Abstract
Objective
Delta-9-tetrahydrocannabinol (Δ9-THC), the primary active component of cannabis, was isolated from the cannabis plant in 1964, and ever since then, scientists focused more on compounds that activate this receptor and created synthetic cannabinoid derivatives to study the pharmacological and toxicological effects of cannabinoids. Following this, analogues of Δ9-THC began to be synthesized especially for medical use of cannabis. Synthetic cannabinoids may be employed in autoimmune illnesses, and further research on cannabis derivatives is necessary to understand how they affect immune system cells.
Methods
We have used MDMB-4en-PINACA, which is an indazole-based synthetic cannabinoid. Methods: In our study, the immunomodulatory effects of MDMB-4en-PINACA and its derivative were evaluated on mammalian macrophages. ELISA was used to quantify pro-inflammatory cytokine levels and assess their impact on macrophages. Following that, the intracellular mechanism of action of synthetic cannabis derivative was tested by measuring the amounts of phosphorylated PI3K using flow cytometry.
Results
Our findings imply MDMB-4en-PINACA and its derivative stimulated the anti-inflammatory cytokines in the lipopolysaccharide (LPS)-stimulated macrophages.
Conclusion
Consequently, these compounds may be used as therapeutic possibilities for autoimmune and inflammatory diseases.
Keywords
1. Introduction
The euphoric effects of cannabinoids, such as delta-9-tetrahydrocannabinol (THC) (Δ9-THC) found in cannabis acts by stimulating many receptors, including CB1, CB2 and PPARγ, to provide its neuroprotective, antispasmodic, and anti-inflammatory effects. 1 Since THC, the primary active component of cannabis, was isolated from the cannabis plant in 1964, a wider research has been done on compounds that act on this receptor. Later, Δ9-THC analogues were created for medical use in biomedical and cannabis research. Synthetic cannabinoids are, in general, a broad class of compounds that affect the CB- and CB2 cannabinoid receptors, resulting in different physiological reactions. As indicated by previous studies cannabinoids are known for their anti-inflammatory effects, 2 however, some increase the proinflammatory cytokines via activation of the MAP kinases p38 and c-Jun NH(2)-terminal kinase (JNK).3,4 Synthetic cannabinoids that mimic Delta-9-tetrahydrocannabinol (THC) not only pose significant risks to users but also exhibit significant toxicity. 5
MDMB-4en-PINACA (5-CI-ADB-A), also known as MDMBPINACA N1-pentyl-4-en isomer, is an indazole-based synthetic cannabinoid structurally related to 5F-MDMB-PINACA. It differs from 5F-MDMB-PINACA due to replacement of 5-fluoropentyl (5F) that is present in 5F-MDMB-PINACA with a pent-4-ene (4-en) moiety; however, MDMB-4en-PINACA shares structural similarities with 5F-MDMB-PINACA. (MDMA 1) MDMB-4en-PINACA was first reported to the EMCDDA’s Early Warning System, which includes the European information system and data base on new drugs (EDND), in August 2018.6,7 Ozturk et al reported the detection of MDMB-4en-PINACA in an herbal product from Turkey in April 2019.
6
In their study, the
In this study, MDMB-4en-PINACA and its metabolite (Figure 1) were tested on the LPS activated and inactivated macrophages for the detection of their immunomodulatory and immunostimulatory effects. Pro-and anti-inflammatory cytokine balance determines the fate of the inflammation.
8
They can also present these foreign invaders to the adaptive immune system cells to further shape the overall immune response. Macrophages have been found in most of the tissue lesions of the autoimmune and inflammatory disorders.9,10 TNF-α, IL-6, GM-CSF and IL-12p40, which are the major pro-inflammatory cytokines, associated with autoimmune and inflammatory disorders, were measured in the presence and absence of Lipopolysaccharides (LPS).11-13 Certain kinase activities initiate inflammatory reactions by transmitting signals in pathways such PI3K intracellularly. To assess its activity, we used flow cytometry and measured the amounts of phosphorylated (active) form of this protein.14-16 The molecular structures of the molecules, (A) MDMB-4en-PINACA and (B) MDMB-4en-PINACA M (derivative)
According to our findings, the synthetic cannabinoid MDMB-4en-PINACA and its metabolite had varying impacts on the activated macrophages’ levels of production of various pro-inflammatory cytokines. The PI3K pathway was not involved in the actions of the synthetic cannabis or its metabolite.
2. Materials and Methods
2.1. Toxicological Analysis in Synthetic Cannabinoids
LC/MS-MS Gradient Program for Synthetic Substance Analysis

The precursor ion of the MDMB-4en-PINACA substance with a retention time of 3.921 min is 358.05, the fragmentation ions are 213.1, 298.2 and 145.05, and the chromatogram is shown in Figure 2. Calibration curve range; 0,5-200 ng/L was formed from ten points by adding the internal standard (20 ng/L). The calibration coefficient (R2) is > 0.99. Chromatogram of MDMB-4en-PINACA
2.2. Materials for Biological Activity Tests
J774.2 murine macrophage cell line was purchased from Sigma Aldrich and cultured according to its manufacturer under aseptic conditions. Cells were grown in Roswell Park Memorial Institute (RPMI 1640) (R8758, Sigma-Aldrich, USA) media containing 10% fetal bovine serum (F7524, Sigma-Aldrich, USA) with 1% antibiotics (100 μg/cm3 penicillin and 100 μg/cm3 streptomycin) (P4333, Sigma-Aldrich, USA).17,18
The cells were seeded onto 24-well plates at a concentration of 106 cells/mL/well and incubated overnight in a humidified chamber at 37 °C.17,18 Cannabinoid derivative and its metabolite were water soluble and prepared at various concentrations including 1, 5, 10 and 20 μM and applied onto the cells with or without 1ug/ml of LPS (L5293, Sigma-Aldrich, USA). Upon 24 hours of induction, several assays such as Enzyme-Linked ImmunoSorbent Assays (ELISAs), cell counting, and flow cytometry experiments were conducted.17,18 To develop a anti-inflammatory model, we treated the cells with 10 μg/mL of Salicylic acid (247588, Sigma-Aldrich, USA) along with 1μg/mL of LPS (L5293, Sigma-Aldrich, USA) from Escherichia coli and named this group as negative control cells. The SA is known for its anti-inflammatory effects and LPS for its inflammatory effects. By using these two molecules together it is possible to develop an anti-inflammatory model. The positive group cells were treated with 1μg/mL of LPS to trigger the inflammation only. As for the adjuvant control group, the cells were treated with 1μg/mL of LPS along with 5µg/mL of Al2O3,17,18 since the Al2O3 is known to increase inflammation more. We wanted to detect whether the cannabinoids of interest might be considered as an adjuvant. The media in which the cells treated with the agents were collected and the levels of different cytokines including TNFα (cat no: 555212), IL-6 (cat no: 555183), GMCSF (Cat no: 555126), and IL12p40 (Cat no:555220) were measured by ELISA kits (BD Biosciences, CA, USA) according to the manufacturer’s guidelines. 18 Briefly, anti-cytokine antibodies used in the ELISA kit are noncovalently bonded to plastic microwell plates. The plates were washed and the soluble cytokine proteins present in the samples were applied to the plate and captured by the immobilized antibodies. The unbound antibodies were decanted and biotin-conjugated anti-cytokine antibodies also known as detection antibodies were used to identify the captured cytokine proteins. This is followed by an enzyme-labeled streptavidin binding step. Upon binding, the amount of colored product which was produced were detected spectrophotometrically using an ELISA-plate reader at 405 nm. The levels of cytokines produced were calculated. The viability of the cells treated with the concentrations (1, 5, 10, and 20 µM) (chosen according to -the tetrazolium salt MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) assay-, data not shown) was confirmed by Trypan assay (93595, Sigma-Aldrich, USA) in which the cells were trypsinized and stained with Trypan blue dye according to the manufacturer’s instruction. Upon staining with the dye, both the total number of cells and the number of blue cells (dead cells) were counted. The formula for calculating cell viability was as follows: % cell viability = (total number of cells - number of dead cells)/total number of cells x 100. Each data set had three biologically independent experiments and Graph Pad Prism 5 was used to plot the graphs and conduct the Kruskal-Wallis test for the statistical analyses. 19
2.3. Flow Cytometry Analysis for PI3K Intracellular Pathways
As described in our previous study, the cells were fixed and permeabilized according to the BD (BD Fix Buffer I -557,870) protocol. It was then stained with Anti p-PI3K (Invitrogen PE Mouse, p85/p55, cat no: MA5-36,954). Phosphorylated PI3K levels were measured by flow cytometry (BD FACS ARIA III) analysis. Each experiment was repeated thrice.
2.4. Statistical Analysis
GraphPad Prism was used to analyze the data. Statistical significance for all the assays among the groups was determined by using Kruskal-Wallis test followed by Dunn test. Statistical significance was defined as
3. Results
3.1. Cell Viability in the Presence of Synthetic MDMB-4en-PINACA and Its Metabolite
All the Control Groups: Blank: The Cells Were Treated With Only Culture Media (Untreated Cells). Positive Control Cells: The Cells Were Treated With LPS only. Negative Control Cells: The Cells Were Treated With LPS and Salicylic Acid Simultaneously. Adjuvant Control: The Cells Were Treated With LPS and Al2O3 Simultaneously

According to our findings, there was no difference between the control groups (blank group in which the cells were treated with only media) and the study groups (the negative, positive, and adjuvant control cells) that had cannabinoid derivative and its metabolite as depicted in Figure 3. The negative control is the one in which the cells were treated with LPS and Salicylic acid (SA). This way we were able to model the anti-inflammation model by triggering the cells with LPS and inhibiting the inflammation by SA. The positive control cells were designated as the cells treated with only LPS (as an inflammatory agent). In addition, we have added an adjuvant control group in which the cells were treated with an adjuvant agent such as Al2O3 to enhance the inflammation. Therefore, the concentrations which were applied onto the cells were considered to be non-toxic. The cell viability was measured by Trypan blue staining of the cells. MDMB-4en-PINACA and its metabolite MDMB-4en-PINACA M were applied at concentration of 1, 5, 10 and 20 µM with or without 1ug/ml of Lipopolysaccharide (LPS). (* 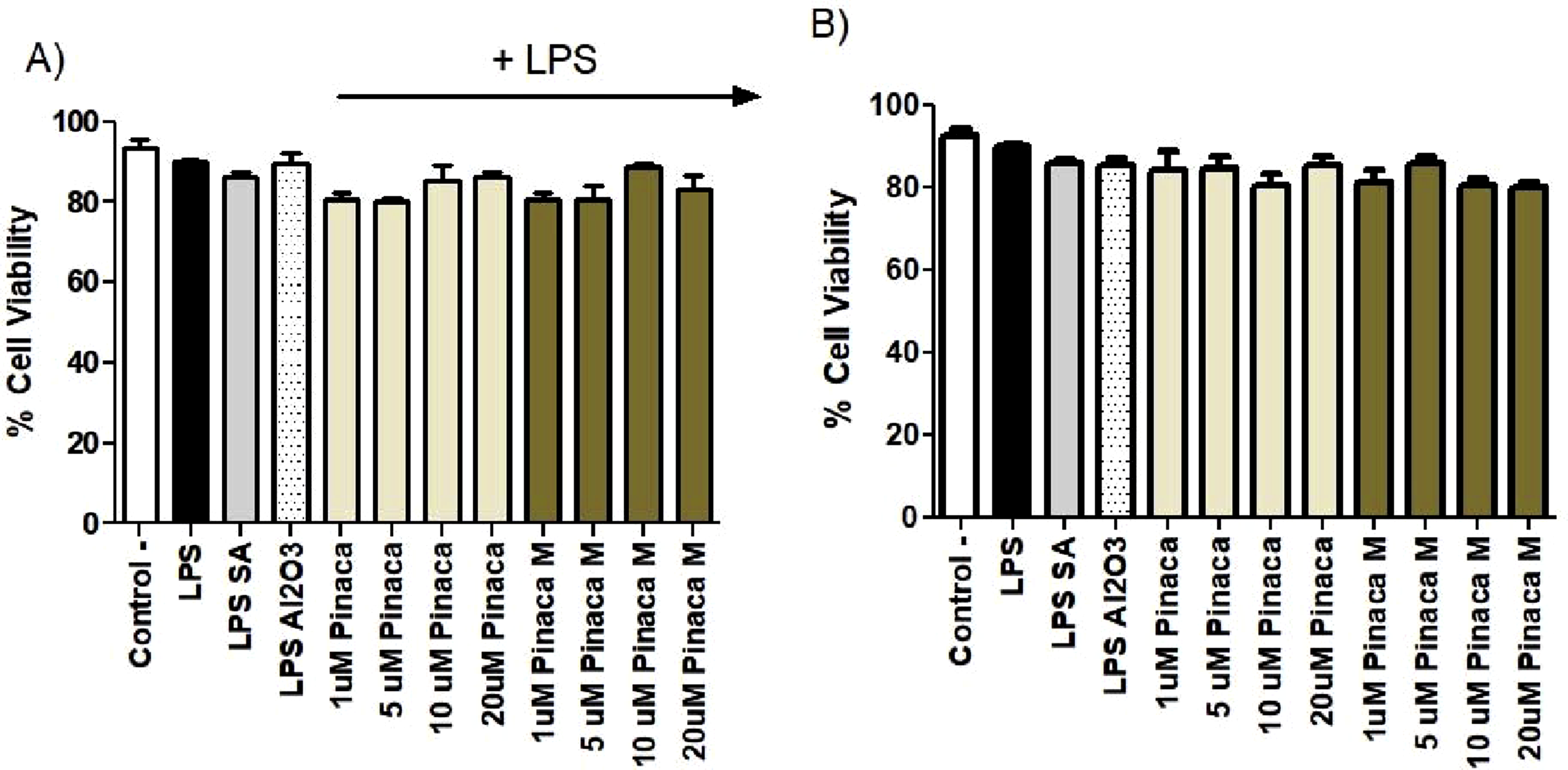
3.2. MDMB-4en-PINACA and Its Metabolite (MDMB-4en-PINACA M) Did Not Have an Immunostimulatory Effect
In the absence of a known stimulatory molecule, some molecules can induce an inflammatory response by the macrophages or any other immune cells. In our experimental set up we used LPS as a stimulatory molecule and in its presence, macrophages produced substantial amounts of TNF-α (Figure 3A), IL-6 (Figure 3B), GM-CSF (Figure 3C) and IL-12p40 (Figure 3D) cytokines (Figure 3). When the MDMB-4en-PINACA and its metabolite MDMB-4en -PINACA M-were applied to the cells without LPS, they did not cause any immunostimulatory effect (Figure 4). Therefore, MDMB-4en-PINACA and its metabolite MDMB-4en-PINACA M did not have an immunostimulatory activity on the macrophages. Detection of cytokine levels for TNFα (A), IL-6 (B), GM-CSF (C) and IL-12p40 (D) when the drug molecule was present and the macrophages were not stimulated with Lipopolysaccharide (LPS) (* 
3.3. MDMB-4en-PINACA and Its Metabolite MDMB-4en -PINACA M Had Differential Anti-inflammatory Activities
The inhibition of immune system cells’ production of pro-inflammatory cytokines is known as anti-inflammatory action. In our study, the macrophages were stimulated with LPS in the presence of MDMB-4en-PINACA and its metabolite Detection of cytokine levels for TNF (A), IL-6 (B), GM-CSF (C) and IL-12p40 (D) when the drug molecule was present and the macrophages were stimulated with LPS (* 
4. Discussion
Many people have used and had access to synthetic cannabis. The anti-inflammatory and protective properties of synthetic cannabinoids in inflammatory illnesses and disorders are well-established.20-23 They can therefore be applied as anti-inflammatory therapeutic molecules to treat inflammatory and autoimmune diseases. Understanding their specific impacts on immune system cells is crucial.
Previous studies indicated the inflammatory potential of synthetic cannabinoids in vitro cell models. In a study done by Al’zubi et al, exposure to AB-FUBINACA increased the expression of pro-inflammatory markers including IL-6, TNF-α, and NF-kB. 24 In a mouse model, cannabinoids bound to CB2 receptors reduced inflammation and protected the blood-brain barrier. 25 A recent study also showed that during chronic stress, the endocannabinoid CB1 receptors are highly expressed and increased the vascular-related gene expression, which ultimately decreased the inflammatory response in astrocytes of adult mouses. 26 Despite being present in immune cells, cannabinoid receptors CB1 and CB2 expression levels vary depending on the organism’s physiological conditions, such as infection or immunological activity. 27 Specifically, both receptor types on human peripheral blood mononuclear cells and T lymphocytes upregulate proinflammatory cytokines such TNF-α, IL-1β, and IL-6. 28
In our study, we focused on one of the cannabinoid derivatives -MDMB-4en-PINACA- and its metabolite for its immunomodulatory activity on the mammalian macrophages. Ozturk et al previously described MDMB-4en-PINACA and its metabolite. 6 The effects of this cannabis derivative and its metabolite on immune system cells have not been investigated. Our study focused on macrophages because of their significant involvement in the development of inflammation and illnesses. 29 When examined for their ability to stimulate the immune system, cannabinoid derivatives and their metabolites failed to cause macrophages to create pro-inflammatory cytokines such as TNFα, IL-6, GM-CSF, and IL-12p40. They exhibited varied impacts on the generation of pro-inflammatory cytokines when their immunomodulatory activity was examined in the presence of LPS to activate the macrophages. While its metabolite could only inhibit the synthesis of GM-CSF, MDMB-4en-PINACA exhibited anti-inflammatory effect by lowering the levels of TNFα, IL-6, and GM-CSF production.
PI3K is one intracellular pathway that is crucial for both metabolism and inflammation.14-16 There was no difference between the experimental and control groups when examining the impact of cannabis oil and its metabolite on phosphorylated PI3K levels (Data not shown). They most likely use a different signaling pathway as their mechanism of action.
Cannabinoids that are created externally have a well-established immunomodulatory effect. For instance, the synthetic cannabinoid HU-44 has been shown to have an oral therapeutic effect as antiarthritic in mice with collagen-induced arthritis in vivo and decreases the production of TNFα, a proinflammatory cytokine. 30 This also shows a similarity with our data that MDMB-4en-PINACA exhibited anti-inflammatory effect by lowering the levels of TNFα. Most of the other studies focus on the neurobehavioral activities of the derivative.31,32 Our results together with Haj et al study support the role of this derivative as a potent anti-inflammatory molecule that can be utilized in therapy against autoimmune and inflammatory disorders. CB1 and CB2 receptors play a significant role in immunomodulation. In a study done by Buckley et al, ligands specific to the cannabinoid CB2 receptor could have clinical value in regulating macrophage immune activity and yet having no effect on the central nervous system. 33 Kaplan et al also demonstrated that numerous immune modulatory impacts of cannabinoids are facilitated through CB1. 34 In addition to this, Turcotte et al, indicated that the CB2 receptor has been demonstrated to influence the functions of immune cells, both in laboratory settings and in animal models of inflammatory diseases. Accordingly, mice deficient in the CB2 receptor exhibit an increased inflammatory response which might hold a potential for therapeutic approaches that that will alter CB2 signaling against various inflammatory disorders. 35
Recent studies exhibited that when strong cannabinoid receptors agonists such as MDMB-4en-PINACA are used excessively, their clinical effects could have detrimental repercussions. With varying degrees of severity, the effects are mostly neurological, psychological, cardiovascular, cerebrovascular, and gastrointestinal.36,37
5. Limitations and Future Perspective
Our future studies will focus on the effect of this derivative on other immune system cells as well as on the
6. Conclusion
The results imply that the cannabis compound derivative MDMB-4en-PINACA M exhibits anti-inflammatory effects on macrophages in mammals. This cannabis derivative may have potential for treating autoimmune conditions and various inflammatory diseases, given that macrophages are essential in triggering inflammatory responses.
Footnotes
Acknowledgments
We would like to thank Prof. Dr. Güneş Esendağlı, and Prof. Dr. Juan Anguita for their material support.
Ethical Considerations
Ethical Approval is not applicable for this article.
Consent to Participate
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
The manuscript was collectively written by all authors, and all authors have reviewed and approved the final version of the manuscript. Each author has contributed equally to the work.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Declaration of Conflicting Interests
The authors have no relevant financial or non-financial interests to disclose.
Data Availability Statement
The data is available from the corresponding authors upon reasonable request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
