Abstract
Lung cancer metastasis remains a major cause of mortality and a central challenge in clinical oncology. This review examines lung cancer metastasis through an integrative framework combining Chinese and Western medicine. It emphasizes the role of traditional Chinese cultural philosophy as a theoretical foundation for integrative approaches. Rather than serving merely as a cultural background, philosophical concepts such as holism, dynamic balance, and the unity of heaven and humanity are discussed in relation to contemporary biomedical concepts, including systems thinking, network regulation, and individualized treatment strategies in metastasis management.
Key cellular and molecular mechanisms of metastasis identified by modern biomedical research are summarized and compared with Traditional Chinese Medicine (TCM) approaches that emphasize systemic regulation, syndrome differentiation, and microenvironmental balance. The analysis is organized according to major stages of the metastatic cascade, highlighting the complementary roles of molecularly targeted therapies and holistic regulatory strategies. Such interactions are particularly evident in immune regulation, tumor microenvironment modulation, and treatment tolerance.
Finally, current challenges in integrative oncology are examined, including the lack of unified theoretical and terminological frameworks linking traditional and modern medical systems. The systematic incorporation of traditional Chinese cultural philosophy is proposed as a means to clarify conceptual connections and to support metastasis-focused research and clinical practice.
Keywords
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide. According to the GLOBOCAN 2022 database, approximately 2.48 million new cases and 1.82 million deaths were reported globally in 2022, accounting for 12.4% of all newly diagnosed cancers and 18.7% of cancer-related deaths, respectively.1,2 Despite advances in early detection and therapy, survival remains poor, particularly among patients diagnosed with distant-stage disease.
Approximately 70% of patients with non-small cell lung cancer (NSCLC) are diagnosed at locally advanced or metastatic stages, underscoring the central role of metastasis in poor clinical outcomes. 3 Metastatic spread is common in lung cancer, and brain metastases in particular contribute substantially to morbidity due to neurological sequelae. 4 Although targeted therapies and immune checkpoint inhibitors have achieved important clinical advances, therapeutic resistance and limited durability of response remain major challenges. 5 These limitations highlight the need for more comprehensive and individualized strategies to prevent and manage lung cancer metastasis, providing a rationale for exploring integrative approaches beyond conventional modalities.
Integrative approaches that combine Traditional Chinese Medicine (TCM) with Western medicine have received increasing attention as complementary strategies in the comprehensive management of advanced lung cancer. Modern oncology emphasizes precise diagnosis, standardized treatment protocols, and target-specific interventions, whereas TCM emphasizes holistic regulation, dynamic balance, and individualized therapeutic adjustment. Integrating these two paradigms provides a conceptual framework for addressing both local tumor control and systemic dysregulation associated with metastatic progression. 6 Clinical studies and meta-analyses have suggested that integrative treatment strategies may improve symptom control, enhance quality of life, and reduce treatment-related adverse effects in patients with advanced NSCLC, particularly during prolonged systemic therapy for metastatic disease. 7 Emerging evidence also suggests a possible role in delaying disease progression. 7 Nevertheless, the extent to which integrative Chinese–Western medicine influences metastasis-related biological processes and clinically meaningful metastatic outcomes has not been systematically elucidated.
Increasing experimental evidence has begun to clarify the biological basis by which TCM interventions may influence lung cancer metastasis. Active compounds derived from commonly used medicinal herbs, such as astragalus polysaccharides, ginsenoside Rh2, and ligustrazine, have been reported to regulate key processes in the metastatic cascade through diverse molecular targets and signaling pathways. 7 These effects include modulation of epithelial–mesenchymal transition, inhibition of tumor angiogenesis, remodeling of the tumor microenvironment, and enhancement of antitumor immune responses. 8 Through these multilevel regulatory actions, the multitarget and multipathway characteristics of TCM-based interventions may help address limitations of single-target therapies and mitigate therapeutic resistance during metastatic progression in lung cancer.
Traditional Chinese philosophical concepts underpin the theoretical system of TCM and provide a methodological perspective emphasizing holistic regulation, dynamic balance, and preventive intervention. 9 In this review, these principles are discussed in relation to contemporary biomedical frameworks, including systems biology, tumor microenvironment regulation, immune homeostasis, and preventive medicine.6,10 Within this integrative context, Chinese and Western medical approaches offer a conceptual lens for understanding lung cancer metastasis as a multistep, system-level process involving tumor cells, host immunity, and the metastatic microenvironment, thereby strengthening the scientific rationale for integrated intervention strategies.6,11
In recent years, multiple review articles have summarized the role of TCM in lung cancer treatment, including clinical applications, mechanistic evidence, and stage-specific therapeutic strategies throughout lung cancer progression.6,11 While these studies provide valuable insights, they are predominantly treatment-oriented and typically address metastasis as one component among multiple therapeutic outcomes rather than as an independent, multistep biological process.
By contrast, the present review focuses on lung cancer metastasis as a distinct biological and clinical challenge and organizes the discussion according to key stages of the metastatic cascade, such as epithelial-mesenchymal transition, tumor microenvironment remodeling, tumor-immune interactions in metastasis, and pre-metastatic niche formation. This metastasis-centered framework emphasizes mechanistic complementarity between Chinese and Western medicine in regulating metastasis-related pathways rather than reiterating general antitumor effects. Furthermore, traditional Chinese cultural and philosophical concepts are incorporated as an interpretive framework to contextualize metastasis biology and integrative therapeutic strategies, offering a focused perspective that extends beyond existing reviews.
Therefore, this review aims to examine synergistic mechanisms and clinical strategies underlying the integration of Chinese and Western medicine in the prevention and treatment of lung cancer metastasis, drawing on insights from both modern biomedical research and TCM theory. By incorporating perspectives from traditional philosophy in a cautious and mechanism-oriented manner, this review seeks to clarify conceptual connections and provide practical insights for optimizing integrative strategies to prevent and manage lung cancer metastasis.
Biomedical Mechanisms Underlying Lung Cancer Metastasis
From the perspective of traditional Chinese cultural philosophy, disease progression is viewed as a consequence of dynamic imbalance within an interconnected biological system rather than a series of isolated pathological events. This holistic view aligns with contemporary systems oncology, which conceptualizes lung cancer metastasis as an emergent, multistep process shaped by coordinated interactions among tumor cells, host immunity, inflammatory signaling, and the tumor microenvironment. Accordingly, traditional Chinese cultural philosophy is used here as an interpretive lens to contextualize biomedical mechanisms of lung cancer metastasis and to inform the discussion of integrative therapeutic strategies.
Cellular and Molecular Steps in the Metastatic Cascade
Primary Tumor Invasion and Epithelial–Mesenchymal Transition
The metastatic cascade begins with invasion of the basement membrane at the primary tumor site, followed by infiltration into surrounding tissues (Figure 1). Epithelial–mesenchymal transition (EMT) is a key biological process that supports this early step. 12 During EMT, tumor cells downregulate epithelial markers such as E-cadherin and upregulate mesenchymal markers including vimentin and fibronectin, thereby acquiring enhanced migratory and invasive capacities. 13 Reduced intercellular adhesion and cytoskeletal remodeling facilitate detachment from the primary mass and penetration through the extracellular matrix, enabling entry into blood or lymphatic vessels and initiating metastatic dissemination. 14

Overview of the observed complex and concurrent routes of metastasis. Metastasis involves multiple interconnected pathways. EMT is a critical process in cancer metastasis, characterized by the conversion of epithelial cells into mesenchymal cells.
From a systems perspective informed by traditional Chinese cultural philosophy, EMT can be interpreted not as a discrete molecular event but as a dynamic, adaptive process emerging from continuous interactions between tumor cells and their microenvironment.9,12,15 Mechanical stress, inflammatory mediators, hypoxia, and stromal signaling collectively regulate epithelial–mesenchymal plasticity, consistent with an emphasis on contextual responsiveness and dynamic balance rather than fixed cellular states.12,13,15
The reversible and plastic nature of EMT also suggests potential vulnerabilities at the tumor–host interface. This feature supports intervention strategies aimed at modulating inflammatory tone, stromal activation, and adaptive plasticity, conceptually aligning with holistic approaches that prioritize restoration of systemic equilibrium rather than single-target suppression.,6,12,15
Survival and Homing of Circulating Tumor Cells
Following intravasation, tumor cells enter the circulation as Circulating Tumor Cells (CTCs). 16 In the circulatory environment—characterized by shear stress and continuous immune surveillance—CTC survival depends on adaptive protective mechanisms. 17 Experimental evidence indicates that CTCs frequently form aggregates with platelets and neutrophils, creating a protective niche that promotes immune evasion and enhances adhesion to microvascular beds in distant organs. 18
After arrest at distant sites, CTCs extravasate, infiltrate local tissue, and colonize permissive microenvironments, ultimately forming metastatic lesions. 19 During circulation and subsequent colonization, RNA-based regulatory mechanisms, including tRNA-derived small RNAs (tsRNAs) and related modifications, have been implicated in regulating tumor cell migration, invasion, and stress adaptation.20,21
From a traditional philosophical perspective that emphasizes relational and contextual regulation, CTC fate is shaped not only by intrinsic tumor properties but also by ongoing interactions with immune surveillance, inflammatory signaling, and the circulatory microenvironment.18,19 In this context, tsRNAs may be considered candidate regulators of adaptive balance, potentially coordinating stress responses and immune interactions at a systems level rather than acting as isolated oncogenic drivers.
Together, these findings highlight immune surveillance, inflammatory signaling, and the circulatory microenvironment as system-level determinants of CTC survival and organ-specific homing. This view provides a conceptual rationale for integrative strategies that aim to disrupt metastatic progression through systemic regulation rather than isolated molecular targeting.
Formation of the Pre-Metastatic Niche
Prior to overt metastasis, primary tumors can condition distant organs by remodeling local immune and stromal environments, thereby establishing a pre-metastatic niche (PMN) that supports subsequent colonization. 22 PMN formation involves coordinated contributions from bone marrow–derived suppressor cells, tumor-associated fibroblasts, endothelial cells, and diverse immune populations, collectively reshaping immune tone and stromal architecture at distant sites. 23
This phenomenon is conceptually consistent with both traditional Chinese cultural philosophy and systems oncology in emphasizing that disease progression depends on systemic susceptibility rather than solely on localized cellular events.6,9,15 PMN formation illustrates how remote environmental conditioning can precede and enable metastatic colonization, reinforcing the importance of host permissiveness in cancer progression.15,23 From a translational perspective, PMN biology underscores immune tone, stromal responsiveness, and metabolic state as potentially modifiable determinants of metastatic risk, rather than viewing metastasis exclusively as a tumor cell–intrinsic event.15,23,24
Driver Genes and Signaling Pathways in Lung Cancer Metastasis
Oncogenes and Tumor Suppressor Genes (eg, EGFR, TP53)
Lung cancer metastasis is driven by diverse molecular alterations that converge on shared functional programs (Figure 2). 25 EGFR, one of the most frequently mutated genes in NSCLC, promotes tumor proliferation, migration, and resistance to apoptosis through activation of the PI3K–AKT and RAS–RAF–MEK–ERK signaling pathways, thereby enhancing metastatic potential. 26 Conversely, TP53 mutations impair cell-cycle control and DNA damage responses, leading to genomic instability that can facilitate metastatic dissemination. 26

The main classification and therapeutic targets of lung cancer. Lung cancer is primarily divided into two major types: NSCLC and SCLC. Targeted therapies and immunotherapies have significantly improved treatment outcomes for patients with specific genetic alterations or immune biomarkers.
In addition to oncogenic signaling alterations, matrix metalloproteinases such as MMP-9 contribute to metastatic progression by degrading extracellular matrix components and promoting cellular motility. 27 These genetic and molecular changes converge on extracellular matrix remodeling, stress adaptation, and survival signaling, supporting a network-based view of metastasis and underscoring the limitations of single-gene targeting strategies within a systems-oriented framework.
Core Signaling Networks Driving Metastatic Progression
Key pathways—including TGF-β, NF-κB, Wnt/β-catenin, and STAT3—form an interconnected regulatory network that drives metastatic progression. TGF-β has stage-dependent effects, suppressing early tumor growth while promoting EMT, immune suppression, and metastatic colonization at later stages.28,29 NF-κB activation in inflammatory microenvironments upregulates pro-metastatic mediators such as IL-6 and CXCL8, thereby enhancing tumor cell migration and angiogenesis. 29 STAT3 signaling further sustains metastatic competence through effects on cell survival, immune evasion, and stromal reinforcement. 30
These pathways often operate through positive feedback loops and network-level amplification rather than isolated linear cascades. From a systems-oriented perspective, metastatic progression can be viewed as emerging from chronic inflammation, immune suppression, and microenvironmental reinforcement as dominant system-level phenotypes23,28,29
Recognizing the integrated nature of these signaling networks supports the rationale for multitarget, regulatory intervention strategies. Importantly, a network-based framing also provides a practical way to interpret TCM interventions as modulators of functional balance and network resilience rather than as direct molecular antagonists.
TCM Perspectives and Therapeutic Theories in Lung Cancer Metastasis
Traditional cultural philosophy serves as a methodological bridge between TCM theory and modern oncology, framing TCM interventions not as direct molecular equivalents but as regulatory approaches that target functional vulnerabilities of the metastatic system, particularly immune surveillance, inflammatory amplification, and microenvironmental permissiveness.
Classical TCM Concepts Interpreted Through Modern Oncology
Vital Qi Deficiency and Tumor Immune Escape
In TCM theory, vital qi represents a fundamental physiological capacity that supports internal stability and resistance to disease progression. 31 Vital qi deficiency is therefore associated with impaired host defense, creating conditions that may favor tumor initiation and metastatic spread. 11
When interpreted within a modern oncological framework, vital qi deficiency can be related to immunosuppressive phenotypes, including reduced T-cell and natural killer (NK) cell activity and imbalanced CD4+/CD8 + ratios.32,33 Such immune dysfunction is consistent with established mechanisms of tumor immune escape and metastatic progression.
The TCM principle of “supporting the righteous” focuses on strengthening vital qi through tonifying qi and blood and harmonizing organ function, conceptually paralleling immunomodulatory strategies.31,34 Polysaccharides derived from qi-tonifying herbs such as Astragalus membranaceus and Panax ginseng have been reported to enhance T-cell proliferation, increase NK cell cytotoxicity, and modulate cytokine production (eg, IL-2 and IFN-γ), thereby partially restoring antitumor immune surveillance and counteracting immune evasion.35,36
Phlegm-Stasis Syndrome and Tumor-Associated Fibrosis
In TCM, phlegm–stasis interaction describes pathological accumulation and obstruction associated with impaired qi and blood circulation. 37 In modern oncological terms, this pattern can be discussed in relation to tumor-associated fibrosis and stromal stiffening within the tumor microenvironment. 24
Tumor-associated fibrosis is characterized by excessive extracellular matrix deposition, particularly collagen and fibronectin, which increases tissue rigidity and facilitates tumor invasion and metastatic spread.15,24,38 Within this framework, “stasis” may be interpreted as microcirculatory impairment and hypoxia, whereas “phlegm” can be related to chronic inflammation, fluid retention, and metabolic dysregulation. 29 Preclinical studies suggest that processes associated with phlegm and stasis may promote fibroblast activation, extracellular matrix remodeling, angiogenesis, and tumor infiltration, thereby contributing to metastatic progression.23,24,38
Blood-activating and stasis-resolving herbs such as Salvia miltiorrhiza (Danshen) contain bioactive tanshinones, including tanshinone IIA, which have been reported to inhibit TGF-β–mediated fibroblast proliferation and activation by downregulating Smad signaling.39,40 Through modulation of fibroblast activity and stromal responses, these compounds may help remodel a pro-tumorigenic fibrotic microenvironment.39,40
Intervention Strategies in TCM for Inhibiting Lung Cancer Metastasis
Immune Enhancement and Microenvironment Regulation via Vital Qi Reinforcement
Strengthening vital qi is a foundational therapeutic principle in TCM oncology, emphasizing enhancement of host resistance and restoration of systemic homeostasis. 9 In metastatic lung cancer, this strategy is often discussed in terms of improving antitumor immune function, regulating inflammation, and influencing the tumor microenvironment. 11
Active components from qi-tonifying herbs, including polysaccharides derived from Astragalus membranaceus and Ganoderma lucidum, have been reported to enhance CD4⁺T-cell and NK-cell activity, upregulate IL-2 and IFN-γ, and downregulate immunosuppressive mediators such as TGF-β and IL-10. 41 These coordinated immunological effects may contribute to immune rebalancing and reduced metastatic permissiveness.
Although the primary role of qi-reinforcing therapies is immunomodulation, certain herbs have also been reported to exhibit additional antitumor activities, including induction of apoptosis and modulation of metastasis-related signaling pathways such as STAT3. 33 These effects are consistent with a regulatory, system-level approach rather than serving as substitutes for conventional cytotoxic therapies.
Targeting CTCs and Angiogenesis Through Blood-Activating Therapies
Blood-activating compounds such as tetrandrine and evodiamine have been reported to regulate inflammatory signaling and immune checkpoint–related pathways, potentially influencing tumor–vascular interactions and metastatic dissemination.42,43 In addition, multiple TCM-derived bioactive compounds have been reported to inhibit metastasis-related mediators, including matrix metalloproteinases (MMPs), VEGF-associated angiogenic signaling, and hypoxia-related pathways involving HIF-1α.28,44,45 These actions may suppress extracellular matrix degradation and angiogenesis, providing multilevel interference with metastatic progression.
Clinical data also suggest potential translational relevance. Randomized clinical studies indicate that adding Chinese medicine to standard systemic therapy for advanced NSCLC may improve patient-reported outcomes and may reduce progression risk in certain settings.44,46,47
Taken together, blood-activating therapies exemplify a systems-oriented strategy that may influence circulating tumor cell dynamics and angiogenic support, consistent with an emphasis on coordinated regulation of circulatory function and microenvironmental balance within integrative oncology (Figure 3).
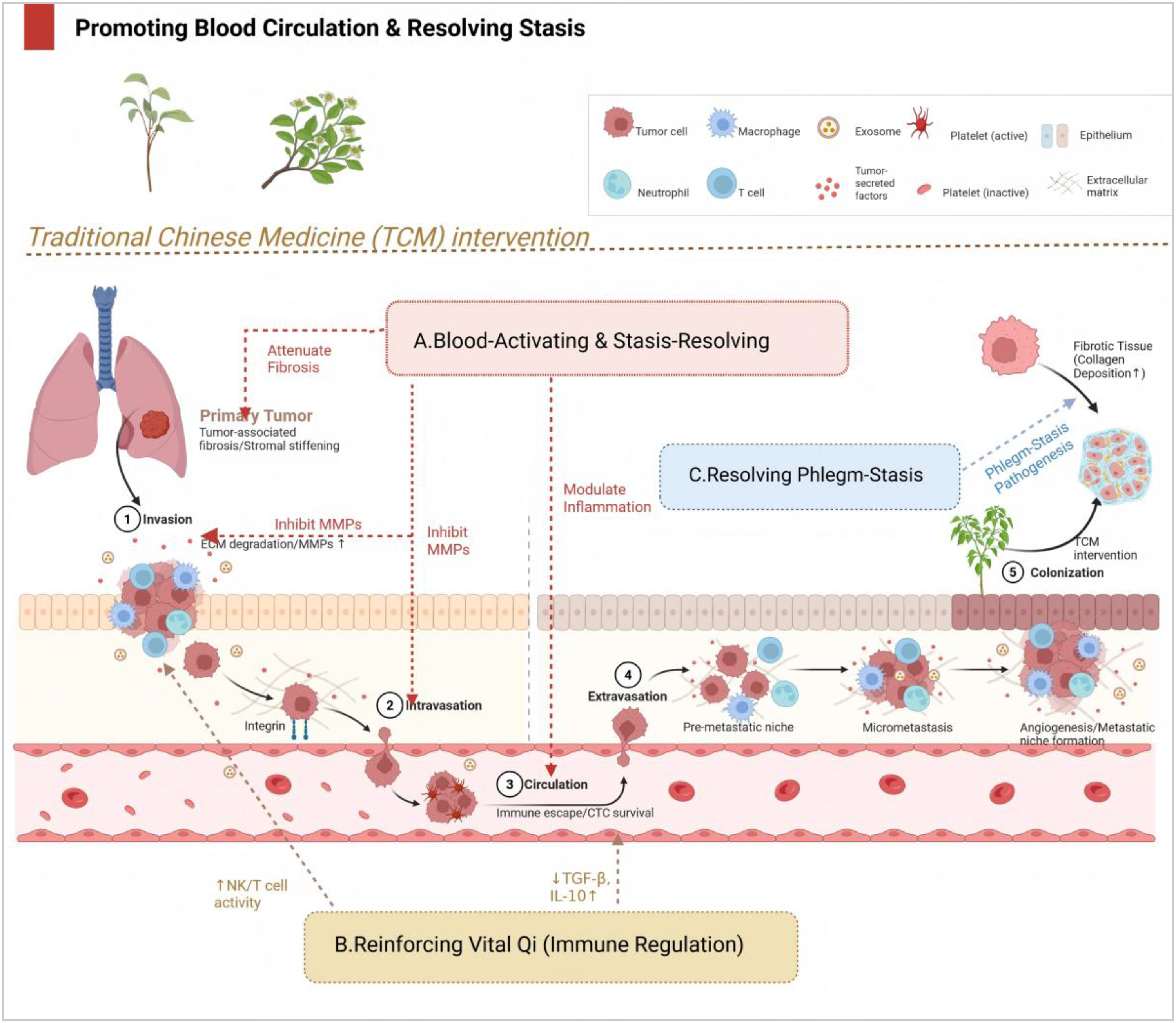
Schematic overview of TCM intervention strategies targeting the tumor microenvironment and metastatic cascade in lung cancer. The diagram illustrates how the TCM principles of Reinforcing Vital Qi and Activating Blood & Resolving Stasis systemically modulate the tumor microenvironment and key steps of the metastatic cascade.
Multi-Herb Formulations as System-Level Interventions in Lung Cancer Metastasis
Unlike reductionist pharmacological approaches that emphasize single agents or isolated bioactive compounds, TCM is commonly practiced using multi-herb formulations that function as integrated therapeutic systems. 31 In lung cancer metastasis, multi-herb formulations are often framed as holistic interventions intended to coordinate functional regulation at the organism level, supporting internal balance and systemic resilience. 48
From a systems perspective, the therapeutic logic of multi-herb formulations lies in functional differentiation and synergistic interactions among constituent herbs, enabling simultaneous regulation of interconnected processes such as epithelial–mesenchymal plasticity, immune surveillance, inflammatory and fibrotic stromal responses, and treatment-related toxicity. 6 This coordinated regulation may allow interference with the metastatic cascade across multiple stages, reducing reliance on single-target inhibition.
Systems-oriented analyses have also been applied to formulation-based strategies in cancer immunotherapy, highlighting multi-target regulation and pathway-level coordination, particularly in immune-related processes. 49 Such network-wide modulation may enhance system stability and limit compensatory activation of alternative pro-metastatic pathways.
Beyond tumor-directed effects, multi-herb formulations may contribute to host-centered regulation by buffering excessive inflammation and supporting immune homeostasis. This property may be relevant in metastatic lung cancer, where disease progression, treatment-related toxicity, and immune exhaustion can coexist.6,11,33 Rather than being viewed solely as direct cytotoxic agents, these formulations can be conceptualized as modulators of tumor–host system dynamics. 50
Although much of the current evidence derives from preclinical and integrative mechanistic studies, systems-level analyses support evaluating TCM interventions at the formula level. 10 Future work integrating multi-omics profiling, network modeling, and experimental validation will be important for translating these systems-level insights into rational integrative treatment strategies.
Synergistic Mechanisms of Integrated Chinese and Western Therapies
From an integrative perspective, synergy between Chinese and Western medicine arises from complementary reductionist and holistic approaches rather than simple co-administration. Western medicine primarily targets discrete molecular drivers of metastasis, whereas TCM emphasizes systemic resilience, adaptive regulation, and restoration of homeostasis. Traditional cultural philosophy is used here as a unifying lens that links the mechanistic discussion in Section 2 with the system-level therapeutic strategies outlined in Section 3.
Chinese Herbal and Non-Pharmacological Modulation of Therapeutic Response
Reversing Drug Resistance: Bufalin as a Chemosensitizer
Chinese herbal medicines have been investigated for their potential to modulate drug resistance and enhance responses to chemotherapy and targeted therapies in lung cancer. 51 Among bioactive compounds, bufalin has attracted attention as a candidate chemosensitizer with multiple proposed mechanisms. 52
Drug resistance remains a major contributor to treatment failure across cancers and has motivated efforts to identify agents that restore drug sensitivity. 53 Preclinical evidence suggests that bufalin may mitigate resistance through modulation of resistance-associated pathways, including PI3 K/AKT signaling, thereby partially restoring tumor cell sensitivity to cytotoxic agents. 52
Available clinical evidence—largely from regional studies—suggests that adding TCM interventions to chemotherapy regimens may improve therapeutic response, reduce adverse effects (including myelosuppression and gastrointestinal toxicity), and improve patient-reported quality-of-life outcomes in advanced lung cancer.44,46,54 Nevertheless, validation in rigorously designed multicenter clinical trials remains necessary.
In addition to bufadienolides, ginsenoside Rg3 has demonstrated chemosensitizing effects in experimental models, including attenuation of resistance to targeted therapy in NSCLC. 55 For example, ginsenoside Rg3 has been reported to enhance the efficacy of EGFR tyrosine kinase inhibitors (EGFR-TKIs) in EGFR-mutant lung cancer, in part by promoting tumor cell apoptosis and attenuating resistance-associated phenotypes.56,57 Astragalus polysaccharides may further contribute by modulating the immune microenvironment and interfering with pre-metastatic niche formation through pathways such as S1PR1–STAT3. 58
Together, these findings illustrate how TCM-derived interventions may complement Western anticancer therapies by influencing resistance-related pathways and host responses, consistent with a homeostasis-oriented and patient-centered integrative oncology framework (Figure 4).

Synergistic mechanism of integrated Chinese and western medicine in lung cancer therapy: targeting both tumor and host. The schematic depicts the complementary actions of Western medicine (targeted attack on tumor cells) and TCM (systemic regulation of the host).
Acupuncture and the Neuroimmune Axis: Inflammation Control in Metastatic Settings
Acupuncture has been investigated as a non-pharmacological intervention that may modulate the neuroimmune axis, including vagus nerve–mediated regulation of inflammatory responses. 59 The vagus nerve provides bidirectional communication between the central nervous system and peripheral immunity and contributes to immune homeostasis and systemic inflammatory control. 59
Experimental work indicates that electroacupuncture at specific acupoints can engage defined neuroanatomical circuits, including vagus-related pathways such as the vagal–adrenal axis, providing a mechanistic basis for systemic immunomodulation. 60 Through these mechanisms, acupuncture may influence the tumor-associated inflammatory microenvironment.
Animal studies further suggest that electroacupuncture can reduce systemic inflammatory responses and immune cell infiltration, potentially attenuating tumor-promoting inflammatory cascades. 59 IIn addition, acupuncture has been reported to affect the hypothalamic–pituitary–adrenal (HPA) axis, reducing stress responses, improving treatment tolerability, and indirectly modulating pathways associated with tumor progression and dissemination. 61
Taken together, these findings support a neuroimmune basis for the adjunctive role of acupuncture in lung cancer care, positioning it as a system-level regulatory intervention that complements tumor-directed therapies rather than functioning as a direct anticancer modality.
Clinical Integration Models of TCM and Western Medicine
Supportive TCM Interventions in Perioperative and Chemoradiotherapy Phases
During surgical treatment and chemoradiotherapy for lung cancer, TCM is commonly used as supportive care to mitigate treatment-related adverse effects and facilitate postoperative and post-therapy recovery. 6
Postoperative patients may experience symptoms described in TCM as qi and blood deficiency and systemic depletion. Accordingly, therapies that tonify qi and blood, regulate organ function, and promote circulation are used to support immune recovery, facilitate wound healing, and reduce postoperative complications. 62 Formulations containing Astragalus membranaceus, Angelica sinensis, and Salvia miltiorrhiza are widely used and have reported antioxidant and anti-inflammatory activities.6,11,36,39,58
During chemoradiotherapy, integrative TCM interventions have been reported to alleviate treatment-related adverse effects through multi-target regulatory actions, including modulation of immune function, hematopoiesis, and organ protection. 46 Meta-analyses and clinical studies suggest that combining TCM with Western medicine is associated with reduced incidences of leukopenia, gastrointestinal toxicity, and hepatorenal impairment, as well as improved chemotherapy completion rates.11,46,63
The Assessment–Support–Control–Protection (ASCP) framework proposed by the Chinese Anti-Cancer Association provides a structured model for integrative lung cancer management. 46 It emphasizes individualized planning that balances tumor control with preservation of physiological function and treatment tolerability. 63
Maintenance Therapy Based on Simultaneous Strengthening and Clearance
In the maintenance phase of lung cancer management, primary lesions may be controlled, yet microscopic metastases or residual tumor-initiating cells can persist. 64 Patients at this stage may continue to exhibit immune dysfunction and systemic depletion, reflecting ongoing vulnerability to disease progression. 65
Integrative maintenance therapy in TCM is commonly guided by the principle of “simultaneous reinforcement and elimination,” aiming to restore host resilience while reducing the risk of recurrence or progression; this approach has been supported by real-world evidence from postoperative NSCLC cohorts. 66 In postoperative maintenance, Fu-Zheng–focused prescriptions often incorporate qi-tonifying herbs such as Codonopsis, Atractylodes, and Poria, and may be supplemented with heat-clearing/detoxifying components according to syndrome differentiation. 66
Several studies and meta-analyses suggest that Chinese herbal medicine as an adjunct to EGFR-TKIs is associated with improved progression-free survival and other survival-related outcomes, along with improved performance status and quality-of-life measures in patients with advanced NSCLC. 53 These approaches emphasize long-term disease control, treatment tolerability, and immune restoration, particularly in settings where resistance to conventional therapies has emerged.
Maintenance-oriented TCM management is commonly guided by principles of holism, syndrome differentiation, and dynamic balance. Therapeutic goals include disease control, symptom alleviation, and functional support, including management of treatment-related comorbidities. 11 Tailored interventions during maintenance are individualized according to patient-specific syndromes, clinical manifestations, and disease status, reflecting an emphasis on context-sensitive and personalized care. 47
Comparative Clinical Evidence of TCM Plus Western Medicine Versus Western Medicine Alone
Although large-scale international randomized controlled trials that directly compare integrated TCM plus Western medicine with Western medicine alone remain limited, real-world comparative studies from East Asia have begun to examine the potential added value of incorporating TCM into standard oncology care. 67
Across these studies, integrative strategies have been associated with improved treatment tolerability, enhanced symptom control, better quality-of-life outcomes, and reduced treatment-related adverse effects, including myelosuppression, gastrointestinal toxicity, and fatigue. 46 Notably, most comparative trials emphasize functional and supportive endpoints—such as performance status, immune indicators, and adverse-event profiles—rather than overall survival, reflecting the adjunctive role of TCM in metastatic disease settings.46,53 These outcomes may be particularly relevant in metastatic lung cancer, where long-term treatment continuity and preserved functional status can influence prognosis.
Meta-analyses of available comparative studies are broadly consistent with these findings but also highlight substantial heterogeneity in study design, herbal formulations, treatment duration, and outcome measures.46,53 Sample sizes in most head-to-head comparisons remain modest, and reported survival benefits are variable or inconclusive, supporting cautious interpretation. 53 Beyond survival-related endpoints, the literature also describes potential supportive and immunoregulatory effects of TCM, including modulation of antitumor immunity and the tumor immune microenvironment. 54
Overall, the existing head-to-head evidence suggests that TCM combined with Western medicine may provide complementary benefits relative to Western medicine alone, particularly in treatment tolerability and patient-centered outcomes, rather than serving as a substitute for standard oncological therapies. This evidence profile is consistent with a systems-oriented integrative oncology approach that emphasizes coordinated regulation, host resilience, and quality of life alongside disease control.
Modern Scientific Re-Interpretation of Classical TCM Philosophy as a Systems Framework for Integrative Oncology
This section reframes classical TCM philosophy as a conceptual scaffold for hypothesis generation and research design in lung cancer metastasis. Rather than introducing new therapeutic claims, the goal is to clarify the internal logic of the strategies discussed above within a systems-thinking framework, rather than to introduce new therapeutic claims.
Importantly, TCM philosophy is not used here to infer direct molecular mechanisms. Instead, it is applied as a methodological lens that emphasizes dynamic balance, multi-component interactions, and individualized regulation. In this sense, it functions as a heuristic framework for organizing biological complexity and informing research design rather than as a causal explanatory model.
Philosophical-Medical Analogies Between TCM and Modern Immuno-Oncology
Yin–Yang Balance and the Dynamic Regulation of Immune Checkpoints
n TCM philosophy, Yin–Yang balance describes a dynamic equilibrium between opposing yet complementary forces that sustain physiological stability. 68 When discussed in relation to modern immuno-oncology, this concept can serve as a metaphor for immune homeostasis and its dysregulation during tumor progression. 65
Immune checkpoints such as cytotoxic T-lymphocyte–associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1), and its ligand PD-L1 play central roles in regulating immune activation and tolerance. 69 In lung cancer, tumor cells can exploit the PD-1/PD-L1 axis—including via exosomal PD-L1—to suppress antitumor T-cell responses and promote immune evasion. 70 From a Yin–Yang perspective, this immunosuppressive state can be interpreted as a shift toward regulatory predominance, rather than as a direct correspondence between philosophical categories and molecular events.71,72
Immune checkpoint inhibitors restore antitumor immunity by disrupting PD-1/PD-L1 or CTLA-4 signaling, but their use is often accompanied by immune-related adverse events (irAEs), reflecting excessive immune activation. 69 This clinical trade-off conceptually parallels the Yin–Yang idea that deviation toward one extreme may generate a secondary imbalance. Accordingly, Yin–Yang balance offers a treatment logic that emphasizes bidirectional immune regulation: enhancing antitumor efficacy while preserving systemic tolerance. 72
The TCM principle of syndrome differentiation and individualized treatment resonates with efforts to personalize immunotherapy based on immune phenotypes and treatment tolerability. 73 Rather than treating constitution-based interventions as established clinical practice, this framework may inform future investigations into symptom-guided and immune-guided integrative strategies—such as supportive herbal therapy, acupuncture, or mind–body interventions—to improve adherence and maintain immune balance during immunotherapy.69,74
Within this interpretive framework, Yin–Yang balance is not regarded as a molecular target but as an organizing principle. This perspective provides a conceptual rationale for integrative strategies discussed earlier in this review, such as combining chemosensitization with toxicity mitigation (Section 4.1), with the aim of maintaining therapeutic intensity without compromising systemic resilience.
Five Elements Theory as a Systems Metaphor for the Tumor Ecosystem
The Five Elements (Wu Xing) theory describes systemic interactions through principles of mutual generation (Sheng) and restriction (Ke), as well as imbalance-related phenomena such as overacting (Cheng) and rebellion (Wu).6,75 When adopted as a systems metaphor, this framework offers a way to discuss the multicellular dynamics of the tumor microenvironment.
In lung cancer, tumor cells, immune cells, stromal components, and the extracellular matrix form an interconnected ecosystem governed by feedback regulation and adaptive remodeling. Tumor-derived cytokines such as TGF-β and IL-10 promote recruitment of immunosuppressive populations, including regulatory T cells and myeloid-derived suppressor cells, thereby reinforcing immune evasion. 74 Conversely, cytotoxic T lymphocytes and natural killer cells exert counterbalancing antitumor effects. These reciprocal interactions can be interpreted through Sheng and Ke as reinforcing and restraining forces within a dynamic system. 65
Stromal remodeling further contributes to metastatic progression. Cancer-associated fibroblasts secrete collagen and matrix metalloproteinases, facilitating extracellular matrix remodeling and invasion, 76 while angiogenesis supplies nutrients and oxygen that support tumor expansion. Under hypoxic conditions, activation of hypoxia-inducible factor 1α (HIF-1α) promotes invasiveness and immune suppression, contributing to self-reinforcing pathological cycles. 45 These processes resemble Five Elements descriptions of excessive domination and loss of regulatory balance.
In this interpretation, the Five Elements theory is not intended to substitute for molecular causality. Rather, it functions as a methodological framework that emphasizes interdependence, feedback regulation, and balance across functional subsystems. This perspective aligns with modern systems biology and network pharmacology approaches, 37 and it provides conceptual support for multi-target and network-based interventions in metastatic and maintenance settings. 77
From Philosophical Heuristics to Testable Research Design
To maintain scientific rigor, philosophical principles such as Yin–Yang balance and Five Elements theory should be translated into testable research frameworks by operationalizing them as design logics rather than explanatory mechanisms. This approach preserves conceptual value while maintaining methodological clarity.
For example, Yin–Yang balance can be reframed as a model of dynamic immune regulation in which antitumor immune activation and immune tolerance are assessed longitudinally using immune phenotyping and inflammatory biomarkers. 78 On this basis, adaptive supportive interventions may be introduced to maintain immune efficacy while limiting toxicity, providing a systems-oriented complement to reactive immunosuppression alone.
Similarly, Sheng–Ke logic can inform network-guided formulation design by conceptualizing the tumor ecosystem as an interacting network of functional modules, including tumor survival, immune suppression, stromal remodeling, angiogenesis, and metabolic regulation. 6 Network pharmacology and systems modeling can then be applied to identify intervention combinations that disrupt pro-metastatic feedback loops while preserving host resilience. 51
From a translational perspective, these frameworks are best implemented through a stepwise and iterative integration pathway rather than fixed decision algorithms. This process may include baseline assessment of disease burden and host condition, identification of dominant system-level vulnerabilities, selective introduction of integrative supportive interventions alongside standard Western therapies, and longitudinal reassessment of clinical response, toxicity, and biomarker dynamics to guide modification or discontinuation.1,6,51,79
This assessment–intervention–monitoring–adjustment cycle aligns with routine oncology practice while avoiding premature reliance on rigid algorithms. By conceptualizing integrative medicine as an adaptive regulatory process rather than a static treatment rule, this approach offers a practical pathway for clinical integration and establishes a foundation for future prospective validation and guideline development.
Implications for Prevention and Supportive Care: A Conceptual Outlook
Beyond therapeutic intervention, traditional philosophical concepts also offer a systems-oriented perspective on cancer prevention and supportive care. The TCM principle of “treating disease before it arises” (Zhi Wei Bing) emphasizes early risk identification, timely intervention, and prevention of disease progression.6,79 This preventive orientation is conceptually aligned with primary, secondary, and tertiary prevention strategies in oncology.
In lung cancer, integrative approaches may support risk stratification, symptom-guided monitoring, and maintenance of long-term resilience during micrometastatic or post-treatment phases.6,51 Emerging technologies such as low-dose computed tomography, liquid biopsy, and AI-assisted risk assessment may provide opportunities to integrate biomedical monitoring with health-centered preventive frameworks. 80 TCM constitution assessment and symptom-pattern analysis have been explored as complementary tools for characterizing patient vulnerability and treatment tolerability. 78
TCM psychosocial approaches, often referred to as Qingzhi therapy, emphasize emotional regulation and mind–body balance as components of comprehensive care. 6 Lung cancer patients commonly experience psychological stress, which may influence disease trajectories through neuroendocrine and immune pathways. 81 From a systems perspective, psychosocial interventions—including acupuncture, Tai Chi, Qigong, and meditation—may support treatment adherence and physiological stability by modulating stress responses and inflammatory tone.82,83
These perspectives are presented as conceptual guides rather than evidence-based mandates. While some studies suggest potential benefits for quality of life and symptom management, further rigorous investigation integrating psychosocial metrics, immune biomarkers, and patient-reported outcomes is needed to clarify their role within integrative oncology.75,82,84
Conceptual Summary of the Integrative Systems Framework
Taken together, this re-interpretation positions classical TCM philosophy not as an alternative explanatory system but as a systems-thinking framework that complements modern oncology by emphasizing relational causality, adaptive regulation, and individualized care. By functioning as a methodological scaffold rather than a mechanistic substitute, traditional philosophy can help organize complexity, motivate integrative hypotheses, and guide the design of multi-level intervention strategies in lung cancer metastasis.68,71,84
Importantly, this framework does not advocate substituting evidence-based oncological interventions, nor does it assign direct antimetastatic causality to philosophical constructs or supportive modalities. Instead, it provides a conceptual structure for integrating mechanistic evidence, clinical strategy, and supportive regulation within a unified research logic.6,78,84
Accordingly, Sections 2 to 4 illustrate integrative strategies at mechanistic and clinical levels, while Section 5 clarifies the philosophical logic that ensures their conceptual coherence, reinforcing traditional cultural philosophy as a bridging framework that supports but does not replace evidence based integrative oncology research (Figure 5).
A conceptual model of TCM in cancer therapy. According to TCM theories, cancer is caused by an imbalance of Yin-Yang at both holistic and local levels. TCM aims to rebalance Yin-Yang, enhance body resistance, eliminate pathogens, activate T cells in the TME, and destroy cancer cells to achieve disease remission.
Toward Integrative Innovation: Challenges and Future Directions
Although integrative approaches combining TCM and Western oncology have shown promise in lung cancer metastasis, broader adoption remains constrained by conceptual, methodological, and translational challenges. Addressing these limitations is important for moving integrative oncology beyond empirical application toward a rigorous and globally communicable research and clinical framework.
Notably, many TCM-related studies cited in this review are limited by small sample sizes, heterogeneous designs, inadequate blinding or randomization, and potential selection and reporting bias, which restrict causal inference.63,85,86 These limitations highlight the need for well-designed, adequately powered, and transparently reported clinical trials to strengthen the evidence base for integrative oncology.
Conceptual and Terminological Translation as a Foundation for Integration
A fundamental barrier to deeper integration between TCM and Western medicine lies in their distinct conceptual and terminological systems. 87 TCM terminology derives from classical philosophy and cumulative clinical experience and emphasizes holism, relational reasoning, and functional pattern recognition, whereas Western oncology relies on standardized, quantifiable descriptors grounded in pathology, molecular signaling, and pharmacology. 6 As a result, core TCM concepts such as qi, syndromes, and phlegm and blood stasis do not translate directly into anatomy-based and molecule-based biomedical language. 88
This gap is particularly evident in integrative lung cancer research, where TCM descriptions of metastatic susceptibility—such as deficiency of righteous qi or phlegm and blood stasis interlocking—coexist with biomedical constructs including epithelial–mesenchymal transition, immune evasion, and tumor microenvironment remodeling.6,85,88 Although prior studies have attempted to relate TCM concepts to biomedical correlates (eg, linking righteous qi with immune competence or phlegm and blood stasis with stromal fibrosis and inflammation), these associations remain largely exploratory, context-dependent, and insufficiently standardized. 88
Future progress will require structured strategies for conceptual translation rather than literal equivalence. Approaches such as ontology-based terminology mapping, interdisciplinary consensus building, and explicit separation between philosophical descriptors and clinical endpoints may provide a more rigorous foundation for communication across paradigms. 88 Within this framework, classical concepts such as Yin–Yang and the Five Elements are best treated as organizing principles for hypothesis generation and system-level reasoning rather than as direct mechanistic or clinical labels.
Systems-Oriented and Interdisciplinary Research Methodologies
Advances in systems biology, network pharmacology, and computational modeling have created opportunities to investigate integrative oncology within empirically testable research frameworks. 89 Moving beyond single-target paradigms, network-based approaches allow multi-component interventions (such as Chinese herbal formulations) to be evaluated in terms of pathway interactions, feedback regulation, and system-level effects. 86
In lung cancer metastasis research, network pharmacology has been used to examine how multi-herb formulations may concurrently modulate signaling pathways involved in epithelial–mesenchymal transition, immune regulation, angiogenesis, and stromal remodeling. 86 More broadly, integration of multi-omics technologies with computational analytics has enabled predictive models that combine molecular data with functional and pattern-based clinical descriptors, including TCM syndrome differentiation.73,88
Future research could prioritize several directions. First, single-cell sequencing and spatial-omics approaches may help identify cellular and microenvironmental targets of Chinese herbal medicine within metastatic niches. 90 Second, predictive models of lung cancer metastasis could be developed that integrate TCM diagnostic indicators—such as tongue and pulse characteristics—with molecular and imaging biomarkers. 91 Third, Digital Twin technologies may be applied to simulate outcomes under different integrative treatment strategies, supporting personalized decision-making and prospective evaluation of therapeutic responses.32,92 Notably, many of these interdisciplinary methodologies operationalize systems-thinking principles in quantifiable and experimentally testable designs.71,73,92,93
Safety, Adaptation, and System-Level Constraints in Integrative Therapy
Despite growing interest in integrative oncology, system-level constraints must be addressed to ensure safe and responsible clinical application. One consideration is biological adaptation during long-term TCM use. 94 Unlike genetic resistance to single-target agents, reduced responsiveness to TCM interventions may be better understood as system-level adaptation, potentially involving immune rebalancing, metabolic compensation, or microbiome-mediated effects.63,88,95 These dynamics underscore the value of longitudinal monitoring and adaptive treatment adjustment.
Toxicological risk and herb–drug interactions represent an additional challenge.8,11,88 Although TCM is often considered relatively safe, certain herbal components may exhibit dose- or duration-dependent toxicity, and some may modulate drug-metabolizing enzymes or transporters, thereby altering the pharmacokinetics or pharmacodynamics of Western anticancer agents.7,37,53 Such interactions may affect both efficacy and safety, particularly in metastatic lung cancer patients receiving complex multimodal regimens.
Beyond biological considerations, economic and implementation constraints also merit attention. 96 While integrative approaches may reduce indirect healthcare burdens by improving treatment tolerability and adherence, they may introduce additional costs related to prolonged therapy, individualized formulations, and safety surveillance.97,98 Accordingly, future studies should incorporate systematic toxicological assessment, pharmacokinetic–pharmacodynamic modeling, and health-economic endpoints to support sustainable and scalable clinical translation. 7
Regulatory and Translational Pathways for Global Integration
Global integration of TCM into oncology practice faces substantial regulatory challenges. Multicomponent formulations, batch variability, and syndrome-based indications do not readily align with conventional regulatory frameworks used by agencies such as the FDA or EMA.11,99 As a result, conceptual and methodological translation—rather than direct transplantation of traditional practices—may represent a more feasible pathway for global integration. 87
Potential strategies include prioritizing standardized extracts or well-characterized formulations, establishing robust quality control and pharmacokinetic profiles, and positioning TCM interventions primarily as adjunctive or supportive therapies targeting treatment tolerability, immune modulation, and quality of life.8,73,85 Systems biology and network pharmacology approaches may also facilitate regulatory evaluation by providing pathway- and network-level evidence that supports biological plausibility.86,89
Unlike single-target anticancer agents, TCM interventions are unlikely to induce classical genetic resistance driven by target mutation or pathway bypass.53,86 However, system-level adaptive responses may emerge during long-term or repeated use, including immune rebalancing, metabolic compensation, or attenuation of regulatory effects.65,100 These dynamics may be better framed as system adaptation rather than pharmacological resistance, reinforcing the need for longitudinal monitoring, adaptive adjustment, and periodic strategy recalibration rather than fixed intervention paradigms.32,65,72
Ultimately, progress will depend on international collaboration among clinicians, basic scientists, and regulatory experts.32,99 Stepwise, indication-specific, and evidence-driven translational strategies may allow selected integrative interventions to be responsibly incorporated into global oncology practice while balancing scientific rigor with therapeutic complexity.25,73,85
Taken together, the challenges and future directions outlined above indicate that integrative innovation in lung cancer metastasis depends not only on therapeutic efficacy but also on conceptual clarity, methodological rigor, safety assurance, and translational feasibility. Addressing these dimensions within a systems-oriented framework may allow integrative oncology to evolve from parallel coexistence toward coherent, evidence-informed collaboration between traditional philosophy and modern cancer science.
Conclusion
Integrative Oncology as a Complementary Strategy in Metastatic Lung Cancer
This review synthesizes current evidence and conceptual perspectives on integrative oncology—combining TCM with Western medicine—in the management of lung cancer metastasis. Rather than serving as an alternative to standard oncological care, integrative approaches are best viewed as complementary strategies that may improve treatment tolerability, symptom control, immune regulation, and quality of life.
Clinical and translational studies reviewed here suggest that selected TCM interventions, used alongside chemotherapy, targeted therapy, or immunotherapy, are associated with improved treatment adherence, reduced therapy-related toxicity, and better functional outcomes. From a metastasis-oriented perspective, integrative oncology pairs tumor-directed interventions with system-level regulation to support therapeutic continuity and long-term disease management without replacing evidence-based cancer treatments. In practice, the potential clinical value of integrative strategies lies in improving tolerability and continuity of standard therapy—factors that strongly influence real-world outcomes in metastatic lung cancer.
From Experience to Evidence: Cultural Paradigm Shift in Integrative Oncology
The development of integrative oncology reflects a methodological shift from experience-based practice toward evidence-informed, systems-oriented research. TCM is rooted in empirical observation and philosophical reasoning and emphasizes holism, dynamic balance, and individual variation, whereas Western medicine relies on anatomical, molecular, and evidence-based frameworks. Integrative oncology therefore advances through translation and alignment of concepts rather than direct equivalence.
Advances in systems biology, network pharmacology, and computational modeling provide tools to examine classical TCM concepts—such as syndrome differentiation and multi-component regulation—within testable and communicable research designs. Importantly, this approach does not require reducing traditional philosophy to molecular mechanisms. Instead, it reframes traditional concepts as methodological resources that can inform hypothesis generation, study design, and integrative clinical reasoning under modern biomedical standards.
Integrating TCM and Western Medicine in Cancer Care: A Concluding Perspective
Integrating TCM and Western medicine offers a systems-oriented perspective for addressing the biological and clinical complexity of lung cancer metastasis. The value of this integration lies not only in specific interventions but also in the conceptual framework used to interpret multilevel interactions among tumor biology, host responses, and treatment effects.
By positioning traditional philosophical reasoning as a methodological bridge rather than a mechanistic substitute, integrative oncology supports evidence-informed collaboration between traditional medical knowledge and modern cancer science. This perspective may help guide future work toward clearer conceptual translation, stronger clinical evidence, and responsible implementation in metastatic lung cancer care.
Footnotes
Acknowledgements
We thank BioRender for providing the graphical tools used to create the figures in this work. Figures were created with BioRender.com.
Author Contributions
Rui Sheng: Conceptualization, literature search, writing of the original draft. Xiuxiu Qiu: Review, editing the manuscript. Zhanxia Zhang: Resources, curation, supervision. Each author contributed significantly to the paper and consented to the submitted version.
Funding
This work was supported by Anhui Provincial Universities’ Humanities and Social Science Major Project (2025AHGXSK20036), Fuyang Normal University's Doctoral Startup Fund (2021KYQD0005) for Turkish foreign policy research, Anhui's Ideological and Political Education Key Project (2022sxzz064), Shanghai Biomedical Science and Technology Support Project (22S11900800), Natural Science Foundation (20ZR1459100).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
No data was used for the research described in the article.
