abstract
Certain ecological traits of bats may be associated with sensitivity to the fragmentation of their habitat, but the relative importance of this is unclear and has been poorly studied in tropical environments. We evaluated the relationship between six ecological traits of phyllostomid bats (body mass, trophic level, vertical foraging area, natural abundance in continuous forest, wing aspect ratio, and relative wing loading) and three measures of sensitivity to habitat fragmentation (species prevalence, change in abundance index, and nestedness ranking) in a fragmented forest in Los Chimalapas, Mexico. Ecological traits were obtained from 20 bat species for a period of 2 years, and their relationship with measures of sensitivity to fragmentation was analyzed using Generalized Linear Models. The analysis suggests that naturally non-abundant species (
Introduction
Disturbances in natural ecosystems due to human activities have increased alarmingly, jeopardizing the preservation of many species [1], particularly in tropical forests where the land-use change from forest to cultivated areas and the increase of human settlements are alarming [1–3]. Habitat fragmentation affects different biological groups and is one of the main causes of increased species extinction rates in recent decades [3, 4].
Phyllostomid bats (Chiroptera: Phyllostomidae) are an ideal taxa to study the effects of habitat fragmentation [5, 6], because of their high species richness (especially in tropical zones [7–9]) and differential response to fragmentation by distinct trophic guilds [5, 7, 9]. The importance of this group in tropical environments is in their role as seed dispersers, pollinators, and arthropod predators [5, 10].
Moreover, certain species-specific ecological traits are related to sensitivity to habitat fragmentation [4, 11]. Traits such as naturally low population size, high habitat specialization, large body size, low mobility, high trophic level and low tolerance to habitat disturbance (
Theoretical and empiric evidence predict that species with natural low abundance are more susceptible to fragmentation than those with high abundance [16]. This is because populations in isolated patches tend to decrease compared to co-occurring species with high abundance [16]. The latter usually have the capacity to tolerate modified habitats and tend to maintain or even increase their abundance, so they are less vulnerable to fragmentation [12, 13]. Species with small populations are more vulnerable to extinction risk caused by demographic and environmental stochasticity and genetic deterioration [16].
Species with larger body size also are more vulnerable to fragmentation, and therefore are prone to extinction, mainly because of greater energy requirements [4, 14, 17]. Wing morphology is a predictor of diverse ecological traits such as habitat preference, foraging strategy, dispersion ability, and home range size [5, 21–23]. Bat species with relatively high wing loading and aspect ratio (long and narrow wings) have a faster and more energy-efficient flight compared to other species with low-aspect (wide and short) and high maneuverability [22]. However, bat species with high loading and aspect ratio are less efficient in long-distance flights, thus limiting their mobility within fragmented landscapes and making them more vulnerable to fragmentation [17].
Likewise, trophic level may be related to sensitivity to fragmentation. Species found on the higher levels of the trophic pyramid (carnivorous) are more susceptible to extinction than species found on the lower levels (frugivorous and nectarivorous) because of more unstable population dynamics [4, 16]. Nevertheless, bat species feeding in the forest canopy and dependent on unevenly distributed resources tend to be more mobile, and thus, less vulnerable to fragmentation than understory-foraging bats that exploit locally restricted resources [17, 23–26].
Traits related to extinction risk in bats have been previously measured, but these approximations have been on large spatial scales (
Meyer
We assessed the relationship of six ecological traits of phyllostomid bats (body mass, trophic level, vertical foraging area, natural abundance in continuous forest, wing aspect ratio, and relative wing loading) with three measures of sensitivity to habitat fragmentation (species prevalence, change in abundance index, and nestedness ranking) in a fragmented forest in Los Chimalapas, Mexico.
Methods
Study site
The study was conducted in Los Chimalapas tropical rainforest (16° 55′-16° 47′ N and 94° 50′-94°40′ W, 160 to 600 m asl), in Oaxaca, Mexico (Fig. 1). The predominant climate is warm and humid [28], and the predominant vegetation is tropical rain forest, which covers approximately 48% of the total surface (590,993 ha) of Los Chimalapas [28]. This vegetation is characterized by trees exceeding 30 m high that maintain their leaves throughout the year. The forest is composed of several strata [28]: high arboreal (40 m), low arboreal (20–30 m), high shrubby (10–15 m) and low shrubby (less than 10 m).

Study area, sampling sites and analysis scale (4 km in diameter) where the sensitivity to fragmentation of phyllostomid bats in Los Chimalapas tropical rainforest was evaluated. The first upper left frame shows (arrow) the Los Chimalapas rainforest in Mexico. The second frame shows the study area in Los Chimalapas.
The study area is a fragmented landscape in early stages of disturbance, with a predominance of continuous forest patches as well as large areas of induced grassland and itinerant farming [29]. The disturbances in the region are the result of forest fires, semi-intensive livestock, and extraction of timber trees [28]. This has resulted in a considerable loss of vegetation cover in a relatively short period, starting in the 1950s.
Site selection
A spectral enhancement was done with orthorectified Landsat 7 ETM+ images using the normalized difference vegetation index (NDVI) as the first step in site selection [30, 31]. We then carried out a supervised classification on the resulting map, generating two categories: continuous vegetation areas and areas without apparent vegetation. We chose this dichotomous classification as we were unable to distinguish categories of vegetation reliably [29]. Upon this classification, 33 potential circular areas were delimited, each with a 4 km diameter. This size is a compromise between establishing a sufficiently small area, inside which biogeographical factors would not differ among the sites, and a sufficiently large area, where parameter measurements of communities (
We obtained seven structure measures for each circular area: forest coverage, patch density, mean patch size, edge density, mean patch form, mean proximity index, and Euclidian mean distance to the nearest neighbor. The measures were obtained using FRAGSTAT v3.3 [34]. All areas were ordered using a Principal Component Analysis (PCA), allowing us to recognize two large groups: continuous forest (CF) and fragmented forest (FF) areas [29]. CF areas were characterized by a larger forest cover proportion (> 84.96%), low patch density (< 1.27 patches/100 ha), and lower edge density (< 38.59 m/ha); while FF areas showed low forest cover (< 75.87%), high patch density (> 2.46 patches/100 ha), and higher edge density (> 62.07 m/ha). Six areas from both CF and FF were chosen based on accessibility (roads, trails) (Fig. 1); sampling sites were located in the geometrical center of each of the six sites.
Capture of bats
The study was conducted from March 2010 to February 2012, and included two dry and two wet seasons. Each of the twelve sites was sampled for three nights in each season. The time interval between each capture session at each site was six months. In total, captures were done during 144 nights (12 sites*3 nights/season*2 wet seasons*2 dry seasons). In each site, eight mist nets (6 × 2.5 m) were placed 20 m apart in a line along the vegetation trails. The nets were checked every 30 minutes during a period of approximately 8 hours each night (1800 h-0200 h), except on heavy rain days, during which the sampling period was shorter. Five nets were placed at the understory level (< 4 m) and three in the canopy level (> 6 m). Total sampling effort was of 125,049.6 m2n·h (square meters of net perhour). No significant differences (
Bats were identified using dichotomous keys [35, 36] and classified using the nomenclature of Simmons [37]. We determined the sex, age, body mass and reproductive condition of each specimen following the recommendations of Kunz
We captured 1,840 individuals from 29 phyllostomid bat species, of which 747 (25 species) were captured in CF and 1,093 (25 species) in FF area. The completeness of inventories was adequate (CF > 78.4%, FF > 77.1%), based on estimates of expected number of species obtained through the first-order jacknife estimator [29]. For purposes of this study, only 20 species were selected and nine species were excluded. Species that showed low abundance (< 4 captures) and low presence (< 4 sites), as well as species exclusive to FF or CF, were excluded (Appendix 2).
Derivation of ecological traits
Natural abundance of each species was calculated as the number of mean captures on each fragmented forest site divided by sampling effort (m2n·h). Before the analysis, we applied a logarithmic transformation to the data. We considered mean body mass (logarithmic transformation) for each species as the mean value of all captured adult organisms, excluding pregnant females. The trophic level was obtained by recognizing three upper trophic guilds (frugivorous, nectarivorous, and omnivorous) [5, 7, 8, 23]. The vertical foraging area of each species was determined from previous studies describing their foraging habits [23–26], as well as from data of our captures in the study area. Therefore, three categories were established: understory species that forage from ground level up to 5 m, canopy species (> 6 m), and species without clear preference. Wing aspect ratio was obtained following Norber & Rayner [22]: A = B2/S, where A = aspect ratio; B = wing span (m); S = wing area (m2), based on rectangular approximation (wing length*wing width). Wing loading was obtained using the following equation [22]: C = Mg/S, where C = wing loading, Mg = mass in grams, g = gravitational acceleration (9.81 m/s2), and S = wing area.
Statistical analysis
We used three measures of sensitivity: i) species prevalence in fragmented sites, expressed as the site occupation ratio [13, 17]; ii) relative changes in abundance expressed as an index in fragmented forests compared to continuous forests [16, 17] and iii) nestedness ranking. The last measure considers the loss of species in a predictable order after fragmentation, caused by differential vulnerability to extinction among species. This selective loss results in a nested structure between ensembles [40, 41]. An ensemble is nested when species inhabiting low species richness sites are a subset of species inhabiting high species richness sites [42], and the nesting range is the order in which species are nested [19]. The nestedness ranking shows that species present in low richness sites also tend to be present at high richness sites (higher ranks). This is because they have less extinction vulnerability than species inhabiting only high diversity sites (lower rank) that have high extinction vulnerability [19, 40].
An arcsine transformation was applied to the fragmented site occupancy ratio (
Three measures of sensitivity to fragmentation of 20 species of phyllostomid bats in Los Chimalapas rainforest, Mexico.

In order to arrange the species according to sensitivity to fragmentation and determine coincidences, a non-metric multidimensional scaling analysis (MDS) of a dissimilarity matrix (Bray-Curtis index) was used [46]. Analysis was run using MASS v7.3–9 package [47] on R v3.0.0 platform [48]. Independent analyses were conducted to evaluate the relationship between the six ecological traits of phyllostomid bats previously mentioned (Table 2), with three measures of sensitivity to fragmentation using Generalized Linear Models (GLM). Gaussian-type error approximation was used to evaluate prevalence and change in abundance, while Poisson-type error was used to evaluate nestedness ranking [49]. The goodness-fit of each response variable was evaluated based on the global model as percentage deviance explained [49].
Ecological traits employed to determine sensitivity to fragmentation of phyllostomid bats in Los Chimalapas tropical rainforest, Mexico.

Where BM = body mass (g); NA = natural abundance in continuous forests; TL = trophic level (1 = frugivorous; 2 = nectarivorous; 3 = omnivorous); WAR = wing aspect ratio; RWL = relative wing loading; VF = vertical foraging area (1 = understory; 2 = without preference; 3 = canopy).
For the model selection procedure, we performed an automated routine to “dredge” with the MuMIn v1.9.5 package [50] on R, which generates all possible combinations of explanatory variables and arranges the best models based on Akaike information criterion corrected for small sample sizes (AICc) [51]. Each model was arranged according to its AICc, ΔAICc (differences between respective and most parsimonious models) and W
Parameter estimation was based on an averaging model procedure, which implies calculating average AICc weight of different candidate models and estimating regression coefficients based on the confidence set [51]. Unconditional standard error is estimated likewise, which evaluates model precision over the set of models and provides a confidence interval in parameters estimation that accounts for uncertainty associated with model selection [51, 52]. Calculations were performed using MuMIn packing.
Results
The MDS analysis shows a high explicative power (stress = 0.063, [54]), which indicates high coincidence in measures of sensitivity to fragmentation. The analysis describes a sensitivity gradient (Fig. 2) determined largely by its first dimension (87% of variance explained). The gradient shows that certain species—Jamaican Fruit-eating Bat (
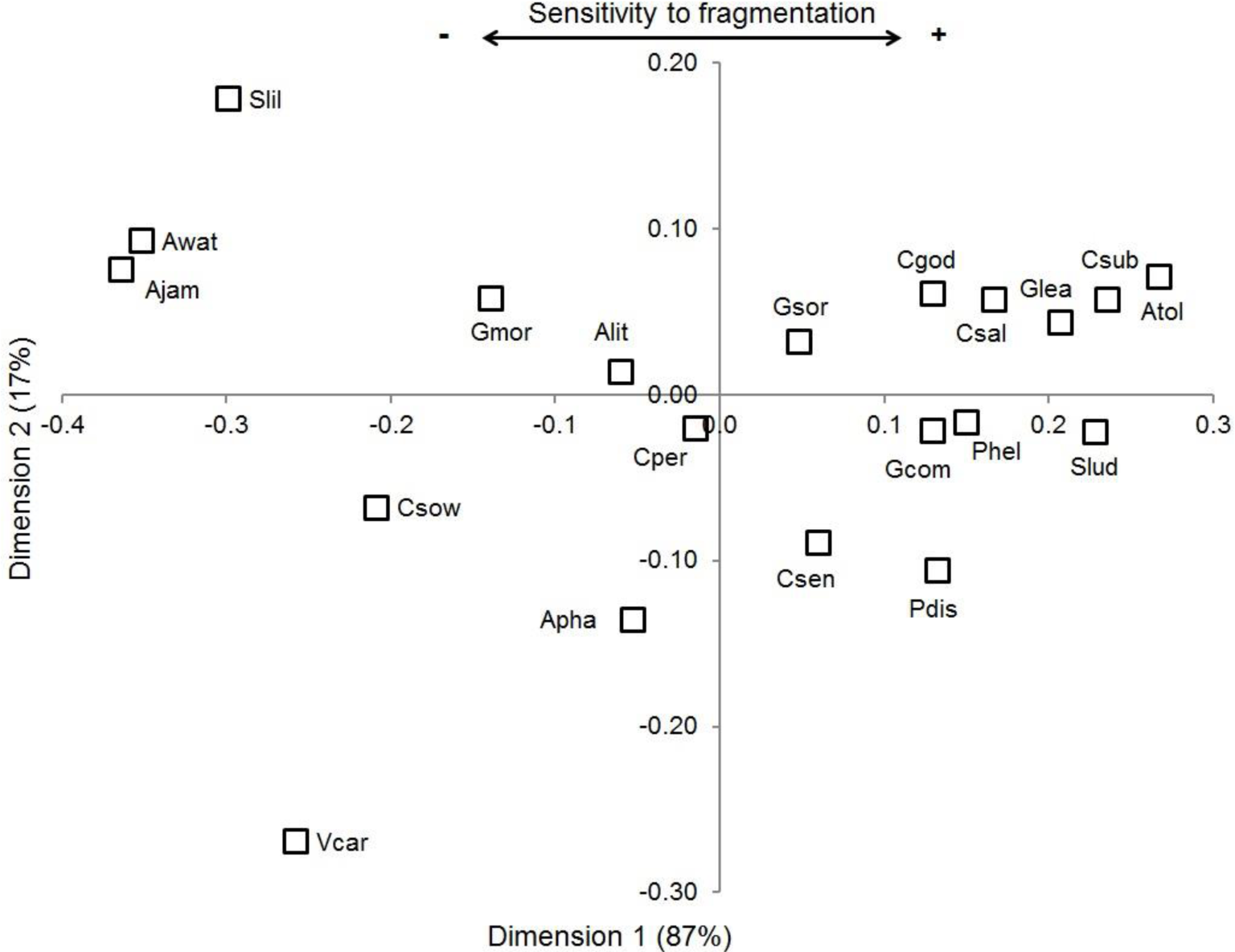
Ordering of phyllostomid bat species based in three measures of sensitivity to fragmentation throughout dimensions of non-metrical multidimensional scaling. The first dimension describes the degree of sensitivity to fragmentation (87% dissimilitude variation). Complete species names and abbreviations are found in Table 1.
The model selection procedure shows that in none of the cases a single best model had high support (
Results of model selection of the relationship between three measures of sensitivity to fragmentation with six ecological traits of phyllostomid bats in Los Chimalapas tropical rainforest, Mexico.

The considered models where those with a ΔAICc < 4. Where K = number of parameters; Log(L) = log-likelihood; AICc = Akaike Information Criterion corrected for small-sized samples; ΔAICc = difference between each model's AICc and best model candidate; Wi = model weight; BM = body mass; NA = natural abundance in continuous forests; TL = trophic level; WAR = wing aspect ratio; RWL = relative wing loading; VF = vertical foraging area.
Estimated parameters for each response variable based on the confidence set. Unconditional standard errors and significance in parenthesis. Significant values are shown in bold.

For change in abundance the best candidate model was the null model (
Discussion
Species prevalence and nestedness ranking showed high coincidence, suggesting that each one could be a reliable predictor of species vulnerability. Species prevalence is an index widely used [13, 53], and is employed as a predictor of metapopulational viability [54]. It also has been observed that nestedness ranking reflects the level of sensitivity to habitat fragmentation in ensembles with nested distribution [19, 40, 55].
The change in abundance index shows little relation to explicative variables. Originally, this index was designed to evaluate changes in abundance in experimentally controlled units (size and composition of patches, [16]) and has been adapted as an indirect measure of sensitivity to fragmentation [17]. The index evaluates the change in abundance of islands compared to continuous forest mainland. This index could show unpredictable behavior in fragmented terrestrial systems, because the change in abundance depends on the intensity of the disturbance, the elements present in the fragmented landscape, and the tolerance of the species and capacity of adaptation to those elements [6, 32, 56].
Species prevalence and nestedness ranking coincide in identifying natural abundance in continuous forests as a variable associated with sensitivity to fragmentation. This suggests that less abundant species, with presumably smaller population sizes, are more likely to decline due to habitat fragmentation, and are therefore at greater risk of local extinction [4, 16, 17]. Species with small population sizes are more vulnerable to phenomena such as demographic and environmental stochasticity, genetic deterioration, and social dysfunction [4, 57]. The importance of natural abundance has been widely shown for bird species [19, 57–59], and partially for tropical bats [17].
In contrast, natural abundance also suggests that species with large population sizes could be less affected by habitat fragmentation [4, 12, 13]. This is because species with higher natural abundance have a greater dispersion capacity amongst patches [17, 27, 60]. In addition, species that tolerate and exploit resources in modified habitats could adapt efficiently to changes in patches (5, 7, 8, 56), particularly to the edge effect.
In insular systems, strong evidence indicates that tropical bats displaying low tolerance to edge effect are predisposed to population declines because of habitat fragmentation [17]. However, the magnitude of edge effect depends on the degree of contrast between matrix and patches; a greater effect is observed in high contrast landscapes [11, 12], and possibly is less severe in fragmented terrestrial landscapes [7, 61–63] because the contrast between border and matrix is less pronounced [63]. Additionally, bats are able to use elements of fragmented landscapes such as corridors, stepping-stones, and even the vegetation matrix [3, 56, 64]. Therefore, it is difficult to expect the same edge effect in terrestrial and insular systems [63], though this still needs to be further studied.
The results suggest that species such as Toltec Fruit-eating Bat (
These less abundant species show low mobility, which limits their movement across the fragmented landscape. This is the case of small frugivorous bats of the genus
In contrast, species such as Thomas's Fruit-eating Bat (
Unlike other studies where morphological wing traits—wing aspect ratio and relative wing loading—are intimately related to the extinction risk of bats [21, 27], we found weak evidence (nestedness ranking) of the effect of wing aspect ratio on extinction risk. In general, these variables had low predictive power, probably because of the range of variation of wing aspect ratio and relative wing loading is low in the analyzed species [17]. These were mostly species dependent on forests or occupants of closed-vegetation environments. We did not include insectivorous open-space species, which would have allowed greater contrast in the analysis [22].
We did not find evidence of a relationship between body mass and sensitivity to fragmentation. Empirical studies amongst mammal and bird communities report a number of different patterns of the relation between body size and extinction risk: positive [71, 72], negative [73], and unrelated [12, 58]. The relationship between body size and extinction rate shows a high degree of complexity, and observed patterns are unclear [16], since body size can be correlated to other more important variables such as abundance, population fluctuations, and population growth rates [4, 16]. The relationship between abundance and body size remains unclear [72, 74].
Evidence of trophic level and vertical foraging area effects on sensitivity to fragmentation was not found, thus coinciding with previous studies [17]. A variable more important and descriptive than trophic level could be diet specialization, but this variable was not included; detailed descriptions of the diets of species within our area of study do not exist, and existing descriptions show high geographical variation [75–77]. On the other hand, vertical foraging area is a highly flexible trait [26] and a sole species can use several forest strata [24, 26]. It is also possible that vertical foraging area does not reflect the degree of habitat specialization. Finally, we recommend the inclusion of other variables in further studies that could have more predictive power, such as edge tolerance, diet specialization, mobility, population fluctuation, and population growth rate.
Implications for conservation
We found natural abundance in continuous forests to be highly related to sensitivity to fragmentation in the species studied (Fig. 3). This finding is relevant because natural abundance and population size are fundamental to determining the degree of sensitivity to fragmentation and local extinction risk of a species. The adequate identification of these traits, not only in bats but also in other groups such as non-flying mammals, birds, reptiles, and amphibians could be essential for effective species-specific conservation strategies [4, 11, 12, 14].

(A) Salvin's Big-eyed Bat (
Conservation strategies that have been proposed include ensuring the presence of a high proportion and density of large patches, as well as maintaining vegetation corridors to promote the effective connectivity and persistence of naturally low abundant species in fragmented landscapes [1, 6, 29]. It is also important to promote the recovery of areas with secondary vegetation in advanced successional stages and the conservation of plant species that are the diet of several phyllostomid bats [7, 77]. The application of these strategies is particularly important in our study area because Los Chimalapas is the last rainforest areas in Mexico with high biodiversity, which is under severe pressure from human activities [28, 29].
Footnotes
Acknowledgements
The Consejo Nacional de Ciencia y Tecnología of Mexico (CONACyT) granted a doctoral studies scholarship (No. 228955) to the first author. This investigation had financial support from the Secretaría de Investigación y Posgrado of the Instituto Politécnico Nacional of Mexico (IPN) through projects SIP-20100377, SIP-20110395 and SIP- 20120962; and from the Programa Institucional de Formación de Investigadores (PIFI). We are grateful for the comments to improve this document of D. Mondragón, G. Ramos, R. del Castillo and R. Solano. We thank the municipal authorities of Santa María Chimalapa and those persons who provided invaluable assistance in the field work.
Appendix 1. Nestedness ranking based on an absence-presence matrix of the phyllostomid bat community in twelve sites in Los Chimalapas rainforest. The matrix is ordered (matrix temperature T = 32.95,P < 0.001) to maximize nesting according to Rodríguez-Gironés & Santamaría (2006). Species vulnerable to extinction are present only in sites diverse in number of species,and they have low rank. Complete species names and abbreviations are found in Appendix 2. * Species excluded from the analysis of sensitivity to fragmentation.

| Nestedness ranking | Species / Sites | F3 | C2 | C5 | C4 | F5 | C6 | F2 | C1 | C3 | F6 | F1 | F4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Slil,Ajam,Awat | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 2 | Vmaj | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 |
| 3 | Csow | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 |
| 4 | Gmor | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 |
| 5 | Apha | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 |
| 6 | Alit | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 7 | Cper | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 |
| 8 | Csen | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 |
| 9 | Pdis | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 |
| 10 | Gsor | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 |
| 11 | Gcom | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 0 |
| 12 | Phel | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 0 |
| 13 | Slud | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 |
| 14 | Cgod | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 0 | 0 |
| 15 | Csal | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 |
| 16 | Glea | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 |
| 17 | Csub | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 0 |
| 18 | Drot* | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 |
| 19 | Atol | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 |
| 20 | Laur* | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| 21 | Msil* | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 22 | Cvil* | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 23 | Hund* | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| 24 | Ubil* | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 25 | Aazt* | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 26 | Vspe* | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 27 | Msch* | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
Appendix 2. List of phyllostomid bat species in fragmented and continuous rainforests in Los Chimalapas. Abbreviations in parentheses.

| Species | Continuous Forests | Fragmented Forests | Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phyllostomidae Family | C1 | C2 | C3 | C4 | C5 | C6 | F1 | F2 | F3 | F4 | F5 | F6 | |
| Subfamilia Desmodontinae | |||||||||||||
| |
0 | 0 | 0 | 0 | 0 | 0 | 5 | 10 | 7 | 10 | 26 | 0 | 58 |
| Subfamilia Glossophaginae | |||||||||||||
| |
0 | 1 | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 0 | 12 | 1 | 18 |
| |
4 | 23 | 1 | 11 | 9 | 1 | 5 | 0 | 25 | 0 | 0 | 0 | 79 |
| |
1 | 0 | 1 | 0 | 5 | 0 | 0 | 2 | 1 | 0 | 0 | 0 | 10 |
| |
2 | 5 | 0 | 4 | 7 | 2 | 23 | 11 | 3 | 80 | 6 | 9 | 152 |
| |
1 | 10 | 0 | 0 | 0 | 1 | 12 | 23 | 5 | 25 | 3 | 0 | 80 |
| |
2 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Subfamilia Phyllostominae | |||||||||||||
| |
0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| |
0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| |
0 | 0 | 0 | 0 | 0 | 0 | 0 | 12 | 6 | 1 | 0 | 0 | 19 |
| |
0 | 7 | 2 | 3 | 2 | 5 | 0 | 0 | 1 | 0 | 2 | 1 | 23 |
| |
0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Subfamilia Carolliinae | |||||||||||||
| |
0 | 1 | 8 | 5 | 8 | 1 | 0 | 1 | 7 | 9 | 21 | 7 | 68 |
| |
21 | 58 | 29 | 12 | 28 | 38 | 0 | 13 | 15 | 95 | 9 | 19 | 337 |
| |
0 | 2 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 5 |
| Subfamilia Stenodermatinae | |||||||||||||
| |
2 | 10 | 3 | 13 | 14 | 13 | 39 | 25 | 35 | 26 | 22 | 47 | 249 |
| |
5 | 0 | 0 | 3 | 5 | 7 | 0 | 0 | 1 | 0 | 0 | 1 | 22 |
| |
9 | 10 | 21 | 11 | 0 | 5 | 17 | 42 | 63 | 15 | 12 | 3 | 208 |
| |
11 | 4 | 58 | 5 | 12 | 9 | 1 | 1 | 30 | 2 | 11 | 40 | 184 |
| |
0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| |
3 | 6 | 3 | 1 | 4 | 2 | 1 | 1 | 1 | 0 | 5 | 0 | 27 |
| |
3 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 1 | 0 | 0 | 0 | 6 |
| |
9 | 11 | 16 | 9 | 16 | 21 | 10 | 26 | 17 | 10 | 20 | 11 | 176 |
| |
1 | 8 | 6 | 1 | 4 | 2 | 1 | 3 | 0 | 0 | 4 | 1 | 31 |
| |
0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 4 |
| |
0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 2 |
| |
0 | 3 | 3 | 1 | 5 | 0 | 2 | 0 | 1 | 0 | 3 | 0 | 18 |
| |
0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 2 |
| |
18 | 6 | 8 | 2 | 1 | 8 | 1 | 3 | 5 | 1 | 1 | 0 | 54 |
| Total | 92 | 166 | 162 | 85 | 123 | 119 | 117 | 174 | 226 | 275 | 159 | 142 | 1,840 |
