Abstract
Objectives
This study aimed to assess the effects of different cooking methods and extraction solvents on the bioactive properties of sweet potatoes (
Methods
Sweet potato samples were processed using an air fryer (150 °C for 15, 20, or 25 min) or a microwave oven (720 W for 2, 3, or 4 min). The samples were cooled to room temperature, oven-dried, and extracts were prepared using either 100% ethanol or 50% ethanol–water. Subsequently, the extracts were analyzed for total phenolic content (TPC), total flavonoid content (TFC), antioxidant activity, reducing power, and antimicrobial activity.
Results
Chlorogenic acid, 3,5-DNSA, and salicylic acid were detected in all samples, with chlorogenic acid being the predominant phenolic compound. The control SCW (extracted in 50% ethanol/water) showed the highest mean total polyphenol content (TPC, 7.53 mg GAE/g DW) and flavonoid content (TFC, 4.39 mg CE/g DW). Compared to microwave cooking, air-fried samples exhibited higher TPC (medium duration) and TFC (short duration). Control SCE (100% ethanol) exhibited higher radical scavenging activity (81.88%), however, the SCW demonstrated greater reducing power (0.92 for SCW vs. 0.89 for SCE). Ethanol/water extraction, significantly (p ≤ 0.05) enhanced antioxidant activity, with air frying showing greater radical scavenging at medium/high cooking and higher reducing power at low cooking times. Both aqueous and ethanol extracts of sweet potato possess notable antimicrobial properties, though the ethanol extract generally exhibited superior efficacy.
Conclusions
The results indicate that different extraction solvents and thermal processing treatments of sweet potatoes have varying effects on the content of bioactive compounds and these results might be beneficial for functional food processors.
Introduction
Sweet potatoes (
Most vegetables require cooking to inactivate anti-nutritional factors and enhance bio-accessibility of some phyto-constituents and antioxidant activity. Different food processing methods used before consumption can significantly influence the bio-accessibility of phytochemicals and their antioxidant potential. 11 The thermal processing of sweet potatoes is used for decades to increase their palatability, digestibility, and safety. Boiling, steaming, frying, roasting, and drying are examples of traditional cooking methods.12,13 Nowadays, vacuum cooking, infrared heating, microwave heating and ultrasonic-assisted cooking technologies are quickly emerging to maximize nutrient retention, starch modification, and bioactivity.12-14 Heat treatment induces changes in the physical and chemical content which in turn influence the compounds bioavailability.15,16 Sometime, it may reduce some nutrients but often enhances the availability and activity of these compounds by breaking down plant cell walls and releasing or transforming them into more active forms. 17 Bengtsson et al, 18 reported that cooking methods except microwaving increased the carotenoids release from orange-fleshed sweet potatoes matrix by softening and degrading cell walls. Similarly, the study conducted by Franková et al, 19 on the effect of different heat treatment methods found that all processed methods positively influenced the total polyphenol content and antioxidant activity in sweet potatoes. Musilova et al, 20 in their study have mentioned that different processing methods including baking, boiling, microwaving, and steaming had varying effect on bioactive compound of sweet potato tubers. An increase in total polyphenol content (TPC), anthocyanins, and antioxidant activity has been observed compared to raw samples while phenolic acids reduced. Similarly, some other studies have also noted increase in TPC of sweet potatoes while certain phenolic derivative such as caffeic acids declines by heat treatment.21,22 Like sweet potato tubers, the leaves polyphenols have also shown strong antioxidant activity and good processing stability.23,24 Now a days air frying and cooking in microwave are the most commonly used household cooking method which are known for providing rapid heating with less cooking time. Solvent choice is also very crucial for extraction of polyphenols from plant materials, as it is a key factor in determining the success of the extraction process. 25 Combining processing techniques with solvent extraction enables the efficient recovery of valuable bioactive compounds. 26 Various thermal processing techniques effect on sweet potato phenolics and antioxidant activity have been reported previously and a number of studies have additionally investigated the impact of solvent on phenolic extraction, but these aspects have largely been studied separately and contradictions have also been reported which might be due to differences in the variety of sweet potato, their processing duration, techniques or temperature. Thus the purpose of this study was to investigate how the most common household technique used at home now a days i.e. air frying and microwave cooking at different time interval and solvent systems affects the phenolic composition and antioxidant activity of sweet potatoes. Their potential uses as natural colorants, antioxidants and anti-inflammatory nutraceutical emphasize the significance of this research in imparting information for food processing techniques, nutritional recommendation, and the development of functional foods.
Materials and Methods
Air Fryer and Microwave Oven Cooking and Sample Preparation
Sweet potatoes (orange fleshed) were obtained from a local market in Riyadh, Saudi Arabia. All control and treated samples were drawn from a single, homogenized batch of sweet potatoes, meaning baseline variables (maturity, harvest time, initial moisture) were internally controlled. The whole sweet potato was cut into uniform pieces (1 cm thickness) and divided into two batches to be cooked in the household air fryer (Philips Avance Collection 1.4 Kg Airfryer XXL - HD9650/94) and microwave oven (MWL311, Kenwood, China; 900 W and 2450 MHz). In air fryer the sweet potato was heated at 150°C for 15 min (low), 20 min (medium) and 25 min (high). Similarly another lot was placed in the middle of microwave oven cavity and treated at 720 W for 2 min (low), 3 min (medium) and 4 min (high). After cooking, the samples were cooled to room temperature (23 °C) and were placed in oven dryer at 40°C for 120 h. Uncooked sweet potato was dried under same conditions and used as control. Because microwave and air-frying treatments differentially alter the moisture content of the sweet potato matrix, samples were oven-dried to a standardized moisture content prior to extraction. Low temperature (40°C) was chosen to reduce heat-sensitive phenolics’ oxidation and thermal degradation 27 while letting complete moisture removal for stable storage and analysis. All samples were subjected to identical drying conditions (40°C for 120 h). As a result, any drying-related degradation would have happened consistently in all treatments and the relative differences observed can still be attributed mainly to the cooking process rather than the drying itself. Consequently, to ensure accurate and reproducible comparisons across all treatment groups, all final concentrations and bioactive yields are reported on a dry weight (DW) basis (e.g., mg/100 g DW). Finally, the dried samples were ground into powder and sieved through 60 mesh and were stored in air-tight plastic bags at room temperature for further use.
Extraction
Two grams powdered sweet potato was extracted with 20 mL of each 100% ethanol and 50% (v/v) ethanol/water employing ultrasound extractions for 1 h at 20°C. Then, centrifugation was performed at 5000 RPM for 10 min at room temperature. The supernatant was then filtered using a Whattman filter paper number 2 and the obtained extracts were stored at 4 °C for further analysis.
The extracts obtained were termed as follows: SCE and SCW- Sweet potatoes (SP) extracted in 100% ethanol and 50% (v/v) ethanol/water respectively SALE, SAME, SAHE- SP air fried for low, medium and high cook duration respectively and extracted in 100% ethanol SMLE, SMME, SMHE- SP microwaved for low, medium and high cook duration respectively and extracted in 100% ethanol SALW, SAMW, SAHW- SP air fried for low, medium and high cook duration respectively and extracted in 50% (v/v) ethanol/water SMLW, SMMW, SMHW- SP microwaved for low, medium and high cook duration respectively and extracted in 50% (v/v) ethanol/water
Figure 1 represents the flowchart illustrating samples preparation and analysis. Flowchart illustrating samples preparation and analysis. 
Total Polyphenol Content and Total Flavonoid Content
The procedure described by Hayat et al, 28 was used to determine the TPC and TFC of sweet potatoes.
DPPH Scavenging
To determine the free radical scavenging capacity of the extract, 2, 2-diphenyl-1-picrylhydrazyl (DPPH) assay was used as described earlier, with some modifications.
29
In this procedure, 0.13 mL of extract was mixed with 2 mL of 0.1 mM DPPH solution. The mixture was incubated for 30 min in dark and then absorbance was read at 510 nm (Jasco, V-630 spectrophotometer, USA). The control was prepared in the same way but extract was replaced by ethanol. The percentage of DPPH radical scavenging activity was calculated as follows:
Reducing Power
Reducing power was determined according to the method used by Sharma and Vig, 30 with some modification. Briefly, 0.5 mL extract was mixed with 1.25 mL of potassium ferricyanide and 1.25 mL buffer (0.2 M, pH 6.6) and incubated at 50 °C for 20 min. Then 1.25 mL trichloroacetic acid was added in the mixture and centrifuged at 3000 rpm for 10 min. Finally, 1.25 mL aliquot was acquired from the supernatant, to which 1.25 mL water and 0.25 mL of ferric chloride was added and absorbance was measured at 700 nm (Jasco, V-630 spectrophotometer, USA).
HPLC Analysis of Phenolic Compounds
Phenolic compounds (tannic acid, resorcinol, 1, 2-dihydroxybenzene (1, 2-DHB), chlorogenic acid, caffeic acid, vanillin, 3, 5-dinitrosalicylic acid (3, 5-DNSA), salicylic acid) analysis was carried out using HPLC analysis, as described earlier 31 with some modifications. The HPLC (Prominence) system Shimadzu (Kyoto, Japan) was equipped with LC-20AB binary pump and variable Shimadzu SPD-10A UV detector. The column used was Zorbax SB-C18 (250 x 4.6 mm, 5µm; Agilent, Santa Clara, CA, USA) and mobile-phase was Milli Q water (1% acetic acid, A) and MeOH (B). The binary gradient program used was 0−10 min. 15−30% B; 10−20 min. 30−40% B; 20−30 min. 40−50% B; 30−41 min. 50−60% B; and 41−45 min. 15% B. The flow rate was 1.0 mL/min. The injection volume was 10 μL, and the detector was set at 280 nm. Compounds were identified by comparing their peak retention time with those of standards analyzed under identical chromatographic condition i.e same column, mobile phase composition, gradient program, detection wavelength.
Antimicrobial Activity
Antimicrobial activity was assessed against
Assessment of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
MIC of the sweet potato extracts was determined using the microbroth dilution method while the dilution in broth method was used to calculate the MBC for the antimicrobials described previously. 33
Statistical Analysis
One-way analysis of variance (ANOVA) was performed to identify the differences among the treatment groups by SAS (Version 9.2, 2000-2008; SAS Institute Inc., Cary, NC, USA) at a significance level of p ≤ 0.05. A post-hoc analysis using Duncan’s multiple range tests was performed if any significant differences were found, at a confidence interval of 95%. All analysis was performed in triplicate ± standard deviation.
Results and Discussion
Quantification of Phenolic Acid Compounds of Sweet Potatoes
Effect of Extraction Solvents on the Phenolic Compounds of Sweet Potatoes Subjected to Different Cooking Methods (mg/100 g DW)
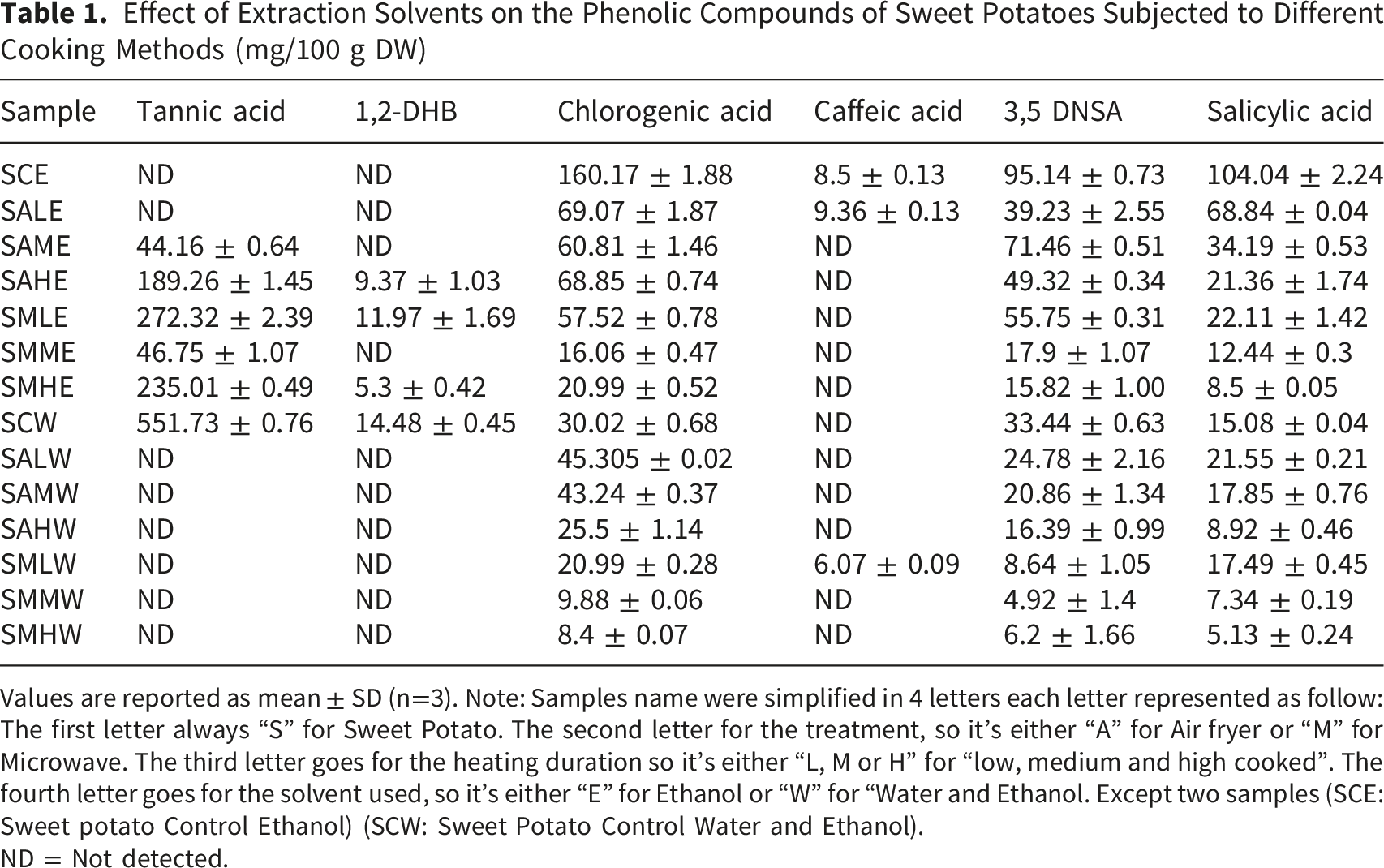
Values are reported as mean ± SD (n=3). Note: Samples name were simplified in 4 letters each letter represented as follow: The first letter always “S” for Sweet Potato. The second letter for the treatment, so it’s either “A” for Air fryer or “M” for Microwave. The third letter goes for the heating duration so it’s either “L, M or H” for “low, medium and high cooked”. The fourth letter goes for the solvent used, so it’s either “E” for Ethanol or “W” for “Water and Ethanol. Except two samples (SCE: Sweet potato Control Ethanol) (SCW: Sweet Potato Control Water and Ethanol).
ND = Not detected.
The highest amount of chlorogenic acid, 3, 5 DNSA and salicylic acid was observed in samples extracted in ethanol. Generally, the extraction of bioactive compounds from plant material is carried out either using a single solvent or a mixture of two solvents or mixtures of water with the alcohols, at variable ratios. 37 Jeon et al, 38 in their study have reported that as compared to pure water, chlorogenic acid was more soluble in pure ethanol, and the solubility increased with temperature. They also reported that the solubility of chlorogenic acid in mixed solvent exhibited a gradual rise as the water content increased, reaching a maximum at a specific water weight fraction and then decreased sharply as the water weight fraction increased further. A previous study that aimed to determine the best extraction method for phenolic compounds in the New Zealand damson plums reported that neo-chlorogenic acid was extracted better using ethanol while epicatechin and catechin were extracted more efficiently using water, which highlights the prominence of the role of extraction solvent in determining the yield of desired compounds. 39 Various parameters influence the phenolic compounds extraction, with solvent characteristics considered as the most crucial because of their polarities and chemical properties, 40 and ethanol-water mixtures are ideal for plant extracts because of their efficiency to get combined in any ratio, offer different polarities, and prevent the toxic residues associated with solvents like acetone and methanol, so even considered as safe for human consumption. 25 Almost all processing methods induced reductions in phenolic compounds when compared to control, but as compared to microwave cooking, more chlorogenic acid, caffeic acid, 3,5-DNSA and salicylic acid was exhibited in samples cooked in air fryer and a reduction in almost all phenolic compound has been noted with increase in cooking duration in both air fried and microwave cooked sweet potatoes. In a previous study, caffeic acid was significantly affected by the cooking methods (p < 0.05), particularly those involving cooking for a long time and at high temperature, such as air-frying and baking. 41
Effect of Extraction Solvents on the TPC and TFC of Sweet Potatoes Subjected to Different Cooking Methods
Phenolic compounds have been identified to contribute to the color, flavor, and antioxidant activity of the plants.
42
Figure 2A and B depicts the effect of extraction solvents (100% ethanol and 50% (v/v) ethanol/water) on TPC and TFC respectively of sweet potatoes subjected to different cooking methods. The Control SCW exhibited the highest mean value of TPC [7.53 milligrams of gallic acid equivalents per gram of dry weight (mg GAE/g DW)] and TFC [4.39 milligrams catechin equivalents per gram dry weight (mg CE/g DW)]. TPC and TFC exhibited by SCE was 4.37 mg GAE/g DW and 3.78 mg CE/g DW respectively. It has been observed that the 50% (v/v) ethanol/water extraction showed significantly (p≤0.05) more total TPC and TFC than 100% ethanol and as compared to microwave the samples cooked in air fryer showed better and higher TPC especially under medium duration cooked condition (cooked at 150 °C for 20 min) and TFC especially under low duration cooked condition (cooked at 150 °C for 15 min). The TPC and TFC results align with previous findings that binary solvents are more effective than mono-solvents for extracting phenolic compounds from plants,
43
as the addition of water to the organic solvents forms a more polar medium that simplifies the extraction of phenolic compound.
44
All the treated sample (air fried and microwaved) extracted in 50% (v/v) ethanol/water exhibited more TPC, while air fried samples exhibited more TFC than their corresponding ethanol extracted treatment. Least TPC (2.73 mg GAE/g DW) was exhibited by the high duration cooked, microwaved samples extracted in ethanol (SMHE), while least TFC (1.51 mg CE/g DW) was exhibited by the medium duration cooked, microwaved samples extracted in 50% (v/v) ethanol/water (SMMW). During processing the change in phenolic is more likely due to a combination of degradation from the effects of heat, oxidation by polyphenol oxidase, isomerization and even due to leaching into water.
21
Donado-Pestana et al,
15
in their study in raw roots of the four cultivars of sweet potato reported that the total phenolic content ranged from 1.30 to 1.93 mg/g DW. Padda and Picha
35
reported that phenolic content varied between 1.4 and 4.7 mg/g DW with the highest values credited to a purple-fleshed cultivar which is lower than the values reported in this study. Various factors such as sweet potato genotypes, phenolic extraction methods, and growing conditions
45
with variety being the most significant, contribute to the wide range of TPC values in sweet potato.
8
In sweet potato, substances such as aromatic amines, ascorbic acid, sulfur oxide and other compounds can interfere with the Folin-Ciocalteu assay, thus giving an inaccurate and higher than the real phenolic content, which is a drawback of Folin-Ciocalteu assay.
46
So, comparing these data is challenging due to variations in extraction techniques, solvents, and analytical methods used across studies.
47
In a previous study,
35
the TFC of the different sweet potato cultivars varied according to heat treatments with distilled water (2.44–54.39 mg CE/g ER) and ethanol A (5.37–65.43 mg CE/g ER), and were higher than that of untreated sweet potato (0.70 -29.78 mg CE/g ER). In a latest study the TPC and TFC significantly (p < 0.05) increased in all genotypes of sweet potatoes, irrespective of the cooking method applied.
48
Vegetables TFC can be influenced by both intrinsic and extrinsic factors, including variety, maturity stage, cultivation location, and other processing conditions, such as temperature, pH, and pressure.
49
Some studies,1,22,35 reports increase in phenolic content in cooked samples which can be attributed to the release of bound phenolic and inactivation of polyphenol oxidase affected by heat treatment, bust most of these studied compared the cooked/heat treatment with raw samples, but in the present study even the control sample like all other treated sample after treatment was dried at 40 °C for 120 h, so in the present study control sample was not actually the raw sample Effect of extraction solvents on (2A) TPC (mg GAE/g DW) and (2B) TFC (mg CE/g DW) of sweet potatoes subjected to different cooking methods. Values are reported as mean (n=3) ± SD. Different letters indicates significant differences at p≤0.05 according to Duncan’s multiple range test. 
Research suggests that the best solvent system for the extraction of phytochemicals is hydro alcoholic mixtures such as water and ethanol. 25 Generally, 40–50% ethanol has a greater effectiveness for the extraction of polyphenolic compounds when compared to pure ethanol. In a study, extraction yield increased with ethanol concentration, peaking at 50:50 (v:v), likely due to enhanced solubility of phenolic, tannins, flavonoids, and polysaccharides in ethanol–water mixture, but declined beyond this ratio. 52 The use of higher temperatures (>140 °C) for shorter periods in air-frying, combined with the constant circulation of hot air, makes it an ideal method for preserving nutrients and phenolic compounds without causing them to leach out. 53
Effect of Extraction Solvents on the DPPH Radical Scavenging and Reducing Power of Sweet Potatoes Subjected to Different Cooking Methods
Figure 3A and B depicts the effect of extraction solvents on DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging and reducing power of sweet potatoes. Control, SCE extracted in 100% ethanol exhibited more radical scavenging activity (81.88%) when compared with SCW i.e. 50% v/v ethanol/water (70.16%), while, the control SCW exhibited more reducing power than SCE (0.92 SCW vs 0.89 SCE). The sweet potatoes reducing power aligned closely with the results of TPC and TFC, representing that the extracted bioactive compounds have a significant influence on reducing power. It has been observed both air fried and microwaved samples extracted in 50% (v/v) ethanol/water showed significantly (p≤0.05) more radical scavenging activity, which ranged from 60.2%-77.52% indicating their higher antioxidant potential when compared with the air fried and microwaved samples extracted in 100% ethanol which ranged from 19.99%-45.53% and same trend was followed in reducing power and as compared to microwave the samples cooked in air fryer showed better and higher radical scavenging activity especially under medium and high cooked condition and reducing power especially under low time cooked condition (SP cooked 150 °C for 15 min). More than 50% radical scavenging activity was reported for air fried sample, showing that thermal processing has favorable conditions toward antioxidant activity which is in line with a previous study in Effect of extraction solvents on (3A) DPPH radical scavenging (%) and (3B) reducing power of sweet potatoes subjected to different cooking methods. Values are reported as mean (n=3) ± SD. Different letters indicates significant differences at p≤0.05 according to Duncan’s multiple range test. 
Effect of Extraction Solvents on the Antimicrobial Activity, Minimum Inhibitory Concentrations (MIC) and Minimum Bactericidal Concentrations (MBC) of Sweet Potatoes
Effect of Extraction Solvents on the Antimicrobial Activity of Sweet Potatoes
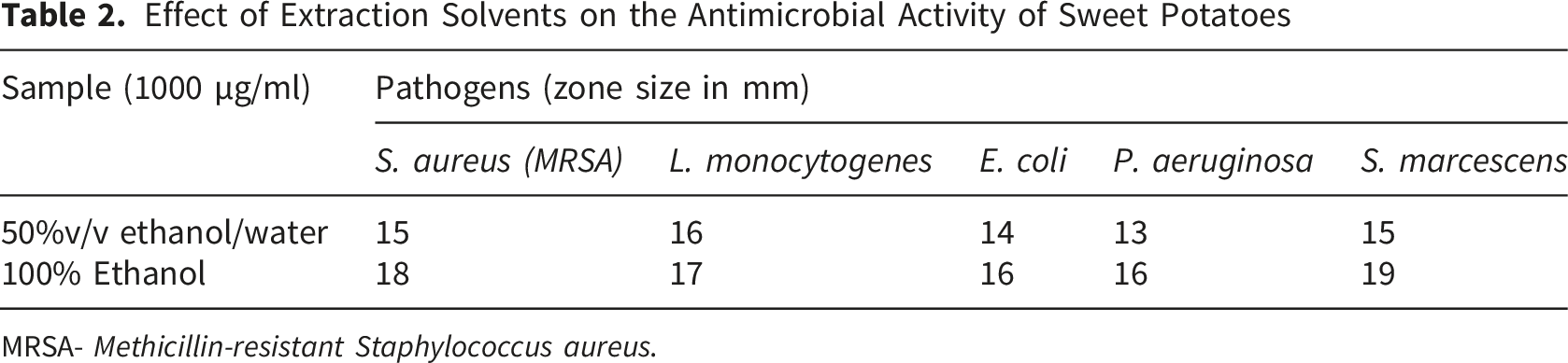
MRSA-
Effect of Extraction Solvents on the Minimum Inhibitory Concentrations (MIC) and Minimum Bactericidal Concentrations (MBC) of Sweet Potatoes
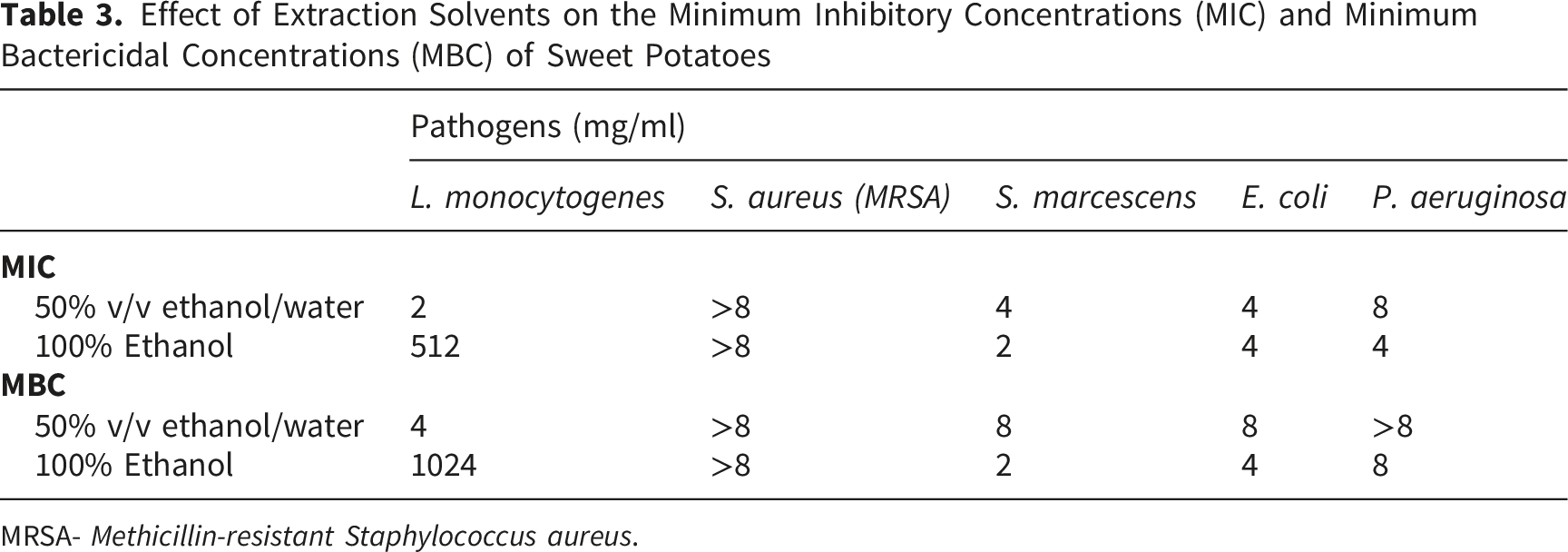
MRSA-
The 100% ethanol extract exhibited bactericidal activity (Table 3) against
The findings demonstrate that both 50% v/v ethanol/water and ethanol extracts of sweet potato possess notable antimicrobial properties, though the ethanol extract generally exhibited superior efficacy, indicating that ethanol is more effective at extracting phytochemicals with antimicrobial properties such as phenolics, flavonoids, and anthocyanins, particularly from root vegetables like sweet potatoes. The ethanol extract’s strong activity against
This study has several limitations that needs to be acknowledged. A major limitation of the present study is the absence of in vitro digestion or bioaccessibility analysis. While total phenolic content and antioxidant activity provide useful indicators of potential functionality, they do not fully reflect absorption, metabolism, or biological activity in humans. Another limitations are the absence of recorded moisture content after cooking and extraction yield efficiency as well as lack of normalization to fresh weight.
Conclusions
The results indicate that different extraction solvents and thermal processing treatment of sweet potatoes have varying effects on bioactive compounds content, including phenolic acids, polyphenols, flavonoids, as well as on their antioxidant properties. It has been observed that all extraction solvents significantly impacted the properties of sweet potatoes and among solvents, the 50% (v/v) ethanol/water extract of sweet potato air fried at low to medium heat duration showed the highest bioactive characteristics. The findings also demonstrated that both aqueous and ethanol extracts of sweet potato possess notable antimicrobial properties, though the ethanol extract generally exhibited superior efficacy.
Footnotes
Acknowledgments
The author- extend thanks to Ongoing Research Funding program, (ORF-2026-437), King Saud University, Riyadh, Saudi Arabia. I would like to express my sincere gratitude to Mrs. Shaista Arzoo, a researcher in Department of Food Science- and Nutrition, College of Food and Agriculture Sciences, for her valuable contribution to the successful completion of this research paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Ongoing Research Funding program, (ORF-2026-437), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
