Abstract
Removal of major xenoantigens of the Galα1-3Gal (α-Gal) epitope and the nonhuman sialic acid
Impact Statement
We identified a method for
Introduction
Finding an ideal heart valve is a critical issue in treating patients with heart disease, especially for those with congenital heart disease who had disease specifically related to right ventricular outflow tract abnormalities, because they require repeated reoperations as they grow older. The ideal xenogeneic valve should be nonthrombogenic, nonimmunogenic, durable, and have the potential to grow. Xenogeneic decellularized heart valves have been extensively studied as an attractive alternative for the development of viable biological valves capable of remodeling and regeneration. Decellularization protocols included a process to remove the cellular components of the original tissue while preserving the three-dimensional extracellular matrix (ECM) of the biological tissue.
1
Although a consensus on decellularization protocols has not yet been reached, we previously developed an optimal decellularization protocol using sodium dodecyl sulfate (SDS) and Triton X-100 through a multistep method with hypotonic, isotonic, and hypertonic buffer solutions.2–4
This protocol has been shown to preserve the microstructure, the degree of cross-linking, and tissue strength; reduce cytotoxicity; and inhibit
There is a significant clinical need for valve tissues that can prevent degeneration, provide growth characteristics, and reduce the replacement cycle, particularly for pediatric patients. The recellularization of decellularized xenografts is believed to have the potential to shift xenoreactive immune responses toward an anti-inflammatory phenotype and enhance cellular regeneration, making it a promising candidate for an ideal valve tissue.
In this study, we aimed to identify a method for
Materials and Methods
Tissue preparation and decellularization
This study was approved by the Institutional Animal Care and Use Committee of Clinical Research Institute, Seoul National University Hospital (IACUC No. 24-0081-S1A0). Porcine pericardia were obtained from Taewoong Medical Inc., (South Korea). After fat and connective tissue removal on the surface of pericardium, these tissues were trimmed. The tissues were sterilized with 0.1% PAA for 4 h at room temperature (RT) and then washed with phosphate buffered saline solution (PBS) under agitation at RT. The porcine pericardium was directly decellularized using various solutions. Each process of decellularization was carried out under an agitator (180 rpm) in an orbital shaker (SHO-2D, DAIHAN).
The porcine pericardial tissues were initially decellularized with hypotonic solution for 14 h at 4°C and hypotonic solution with 0.25% SDS (Sigma, 436143) in diH2O containing 0.5% (v/v) Triton X-100 for 24 h at 4°C while agitating, and then washed with distilled water for 12 h at 4°C. After washing, these tissues were decellularized with isotonic solution for 48 h at 4°C. The tissues were treated in PBS with 30% polyethylene glycol 1000 and 1% antibiotic/antimycotic for 48 h, using an agitator at 4°C. The tissues were finally treated with a hypertonic solution for 3 h at 4°C. The tissues were washed in PBS for 24 h. Decellularized samples were prepared by our decellularization protocols as described by Lim HG, et al. 2
Enzymatic digestion
Native and decellularized porcine pericardia were treated with PNGase-F (#P0704L, 1000U, NEB) to obtain PNGase-F concentrations of 500, 1000, 1500, and 2000 unit/mL. In addition, native and decellularized porcine pericardia were treated with recombinant α-galactosidase (G7163, Sigma) to obtain recombinant α-galactosidase concentrations of 0.1 and 0.2 unit/mL. Native tissues served as an untreated control or negative control. Native and SDS + Triton X-100 (Tx)-decellularized porcine pericardia were treated with different enzymatic treatments: α-galactosidase, PNGase-F, or α-galactosidase in combination with PNGase-F. The sliced porcine pericardial tissues of 5 × 5 mm size in tubes with isotonic solution were conducted for 24 h in 4°C with agitation. Each enzymatic digestion with sliced tissues was treated at 37°C for 24 h under agitation, and then the tissues were rinsed in PBS under agitation (120 rpm) at RT. Finally, the tissue samples were stored in sterile PBS supplemented with 1% (v/v) penicillin/streptomycin and 400 μL/L amphotericin B for 24 h at 4°C.
Biomechanical analysis
After enzymatic digestions, all samples were stored at 4°C for 24 h and then rinsed in PBS. Biomechanical analyses were performed on native pericardium with or without enzymatic treatments. According to manufacturer’s instruction, the sliced samples were cut into approximately 5.0 × 20 mm (test length × width,
Histology and lectin histochemistry for staining
For histological staining, native and SDS + Tx-decellularized porcine pericardia were treated with different enzymatic treatments: α-galactosidase, PNGase-F, or α-galactosidase in combination with PNGase-F, and these sliced samples were fixed at 4% paraformaldehyde for 24 h in 4°C. Histological sections (5 μm) of each sample were stained for hematoxylin and eosin (H&E) and lectins staining. The tissue sections were incubated with lectins at 1:250 overnight at 4°C. Effects of the enzymatic treatments on carbohydrate-binding lectins such as isolectin B4 (IL-B4/GSL-IB4) (#B-1205, Vector Laboratories Inc), specific for α-galactose residues; Wheat Germ Agglutinin (WGA) (#RL-1022, Vector Laboratories Inc); Datura Stramoniumlectin (DSL) (#B-1185, Vector Laboratories Inc); Ricinus Communis Agglutinin I (RCA-I) (#RL-1082, Vector Laboratories Inc); Soybean Agglutinin (SBA) (#B-1015, Vector Laboratories Inc); Wisteria Floribunda Lectin (WFA) (#B-1355, Vector Laboratories Inc); Peanut Agglutinin (PNA) (#B-1075, Vector Laboratories Inc); Maackia Amurensis lectin I (MAL I) (#B-1315, Vector Laboratories Inc); Jacalin (#B-1155, Vector Laboratories Inc); Erythrina Cristagalli Lectin (ECA) (#B-1145, Vector Laboratories Inc). Stained sections were additionally stained with avidin-linked Texas Red (#A-2006; Vector Laboratories Inc). The sections were washed 3 times with PBS, and stained sections were imaged using an inverted fluorescence microscope (DMI4000B, Leica, Germany). Stained images were obtained using Application Suite X Image Viewer (Leica).
Lectin binding assay
Lectin binding analysis was determined for porcine pericardial tissues with or without enzymatic treatments: α-galactosidase, PNGase-F, or α-galactosidase in combination with PNGase-F, and these sliced samples were homogenized in Radio-Immunoprecipitation Assay (RIPA) lysis buffer (ATTO) and stored in frozen condition. Tissue homogenates were coated onto a 96-well plate (Corning) in duplicates with coating buffer (Biosesang), and incubated overnight at 4°C, and then each wells was blocked with carbo-free blocking solution (#SP-5040-125, Vector Laboratories Inc.) for 1 h at RT. Wells were aspirated and incubated with diluted primary lectins (1:500) at 100 μL per well in the carbo-free blocking solution for 3 h at RT. After the primary lectins incubation, the plates were washed 3 times with PBS containing 0.05% Tween 20 (PBST, Biosesang). Subsequently, the wells were incubated for 30 min at RT with streptavidin-conjugated peroxidise (1:1000, #SA-S0004, Vector Laboratories Inc.) in blocking solution. Following several washes with PBST, the reaction was developed with a 3,3′,5,5′-tetrame-thylbenzidine (TMB) solution (#34028, Thermo Scientific). The absorbance of lectins was measured at 450 nm using an ELISA reader (TECAN, Spark).
In vitro culture and expansion of HUVECs and human adipose tissue-derived stem cells (ADSCs)
HUVECs (#C2517A, Lonza) were purchased from ATCC (PCS-100-010, USA). HUVECs were used as pooled primary cells frozen after the first subculture. HUVECs were cultured at 37°C in a humidified atmosphere of 5% CO2 with Endothelial Growth Medium-2 BulletKitTM (EGM, CC-3162, Lonza). Human ADSCs were purchased from Thermo Scientifics (#R7788115, USA). ADSCs were cultured at 37°C in a humidified atmosphere of 5% CO2 with Dulbecco’s Modified Eagle Medium (DMEM) medium containing 10% fetal bovine serum (FBS), transforming growth factor beta 1 (TGF-β1, 2.5 ng/mL, ab50036, Abcam), and bone morphogenetic protein 4 (BMP-4, 2.5 ng/mL, SRP6156, Sigma). The cell culture medium of both cells was changed every 3 days.
Pericardium modification
For recellularization, modification of porcine pericardium was performed on native, decellularized, and decellularized porcine pericardium treated with α-galactosidase in combination with PNGase-F. The pericardial samples were cut into approximately 1 × 1 cm (length × width,
ADSCs and HUVECs seeded onto modified pericardium (HUVECs/ADSCs coculture)
For endothelialization of the pericardium, all pericardial samples were seeded on both sides with ADSCs and HUVECs coculture. ADSCs (passage 3) were seeded on top of all modified pericardial samples (P- or DP-Fb + H + VEGF) at approximately 1 × 105 cells (day 0) per well in a 12-well plate. The seeded samples were turned over, and ADSCs were seeded on the bottom of samples in DMEM medium supplemented with 2% FBS, TGF-β1, and BMP-4 (day 2). After ADSCs seeding on both sides, subsequently, HUVECs (passage 6) were seeded on the upper side of these samples at 2 × 105 cells (day 16) and then seeded on the lower side of the pericardium at 2 × 105 cells (day 18) in a 12-well plate. These samples were incubated in EGM-2 medium supplemented with TGF-β1 (2.5 ng/mL) and BMP-4 (2.5 ng/mL), and these pericardial samples were cultured until day 28.
Immunofluorescence staining of recellularized pericardium
Native, decellularized, and decellularized porcine pericardia treated with α-galactosidase in combination with PNGase-F, which had been seeded with ADSCs and HUVECs, were fixed in 4% paraformaldehyde solution and sectioned into 5 μm on microtome. Paraffin sections were processed for H&E and immunefluorescence (IF) staining. IF staining was performed using embedded sections and deparaffinized. Tissue samples were incubated in antigen retrieval buffer (10 mM citrate buffer, pH 6.0) at 95°C for 10 min. Tissue sections were then blocked with 5% blocking serum. Tissue slides were then incubated with primary antibodies for vimentin (ab16700, Abcam, dilution 1:200), calponin (ab227661, Abcam, dilution 1:100), fibronectin (F0916, sigma, dilution 1:100), and CD31 (ab182981, Abcam, dilution 1:2000) at 4°C overnight and were washed 3 times with PBST. The tissues were then incubated with antirabbit secondary antibody conjugated with Alexa Fluor 488 (A11008, Invitrogen, 1:500) for 2 h and antimouse secondary antibody conjugated with Alexa Fluor 488 (AB150113, Invitrogen, 1:500) for 2 h, and were rinsed 3 times in PBST. Nuclei were counterstained with DAPI (Invitrogen, 1:1000) for 1 min. The sections were washed 3 times with PBST, and stained sections were imaged using an inverted fluorescence microscope (DMI4000B, Leica, Germany). Stained images were obtained using Application Suite X Image Viewer (Leica).
Results
Histology and biomechanical property in decellularized porcine pericardium with enzymatic digestion
Porcine pericardial tissues were processed according to our developed decellularization protocol using 0.25% SDS + Triton X-100 and subjected to a multistep method using hypotonic, isotonic, and hypertonic buffer solutions (Supplementary Fig. S1) (Supplementary Fig. S2). Separate samples were prepared for each enzyme treatment, which was treated with or without 0–2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1–0.2 unit/mL) for 24 h. H&E staining was used to examine the fiber structure, such as the collagen fiber pattern, structural loosening, and the presence of cells, and in all cases, a normal collagen structure similar to that of fresh tissue was observed regardless of enzyme concentration (Fig. 1) (Supplementary Fig. S3).
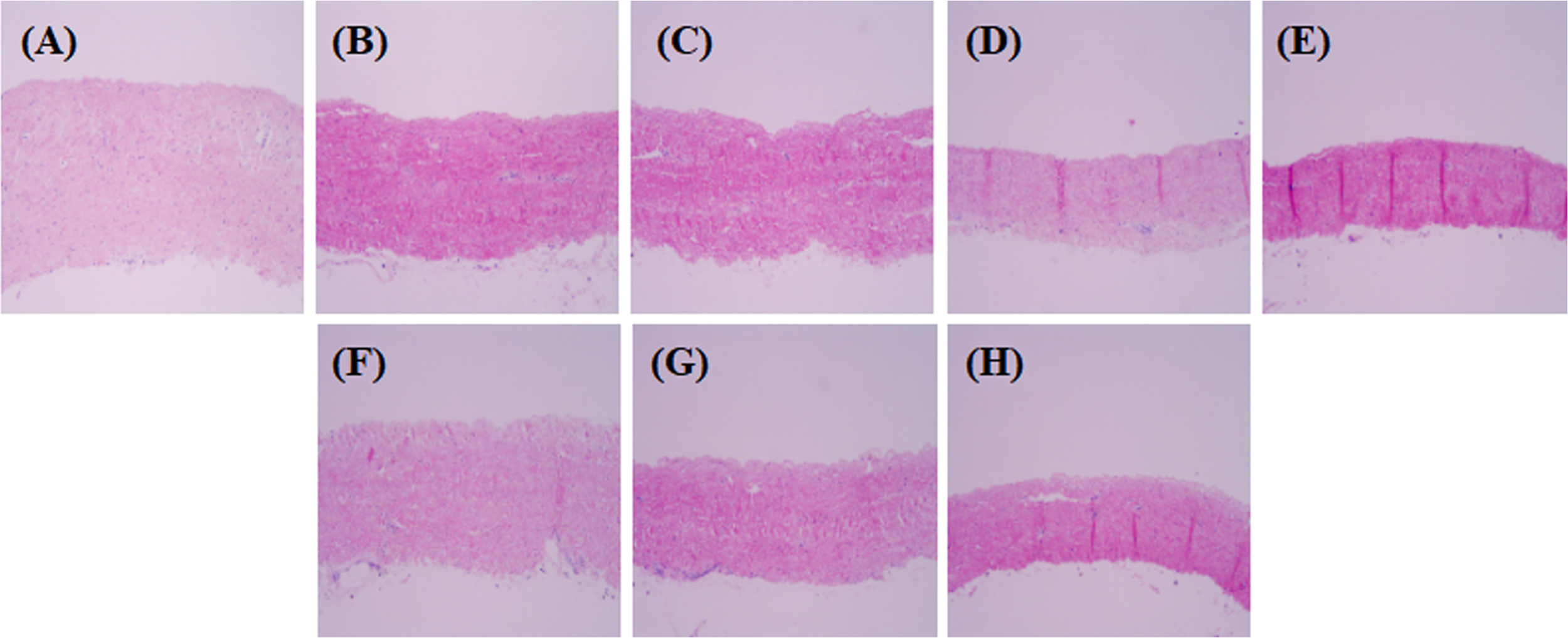
Histological observation of native, α-galactosidase, and PNGase-F-treated group.
Biomechanical properties were determined with a uniaxial tensile test and a permeability test. The tensile stress and permeability test showed no significant differences between native porcine tissue and the differently (0–1000 unit/mL) PNGase-F-treated porcine tissue and differently (0.1–0.2 unit/mL) α-galactosidase-treated porcine tissue and PNGase-F and α-galactosidase both treated porcine tissue (Fig. 2). These findings suggested that the effects of PNGase-F/α-galactosidase treatment concentration and combination were not reflected in the biomechanical properties of the porcine pericardium.
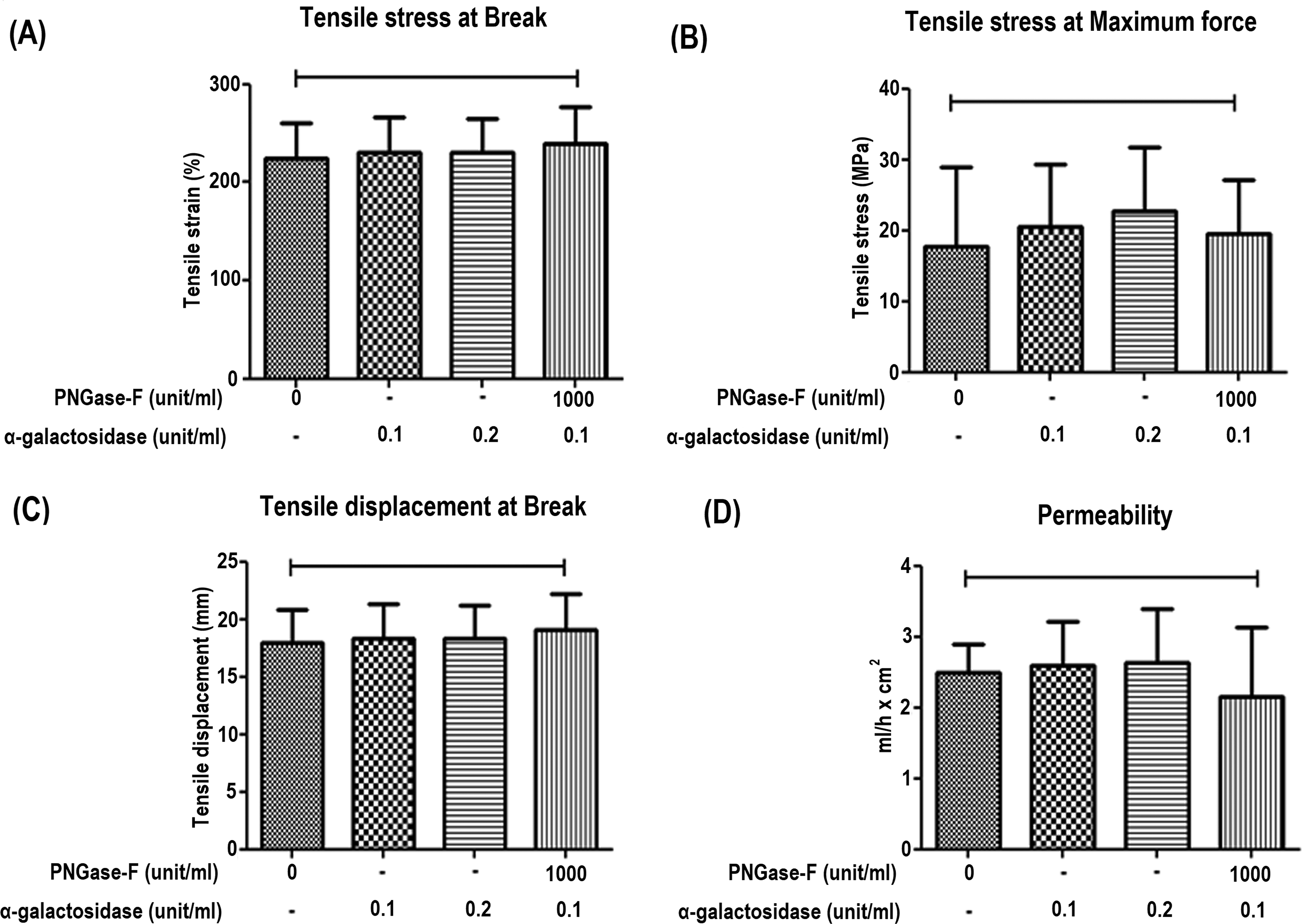
Biomechanical properties of the native, α-galactosidase, and PNGase-F-treated group.
Lectin histochemistry and lectin binding assay in decellularized porcine pericardium with enzyme digestion
In xenotransplantation, carbohydrate components such as lectins play a significant role, with elevated lectin levels being closely associated with immune rejection. We investigated the effects of enzymatic digestion using PNGase-F and α-galactosidase in fresh pericardium on carbohydrate removal through the expression of lectins. Native tissue, tissues treated with varying concentrations of PNGase-F and α-galactosidase, and tissues treated with both PNGase-F and α-galactosidase were stained with Jacalin, MAL-I, WGA, RCA-I, GSL, ECA, PNA, SBA, WFA, and DSL. Each enzymatic treatment showed a decrease in lectin expression as the concentration increased, indicating effective removal of carbohydrates in lectin histochemistry. In addition, when α-galactosidase and PNGase-F were treated together, a more significant reduction in lectin expression was observed, indicating that both α-Gal and non-α-Gal epitopes were more effectively removed (Fig. 3).

Lectin histochemistry of native, α-galactosidase, and PNGase-F-treated group.
To quantitatively determine the expression of MAL-I, WGA, RCA-I, GSL, ECA, PNA, SBA, WFA, and DSL, a lectin binding assay for native, PNGase-F (0–1000 unit/mL), and α-galactosidase (0.1–0.2 unit/mL), α-galactosidase 0.1 + PNGase-F 1000 was performed. PNGase-F treatment significantly inhibited lectin binding levels in a concentration-dependent manner. Interestingly, when α-galactosidase was treated simultaneously with PNGase-F at the same concentration, most lectins showed reduced binding more synergistically compared to PNGase-F treatment alone (Fig. 4).
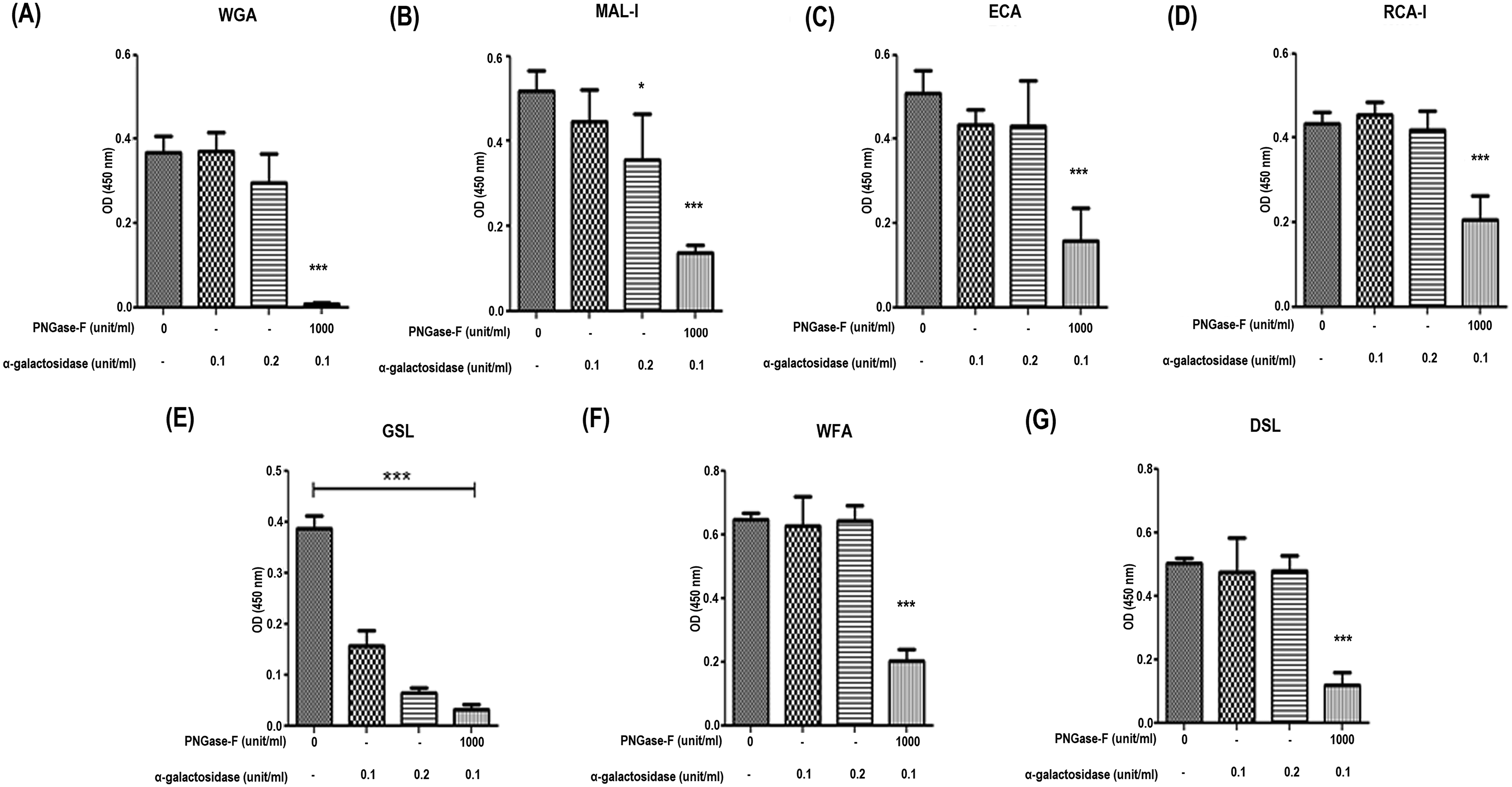
Quantitative evaluation of lectins binding to solubilized proteins of porcine pericardium. The porcine pericardium was treated with or without 0–1000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1–0.2 unit/mL) for 24 h. Each tissue samples were homogenized with lysis buffer, and detection of lectin-binding to solubilized extracellular matrix proteins on the surface of the tissue was performed to detect the expression of WGA
We applied an additional decellularization protocol and observed through histochemical staining that the group treated with α-galactosidase 0.1/PNGase-F 2000 showed the most effective removal of lectin binding level (Fig. 5).
Expression of lectins on decellularized pericardium with or without enzymatic treatment.
In vitro evaluation of recellularized porcine pericardium with ADSC and HUVEC coculture
Histological observation in both ADSC and HUVEC seeded onto modified pericardium
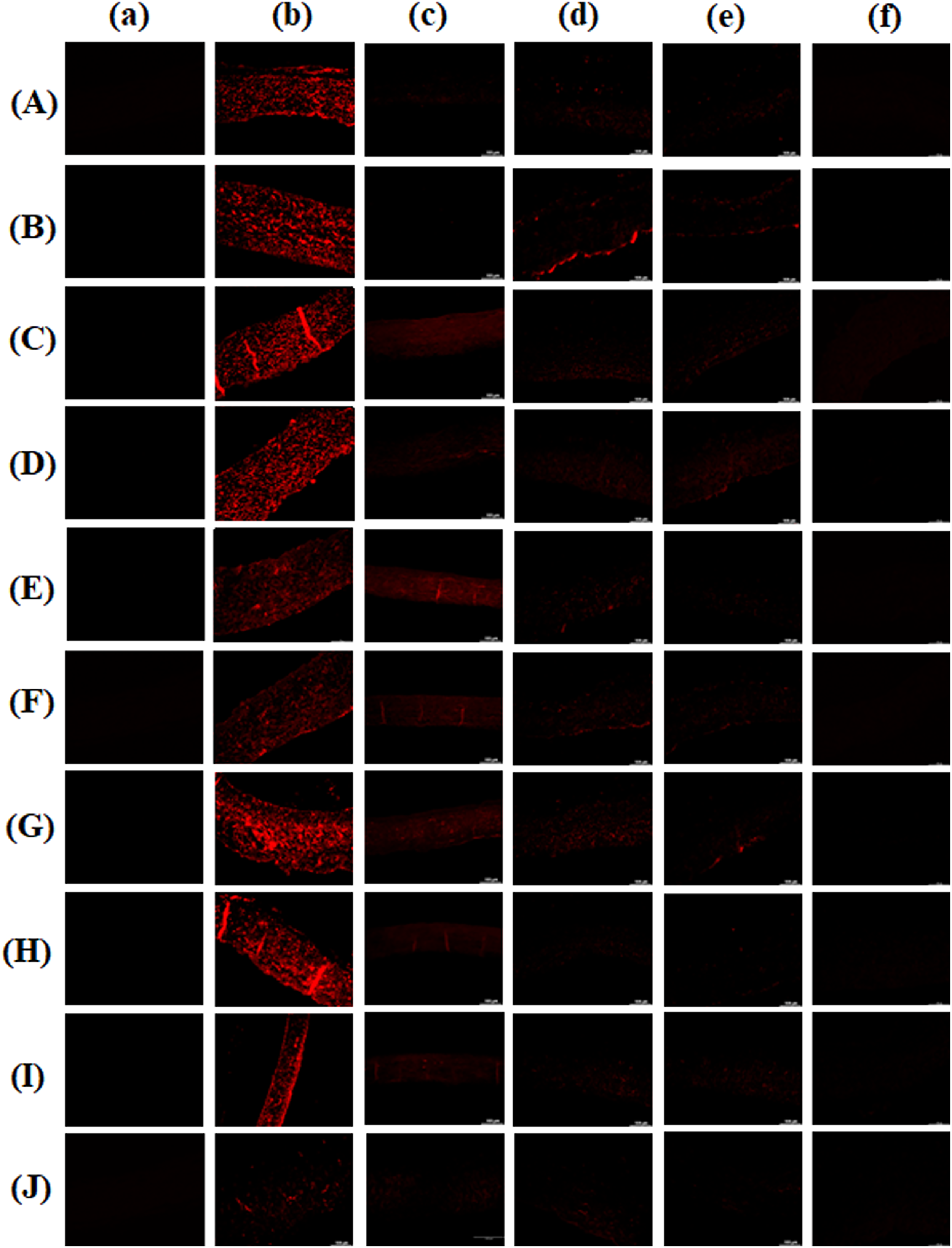
First, decellularized porcine pericardium was treated with or without 2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1 unit/mL) for 24 h. Native, decellularized, and decellularized-α-galactosidase/PNGase-F combination pericardial samples were coated with a fibrin mesh + heparin (Fb + H) and incubated with VEGF (Fb + H + VEGF). Then, all modified pericardial samples were seeded with human ADSCs and subsequently with HUVECs. These modified pericardial samples were incubated on day 7 and on day 28, and these samples were stained with H&E. In the native, decellularized, and decellularized-α-galactosidase/PNGase-F combination pericardial samples, the tissue structure was preserved in all groups. In the group treated with and without the α-galactosidase/PNGase-F combination after decellularization, cell infiltration was observed. In the decellularized tissue, cells were observed to line the outer edges of the tissue at day 7, while cells were observed to infiltrate into the tissue by day 28. In the α-galactosidase/PNGase-F combination-treated group, cells had already infiltrated into the tissue by day 7, and by day 28, a significant number of cells were found to infiltrate into the tissue (Fig. 6).
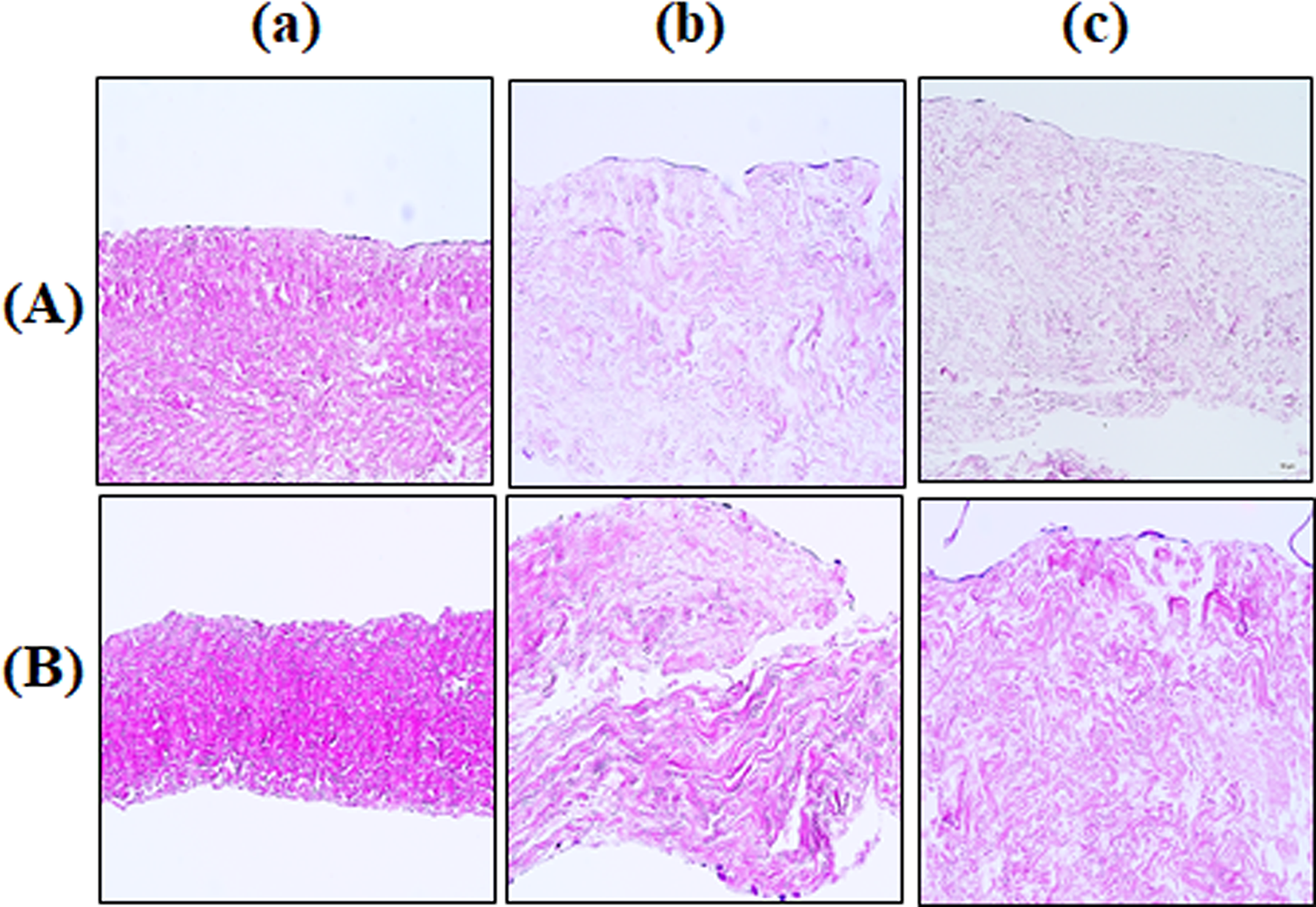
Histological observation in both ADSC and HUVEC seeded onto modified pericardium. The decellularized porcine pericardium was treated with or without 2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1 unit/mL) for 24 h. Native
Immunofluorescence staining in both ADSCs and HUVECs seeded onto modified pericardium
Native, decellularized, and decellularized-α-galactosidase/PNGase-F combination samples were incubated for 7 days and 28 days, and these samples were stained with vimentin, which is a marker of mesenchymal cells. Vimentin-positive cells were present on the surface of the pericardium on day 7. On day 28, a significant number of cells were observed to line the surface and penetrate deeper into the tissue. In particular, the decellularized-α-galactosidase/PNGase-F combination sample showed faster mesenchymal cell infiltration into the tissue, leading to accelerated recellularization (Fig. 7).
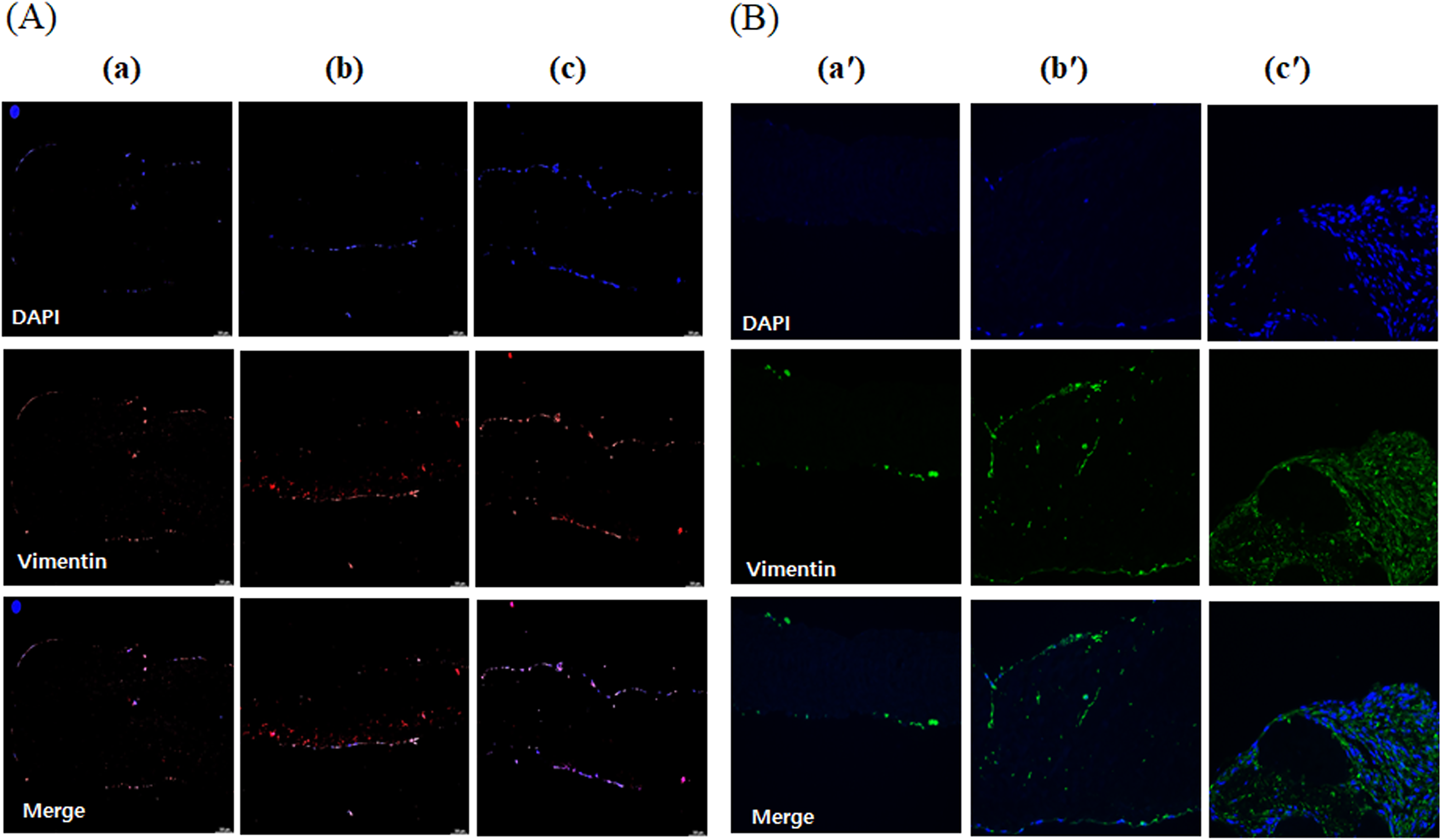
Immunofluorescence staining of vimentin in ADSC and HUVEC coculture on to modified pericardium. The decellularized porcine pericardium was treated with or without 2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1 unit/mL) for 24 h. All porcine pericardium samples modified with fibrin, heparin, and VEGF (Fb + H + VEGF). Then, all modified pericardium samples were seeded with ADSCs and HUVEC. Native
Calponin is an intermediate marker indicating cell differentiation into vascular smooth muscle cells. On day 7 of culture, minimal stainings were observed. On day 28, calponin-positive cells began to appear on the surface of the pericardium. In the decellularized-α-galactosidase/PNGase-F combination sample, calponin-positive signals were observed inside the tissue, suggesting that the infiltrating cells are differentiated into smooth muscle cells (Fig. 8).
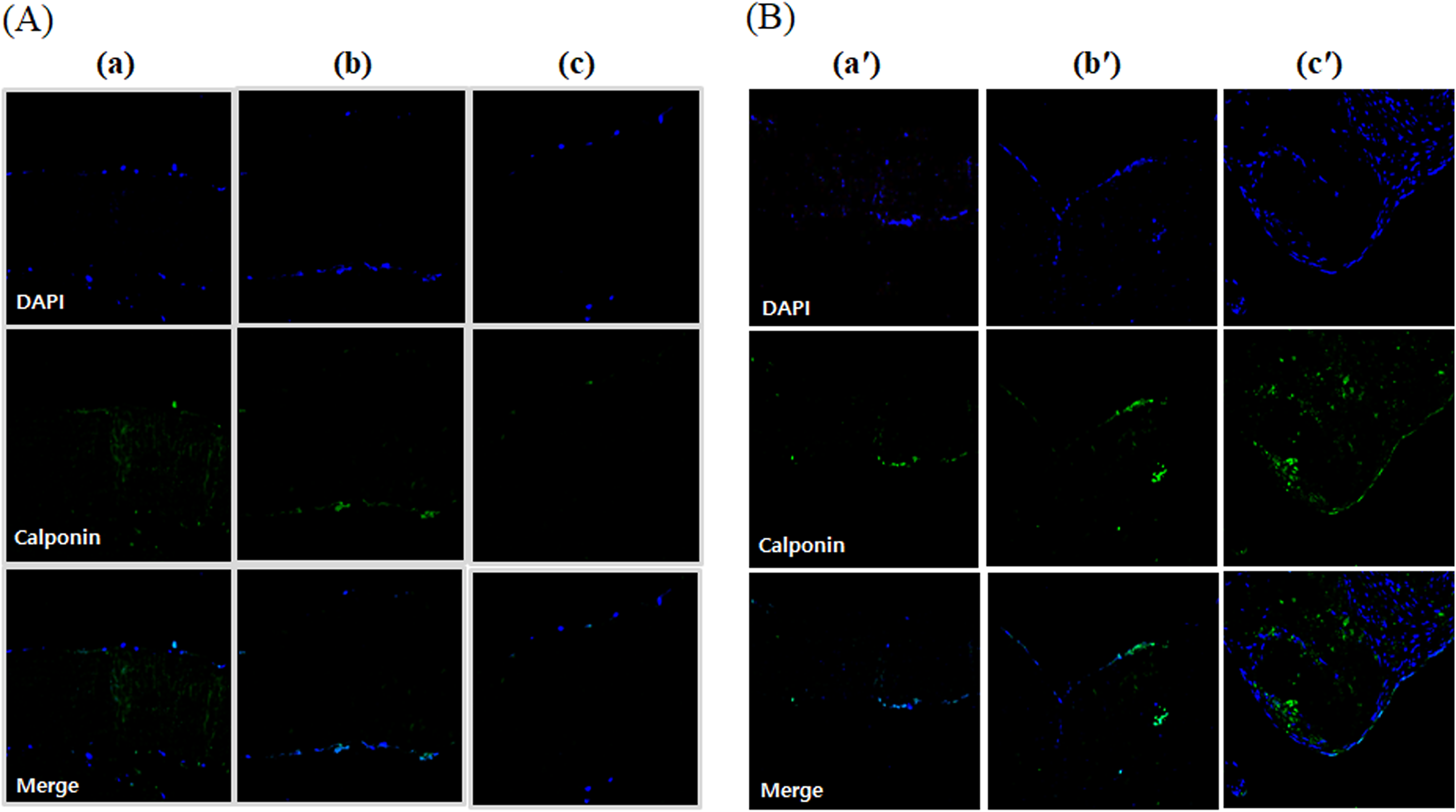
Immunocytochemical staining of calponin in ADSC and HUVEC coculture onto modified pericardium. The decellularized porcine pericardium was treated with or without 2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1 unit/mL) for 24 h. All porcine pericardium samples were modified with fibrin, heparin, and VEGF (Fb + H + VEGF). Then, all modified pericardium samples were seeded with ADSCs and HUVEC. Native
Fibronectin staining serves as a key component of the ECM, promoting cell attachment and regeneration. Intense fibronectin staining was observed, and stronger signals in tissue were detected in the decellularized group cultured for 28 days compared to 7 days, as well as in the decellularized group treated with both α-galactosidase and PNGase-F simultaneously (Fig. 9).
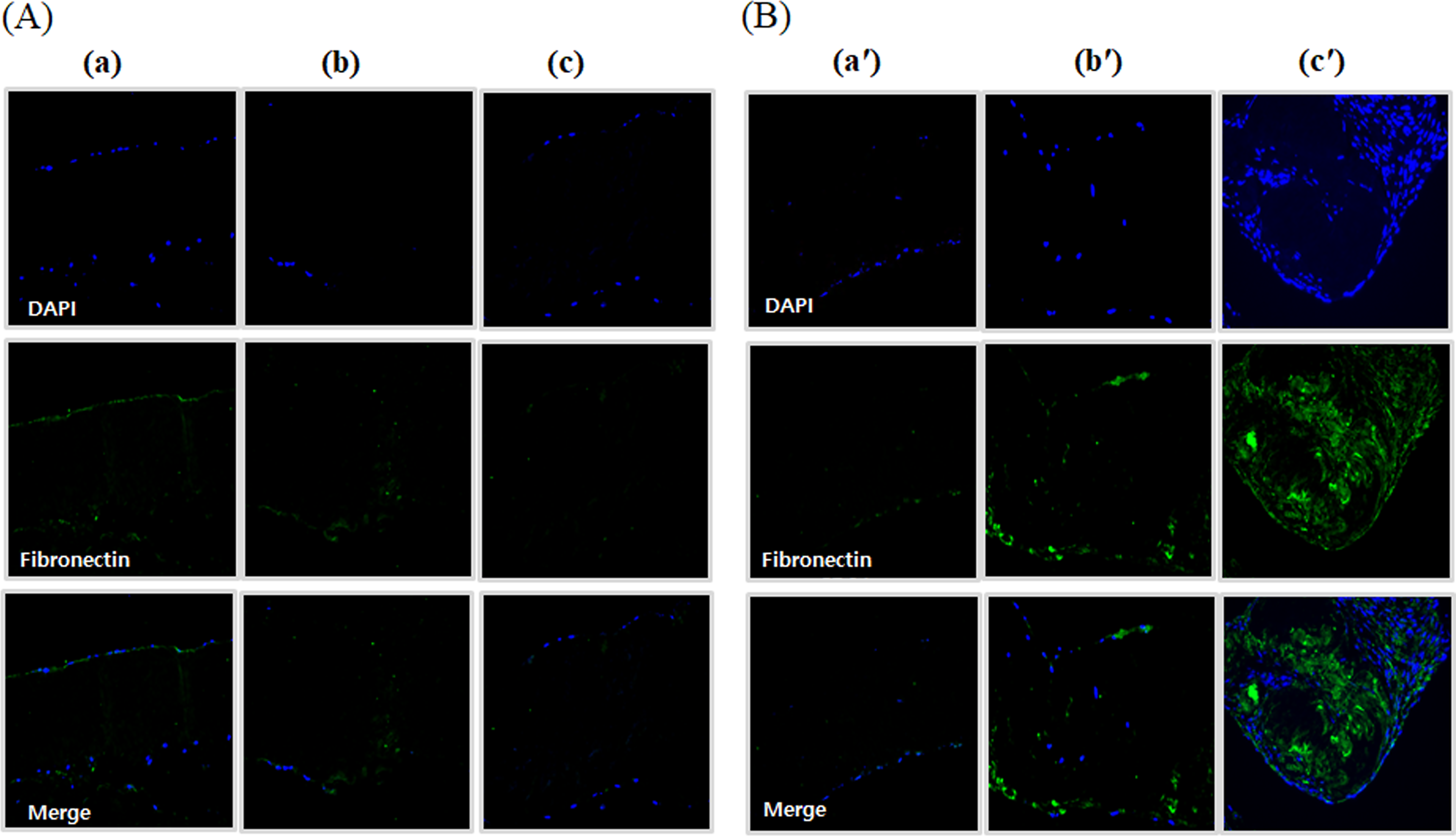
Immunocytochemical staining of fibronectin in ADSC and HUVEC coculture onto modified pericardium. The decellularized porcine pericardium was treated with or without 2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1 unit/mL) for 24 h. All porcine pericardium samples were modified with fibrin, heparin, and VEGF (Fb + H + VEGF). Then, all modified pericardium samples were seeded with ADSCs and HUVEC. Native
CD31 positivity indicates the expression of CD31 (platelet endothelial cell adhesion molecule-1, PECAM-1) protein in cells or tissues. CD31 is primarily expressed in endothelial cells and some immune cells, playing a critical role in angiogenesis, cell-cell adhesion, and inflammatory responses. Similar to the staining of other markers, a strong signal was observed within the tissue on day 28 in the decellularized-α-galactosidase/PNGase-F combination group, suggesting that new blood vessels are forming in the recellularized graft (Fig. 10), (Supplementary Fig. S4).
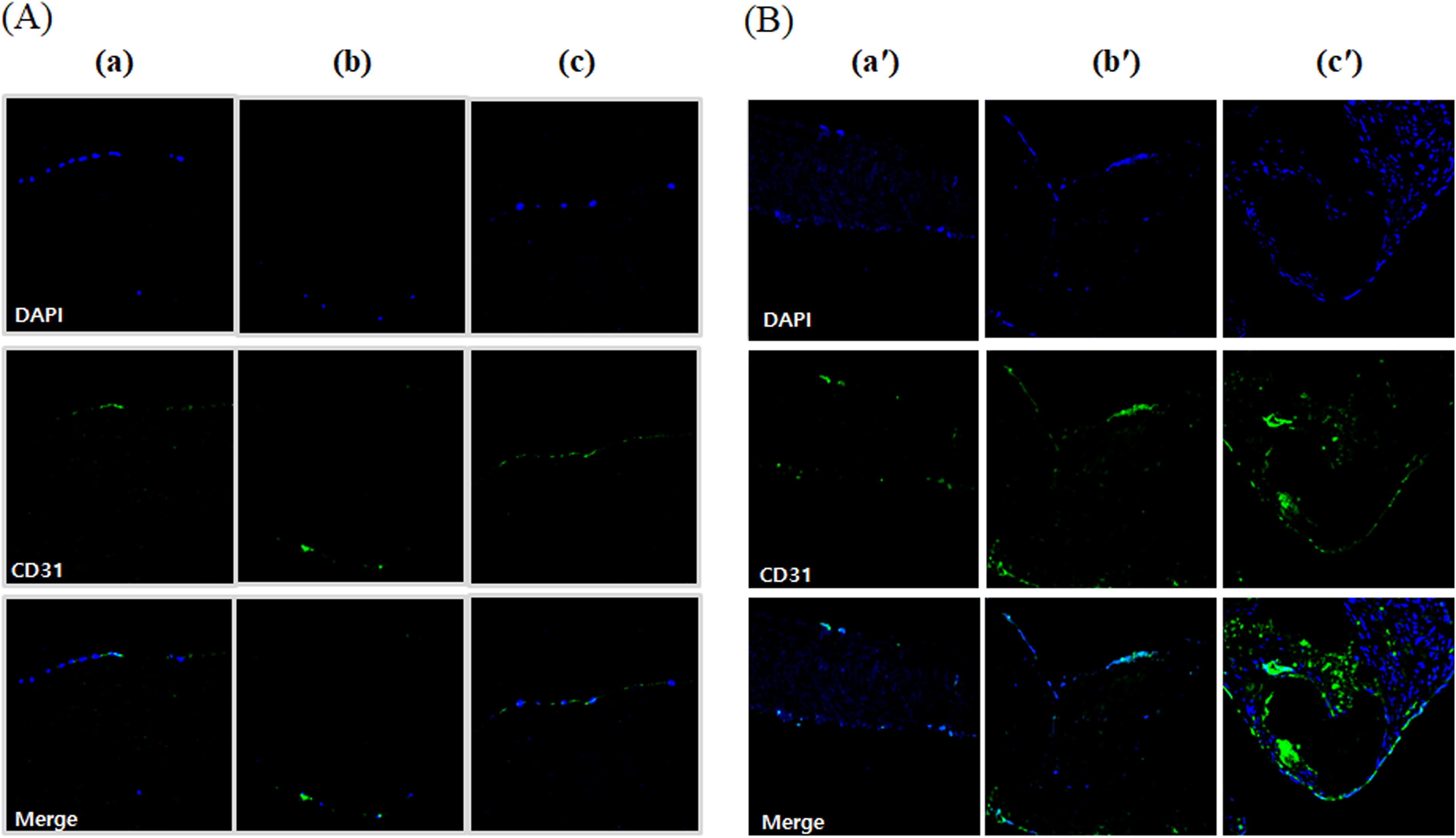
Immunocytochemical staining of CD31 in ADSC and HUVEC coculture onto modified pericardium. The decellularized porcine pericardium was treated with or without 2000 unit/mL PNGase-F, respectively, in the absence and presence of α-galactosidase (0.1 unit/mL) for 24 h. All porcine pericardium samples were modified with fibrin, heparin, and VEGF (Fb + H + VEGF). Then, all modified pericardium samples were seeded with ADSCs and HUVEC. Native
Discussion
Significant research has been conducted to develop an ideal tissue-engineered heart valve by selecting suitable tissues to serve as scaffolds for optimal valve structures. In the case of xenogeneic tissues, efforts have focused on appropriately removing cells that may trigger immune responses while minimizing damage to the ECM.9,10 Decellularization protocols have been developed to process the ECM into an immunologically tolerable state while preserving its functionality. Generally, decellularization is achieved using chemical methods (alkaline/acid, detergents, alcohols), physical methods (electroporation, pressurization, and freeze/thaw), and biological methods (enzymes, etc.).1,11 The removal of nucleic acid residues is crucial in all decellularization processes because these residues tend to adhere to ECM and attract circulating calcium salts, leading to calcification and subsequent tissue degeneration. Therefore, during tissue decellularization, proteolytic enzymes are used to break down proteins and effectively separate cellular components from the connective tissue.
In particular, when using pig or bovine pericardial tissues, it is necessary to effectively remove α-Gal (xenoepitope), which induces xenogeneic immune rejection in humans.
5
We have previously developed a decellularization protocol incorporating an α-galactosidase, which has been utilized to create artificial pericardial pulmonary valves. These valves have been successfully applied in clinical practice, and the outcomes to date have been satisfactory.6,12 However, it has been found that the presence of non-Gal antigens (Neu5Gc) in the GalT-KO area does not trigger hyperacute xenorejection but can mediate acute graft damage to both vascularized organs and cellular grafts in xenotransplantation.
13
Furthermore, anti-non-Gal antibodies will be induced upon xenograft or tissue, and it can be anticipated, analogously to the human allo-situation, that these will have an impact on xenograft or tissue survival and durability. In xenotransplantation, lectins are carbohydrate-binding protein components that are deeply involved in immune rejection, and increased lectin levels are closely associated with immune rejection. The effect of enzymatic treatment using PNGase-F on carbohydrate removal was investigated by analyzing lectin expression. In our previous studies, we demonstrated that combining decellularization with PNGase-F treatment effectively removes non-Gal antigens without affecting the mechanical stability of the tissue.
14
The removal of non-Gal antigens, such as Neu5Gc, primarily requires deglycosylation treatment, which effectively eliminates carbohydrates from
The decellularized xenograft scaffold is recellularized with host cells in an
However, when a decellularized xenograft valve tissue is implanted to enable host cell recellularization, the absence or delayed recruitment of cells capable of repairing and remodeling the ECM during the valvular cycle may lead to tissue failure or degradation due to valve tissue damage.
For
Fibronectin mediates various cell interactions with the ECM, playing a crucial role in cell adhesion, migration, growth, and differentiation.
22
When decellularized tissue treated with a combination of α-galactosidase and PNGase-F was cocultured with MSCs and HUVECs, the expression of fibronectin was elevated, suggesting that MSCs infiltrated the tissue more rapidly and abundantly.
23
Fibronectin may have originated from the serum contained in the culture medium or from the MSCs. Some studies have suggested that preimplant conditioning with fibronectin alone or in combination with other growth factors can enhance
Through vimentin staining, we confirmed that MSCs migrated into the tissue, and the activation of interstitial cells was initiated, indicating the beginning of tissue remodeling. Particularly, as myofibroblasts can also exhibit vimentin positivity, this suggests that it represents an early stage of ECM remodeling and tissue repair. However, to confirm whether these cells are in the early stages of myofibroblasts, it is necessary to check for coexpression of α-SMA positivity.
Calponin staining suggested that MSCs interacted with the surrounding ECM and transitioned into myofibroblasts, exhibiting a mature smooth muscle cell phenotype. This was particularly evident after 4 weeks of coculturing MSCs and HUVECs on tissue treated with α-galactosidase and PNGase-F, where calponin expression was significantly increased. These findings indicate that multipotential stem cells undergo recellularization at the valve site and differentiate into mature cell phenotypes.
However, excessive activation of myofibroblasts can lead to tissue fibrosis and valve stiffening, which may result in functional impairment over the long term.
When the graft achieves sufficient endothelialization in the human body and therapeutic angiogenesis occurs, the incidence of thrombosis is reduced. In addition, paracrine signaling promotes recellularization, enabling differentiation into improved tissue structures. Previous studies have observed an increase in markers expressed in vascular endothelium, CD31, through the coculture of HUVECs and MSCs, demonstrating that coculture is effective for the rapid endothelialization of vascular substrates.25,26 This finding was also replicated in our study. Thus, xenografts recellularized with MSCs can regulate the host immune response, enabling effective tissue remodeling, delaying the biodegradation process, and extending the survival of the tissue until successful ECM replacement is achieved in the recipient.
Compared with recent TRICOL decellularization based on osmotic shock, detergents, and nuclease treatment, 27 our decellularization protocol using SDS and Triton X-100 through a multistep method with hypotonic, isotonic, and hypertonic buffer solutions was developed without nuclease and successfully commercialized due to its economic feasibility and mass productivity. Lately, decellularization and subsequent recellularization particularly focus on the obstacles and unresolved issues that must be overcome toward optimal tissue engineering. 28 The glycans with terminal α-Gal and Neu5Gc are known to play a major role in xenoimmunogenicity, ultimately leading to graft rejection. 29 Similar to our results, the decellularization protocols were revised in combination with enzymatic deglycosylation to reduce xenoimmunogenicity, by means of cells, carbohydrates, and primarily α-Gal epitope removal, and decellularized xenografts treated with PNGase-F resulted in potentially reduced immunogenicity with mechanical stability. 30
In the development of tissue-engineered heart valves, strategies to recellularize the entire valve, including decellularized xenoantigen-free scaffolds, with an appropriate cell population face numerous challenges. This experiment suggests the potential for promoting recellularization of xenogeneic valvular tissue
In conclusion, we have successfully produced xenoantigen-free scaffolds by demonstrating the safety and the synergistic effect of α-galactosidase and PNGase-F treatments and proved effective recellularization for the xenoantigen-free scaffolds not previously reported in the literature.
Authors’ Contributions
J.K.Y.: Contributed to the conceptualization, generating figures of the article, writing of the article. S.Y.K.: Contributed to the conceptualization, generating figures of the article, and writing of the article. S.Kim.: Contributed to the generating figures of the article. K.M.L.: Contributed to the generating figures of the article. S.Ko.: Contributed to the generating figures of the article. G.B.K.: Contributed to the conceptualization, and writing of the article. H.G.L.: Contributed to the conceptualization, generating figures of the article, and writing of the article. Y.J.K.: Contributed to the conceptualization, and writing of the article.
Footnotes
Funding Information
This research was supported and funded by SNUH Lee Kun-hee Child Cancer & Rare Disease Project, Republic of Korea (grant number: 25C-022-0100).
Disclosure Statement
No competing financial interests exist.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
