Abstract
Objective
This study aimed to determine pre-processing parameters for Armeniacae Semen Amarum (ASA), specifically the optimal particle size and appropriate packaging conditions, in order to support intelligent dispensing and ensure product quality.
Method
The physical properties of 10 batches of raw, blanched, and fried ASA, including moisture content, hardness, relative density, and extract yield, were evaluated. A validated HPLC method was used to determine the contents of amygdalin and prunasin in water decoctions prepared from whole seeds and from five crushed particle size fractions (< 0.85, 0.85–2, 2–4, 4–5, and 5–6 mm). Accelerated stability testing was conducted according to Chinese Pharmacopoeia guidelines at 40 °C ± 2 °C and 75% ± 5% relative humidity for 30 days. Paper bags, polyethylene bags, and aluminum foil bags were evaluated under both vacuum and non-vacuum conditions. Key stability indicators—moisture content, fatty oil content, acid value, and amygdalin content—were monitored throughout the storage period.
Result
Processing significantly altered the physical properties of ASA (P ≤ 0.001). The contents of amygdalin and prunasin in water decoctions prepared from crushed samples of all three processed ASA products were significantly higher than those from whole seeds (P < 0.001). The recommended optimal particle size ranges were 4–5 mm for raw ASA, 2–4 mm for blanched ASA, and 0.85–2 mm for fried ASA. In the stability study, samples packaged in paper bags showed marked deterioration: after 30 days, moisture content increased to 9.03%, the acid value rose to 2.84, and amygdalin content decreased by 15.41%. In contrast, polyethylene and aluminum foil maintained all stability indicators within acceptable ranges, with no significant benefit from vacuum treatment.
Conclusion
Processing altered the physical properties of ASA, and crushing enhanced the dissolution of its active components. Optimal particle size ranges and suitable packaging materials for processed ASA were identified.
Introduction
Armeniacae Semen Amarum (ASA) is the dried mature seed of Prunus armeniaca L. var. ansu Maxim., Prunus sibirica L., Prunus mandshurica (Maxim.) Koehne, or Prunus armeniaca L., and is widely used in clinical medicine. This herb was first recorded in the Shennong Bencao Jing under the name “Xingheren”. It has a long history of medicinal use and is described as having a bitter taste and a mild temperature. 1 ASA is traditionally used to lower qi, relieve cough and asthma, and moisten the intestines to promote laxation. It is commonly prescribed for cough and asthma, chest congestion with phlegm, and intestinal dryness with constipation. 2 As a commonly used drug for respiratory diseases, ASA played an important role during the prevention and control of the COVID-19 epidemic, becoming one of the most frequently used Chinese herbal pieces in anti-epidemic compound prescriptions. 3 ASA is rich in lipids, proteins, dietary fiber, minerals, and various bioactive compounds. In addition to its traditional medicinal use, it has attracted increasing attention for its potential applications in human nutrition, health, and in the pharmaceutical and food industries.4-6
According to the Pharmacopoeia of the People’s Republic of China (2025 edition), ASA is classified as “slightly toxic”. 2 Its toxicity is mainly attributed to the active components amygdalin and prunasin. 7 These compounds can be hydrolyzed by specific β-glucosidases, releasing toxic hydrocyanic acid, 8 which may cause cyanide poisoning and, in severe cases, death.9-11 Among these constituents, amygdalin accounts for approximately 4.9% of the total component content. 7 The core principle of ASA processing is “enzyme inactivation and glycoside preservation”, which aims to inhibit the enzymatic hydrolysis of amygdalin. To directly evaluate the effectiveness of processing and subsequent storage, this study selected amygdalin and its primary enzymatic degradation product, prunasin, as chemical markers. Amygdalin represents the bioactive and toxic precursor, whereas prunasin serves as a sensitive indicator of insufficient enzyme inactivation or degradation during storage. Monitoring both compounds provides mechanistic insight into how processing parameters influence the quality and safety of ASA. Notably, beyond its toxicological profile, ASA also exhibits a range of pharmacological activities, including anti-inflammatory, anti-atherosclerotic, anti-tumor, and anti-fibrotic effects.9,12-14
As an important traditional Chinese medicinal material, ASA possesses both therapeutic efficacy and potential toxicity. To ensure its safety and effectiveness, traditional Chinese medicine emphasizes control through “processing” and “time-consuming mashing”.15-18 At present, the commonly used processed products of ASA include blanched bitter almond (BASA), fried bitter almond (FASA), bitter almond cream, steamed bitter almond, and honey-processed bitter almond. 17 Among these, BASA and FASA have been consistently included in the Chinese Pharmacopoeia and represent the two most widely used processed forms in clinical practice. 2 As an important complement to conventional processing methods, prescription processing not only helps preserve traditional processing techniques but also better meets the needs of individualized diagnosis and treatment in traditional Chinese medicine and the development of intelligent dispensing systems. 19
According to the Chinese Pharmacopoeia, ASA should be mashed before use. At present, the commonly used clinical mashing methods mainly include “time-use mashing” and “pre-mashing”. “Time-use mashing” facilitates the identification and preservation of the characteristics of decoction pieces and promotes the extraction of active components during decoction, thereby improving the quality and efficacy of the decoction.20,21 However, in modern clinical practice this method is associated with several limitations, including cumbersome procedures and time-consuming operation, which reduce dispensing efficiency and negatively affect the patient experience.
“Pre-mashing” has emerged as an adaptive strategy in the modernization of traditional Chinese medicine. In this approach, decoction pieces that require mashing are processed in advance for later use. However, this approach faces significant quality control challenges, including susceptibility to deterioration, loss of active components, and difficulties in storage and preservation. 20 In this context, “linfang mashing” has been proposed as a new storage and processing strategy. This approach involves immediate mashing and short-term standardized storage of decoction pieces according to clinical prescription requirements. It combines the quality advantages of “time-use mashing” with the efficiency benefits of “pre-mashing”, and is particularly suitable for traditional Chinese medicines that are used in large doses and must be mashed before administration. 21 The modernization of traditional Chinese medicine has also promoted the development of intelligent dispensing systems, including automated herbal cabinets and robotic dispensing units designed to improve dispensing accuracy, efficiency, and traceability. 22 However, the effective operation of such systems depends on well-defined physical parameters of herbal materials, including uniform particle size for accurate weighing and flow, as well as validated stability data to ensure consistent quality during storage and dispensing. 23 Prescription mashing therefore represents a critical link between the inheritance of traditional processing techniques and the development of personalized and intelligent dispensing systems. Despite the promising prospects of prescription-based mashing, studies on key parameters—such as suitable storage duration, optimal particle size, and appropriate packaging materials and forms prior to mashing—remain limited. Previous studies have shown that inappropriate packaging may lead to increased moisture absorption, lipid oxidation (rancidity), and degradation of bioactive components. For ASA, environmental factors such as humidity and temperature have also been shown to influence its chemical profile during storage. However, these studies have not addressed the combined challenges associated with pre-mashing and short-term storage under conditions that simulate modern prescription-based or intelligent dispensing systems. Consequently, the optimal particle size for efficient dissolution and the most suitable packaging conditions for preserving the quality of pre-crushed ASA remain unclear. Specifically, there is a lack of standardized particle size criteria for crushed ASA, and sufficient stability data for pre-mashed ASA under conditions relevant to modern dispensing systems are currently unavailable, highlighting a critical knowledge gap that must be addressed to ensure quality standardization and medication safety.
The toxicity mechanism of ASA provides a strong rationale for investigating these parameters. Crushing disrupts the cellular compartmentalization that normally separates cyanogenic glycosides from their degrading enzymes, bringing them into contact and initiating potential degradation. The subsequent enzymatic hydrolysis, which releases toxic hydrocyanic acid, is strongly influenced by moisture and temperature.24,25 Therefore, the degree of crushing (particle size) determines the initial surface area and the extent of cellular disruption, directly affecting the release and dissolution of these components during decoction. At the same time, the packaging conditions after crushing determine the levels of ambient moisture and oxygen during storage, which may either promote or inhibit ongoing enzymatic degradation and the associated toxicity risk. Optimizing particle size and packaging is therefore not only important for maximizing efficacy but is also closely related to controlling and minimizing potential toxicity.
Guided by the principles of traditional Chinese medicine processing and the demands of intelligent and scientifically regulated clinical dispensing, this study investigated the effects of processing on the storage and preparation of ASA decoction pieces from the perspective of physical properties. The study focused on raw ASA (RASA) and two commonly used processed forms: blanched ASA (BASA) and fried ASA (FASA). High-performance liquid chromatography (HPLC) was employed to analyze the effects of crushing and particle size on the contents of the main components in aqueous extracts of these products. The reliability of chromatographic techniques for analyzing complex plant matrices has been well demonstrated in previous studies.26,27 Following these established principles, the HPLC method used in this study was fully validated to ensure accurate and reproducible quantification. The primary aim was to optimize the crushing protocol in order to improve the efficacy and quality of the processed products. In addition, an accelerated stability test using FASA was conducted to compare the effects of different packaging materials and packaging conditions on the chemical stability of the processed products. This approach aimed to maintain the quality stability of decoction pieces and support standardized dispensing and medication safety. Experimental data were analyzed using appropriate statistical methods based on their relevance, distribution characteristics, and homogeneity of variance. The findings of this study are intended to provide a scientific basis for the quality control and intelligent, standardized dispensing of ASA in clinical practice.
Materials and Methods
Samples and Reagents
ASA Decoction Pieces Sample Information

Amygdalin (Shanghai Yuanye Biotechnology Co., Ltd., Batch No.: Y08O10K96808, purity ≥ 98%); prunasin (Shanghai Yuanye Biotechnology Co., Ltd., batch number: M27GB143237, purity ≥ 98%); methanol (Tianjin Siyou Fine Chemicals Co., Ltd., batch number: 220830); acetonitrile (Fisher Chemical, lot number: F22M9J201); purified water (homemade).
Apparatus
UltiMate-3000 high performance liquid chromatograph; phenyl-Hexyl chromatographic column (250 mm × 4.6 mm, 5 μm, Feinuomei); high-speed universal pulverizer (Beijing Kewei Yongxing Instrument Co., Ltd., Model: FW-400); multifunctional health pot (Shenzhen Zhengyun Technology Co., Ltd., Model: 30MF5 3L); electric hot blast drying oven (Shanghai Yiheng Scientific Instrument Co., Ltd., Model: DHG-9023A); ten thousandth of a balance (Shanghai Precision Balance Co., Ltd., Model: FA2004); traditional Chinese medicine rolling flat machine (Shanghai Kangjian Traditional Chinese Medicine Machinery Manufacturing Co., Ltd., model: ZYJ-160); texture analyzer (Tianjin Kexing Electronic Equipment Manufacturing Co., Ltd., Model: CTA-10HD), etc.
Sample Preparation
Preparation of Different Processed Products
BASA
According to the method described in the literature, 28 RASA decoction pieces from each batch (S1–S10) were added to 1000 mL of boiling deionized water (10 times the volume) in a stainless steel vessel. The water was maintained at a rolling boil (approximately 100 °C). After exactly 10 minutes of boiling, the seeds were immediately removed and immersed in cold water for 1 minute. The seed coats were then manually removed by rubbing, and the resulting kernels were dried in a hot-air drying oven at 60 °C for 6 hours until a constant weight was reached.
FASA
Ten pieces of BASA decoction material were used for stir-frying according to the method described in General Rule 0213 of the Chinese Pharmacopoeia. The decoction processing equipment was preheated to 150–160 °C, after which BASA was added and continuously stirred during heating. The material was stir-Fried for 8–10 minutes. The endpoint of processing was determined when the surface color changed from light yellow to a uniform light brownish yellow, no white core remained in the cross-section, and a distinct, strong nutty aroma was produced. The processed samples were then removed and allowed to cool to room temperature.
Preparation of Prescription-Processed Products With Different Crushing Particle Sizes
The raw and processed ASA products were crushed using a traditional Chinese medicine rolling mill. The crushed materials were then separated into different particle size fractions using sieving and manual measurement. The fractions were defined as follows: < 0.85 mm, material passing through a No. 2 sieve; 0.85–2 mm, material passing through a No. 1 sieve but retained on a No. 2 sieve (24 mesh, aperture 850 μm); 2–4 mm, particles with diameters between 2.0 and 4.0 mm; 4–5 mm, particles between 4.0 and 5.0 mm; and 5–6 mm, particles between 5.0 and 6.0 mm.
Preparation of Water Decoctions From Different Processed Products and Crushing Particle Sizes
The preparation of decoctions was conducted in accordance with the Decocting Technical Specification for Traditional Chinese Medicine Decoction (T/CACM 1366–2021) issued by the Chinese Association of Traditional Chinese Medicine, as well as relevant literature. 29 Samples (50 g) of uncrushed RASA, BASA, and FASA, as well as samples crushed to different particle sizes, were placed in a multifunctional decoction pot and extracted with seven times the volume of water. The materials were soaked for 30 minutes before filtration, and the filtrate was temporarily reserved. The filtrate was then heated to boiling and returned to the filter residue. After the mixture reached boiling again, it was decocted for 15 minutes and filtered, and the filtrate was collected. The residue was subsequently extracted with six times the volume of water, first heated with high heat and then decocted with gentle heat for 20 minutes. After filtration, the two filtrates were combined and diluted with water to a final volume of 1,000 mL. Decoctions were thus obtained for the uncrushed RASA, BASA, and FASA samples, as well as for samples with different crushing particle sizes.
Preparation of Packaged Samples With a Defined Crushing Particle Size
An appropriate amount of crushed FASA was divided into five groups for packaging: paper bag packaging (ZD), polyethylene plastic vacuum packaging (SL-K), polyethylene plastic non-vacuum packaging (SL-F), aluminum foil vacuum packaging (LB-K), and aluminum foil non-vacuum packaging (LB-F).
Preparation of Test Solutions for Content Determination
A 5 mL aliquot of the decoction prepared from the raw and processed ASA samples (Section Preparation of Water Decoctions from Different Processed Products and Crushing Particle Sizes) was transferred to a 50 mL volumetric flask. Sixty percent methanol was added to volume, the solution was thoroughly mixed, and then filtered. The filtrate was collected and used as the test solution for subsequent content determination.
Determination of Physical Parameters of Different Processed Products
Moisture Determination
An appropriate amount of powdered raw and stir-fried ASA samples (passed through the No. 2 sieve of the Chinese Pharmacopoeia) was collected. The moisture content was determined according to the second method (drying method) described in General Rule 0832 of the 2020 edition of the Chinese Pharmacopoeia.
Hardness Measurement
Appropriate amounts of RASA, BASA, and FASA decoction pieces were selected, and their hardness was measured using a texture analyzer. Prior to testing, the measuring probe was installed and the instrument parameters were set as follows: pre-test speed 1.5 mm/s, test speed 1 mm/s, post-test speed 1 mm/s, trigger force 5 g, and deformation distance 1.5 mm. Measurements were performed using the TPA (Texture Profile Analysis) mode. Each sample was tested six consecutive times, and the average value of the six measurements was recorded as the final hardness value.
Determination of Relative Density of the Decoction Liquid Phase at Specific Crushing Particle Sizes
The weight of the empty specific gravity bottle (m1) and the weight of the bottle filled with purified water at 20 °C (m2) were accurately measured. The decoction sample was then added to the empty bottle, and the weight of the bottle containing the sample at 20 °C (m3) was determined. The relative density of the decoction was calculated using the following formula: Relative Density = (m3 - m1)/(m2 - m1).
Determination of Extract Yield of Decoctions With Specific Crushing Particle Sizes
A 25 mL aliquot of the decoction obtained in Section Preparation of Water Decoctions from Different Processed Products and Crushing Particle Sizes was accurately measured and transferred into a previously dried and weighed evaporating dish. The liquid was evaporated to dryness in a water bath. The residue was then dried in an oven at 105 °C for 3 hours, transferred to a desiccator, and allowed to cool to room temperature. The sample was weighed accurately until a constant weight was obtained. The extract yield was calculated based on the weight of the dried residue using the following formula: Paste Yield Rate (%) = Dry Paste Weight/Decoction Piece Weight × 100%.
Determination of Index Component Content in Decoctions Before and After Crushing and at Different Crushing Particle Sizes of Clinical Prescription Preparations
Preparation of Test Solutions
The raw ASA products and their processed forms prepared in Section Sample Preparation were used. Samples with different crushing particle sizes were transferred into 50 mL volumetric flasks, 60% methanol was added, and the mixtures were thoroughly shaken. The solutions were then diluted to volume with 60% methanol, filtered, and the filtrates were collected as the test solutions.
Preparation of Reference Solutions
A total of 21.33 mg of the amygdalin reference standard was accurately weighed, and methanol was added to prepare a stock solution with a concentration of 2.133 mg/mL. An appropriate volume of this stock solution was then diluted with methanol to obtain a reference solution containing amygdalin at a concentration of 42.66 μg/mL.
Chromatographic Conditions
Chromatographic separation was performed using a Phenomenex luna Phenyl-Hexyl column (250 mm × 4.6 mm, 5 μm). The mobile phase consisted of acetonitrile–water (6:94, v/v). The flow rate was set at 1.0 mL/min, the column temperature at 30 °C, and the injection volume at 10 μL. Detection was performed at a wavelength of 207 nm, and isocratic elution was employed throughout the analysis.
Method Validation of HPLC
The HPLC method was validated in terms of precision, repeatability, stability, linearity, and accuracy.
Precision
Six consecutive injections of the same RASA sample solution were analyzed within 24 hours. The relative standard deviations (RSDs) of the relative retention times of common peaks ranged from 0.10% to 0.63%, and those of relative peak areas ranged from 0.14% to 3.09%. The RSDs for the relative retention times of amygdalin and prunasin were 0.16% and 0.10%, respectively, while the RSDs for their peak areas were 0.14% and 0.95%, indicating good instrument precision.
Repeatability
Six replicate sample solutions were independently prepared from the same batch of RASA and analyzed. The RSDs of relative retention times ranged from 0.10% to 0.20%, and those of relative peak areas ranged from 0.09% to 2.04%. The RSDs for the peak areas of amygdalin and prunasin were 0.09% and 0.55%, respectively, demonstrating good method repeatability.
Stability
The stability of the sample solution was evaluated by analyzing it at 0, 2, 4, 6, 8, 12, 18, and 24 hours after preparation. The RSDs of relative retention times ranged from 0.30% to 0.67%, and those of relative peak areas ranged from 0.17% to 3.39%. The RSDs for the peak areas of amygdalin and prunasin were 0.17% and 0.60%, respectively, indicating that the sample solution remained stable for at least 24 hours.
Linearity
Calibration curves were established using a series of standard solutions. Amygdalin showed excellent linearity (y = 162.91x + 0.0880, R2 = 1.000) within the concentration range of 0.002133–0.53325 mg/mL. Prunasin also exhibited excellent linearity (y = 291.64x − 0.0037, R2 = 1.000) over the concentration range of 0.000171–0.085600 mg/mL.
Accuracy
Recovery experiments were performed by spiking a known sample solution (n = 6) with amygdalin (1.5064 mg) and prunasin (0.1195 mg) reference standards. The average recoveries were 98.67% (RSD = 0.73%) for amygdalin and 101.25% (RSD = 1.19%) for prunasin, indicating excellent accuracy of the method.
Determination of Index Component Content in Decoctions Before and After Crushing of Different Processed Products
The test solution and reference solution were analyzed under the chromatographic conditions described in Section Chromatographic Conditions, and the corresponding peak areas were recorded. The content of the index components was calculated using the external standard method after organizing and processing the obtained data.
Determination of Component Content in Decoctions at Different Crushing Particle Sizes
The test solutions and mixed reference solutions were analyzed under the chromatographic conditions described in Section Chromatographic Conditions, and the chromatograms were recorded and saved. The chromatographic data were imported into the Similarity Evaluation System for Fingerprint of Traditional Chinese Medicine (2012 Edition). A reference chromatogram was established with the time window width set to 3 min. Multi-point correction was applied for automatic peak matching to obtain chromatographic fingerprints of raw ASA, processed ASA, and fried ASA samples. The median method was used to generate a representative chromatographic profile from the original experimental data, which was designated as the control fingerprint. This control fingerprint was subsequently used for similarity evaluation.
Determination of the Stability of FASA Processed Products With a Specific Particle Size Under Different Packaging Conditions
Appearance Evaluation Under Different Packaging Conditions
Samples prepared as described in Section Preparation of Packaged Samples with a Defined Crushing Particle Size were stored at 40 °C ± 2 °C and a relative humidity of 75% ± 5% for 30 days. Observations were conducted on days 0, 5, 10, 15, 20, 25, and 30 to evaluate changes in appearance, including color, mildew formation, and insect infestation.
Moisture Determination Under Different Packaging Conditions
Samples prepared according to Section Preparation of Packaged Samples with a Defined Crushing Particle Size were stored at 40 °C ± 2 °C and a relative humidity of 75% ± 5% for 30 days. Measurements were conducted on days 0, 5, 10, 15, 20, 25, and 30. Moisture content was determined using the same method described in Section Moisture Determination.
Determination of Fat Oil Content Under Different Packaging Conditions
Fatty oil content was determined according to General Rule 2303 of the 2020 edition of the Chinese Pharmacopoeia. Sample powder was placed in a Soxhlet extractor, and 100 mL of n-hexane was added. The mixture was heated and refluxed in a water bath for 2 hours, then allowed to cool and filtered through a No. 3 vertical melting glass funnel. The filtrate was concentrated in a water bath under reduced pressure to recover the solvent completely. The remaining residue was the extracted oil, which was accurately weighed. The fatty oil content was calculated using the following formula: Oil Content = Oil Weight/Decoction Pieces Weight × 100%.
Determination of Acid Value Under Different Packaging Conditions
The acid value represents the amount of potassium hydroxide (mg) required to neutralize the free fatty acids present in 1 g of the sample. Samples were stored at 40 °C ± 2 °C and a relative humidity of 75% ± 5% for 30 days, with measurements conducted on days 0, 5, 10, 15, 20, 25, and 30. The acid value was determined according to the method for fats and fatty oils described in General Rule 0713 of the 2020 edition of the Chinese Pharmacopoeia. An accurately weighed sample was placed in a 250 mL conical flask. A total of 50 mL of an ethanol–ether mixture (1:1) was added; prior to use, 1.0 mL of phenolphthalein indicator solution was added to the mixture and adjusted to a faint pink color using sodium hydroxide titration solution (0.1 mol/L). The sample was shaken until completely dissolved. If dissolution was difficult, the mixture was gently heated under reflux until dissolution was achieved. The solution was then titrated with sodium hydroxide titration solution (0.1 mol/L) until a stable pink color persisted for 30 seconds. The acid value was calculated using the following formula: Acid Value = A × 5.61/W. Where A represents the volume of sodium hydroxide solution consumed (mL), and W represents the weight of fatty oil in the sample (g).
Determination of Amygdalin Content Under Different Packaging Conditions
Preparation of Test Solutions: Sample powder prepared as described in Section Preparation of Packaged Samples with a Defined Crushing Particle Size was accurately weighed (0.25 g, passed through the No. 2 sieve) and placed in a stoppered conical flask. Twenty-five milliliters of methanol was accurately added, and the total weight was recorded. The sample was subjected to ultrasonic extraction for 30 minutes (power 250 W, frequency 50 kHz). After cooling, methanol was added to compensate for any loss of weight. The mixture was shaken and filtered. Five milliliters of the continuous filtrate was transferred and diluted to 50 mL with 50% methanol, shaken, and filtered again. The resulting filtrate was used as the test solution. Samples collected on days 0, 5, 10, 15, 20, 25, and 30 were labeled as ZD-0, ZD-5, ZD-10, ZD-15, ZD-20, ZD-25, and ZD-30, respectively, and samples from other packaging groups were labeled using the same method.
Determination of Amygdalin Content: The preparation of the reference solution and the chromatographic conditions were the same as those described above. The amygdalin content in each sample was calculated using the external standard method.
Statistical Analysis
Statistical analyses were performed using SPSS software (version 26.0, IBM Corp., Armonk, NY, USA). Data were analyzed using one-way analysis of variance (ANOVA) or the Kruskal–Wallis H test, as appropriate. To more intuitively reveal the correlations among the physical properties and chemical components of FASA samples under different packaging conditions during the accelerated stability study, Pearson correlation analysis was performed using Origin Pro 2021 software.
Results
Determination Results of Physical Parameters of Different Processed Products
Moisture Determination Results
Characterization of the Physical Properties of BA Processed With Different Methods (n = 10)
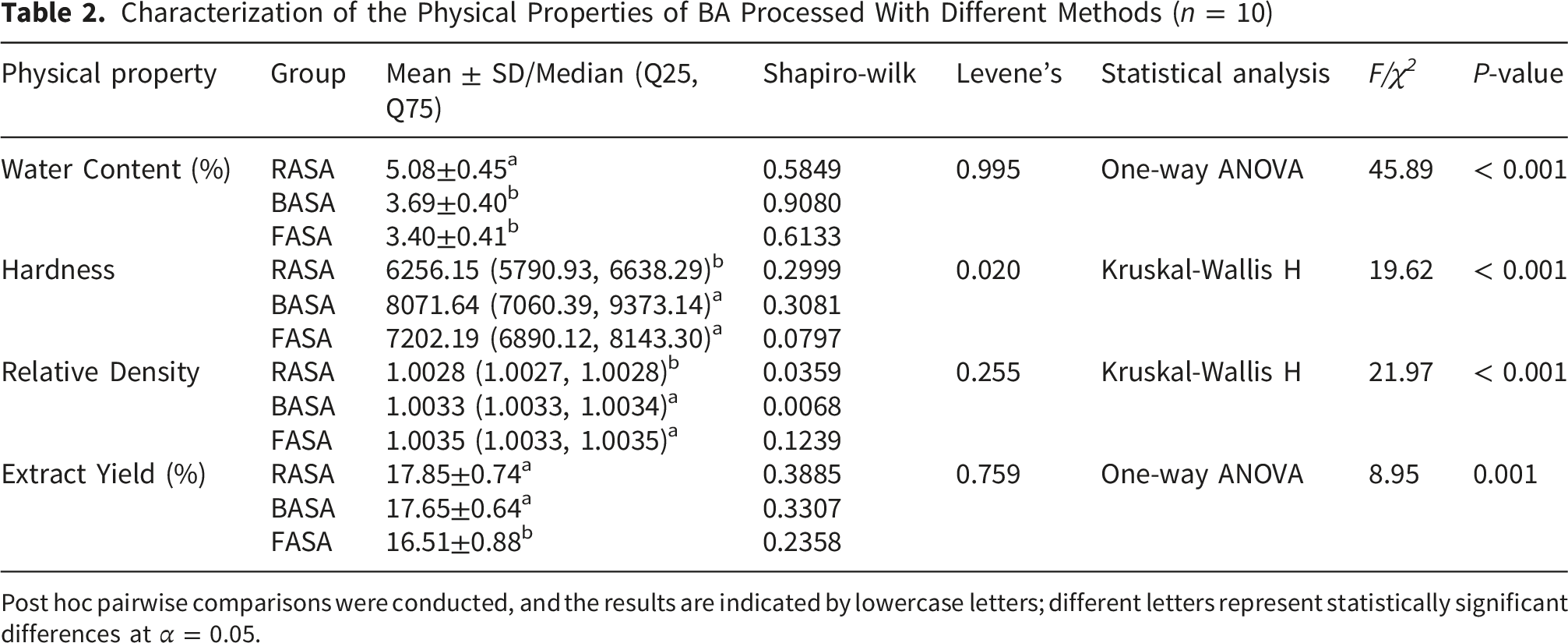
Post hoc pairwise comparisons were conducted, and the results are indicated by lowercase letters; different letters represent statistically significant differences at α = 0.05.

Comparison of the physical properties of different ASA processed products. (A) Moisture content, (B) hardness, (C) relative density and (D) extract yield. Different lowercase letters indicate statistically significant differences at α = 0.05
Hardness Measurement Results
As shown in Table 2 and Figure 1B, the median hardness values for the 10 batches of RASA, BASA, and FASA were 6252.15, 8071.64, and 7202.19, respectively. The hardness data conformed to normal distribution according to the Shapiro–Wilk test but did not satisfy the assumption of homogeneity of variance based on Levene’s test. Therefore, the Kruskal–Wallis H test was applied. The results revealed significant differences in hardness among the different processing groups (P < 0.001), with the order BASA > FASA > RASA. Post hoc analysis with Bonferroni correction showed that the hardness of RASA differed significantly from that of BASA and FASA (P < 0.01). Although the hardness of BASA was higher than that of FASA, the difference between these two groups was not statistically significant (P = 0.929). These findings suggest that processing significantly increases the hardness of ASA decoction pieces, whereas stir-frying does not produce a further significant change in hardness compared with the processed product.
Relative Density of Decoction Liquid Phase at Specific Crushing Particle Sizes
As presented in Table 2 and Figure 1C, the median relative densities of the decoctions prepared from crushed RASA, BASA, and FASA were 1.0028, 1.0033, and 1.0035, respectively. The Shapiro–Wilk test indicated that the data followed a normal distribution, whereas Levene’s test showed that the assumption of homogeneity of variance was not met. Therefore, the Kruskal–Wallis H test was used for analysis. The results demonstrated significant differences in the relative density of the decoction liquid among the different processing groups (P < 0.001). After Bonferroni correction for multiple comparisons, the results showed that the relative densities of decoctions prepared from BASA and FASA differed significantly from those of RASA (P < 0.01). However, no significant difference was observed between BASA and FASA decoctions (P = 0.448).
Extract Yield of Decoctions With Specific Crushing Particle Sizes
As shown in Table 2 and Figure 1D, the average extract yields of the 10 batches of RASA, BASA, and FASA were 17.85%, 17.65%, and 16.51%, respectively. The Shapiro–Wilk and Levene’s tests confirmed that the data satisfied the assumptions of normal distribution and homogeneity of variance. One-way ANOVA showed significant differences in extract yield among the different processed products (P = 0.001), with the order RASA > BASA > FASA. Further multiple comparisons using the LSD method indicated that the extract yield of BASA decoctions did not differ significantly from that of RASA (P = 0.573). However, the extract yield of FASA decoctions was significantly lower than that of the other groups (P < 0.001), indicating that stir-frying significantly reduced the extract yield of the decoction.
As shown in Table 2 and Figure 1, the physical properties of ASA changed after processing. Specifically, the moisture content and the extract yield of the water decoction decreased, whereas hardness and the relative density of the decoction increased. Except for the extract yield, significant differences were observed in moisture content, hardness, and relative density of the decoction between the processed products (BASA and FASA) and the raw product (P < 0.01). However, no statistically significant differences were found between the physical properties of the two processed products (P > 0.05). These findings provide a basis for understanding how processing affects the storage and handling characteristics of ASA, and they set the stage for evaluating the influence of crushing and packaging on product quality in the following sections.
Determination of Index Component Content in Decoctions of Different Prescription-Processed Products Before and After Crushing and at Different Crushing Particle Sizes
Changes in Index Component Content in Decoctions Before and After Crushing of Different Processed Products
Comparative Analysis of Marker Component Content in Water Decoctions From Different Clinical Preparations of ASA: Effect of Crushing (n = 10)
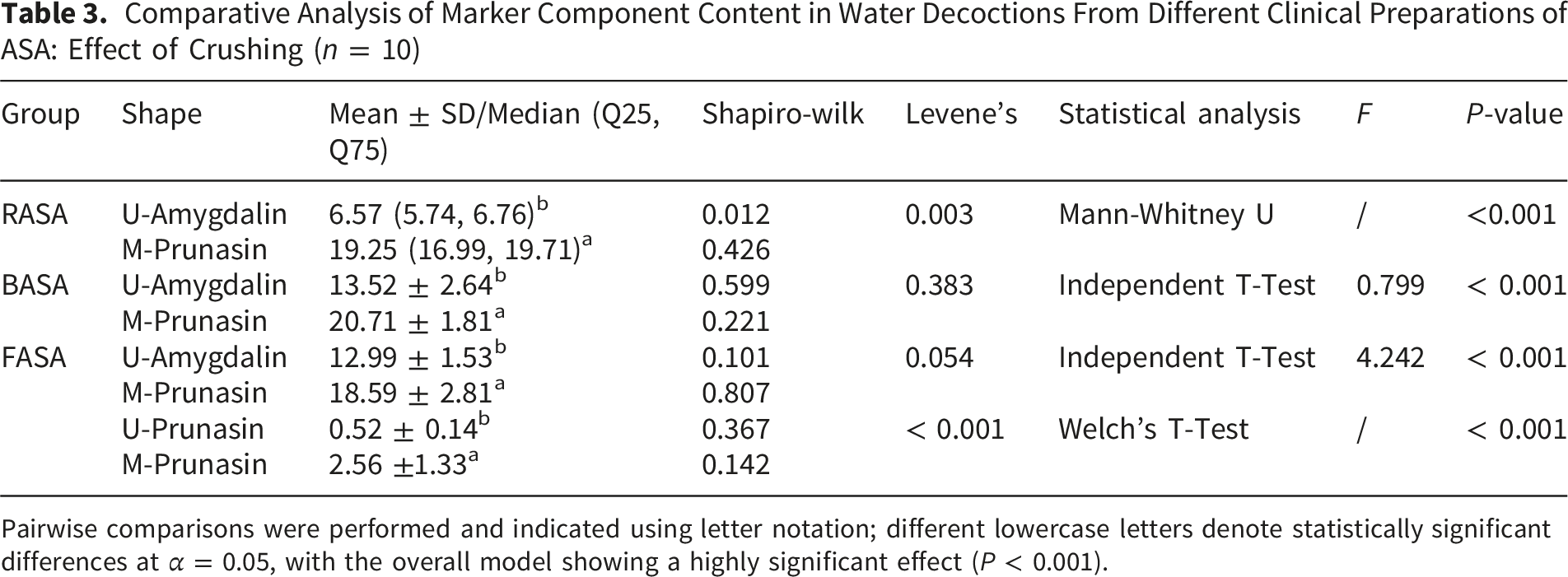
Pairwise comparisons were performed and indicated using letter notation; different lowercase letters denote statistically significant differences at α = 0.05, with the overall model showing a highly significant effect (P < 0.001).

Contents of amygdalin and prunasin in ASA samples (Mean ± SD, n = 10). Different lowercase letters indicate significant differences within the group compared with the uncrushed samples (P < 0.001)
Before crushing, only prunasin was detected in the fried products, with an average content of 0.52 mg/g. After crushing, the prunasin contents in RASA, BASA, and FASA were 0.51 mg/g, 1.41 mg/g, and 2.56 mg/g, respectively, representing significant increases (P < 0.001). These results indicate that crushing significantly improves the extraction efficiency of the index components in the decoctions of raw, processed, and fried ASA (P < 0.001). In addition, the contents of amygdalin and prunasin in BASA and FASA, both before and after crushing, were higher than those observed in RASA.
Changes in Index Component Content in Decoctions at Different Crushing Particle Sizes for Different Processed Products
Content of Marker Components in Water Decoctions of RASA, BASA and FASA at Different Crushing Particle Sizes

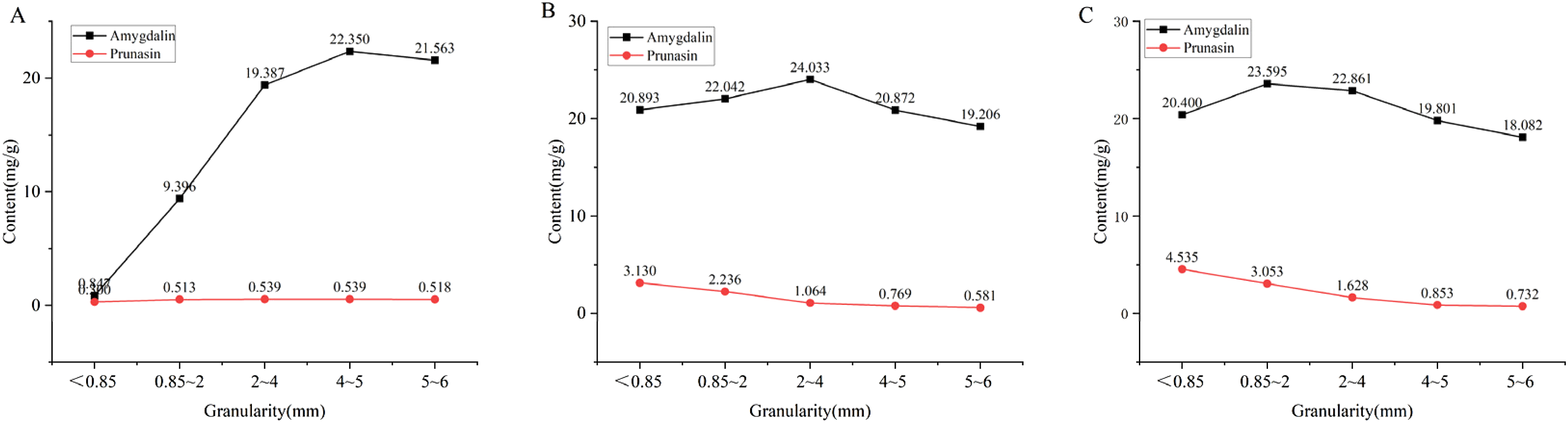
Determination of marker component contents in water decoctions of different processed ASA products at various crushing particle sizes: (A) RASA, (B) BASA and (C) FASA
As shown in Figure 2B, for BASA, comparison of the index component contents in decoctions prepared from different particle sizes showed that the amygdalin content was highest when the particle size was 2–4 mm, while the prunasin content increased progressively as the particle size decreased. Considering that amygdalin is the principal active component of ASA, the optimal crushing particle size for BASA was determined to be 2–4 mm.
Similarly, for FASA, analysis of decoctions prepared from different particle sizes showed that the amygdalin content reached its maximum when the particle size was 0.85–2 mm, while the prunasin content continued to increase as the particle size decreased. As shown in Figure 3C, the amygdalin content at a particle size of 2–4 mm was close to the maximum value. Therefore, the recommended crushing particle size range for FASA was determined to be 0.85–4 mm. Based on these findings, the optimal particle size ranges for each processed ASA product were established.
Determination Results of Stability of FASA Processed Products With a Specific Particle Size Under Different Packaging Conditions
Appearance Evaluation Under Different Packaging Conditions
Macroscopic Characteristics of Processed Herbal Decoction Pieces Stored Under Different Packaging Conditions

Moisture Determination Results Under Different Packaging Conditions
As shown in Figure 4A, the moisture content of FASA packaged in ZD (paper bags) increased markedly during storage, rising continuously from day 5 to day 30. After 30 days, the moisture content reached 9.03%, which exceeded the upper limit of 6.0% specified in the Chinese Pharmacopoeia. These results indicate that paper bag packaging readily allows moisture absorption and is therefore unsuitable for maintaining the quality of fried ASA. In contrast, when FASA samples were packaged using SL-K, SL-F, LB-K, and LB-F, the moisture content increased only slightly during storage and remained below 4.0% after 30 days, meeting the pharmacopoeial requirements. These findings suggest that both polyethylene plastic bags and aluminum foil packaging provide effective protection for the quality of the decoction pieces. No substantial differences were observed between vacuum and non-vacuum packaging, and moisture changes remained minimal after 30 days of storage under high temperature and high humidity conditions using SL-K, SL-F, LB-K, and LB-F packaging methods. Stability trends of stir-fried ASA under accelerated storage conditions. (A) Moisture content, (B) fatty oil content, (C) acid value and (D) amygdalin content. SL and LB represent polyethylene plastic bag and aluminum foil packaging, respectively, while F and K denote non-vacuum and vacuum conditions, respectively
Determination Results of Fat Oil Under Different Packaging Conditions
As shown in Figure 4B, the initial fatty oil content of FASA was 57.53%. After 30 days of storage in paper bag packaging, the fatty oil content decreased to 53.06%. In comparison, when samples were packaged using SL-K, SL-F, LB-K, and LB-F, the fatty oil content decreased only slightly during storage and remained above 55.00% after 30 days, which was higher than that observed with paper bag packaging. These results indicate that both polyethylene plastic bags and aluminum foil packaging help preserve the quality of the decoction pieces. No significant differences were observed between vacuum and non-vacuum packaging, and the fatty oil content showed only minor changes after 30 days of storage under high temperature and high humidity conditions when SL-K, SL-F, LB-K, and LB-F packaging methods were used.
Determination of Acid Value Under Different Packaging Conditions
The acid value results for FASA decoction pieces stored under accelerated test conditions are shown in Figure 4C. The initial acid value was 1.21. Under accelerated storage conditions, the acid value of samples packaged in paper bags increased significantly, reaching 2.84 after 30 days, indicating considerable deterioration and demonstrating that paper bag packaging cannot effectively preserve the quality of FASA. In contrast, when SL-K, SL-F, LB-K, and LB-F packaging methods were used, the acid value increased only slightly and remained below 1.60 after 30 days. The acid values under these packaging conditions were markedly lower than those observed for paper bag packaging, indicating that polyethylene plastic bags and aluminum foil packaging significantly improve the storage stability of FASA decoction pieces. In addition, no significant difference was observed between vacuum and non-vacuum packaging, and the acid value of FASA with a defined crushing particle size showed only minor changes after 30 days of storage under high temperature and high humidity conditions using SL-K, SL-F, LB-K, and LB-F packaging methods.
Determination of Amygdalin Content Under Different Packaging Conditions
Determination of Amygdalin Content in Processed ASA Stored Under Different Packaging Conditions

Furthermore, vacuum packaging did not show a significant advantage over non-vacuum packaging in maintaining amygdalin content. During storage for 30 days under high temperature and high humidity conditions using SL-K, SL-F, LB-K, and LB-F packaging methods, fluctuations in amygdalin content were relatively small, and the amygdalin levels in FASA remained close to the initial values at approximately 15 and 30 days of storage.
Correlation Analysis Among Stability Indicators
To further elucidate the relationships among different stability indicators during the accelerated storage of pre-mashed fried ASA, a Pearson correlation analysis was performed on the measured parameters, including moisture content, fatty oil content, acid value, and amygdalin content. The correlation coefficients are presented in Figure 5. Moisture content showed a strong positive correlation with acid value (r = 0.84, P < 0.01) and a significant negative correlation with fatty oil content (r = -0.65, P < 0.01). These results indicate that moisture increase during storage promotes triglyceride hydrolysis, leading to the release of free fatty acids (elevated acid value) and a corresponding decline in fatty oil content. Acid value exhibited a strong negative correlation with fatty oil content (r = -0.73, P < 0.01), further confirming that lipid hydrolysis is a primary degradation pathway in fried ASA during storage. Notably, amygdalin content showed no significant correlation with moisture content or acid value, and only a weak, non-significant positive correlation with fatty oil content. This suggests that the degradation mechanism of amygdalin may be relatively independent from lipid hydrolysis, possibly due to differences in their degradation pathways or the influence of other factors not captured in this analysis. Correlation plot of physical properties and chemical components of FASA decoction pieces under different packaging conditions and storage durations
Discussion
Processing Significantly Alters the Physical Properties of ASA With Practical Pharmaceutical Implications
The results for the physical properties of the different processed products showed that, compared with RASA, the moisture contents of BASA and FASA decreased significantly after processing (P < 0.01), whereas hardness and the relative density of the decoction liquid phase increased significantly (P < 0.01). The marked reduction in moisture content from RASA (5.08%) to BASA (3.69%) and FASA (3.40%) is of considerable practical importance. Lower moisture levels are a key factor in extending shelf life by inhibiting microbial growth (including molds and bacteria), reducing enzyme activity, and slowing the hydrolytic rancidity of the abundant fatty oils. These findings support the traditional practice of processing ASA to improve its storability.
The significant increase in hardness observed after processing reflects a densification of the tissue structure, likely resulting from heat-induced starch gelatinization and protein coagulation. From a pharmaceutical perspective, increased hardness may influence the efficiency of the crushing process and the resulting particle size distribution. Softer materials such as RASA may be easier to crush but tend to produce a higher proportion of fine powders, whereas harder materials such as BASA and FASA may require greater crushing force but are more likely to yield a uniform particle size distribution, which is advantageous for standardized dispensing.
The increased relative density of decoctions prepared from processed products indirectly indicates a higher concentration of soluble solids in the decoction, which is directly related to the effective dose received by patients. This change may be associated with the high temperatures used during processing, which promote moisture evaporation and cause tissue contraction, resulting in a denser structure and consequently increased hardness and relative density. In addition, the extract yield of FASA decreased significantly after processing of RASA (P < 0.01). This reduction may be related to the degradation or denaturation of certain protein components at high temperatures, which could reduce the dissolution of water-soluble substances. In contrast, no significant change was observed in the extract yield of BASA (P > 0.05), likely because the processing method involves boiling in water at relatively lower temperatures, leading to milder reactions and less pronounced changes.
Moisture content plays a critical role in the quality and storage stability of ASA. Increased moisture can promote lipid hydrolysis and accelerate rancidity. 30 In this study, the moisture contents of BASA and FASA decreased significantly after processing, which helps delay lipid oxidation and rancidity during storage. This characteristic is therefore beneficial for maintaining the chemical stability of decoction pieces and extending their storage period.
Crushing Enhances the Dissolution of Active Components, Balancing Efficacy and Safety
The analysis of component dissolution in different prescription-processed products before and after crushing, as well as at different particle sizes, showed that the contents of amygdalin and prunasin in BASA and FASA increased after processing. Notably, in decoctions prepared from uncrushed samples, the amygdalin content nearly doubled after RASA was processed into BASA and FASA, indicating that processing promotes the retention of amygdalin in ASA. Amygdalin is not only the principal bioactive component responsible for relieving cough and asthma but also the precursor of toxic hydrocyanic acid. However, an increase in amygdalin content does not necessarily imply an increased risk of toxicity. This is attributed to the fundamental processing principle of “enzyme inactivation and glycoside preservation”. β-glucosidase is the key enzyme responsible for hydrolyzing amygdalin and releasing hydrogen cyanide, and the high temperatures used during processing can effectively inactivate this enzyme. Consequently, although more amygdalin may dissolve into the decoction and enhance therapeutic efficacy, the enzymatic pathway leading to the formation of toxic products remains inhibited.
This phenomenon may be explained by two possible mechanisms. First, processing effectively inhibits the activity of amygdalin-degrading enzymes through enzyme inactivation, thereby reducing the decomposition of amygdalin and preserving a greater proportion of the active compound. Second, some studies have suggested that transformation reactions may occur during processing, potentially converting compounds such as neoamygdalin or amygdalin amide into amygdalin36. In addition, the contents of amygdalin and prunasin in the three ASA products increased significantly after crushing (P < 0.001). These experimental results confirm that crushing promotes the dissolution of index components in RASA, BASA, and FASA. This effect is likely related to the increased specific surface area and the disruption of cellular structures caused by crushing, which facilitate the release of chemical constituents. These findings are consistent with the traditional Chinese medicine processing theory of “reducing toxicity and enhancing efficacy”.
Particle Size Nonlinearly Affects Dissolution: Mechanisms and Optimal Ranges
The degree of fragmentation of processed products is closely correlated with the dissolution of their index components, which directly influences both quality and clinical efficacy. 19 Based on a comprehensive evaluation, the optimal particle size for RASA was determined to be 4–5 mm. For BASA, the amygdalin content in the decoction reached its maximum when the particle size was 2–4 mm, whereas for FASA the highest amygdalin content was observed at a particle size of 0.85–2 mm. In contrast, the prunasin content increased progressively as the particle size decreased. The relationship between the dissolution of amygdalin and particle size exhibited a significant nonlinear pattern. The dissolution did not continuously increase as particle size decreased; instead, it showed a trend of initially increasing and then decreasing. When the particle size was reduced from large fragments (5–6 mm) to the optimal ranges for each processed product (raw ASA 4–5 mm, blanched ASA 2–4 mm, and fried ASA 0.85–2 mm), the increase in specific surface area became the dominant factor. A larger specific surface area enhances the contact between solute and solvent, thereby promoting faster and more complete dissolution of the components. This stage corresponds to the rising portion of the dissolution curve.
However, when the particle size becomes excessively fine (all processed products <0.85 mm), several factors collectively inhibit dissolution efficiency. First, ultrafine powders possess high surface energy and tend to agglomerate upon wetting. As a result, the effective specific surface area participating in dissolution is greatly reduced, and solvent penetration into the aggregates becomes limited. Second, the dissolution rate of fine particles is relatively rapid, leading to the formation of a high-concentration, high-viscosity solute diffusion layer around the particle surface. This diffusion layer can hinder the subsequent diffusion of solute molecules into the bulk solution, a phenomenon commonly observed during the dissolution of highly soluble compounds. Third, ASA is a complex medicinal matrix containing multiple components. Excessive crushing may release large amounts of gelatinous substances, such as mucilage and pectin, which can form a physical barrier that encapsulates glycosides and restricts their release into the solvent. Considering that amygdalin is the principal active component of ASA and that its content changes only slightly when the particle size of RASA is within 4–5 mm, the optimal crushing particle size ranges are recommended to be 4–5 mm for RASA, 2–4 mm for BASA, and 0.85–2 mm for FASA.
Packaging Material Critically Determines Stability: Moisture as the Primary Driver
The quality of ASA is significantly influenced by its moisture content, lipid content, and oil composition. 31 Although the Chinese Pharmacopoeia does not specify clear limits for the fatty oil content or acid value of ASA, previous studies have shown that bitter almond oil possesses pharmacological activities such as anti-inflammatory and moisturizing effects. 17 Because FASA and other traditional Chinese medicine decoction pieces rich in oils and proteins are susceptible to problems such as lipid loss and mold growth—which may lead to potential toxic risks—the selection of appropriate packaging materials is particularly important for maintaining their quality. 32 Polyethylene is the most widely produced plastic in the world and is commonly used as a packaging material in the food and pharmaceutical industries. 33
The results of the accelerated stability test showed that the quality of FASA packaged in paper bags deteriorated significantly during storage. This deterioration is likely attributable to the poor barrier properties of paper bag materials against moisture and oxygen, which allow increased moisture absorption and oxidation of the decoction pieces, thereby promoting lipid rancidity and degradation of chemical components. Previous studies have shown that increased moisture content promotes the hydrolysis of lipids and accelerates the rancidity process, 34 while elevated temperature further accelerates rancidity by promoting the decomposition of triglycerides into free fatty acids. 35 In contrast, the quality of decoction pieces packaged in polyethylene bags or aluminum foil (both vacuum and non-vacuum conditions) changed only minimally during storage. The increase in moisture observed in paper packaging was the primary factor driving chemical degradation. Rising moisture levels after day 5 were accompanied by parallel increases in acid value (from 1.21 to 2.84) and accelerated loss of amygdalin (15.41% by day 30).
In this experiment, the fatty oil content decreased from 57.53% to 53.06% and the acid value increased from 1.21 to 2.84 after 30 days of storage. The decline in fatty oil content reflects chemical transformation rather than physical loss and involves three main pathways36,37: autoxidation (formation of hydroperoxides), hydrolytic rancidity (release of free fatty acids, reflected in increased acid value), and secondary oxidation (decomposition into volatile products that may be lost during drying). The pronounced deterioration observed in the paper-packaged samples is consistent with the higher permeability of paper to moisture and oxygen, which accelerates all three degradation pathways. These results indicate that paper bag packaging cannot effectively maintain the quality stability of crushed FASA. Although the Chinese Pharmacopoeia does not specify a limit for the acid value of ASA, previous studies have indicated that acid values of 2.0–3.0 mg KOH/g represent significant rancidity in oil-rich seeds. 38 In the present study, the paper-packaged group reached an acid value of 2.84 by day 30, exceeding this threshold and correlating with visible sensory deterioration, including color darkening and mold growth. In contrast, all other packaging groups remained below 1.6.
Correlation Analysis Confirms Mechanistic Links Between Moisture and Degradation
Correlation analysis further confirmed these relationships: moisture content showed a strong positive correlation with acid value (P < 0.01) and a significant negative correlation with fatty oil content (P < 0.01), indicating that elevated moisture promotes triglyceride hydrolysis. However, no significant correlation was observed between moisture content and amygdalin content (P > 0.05), suggesting that the degradation of amygdalin may be influenced by factors beyond moisture alone, such as localized enzyme activity variations or sample heterogeneity. In contrast, polyethylene and aluminum foil packaging maintained moisture levels below 4.0%, resulting in minimal changes in acid value (< 1.6) and relatively stable amygdalin content throughout the storage period.
Dynamic Equilibrium Explains Fluctuations in Amygdalin Content Under Protective Packaging
The fluctuations observed in amygdalin content under polyethylene and aluminum foil packaging may be attributed to dynamic equilibrium processes within the herbal matrix. Under low-moisture conditions, where enzymatic hydrolysis is largely inhibited, reversible adsorption–desorption interactions between amygdalin and matrix components such as cellulose, pectin, and proteins may occur. In addition, trace reversible transformations between amygdalin and related glycosides may also contribute to these variations. Such equilibrium-driven fluctuations are random and non-directional, which explains why the measured values oscillated around the baseline and returned to levels close to the initial content by day 30. These fluctuations do not alter the primary conclusion that polyethylene and aluminum foil packaging effectively preserve amygdalin content. Although vacuum packaging is generally considered to reduce oxygen exposure and thereby slow oxidation reactions, no significant differences were observed between vacuum and non-vacuum packaging in terms of the overall quality indicators in this study. This finding suggests that vacuum treatment provides limited additional benefit for maintaining the stability of crushed FASA under the tested conditions. It should also be emphasized that these conclusions are based on accelerated stability testing. Comprehensive validation of long-term storage stability will require further real-time long-term stability studies.
Limitation
Several limitations of the present study should be acknowledged. First, the relationship between geographic origin, cultivation conditions, and the physicochemical properties of ASA was not systematically investigated, although such information would contribute to the development of raw-material-based quality standards. Second, objective colorimetric analysis combined with high-resolution photographic documentation was not applied to quantify changes in sample color, and visual observation was therefore used instead. Third, comprehensive dissolution kinetic studies employing appropriate mathematical models were not conducted to quantitatively describe the relationship between particle size and extraction efficiency. Fourth, the methodologies employed in this study—including traditional water decoction and extended drying processes—are relatively time-consuming and resource-intensive. While these approaches were adopted to closely reflect clinical practice, they may not be optimal for routine quality control purposes. Future studies should explore more efficient and practical methodologies that balance scientific rigor with operational feasibility. Future studies addressing these aspects would further strengthen the scientific basis for quality control and standardized processing of ASA.
Conclusions
In summary, the results of this study demonstrate that processing significantly alters the physical properties of ASA (P ≤ 0.001), while the extract yield decreased significantly only in fried products (P = 0.001), indicating that processing plays an important role in determining the quality and storage stability of decoction pieces. The contents of amygdalin and prunasin in the water decoctions of all three crushed ASA products increased significantly compared with those of whole seeds (P < 0.001). The recommended optimal particle size ranges are 4–5 mm for RASA, 2–4 mm for BASA, and 0.85–2 mm for FASA.
With regard to packaging, samples stored in paper bags showed severe deterioration after 30 days, with moisture content increasing to 9.03%, acid value rising from 1.21 to 2.84, and amygdalin content decreasing by 15.41%. In contrast, all stability indicators in the polyethylene and aluminum foil packaging groups remained within acceptable ranges throughout the 30-day storage period, demonstrating that these materials—particularly under non-vacuum conditions—effectively preserve the quality of pre-crushed FASA. Although 10 batches were analyzed to account for batch-to-batch variability and some data dispersion was observed, the consistent trends across batches support the robustness and reliability of the main conclusions.
Study limitations: This study focused on only two chemical markers (amygdalin and prunasin), and the stability evaluation was conducted under a single accelerated storage condition without confirmation from long-term real-time stability studies.
Footnotes
Ethical Considerations
Ethical Approval is not applicable for this article.
Consent to Participate
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Qi Wang, Jiuhui Gong and Tan Xue: Writing—Original Draft, Methodology, Visualization, Writing—Review and Editing; Xiaopeng Wang: Writing—Review and Editing; Xiaoming Wu: Writing—Review and Editing; Jianing Sun: Writing—Review and Editing; Junhan Shi, Xinjing Gui and Ruixin Liu: Project Administration, Conceptualization, Supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the “National Administration of Traditional Chinese Medicine Young Qihuang Scholars Support Program” (2022J056); “Henan Provincial Natural Science Foundation Outstanding Youth Science Fund Project” (242300421023); “Henan Provincial University Science and Technology Innovation Team Support Program” (26IRTSTHN019); “Zhongjing Three Distinguished Talents Project ‘Zhongjing Leading Expert Project’ ”(03104168X-4); “Henan Provincial Natural Science Foundation Youth Program ”(252300423791); “Doctoral Research Start-up Fund of The First Affiliated Hospital of Henan University of Chinese Medicine ”(CZ0377-31); “2025 National Administration of Traditional Chinese Medicine Monitoring and Statistical Research Project” (2025JCTJE72); “Science and Technology Innovation Team in Universities of Henan Province” (23IRTSTHN026); “Central Plains Talent Plan-Central Plains Leading Talents (Science and Technology Innovation Field)” (264200510030).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
