Abstract
Background
Teucrium divaricatum subsp. canescens, an endemic plant of Cyprus, has been employed in folk medicine for wound healing and digestive ailments. Despite the documented anticancer properties of other Teucrium species, no studies have examined this endemic subspecies’ biological activities or chemical profile. This study aimed to evaluate the phytochemical profile and anticancer activities of T. divaricatum subsp. canescens using cytotoxicity, ROS scavenging, and wound-healing assays.
Method
Plant were extracted with 70% methanol (TM) and sub-fractionated into hexane (TM-H), ethyl acetate (TM-E), butanol (TM-B), and water (TM-W). Chemical composition was analyzed with LC-MS/MS. Cytotoxicity, intracellular ROS scavenging, and motility inhibition assays were carried out on the SK-HEP-1 liver adenocarcinoma cell line. IC50 values were calculated from MTT and DCFH-DA assays, and wound healing activity was evaluated for TM-E and TM-B fractions.
Results
Luteolin, cirsimaritin, cirsiliol and forsythoside B are dominant bioactive compounds. Although the crude TM exhibited relatively weak cytotoxicity (IC50 = 628.5 µg/mL), fractionation markedly enhanced this effect. Among the subfractions, the TM-E showed the strongest cytotoxicity (IC50 = 47.75 µg/mL), followed by the TM-H (IC50 = 82.27 µg/mL) and the TM-B (IC50 = 139.4 µg/mL). TM-E and TM-B fractions exhibited significant antioxidant activity, effectively reducing both basal ROS levels (IC50 = 118.8 µg/mL and 17.28 µg/mL, respectively) and induced oxidative stress. Under induced conditions, ROS levels were also markedly decreased. Furthermore, both fractions inhibited cell motility, with TM-E being more potent. The cytotoxicity and anti-motility effects were consistent with flavonoid content and potential pro-oxidant behavior of luteolin derivatives.
Conclusion
Extracts exhibit significant cytotoxic, antioxidant, and anti-motility activities against SK-HEP-1 cells, attributed to its high flavonoid content. These findings highlight its potential to be a candidate for further studies as discovering novel anticancer agents. Further studies on molecular mechanisms and active compounds are warranted to validate its therapeutic applications.
Keywords
1. Introduction
Cancer is considered one of the most life-threatening diseases in both developed and developing countries, causing significant health risks to the population. 1 Despite all the advances in cancer treatment, it is still the world’s second-biggest cause of death. Radiotherapy and chemotherapy are considered the most effective current therapies, but unfortunately, they have serious adverse effects because of toxicity to healthy cells. 2 Hepatocellular carcinoma (HCC) is the most common type of liver cancer and the second main cause of cancer-related deaths, with a rising number of cases. 3 The available treatments for HCC cover a wide spectrum of surgical interventions, from loco-regional to ablation therapy, and these are primarily suitable for early-stage cases. However, most patients are diagnosed at advanced stages, making them ineligible for these interventions. 4 Unfortunately, current chemotherapy treatments used at late stages of HCC are often inadequate, as traditional chemotherapy can significantly impact both the patient’s quality of life and treatment outcomes. This situation has raised the demand for the development of new natural anti-cancer agents. In the past few decades, research has focused on the therapeutic applications of natural products for cancer management. Some findings indicate that using herbal medicine with multi-target and multi-action mechanisms may be an effective treatment for HCC management.5-8 It has been found that dietary phytochemicals such as curcumin, resveratrol, quercetin, silibinin, lycopene, emodine, urolithin A and phloretin are useful in the treatment of HCC and other cancers. 9
Over 1,000 plant varieties today are known to have potent anti-cancer properties. According to a recent study, 60% of the global use of anticancer drugs has been produced from natural sources. Food and Drug Administration data shows that 74% of the approved natural molecules are used in cancer therapy.6,7,10
The Lamiaceae family, is one of the major families of medicinal plants that include basil (Ocimum spp.), mint (Mentha x piperita L.), rosemary (Rosmarinus officinalis L.), sage (Salvia officinalis L.), oregano (Origanum majorana L.), thyme (Thymus vulgaris L.). Members of the Lamiaceae family, known for their wide range of secondary metabolites, have demonstrated various biological activities, including antibacterial, antioxidant, anti-inflammatory, antimicrobial, antiviral, and anticancer properties, with the anticancer effects attributed to the phenolic compounds present in these plants, which also offer potential applications in the pharmaceutical field.11-16
The genus Teucrium, which includes many subspecies, including Teucrium divaricatum Kotschy subspecies, belongs to the Lamiaceae family. Teucrium divaricatum Kotschy subsp. canescens is endemic to Cyprus and used as a folk medicine in Cyprus, as well as in Palestine and Jordan. The plant infusion primarily uses traditional Cyprus medicine for stomachic purposes and also against the common cold and fever. 17 Anti-cancer studies with different species of Teucrium show that this species has promising effects against cancer cell proliferation.18-20 In a study, the methanol extract of Teucrium chamaedrys L. showed selective anticancer potential in SW480 colorectal cancer cells by inducing apoptosis and suppressing the expression of drug resistance genes (CYP1A1, CYP1B1, GSTP1, MRP1, and MRP2). 21 Teucrium polium L. and Teucrium persicum Boiss. extract has been shown to increase cell cycle arrest and decrease cell viability, invasion, and motility in PC3 and DU145 prostate and T47D breast cancer cells.22,23 Studies showed that oily fractions of Teucrium alopecurus de Noé scavenge free radicals and inhibit the proliferation of different cancer cell lines by suppressing gene expression related to survival, proliferation, invasion, angiogenesis, and metastasis processes. 24 Flavonoids play an important role in the antioxidant and anticancer activities of Teucrium species and they may have potential applications in anti-cancer therapies. 14 Although many Teucrium species such as T. polium, T. chamaedrys, and T. alopecurus have been extensively investigated for their antioxidant and anticancer properties, T. divaricatum subsp. canescens represents a taxonomically distinct endemic subspecies restricted to the Mediterranean microclimate of Cyprus. Its unique ecological conditions, including high sunlight exposure, dry limestone soils, and limited water availability, are known to influence secondary metabolite biosynthesis, particularly the accumulation of flavonoids and phenylethanoid glycosides. Therefore, its phytochemical profile and associated biological activities are expected to differ substantially from those of other well-studied Teucrium taxa.
Exploring this endemic plant provides not only a new perspective within the Teucrium genus but also contributes to the broader understanding of biodiversity-driven chemical diversity with biomedical relevance. In this context, the present study focuses on T. divaricatum subsp. canescens to fill this gap in the current knowledge of Cypriot endemics. While information on Teucrium species’ anticancer and antiradical effects is available, no reports are available on Cyprus endemics. To our knowledge, this is the first-ever study on Cyprus endemic Teucrium divaricatum subsp. canescens (Čelak.) Holmboe and its composition and biological activities on liver cancer SK-HEP-1 cells. This study followed a fractionation strategy to enhance the exploitation of antioxidant and anticancer compounds. Liquid Chromatography–Tandem Mass Spectrometry (LC-MS/MS) profiled extracts to determine possible chemicals responsible for biological activities and specify the most effective subfraction in managing cancer cells. Cytotoxicity, Reactive Oxygen Species (ROS) scavenging, and wound healing tests were applied to the methanolic extract, and the fractions were derived.
2. Materials and Methods
2.1. Chemicals
All used chemicals purchased from Sigma-Aldrich.
2.2. Plant Material
Aerial part of Teucrium divaricatum ssp. canescens (Čelak.) Holmboe was collected from Kyrenia region by (May 21, 2022). Botanist Prof. Dr. F. Neriman Ozhatay identified the samples. A voucher specimen of plant (Voucher No: NEUN20123) was deposited at the Near East University Herbarium (NEUN), Nicosia, Northern Cyprus.
2.3. Preparation of Extract
Air-dried herbal part of T. divaricatum subsp. canescens (50 g) was powdered and successively extracted with 70% aqueous methanol at room temperature. After 24 hours of incubation in aqueous methanol, the solvent was filtrated, and the extraction procedure was repeated 2 more times. Combined extracts were evaporated under vacuo at 40°C. Crude methanol extract (6,35 g) was sequentially fractionated using hexane (TM-H, 0.04 g), ethyl acetate (TM-E, 1.81 g), and butanol (TM-B, 2.81 g). The liquid portion left after sub-fractions was named as water fraction (TM-W, 4,63 g).
Each extract was separated and evaporated under vacuo at 40°C. The remaining water was removed by lyophilization. All samples were kept at -20°C until further analyses.
2.4. LC-MS/MS Analysis
LC-MS/MS analysis method was used for the chemical compound analysis of samples as previously described. 25 The Absciex 3200 MS/MS detector and negative mode ionization were used. Chromatographic separations were made with ODS 150 x 4.6 mm, i.d., 3μm column using Shimadzu 20A HPLC (High Performance Liquid Chromatography).
The temperature was set at 40°C for the column oven and the flow rate was changed to 0.5 mL/minute. Methanol: water: formic acid (10: 89: 1,v/v/v) and (B) methanol: water: formic acid (89: 10: 1,v/v/h). In 40 minutes, the concentration of B increased from 10 to 100 percent. A mass range of 100-1000 amu was selected for mass scanning (EMS).
2.5. Cell Culture
The human liver adenocarcinoma cell line SK-HEP-1 (ATCC® HTB-52™) was obtained from the American Type Culture Collection (Manassas, VA, USA). SK-HEP-1, was used to test the anti-cancer effects of methanol extract and its fractions of T. divaricatum ssp. canescens. The cells were maintained in Dulbecco`s Modified Eagles Medium (DMEM) supplemented with 10% heat-inactivated Fetal Bovine Serum (FBS), 100µ/ml penicillin, 2mM L-glutamine, 100mg/ml streptomycin, and 1X non-essential amino acids (NEAA) at 5% CO 2 at 37°C. Plant extracts dissolved in dimethyl sulfoxide (DMSO) before being applied to the cells. DMSO is used as solvent control in all experiments.
2.6. Cytotoxicity Assay
The MTT assay was used to assess the cytotoxic effects of tested extracts on the SK-HEP-1 cell line. 16 Briefly, 2x104 cells/well were seeded at 48-well plates. After overnight incubation cells were treated with plant extracts in a concentration range of 5 μg/mL-1000 μg/mL. Following a 48 hours incubation of extract treatment 0.5 mg/mL MTT was added and after 4 hours of incubation, formazan crystals were dissolved in 100μL of DMSO and absorption was measured using a microplate reader at 570nm and 640nm wavelengths for background control. Cell viability was calculated and was demonstrated as the percentage of viable cells.
2.7. Intracellular ROS Scavenging Assay
DCFH-DA assay was carried out to evaluate extracts’ capacity to scavenge basal and induced ROS activity in SK-HEP-1 cells.1 16 SK-HEP-1 cells were plated on a 96-well plate at a density of 1x104 cells/well. After overnight incubation, cells were treated with different concentrations of plant extracts (ranging between 5 μg/mL and 1000 μg/mL) and 20 μM DCFH-DA dissolved in the antioxidant treatment medium (DMEM-0 plus 10 m HEPES) and incubated for 45 minutes. The medium was replaced by non-appended medium or medium containing 250μM of H2O2. Following one hour of incubation, absorbance values were measured by Thermo Varioskan Flash Multimode microplate reader (Thermoscientific) under fluorescence absorption with an excitation-emission of 485-528 nm.
2.8. Wound Healing Assay
A wound healing assay was performed to evaluate the effect of TM-E and TM-B extracts which shown high basal and induced ROS scavenging activity on the motility of SK-HEP-1 cells. 16 The concentrations that did not affect cell viability were used in the wound healing assay. 50x104 cells were cultured in 6-well plates. After overnight incubation, cells reached 90% of confluency and wounds have been created by a sterile pipette tip. After washing the wound with PBS, extracts were applied to the cells in the concentration ranging from 10 μg/mL to 250 μg/mL. Following 24 hrs incubation, cells were fixed with ice-cold methanol and stained with 0.5% crystal violet. Images of wounds were taken using the CMEX-5.0 camera adjusted on EUROMEX light microscope. Wound-healing gap areas were quantified using ImageJ software to calculate the percentage of wound closure and generate semi-quantitative data for graphical analysis. The motility rates (%) of different HCC cell lines have been semi-quantitatively calculated as in Equation 1. (%Motility rate = [(Gap are t0 – Gap area t24/48 hrs/Gap are t0)] x100). Representative wound-healing images were presented, and red reference lines were added on the micrographs to facilitate visual comparison of the wound closure among experimental groups.
2.9. Statistical Analysis
Experiments were performed independently three times. Results were presented as mean ± SE from six measurements for DCFH-DA and MTT analyses. All the results were normalized to 100 and were stated as a percentage of the control. Data normality was verified prior to performing statistical analysis. One-way ANOVA test with Tukey’s correction for multiple comparisons was used. A p< 0.05 value was considered as statistically significant. IC50 values were determined by using the dose-response inhibition curve for both MTT and DCFH-DA assays. All analyses were performed by GraphPad Prism 8 program.
3. Results
3.1. LC-MS/MS Analysis
TM, TM-H, TM-E, TM-B, and TM-W were analyzed by LC-MS/MS Liquid Chromatography Spectrometry. Due to its composition of predominantly non-polar compounds, the TM-H fraction is not suitable for analysis using the current LC-MS/MS methodology. Compounds existing in TM, TM-E, TM-B, and TM-W were specified in chromatograms as shown in Figures 1A and 2B-D. respectively. The main compounds and their fragmentation details obtained using LC-MS/MS analysis of the extracts are represented in Table 1. All extracts contain polyphenolic compounds mainly flavonoid derivatives. The composition differences of sub-fractions are indicated in Table 1. Total LC–MS/MS chromatograms of (A) TM, (B) TM-E, (C) TM-B, and (D) TM-W fractions. Representative chromatograms show the distribution of major bioactive compounds detected in each fraction Effect of DMSO on SK-HEP-1 cell viability. Data represent the mean ± SE of three independent experiments (n = 3). Statistical significance was determined using one-way ANOVA followed by Tukey’s post hoc test after confirming data normality (MTT assay; ****p < 0.0001) Main Chemicals of Plant Extracts Obtained From LC-MS/MS Composition Analysis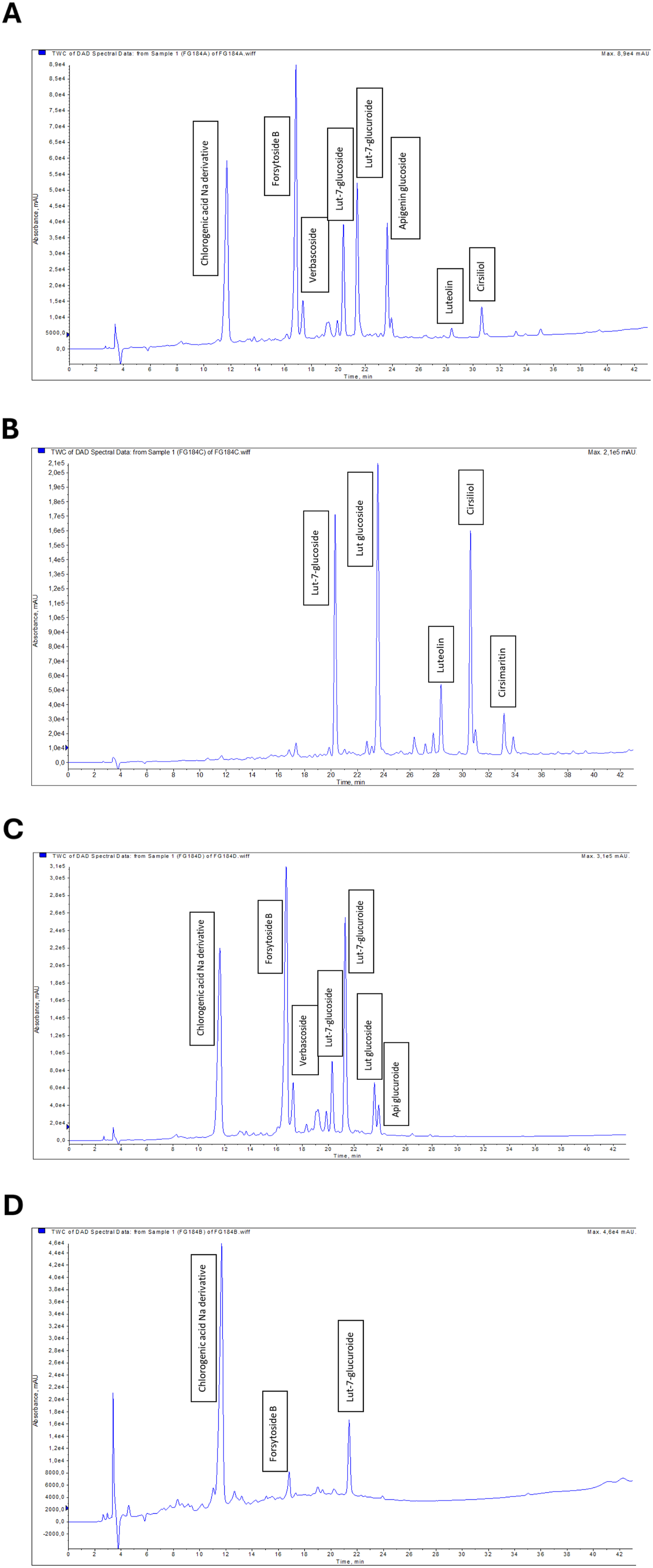


3.2. The Effect of DMSO as Solvent on SK-HEP-1 Cells
All extracts used in cell culture experiments, except for TM-W, were dissolved in DMSO, which served as the solvent control (negative control). The maximum concentration of DMSO reached in the cell culture medium did not exceed 1%, which had no effect on cell viability. Although the maximum concentration of DMSO in all treatments was kept below 1%, its effect was evaluated for SK-HEP-1. At the maximum PBM concentration, the effect of DMSO on cell viability was insignificant (Figure 2).
3.3. The Effect of TM and all Sub-fractions on SK-HEP-1 Cell Viability
The cytotoxic effect of all the extracts on SK-HEP-1 cells increased significantly in a concentration-dependent manner as it shown in Figure 3 (**** p< 0.0001). The IC50 value of TM was calculated as 628.5μg/mL. TM-H, TM-E, and TM-B sub-fractions were more effective in terms of cytotoxicity, with 82.27 μg/mL, 47.75 μg/mL and 139.4 μg/mL, IC50 values, respectively. TM-W fraction was the least effective with 705.4 μg/mL IC50 value possibly because of poor polyphenolic compounds. All IC50 values are listed in Table 2. Effects of TM and its sub-fractions on the viability of SK-HEP-1 cells. Data represent the mean ± SE of three independent experiments (n = 3). Statistical significance was determined using one-way ANOVA followed by Tukey’s post hoc test after confirming data normality (MTT assay; ****p < 0.0001) IC50 Values of TM, TM-H, TM-E, TM-B, TM-W on Viability and Basal Oxidative Stress of the SK-HEP-1 Cells

3.4. The Effect of Teucrium Extracts on Scavenging Intracellular ROS Activity of SK-HEP-1 Cells
The intracellular ROS scavenging effect at basal and induced conditions of all the extracts on SK-HEP-1 cells was analyzed using DCFH-DA assay and is shown in Figure 4. (**p<0.01, ***p<0.001, ****p<0.0001) The highest concentration used in this experiment was the maximum non-cytotoxic concentration that does not affect cell viability. TM-H fraction did not significantly reduce the basal ROS activity. The IC50 value of TM, TM-E, TM-B and TM-W in reducing basal oxidative state was calculated as 234.1μg/mL, 118.8μg/mL, 17.28μg/mL, and 26.65μg/mL, respectively. The induced levels of ROS have scavenged moderately with TM-H, TM-E, and TM-W in a concentration-dependent manner, but the effect of TM and TM-B on induced ROS activity was not statistically significant. In induced oxidative conditions, neither TM nor TM-B exhibited a clear dose-dependent ROS scavenging pattern. The extracts showed both statistically significant and non-significant scavenging effects across different concentrations, which did not follow a typical dose–response trend. The observed data did not exhibit the typical characteristics required for fitting to a sigmoidal dose-response model, thus preventing reliable estimation of IC50 values. This outcome may be attributed to the relatively limited scavenging capacity of the extracts, which was insufficient to neutralize the elevated ROS levels under induced oxidative stress conditions. The effect of TM and its sub-fractions on (A) basal oxidative stress and (B) H2O2-induced oxidative stress in SK-HEP-1 cells. Data represent the mean ± SE of three independent experiments (n = 3). Statistical significance was determined using one-way ANOVA followed by Tukey’s post hoc test after confirming data normality (DCFH-DA assay; **p < 0.01, ***p < 0.001, ****p < 0.0001)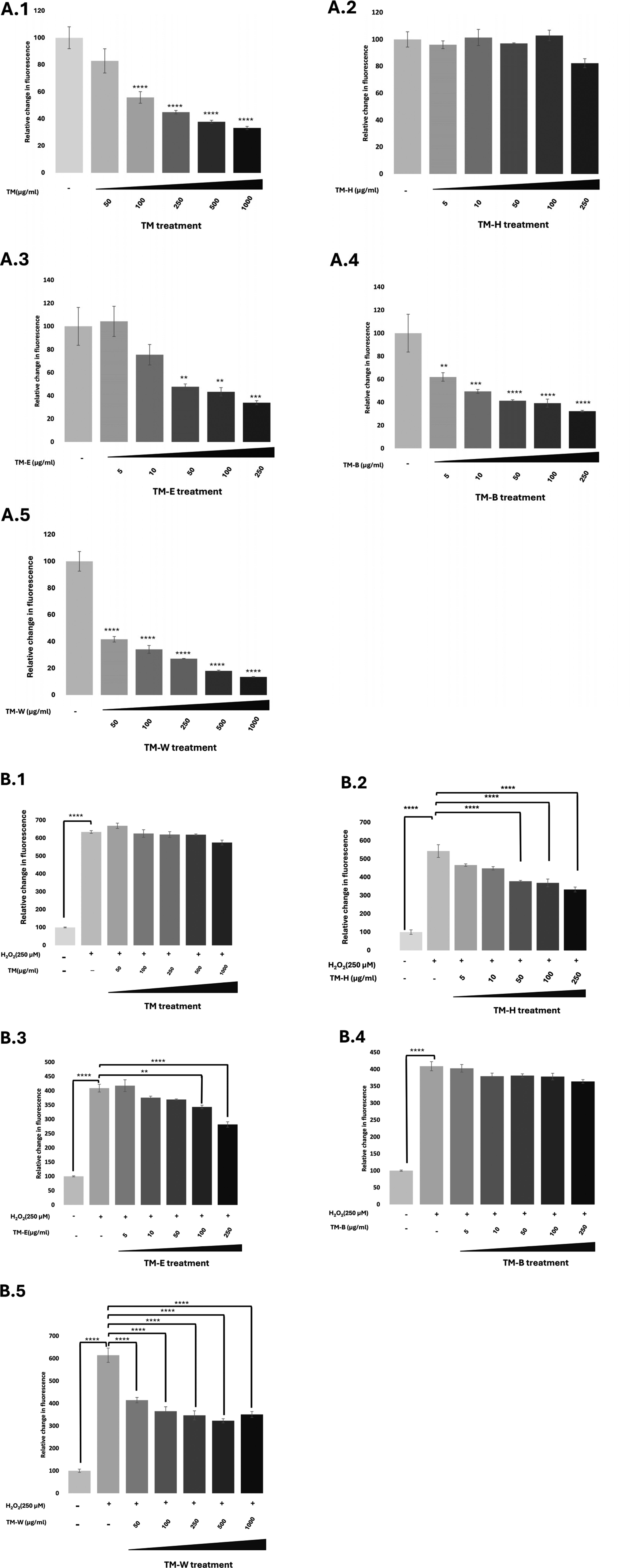
The effect of TM and its sub-fractions on the basal and induced oxidative stress are given in Figure 4.
3.5. Effect of TM-E and TM-B on the Motility of the SK-HEP-1 Adenocarcinoma Cell Line
For wound healing analysis, only TM-E and TM-B were used, as both exhibit cytotoxic and antioxidant scavenging activities. The highest concentration used in this experiment was the maximum non-cytotoxic concentration that does not affect cell viability. Besides cytotoxic and antioxidant activities both TM-E and TM-B extracts showed concentration-dependent inhibitory effects on the motility of the SK-HEP-1 cells. The results, presented semi-quantitatively, indicate an increase in the unhealed wound area, as shown in Figure 5. A significant reduction in cell motility was observed at concentrations exceeding 50 μg/mL for TM-B and 10 μg/mL for TM-E extracts. Effect of TM-E and TM-B on the motility of SK-HEP-1 adenocarcinoma cells, shown with semiquantitative analysis and representative images. Data are presented as mean ± SE of three independent experiments (n = 3). (Scratch wound assay)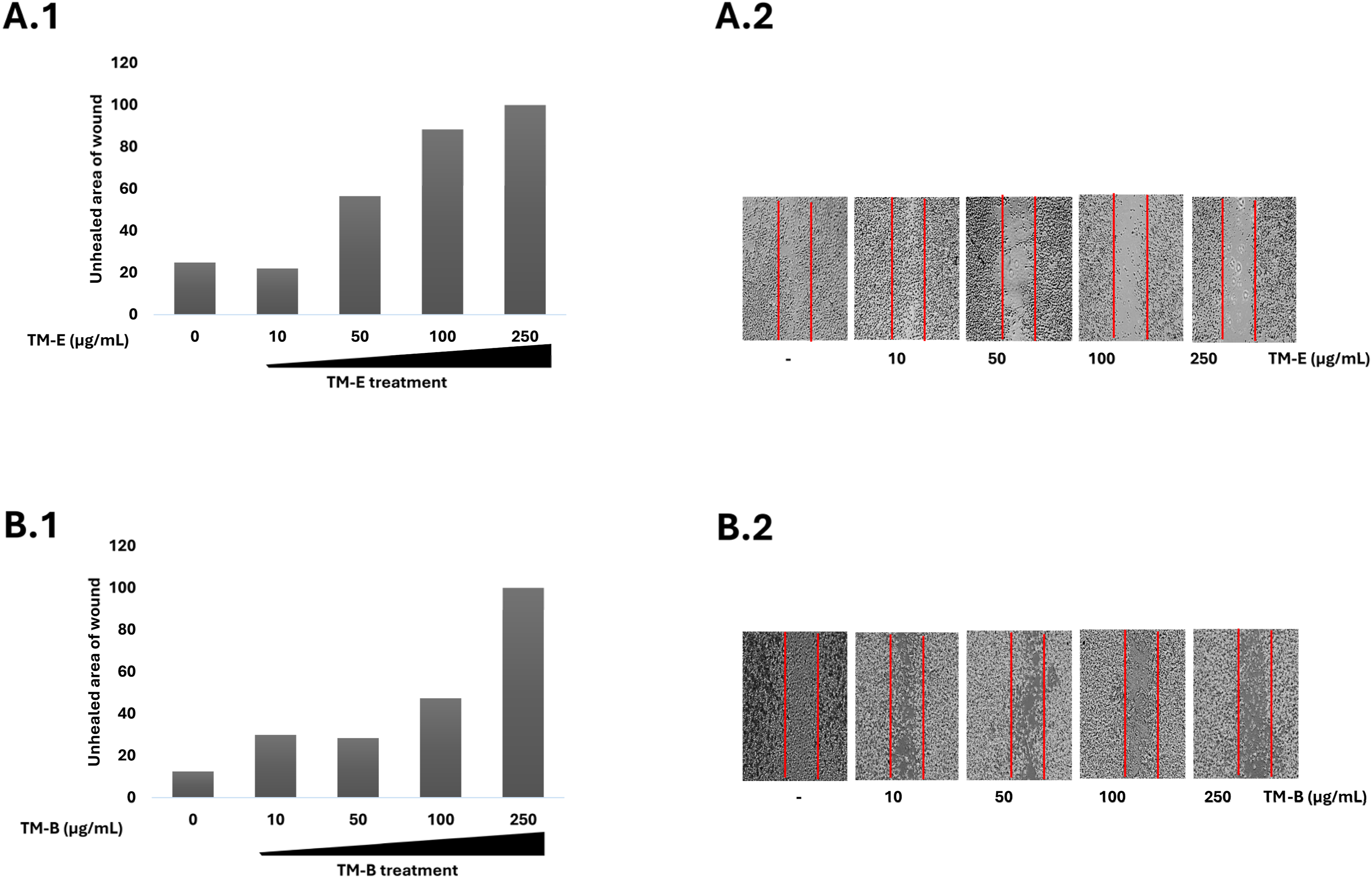
4. Discussion
T. divaricatum subsp. canescens methanolic extract (TM) was fractionated into non-polar, semi-polar, and polar components using different polarity solvents such as n-hexane, ethyl acetate, and butanol, respectively. Qualitative and quantitative analyses of the phytochemical composition by LC/MS-MS of TM and its sub-fractions; butanol fraction (TM-B), ethyl acetate fraction (TM-E), and water fraction (TM-W) revealed the presence of 14 compounds possibly related to either antioxidant or anticancer effects.
Our analysis regarding chemical composition clearly shows that even the constituents of chemicals vary, extracts are very rich in bioactive components, and the phytochemical composition is dominated by flavonoids, especially luteolin and its derivatives. Our results are compatible with studies in the literature, which indicate that flavonoids from different Lamiaceae sp. contain chemical constituents that exert good anti-cancer effects through various mechanisms.26,27 In this study, the main crude extract TM and all sub-fractions contain relatively high amounts of phenolic compounds such as forsythoside-B, chlorogenic acid derivatives, and flavonoids such as luteolin, cirsimaritin, and cirsiliol that are popular for their anti-cancer and antioxidant activities.27-30 The presence of Na derivative chlorogenic acid and luteolin is clear evidence of the cytotoxic effect of TM extract.
In the present study, the biological effects of all extracts were analyzed to determine the most effective one for anticancer properties so that we can reduce the biological effects to a certain group of chemicals. Our data showed that all extracts were cytotoxic on SK-HEP-1 cells, resulting in a significant decrease in cell viability in a concentration-dependent manner. The cellular toxicity effects of our plant extracts are similar to those of other Teucrium sp. with respect to cytotoxicity potential.31-34 IC50 cytotoxicity values of all sub-fractions were lower than the main extract TM, indicating that sub-fractionation successfully enriched the active compounds. A threshold suggested in previous studies that crude extracts showing an IC50 ≤ 100 µg/mL can be considered to be cytotoxic and chosen for further studies.35,36 The highest cytotoxicity against SK-HEP-1 cells was obtained with a TM-E fraction of 47.75 μg/mL, followed by TM-H with 82.27 μg/mL IC50 value. The presence of bioactive flavonoids—such as luteolin, cirsimaritin, and cirsiliol—in the studied fractions is noteworthy, as these compounds are known for their anticancer activity against a wide range of human cancers, including lung, breast, glioblastoma, prostate, colon, and pancreatic types. This aligns well with and supports our results.27,28,32,37-40 We believe that the high content of these bioactive flavonoids plays an important role in the antioxidant and anti-cancer activity of extracts and possibly explains the reason for the high cytotoxic effect of TM-E and TM-H. The least cytotoxic extract was TM-W, with a 705.4 μg/mL IC50 value. TM-W contains relatively small amounts of bioactive compounds compared to other fractions, explaining its highest IC50 value. In SK-HEP-1 cells, reported MTT based IC50 values for standard chemotherapeutic agents vary considerably across studies and experimental conditions. Sorafenib and doxorubicin are often reported in the low-micromolar range (Sorafenib 4.60 µM to 13.93 µM, Doxorubicin 0.5 µM to 11.09 µM), whereas 5-fluorouracil and Cisplatin exhibits a markedly broader and sometimes substantially higher range of IC50 values (5-fluorouracil 50 to >400 µg/mL- Cisplatin 14.3-117 µM) under comparable settings. When interpreted within the context of these reference ranges, the IC50 values obtained for our extracts differ from those reported for conventional chemotherapeutic agents in an expected manner. Reference drugs are single, purified molecules and are typically reported in molar units; in contrast, plant extracts are complex mixtures in which the active constituents are present at unknown and variable concentrations, and synergistic or antagonistic interactions may also occur. Therefore, direct numerical equivalence between purified drugs and crude or fractionated extracts should be approached with caution. For a more meaningful translational comparison, activity-guided isolation followed by compound-level testing under matched experimental conditions would be appropriate.41-46
Our results revealed that all extracts, expecting TM-H, have the potential to reduce the basal oxidative stress of the SK-HEP-1 cell. The only extract that had ROS scavenging activity at both basal and induced oxidative stress was TM-W. The existing high levels of ROS scavenger effects at TW may be possibly explained by low luteolin, luteolin derivatives and cirsimaritin content which are known to increase intracellular reactive oxygen species (ROS) levels as pro-oxidant on basal conditions at cell culture.27,29,47 On the other hand, TM-W, TH, and TE had moderate to low ROS scavenging activity, particularly on induced oxidative stress. Notably, as also stated in the Results section, under induced oxidative stress conditions, ROS readouts did not show a consistent monotonic dose–response pattern across concentrations. Accordingly, although statistically significant changes were observed at certain concentrations, the overall response did not fit to a typical sigmoidal dose–response relationship, which limited robust curve fitting; therefore, IC50 values for ROS scavenging could not be calculated for several conditions. This technical constraint should be taken into account when interpreting the antioxidant efficacy of the extracts under induced oxidative stress.
This antioxidant activity effect could be derived from the ROS scavenging effect of the same flavonoid content in these fractions. 48 We believe that the effects of luteolin derivatives and cirsimaritin are bidirectional because any antioxidant can act as an oxidant at high concentrations.
Wound healing assay is an in vitro assay analyzing the sheet migration of cancer cells involved in metastasis. TM-B and TM-E fractions, which showed high activity levels at cytotoxicity and ROS scavenging, were also assessed for their effect against 2D migration. Even though TM-E seems more potent, both fractions successfully inhibited cell migration, which can have the potential for anti-metastatic agent development. Cirsiliol has been shown to suppress epithelial to mesenchymal transition through upregulation of E-cadherin and downregulation of N-cadherin expression at malignant melanoma which explains the high motility inhibitory activity of TM-E. It is also shown that cirsiliol suppresses PI3K/AKT signalling pathways, which also regulates cellular behaviours such as proliferation, motility, and survival. 49 Motility and proliferation inhibition by tested extracts support these findings in the literature. Forsythoside B was also found to affect the RhoA/ROCK signaling pathway that regulates motility, and the presence of forsythoside B could be related to motility inhibitory actions of TM-B. 50 The antifibrotic effects of luteolin on hepatic stellate cells and liver fibrosis have been previously demonstrated, highlighting its protective role on liver cancer through modulation of the AKT/mTOR and TGF-β signaling pathways. 51 Similarly, the luteolin component of TM-E exerts anti-motility effects by possibly targeting the AKT/mTOR signaling pathway, as supported by existing literature. Although the present study provides novel data on the phytochemical composition and anticancer potential of T. divaricatum subsp. canescens, it has several limitations. The molecular mechanisms underlying the observed cytotoxic and anti-motility effects were not elucidated, as protein expression analyses or pathway-specific markers (e.g., apoptosis-, EMT-, or oxidative stress-related proteins) were not performed. Investigating these molecular signatures through protein expression profiling would further clarify the mechanistic basis of the observed biological effects. Future studies incorporating mechanistic assays (e.g., apoptosis and cell-cycle analyses) and molecular characterization at both the transcriptional (qRT-PCR) and protein levels (Western blotting of key markers) would help clarify the mechanistic basis of the observed biological effects. Also the absence of cytotoxicity data in normal cells is a limitation that requires further investigation to better define the selectivity and safety profile of the extracts. In addition, activity-guided fractionation and isolation of individual bioactive constituents could provide more precise identification of compounds responsible for the activity. However, due to current resource and equipment limitations, these advanced analyses could not be conducted within the present study. In our future studies, we will focus on the compounds thought to be effective and address these aspects to establish a more comprehensive mechanistic understanding.
5. Conclusion
This study on T. divaricatum subsp. canescens, an endemic plant, will pioneer future scientific research on this area. By considering both cytotoxic, antioxidant, and anti-motility effects of the TM and fractions, we can conclude that the great potential of anticancer effects is most likely due to the very high percentage of bioactive flavonoids of T. divaricatum subsp. canescens Furthermore, since TM and its fractions have a strong cytotoxic effect, they may be a good novel candidate for chemotherapeutic findings. Further studies focusing on compound-level validation and key mechanistic endpoints are needed to improve translational relevance and support this endemic plants development as a therapeutic lead.
Footnotes
Acknowledgments
The authors would like to thank Prof. Dr. F. Neriman Ozhatay for plant identification.
ORCID iDs
Ethical Considerations
This study did not involve human participants, human data, or animal subjects. Therefore, ethical approval was not required. The research was conducted using in vitro SK-HEP-1 liver adenocarcinoma cell cultures. Ethical approval is not applicable for this article.
Consent to Participate
This study did not involve human participants.
Consent for Publication
This study does not contain any individual person’s data, images, or videos.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
