Abstract
Objectives
Characterize lignin-derived compounds from the hurd of 10 fiber/grain Cannabis sativa L. cultivars and investigate the antimicrobial activity of select compounds on the wasteful ruminant bacterium, Acetoanaerobium sticklandii SR.
Methods
Hurd was processed using an aqueous ethanol solvent system and analyzed via GC-MS to identify and characterize lignin derivatives. Antimicrobial activity of the hurd and three monomers was assessed using solid (hurd) and liquid medium (monomers) bioassays against SR.
Results
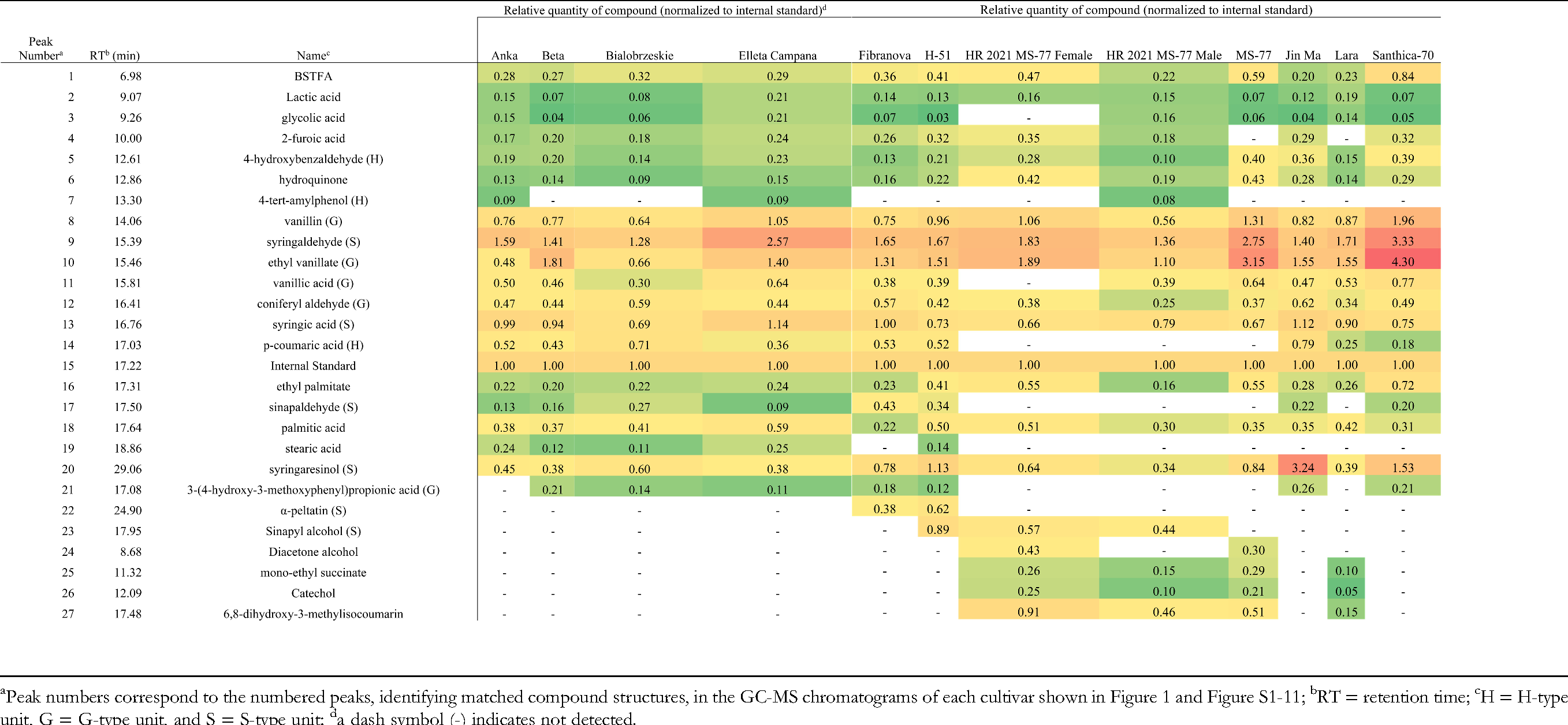
27 compounds were identified, most as monomers but a few were dimers, ethylated derivatives, and organic acids. There were approximately equal G- and S-units with substantially fewer H-units. Several derivatives were present across cultivars, but their abundance appeared varied. Generally, syringaldehyde, vanillin, and syringic acid were most prominent, whereas vanillic acid and p-coumaric acid were less so, and syringaresinol was largely variable. Antimicrobial activity of ground hurd, as well as vanillin, vanillic acid, and p-coumaric acid were tested against SR. Only vanillin exhibited antimicrobial activity (1 mg mL−1) under the testing conditions.
Conclusions
The current work primarily contributes to the characterization and identification of lignin-derivatives in hemp hurd across fiber/grain cultivars with secondary contributions identifying hemp hurd as a potential source of plant-derived antimicrobials for amino-acid fermenting bacteria.
This is a visual representation of the abstract.
Introduction
Cannabis sativa L. (hemp) is an interesting and versatile plant with a rich history across the globe. Today it's grown primarily for the production of four products: fiber, seed, cannabidiol (CBD), and the bioactive substances from the seeds, seed cakes, and leaves. 1 Historically, hemp was cultivated for textile work primarily utilizing the bast fibers (long cellulose-rich phloem fibers; 20-40 wt%) while the hurd (inner woody core; 60-80 wt%) was considered a waste product and used for animal bedding or simply incinerated.2,3 However, new uses and markets for fiber (bast and hurd: abundant in hemp stalks) products are being discovered, e.g., construction materials and biofuels. 2 In 2024, for example, 45,294 total acres of industrial hemp were grown in the United States with 18,855 acres (60.4 million pounds) cultivated for fiber, specifically, according to the National Hemp Report published in April 2025. 4 However, hemp hurds are still largely considered waste products today thereby providing an opportunity to create further value.
Hemp has many bioactive molecules such as the terpenes, phenolics, and cannabinoids all of which are known to be localized largely to the flowers, leaves, and seeds (some phenolics). The antimicrobial capabilities of each of these classes of compounds have been explored against many Gram (+) and (-) bacteria, often times pathogenic. For example, Blaskovich et al reported the antimicrobial properties of CBD on many common pathogens, which revealed activity comparable to existing antibiotics. 5 While the cannabinoids (e.g., CBD) are largely relegated to Cannabis sp., 6 the phenolics and terpenes present are common to many plants of different genera – e.g., limonene, humulene, and bisabolol – and their antimicrobial activity well documented as a result.7,8 Ergo, hemp flowers, leaves, and seeds are well understood to be valuable sources of novel plant-derived antimicrobials and the efficacy of which has been investigated for many microbes of interest. However, a significant portion of the above-ground biomass of hemp has been left under-explored. Lignin, found extensively in woody tissues, is a complex biopolymer, some derivatives of which may possess antimicrobial activity. 9 While lignin structures themselves can be quite diverse, they generally originate from three common lignin monomers: coniferyl (guaiacyl), sinapyl, and coumaryl alcohols. 10 Interestingly, hemp appears to be spatiotemporally sensitive, and these factors can alter the grain, fiber (lignin), and CBD content of different cultivars to such an extent that cultivar-dependent differences are difficult to establish. 11
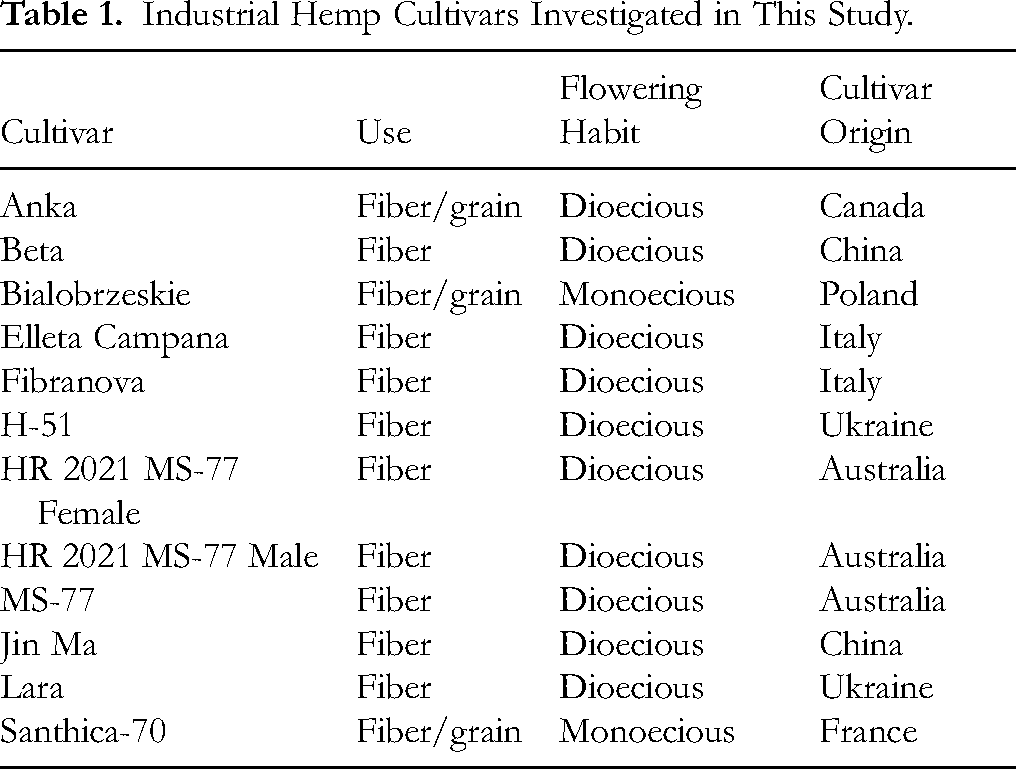
The objective of this study was to characterize and identify differences in the lignin-derived compounds from the hurd tissues of 10 industrial hemp cultivars (monoecious or dioecious) produced for fiber or fiber/grain (listed in Table 1). Additionally, while studies have investigated the antimicrobial properties of the cannabinoids, phenolics, and terpenes within hemp (flowers, leaves, and seeds) there has been far less work on lignin-derivatives (stalks). Therefore, we not only report on the lignin-derivatives present across 10 hemp cultivars, but also on the antimicrobial activity of a few of the identified derivates on Acetoanaerobium sticklandii (previously Clostridium sticklandii 12 ) – a wasteful catabolic amino acid fermenting bacterium common to ruminant livestock such as cattle. 13 This organism was selected as it is a non-pathogenic model organism for amino-acid fermenting bacteria adapted for reliable growth in a laboratory setting. 14
Industrial Hemp Cultivars Investigated in This Study.
Experimental Section
Materials
Syringaresinol (S-β-β′-S) was synthesized in the Lynn lab at the University of Kentucky (Lexington, KY). All other chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA) or Fisher Scientific (Waltham, MA, USA) and used without further purification. 3-(34,5-Trimethoxyphenyl) propionic acid was used as the internal standard for all relative quantification. The 200-proof ethanol used for stock standard preparation in the antimicrobial activity bioassay was purchased from Decon Laboratories Inc. (Prussia, PA, USA).
Planting, Growing, and Harvesting Conditions of Hemp Cultivars
The hurd samples used in this trial were collected from plants grown in a fiber hemp cultivar trial (4 field replications) and a hemp rotation trial (5 field replications) conducted at the University of Kentucky North Farm Complex near Lexington, KY. The soil type was Bluegrass-Maury silt loams (Fine-silty, mixed, active, mesic Typic Paleudalfs). Planting density was adjusted for each cultivar based on seed germination and seed weight to achieve a target density of 2.47 × 106 pure live seeds hectare−1. For the fiber trial, seeds were direct seeded on 5/22/2021 using a cone seeder into a tilled seed bed at a depth of 1.0 to 1.5 cm in rows spaced 20.3 cm apart. For the rotation trial, cultivar MS-77 was drilled into a chemically killed wheat cover crop on 5/14/2021 at a depth of 1.5 to 2.0 cm in rows that were 19 cm apart. Nitrogen, as urea (46-0-0), was applied 2 weeks post-emergence at a rate of 112 kg N hectare−1. Phosphorus and potassium were added as needed based on soil tests. Lime was added to maintain a soil pH of 6.0 to 6.5. Weeds in the plots were managed for the first few weeks by hand weeding.
Fiber trial plants were cut based on maturity: Anka, Bialobrzeskie, Elleta Campana, Fibranova, H-51, Lara and Santhica-70 were cut on 9/16/2021; Beta, Jin Ma and MS-77 were cut on 10/14/2021. Two 1-square meter samples per plot were cut with hand pruners. Stalks were cut at ground level, tied in loose bundles and placed in a grassy area adjacent to the plot to ret for 3 to 4 weeks. The rotation trial plants were cut on 9/31/2021; male and female plants (HR 2021 MS-77 Female and Male) were bundled separately and maintained separately through retting and processing. Following retting, bundles were moved to a green house and allowed to air dry for several weeks until they could be decorticated. The air-dried bundles were weighed and decorticated (separating bast fiber from hurd) between two cleated rollers. Hurd samples were collected from each plot and used to create a composite sample for each cultivar for this analysis. The hurd biomass was stored in a cool room away from light until lignin extraction and characterization was performed in 2023–24.
Ethanosolv Treatment and GC-MS Analysis of Hemp Hurd Biomass
The hemp hurd biomass was extracted using an ethanosolv method adapted from Dorrani (2022). 15 First, 300 mg of hemp hurd biomass was weighed into a small glass vial, purged with nitrogen for 5 min to expel oxygen, and then rapidly sealed. The vial was heated in an oven at 200 °C for 30 min and allowed to cool. After unsealing, the biomass was carefully transferred into a glass pressure tube, and 6 mL of ethanosolv reagent (ethanol/water 1:1 v/v) together with 0.12 M HCl catalyst was added. The tube containing the reaction mixture was then heated at 100 °C in an oil bath under constant stirring for 24 h. After which, the reaction mixture was transferred with 40 mL of ethyl acetate and vacuum filtered using Whatman 50 filter paper. The filtrate was placed in a separatory funnel and extracted twice with water and ethyl acetate (40:60 v/v). The water layer was discarded, and the ethyl acetate layer was then passed through sodium sulfate to remove any residual water. Finally, the solvent was removed using a rotavap, and the residue was reconstituted in a 1:1 mixture of N,O-bis(trimethylsilyl)trifluoroacetaminde (BSTFA) and pyridine for derivatization. The derivatized sample was warmed at ∼ 60 °C for 15 min to ensure complete derivatization before gas chromatography - mass spectrometry (GC–MS) analysis.
GC-MS Run Parameters and Analysis
Analysis of ethanosolv-treated samples was performed using an Agilent 5977C GC-MSD system equipped with a 7693A autosampler. An Agilent HP-5MS UI column (30 m × 0.25 mm i.d. × 0.25 µm film thickness) was used for all injections. The temperature gradient started at 60 °C and was held for 5 min, then ramped to 310 °C at a rate of 15 °C min−1. The temperature was held at 310 °C for an additional 8.5 min. The inlet temperature was set to 250 °C. Helium was used as the carrier gas with a flow rate of 1 mL min−1 through the column. The MS parameters were as follows: transfer line temperature was set to 300 °C, source temperature was 230 °C, and quadrupole temperature was 150 °C. MS scans were performed in the range of 50 to 800 m/z at a scan rate of 2 scans second−1. The solvent delay was set to 6 min.
Data was collected and analyzed using Agilent ChemStation, Agilent MassHunter and Microsoft Excel. For compound identification, a database search was performed by Automated Mass Spectral Deconvolution and Identification System (AMDIS) software v2.4 developed by NIST. Structures and corresponding IUPAC names were generated by ChemDraw software (v20.1.1). For database searches, compounds exhibiting a probability match of ≥ 60% in at least one chromatogram were included. In select cases, compounds with match probabilities < 60% were included following validation through retention time alignment, mass spectral profile evaluation, and reconstructed ion chromatogram comparison against reference standards.
Bacterial Strain and Culture Conditions
Acetoanaerobium sticklandii strain SR was obtained from our culture collection maintained at the Forage-Animal Production Research Unit, ARS – USDA (chain of custody: James Russell, Michael Flythe). SR was maintained and experiments were performed in a reduced basal medium (BM) based on Chen et al. 16 Briefly, the medium composition per 1L was as follows: 240 mg KH2PO4, 240 mg K2HPO4, 480 mg NaCl, 100 mg MgSO4 •7H2O, 516 mg Na2SO4, 64 mg CaCl2 •2H2O, 1 mg pyridoxamine 2HCl, 2 mg thiamine HCl, 2 mg riboflavin, 2 mg nicotinamide, 1 mg lipoic acid, 2 mg CaD pantothenate, 0.05 mg folic acid, 0.1 mg p-aminobenzoic acid, 0.05 mg biotin, 0.05 mg cobalamin, 1 mg pyridoxine, 1 mg pyridoxal HCl, 2.5 mg Na4EDTA, 1 mg FeSO4• 7H2O, 0.01 mg NiCl2 •6H2O, 0.05 mg ZnSO4 •7H2O, 0.1 mg CoCl2 •6H2O, 1 mg MnCl2 •4H2O, 5 μg CuCl2 •2H2O, 0.1 mg H3BO3, 0.015 mg NaMoO4 •2H2O, 600 mg cysteine HCl, and 15 g tryptone peptone (trypticase). The broth was adjusted to a pH of 6.5 via addition of 5 M NaOH, then autoclaved at 121 °C for 20 min to remove O2, after which the medium was cooled under CO2. After cooling, 4.0 g Na2CO3 was added to the broth as a buffer. The media were anaerobically dispensed into Hungate tubes, or into borosilicate serum bottles with agar (0.75% w/v), capped with rubber stoppers and hard plastic or aluminum crimp caps, and autoclaved for sterility. SR was regularly transferred after reaching stationary phase (∼20 h) in BM and incubated in a water bath at 39 °C prior to and throughout the experimentation period.
Antimicrobial Susceptibility Testing – Agar
The presence of any bioactive water-soluble compounds in the ground hemp hurd was tested using agar media prepared as described above. The antimicrobial susceptibility testing in agar described here was adapted from a similar method developed in our laboratory. 17 Briefly, the agar media were melted in a boiling water bath and allowed to cool to touch-temperature then inoculated with stationary phase SR at 2% v/v. The inoculated media were then transferred into the anaerobic chamber (95% CO2/5% H2) and poured into 150 × 15 mm petri plates with sterilized borosilicate scintillation vials (28 × 61 mm) in the plate centers to ensure formation of a well for later addition of the hurd material. After the agar solidified, the scintillation vials were removed aseptically and ∼ 0.2 g of 2 mm ground hurd material for the 10 cultivars was added to the agar wells (1 cultivar sample per plate), after which ∼ 2 mL of sterile water was added to each well to facilitate diffusion of any water-soluble compounds from the hurd into the agar. Two growth controls were prepared where agar was poured around a sterile scintillation vial, the vial removed and the plate left as is, and another where the vial was removed and ∼ 2 mL of sterile water was added to the well. Two sterility controls were prepared as well, in the same manner, but with non-inoculated agar. Two sets of plates, including controls, were prepared and incubated at different temperatures, the first at 39 °C (normal incubation temperature for SR) and the second set at room temperature (RT; ∼ 21 °C) to allow for a longer exposure time to any potentially bioactive compounds. The first set of plates were incubated for 24 h (until turbid growth occurred throughout the agar of the control plates), and the second set for ∼ 48 h.
Antimicrobial Susceptibility Testing – Broth
GC-MS analysis of ground hurd tissue of the 10 cultivars successfully identified several lignin-derived compounds (Table 2), of which three were selected, due to availability and laboratory capability (safe-handling and compatibility with sterilization techniques used - flame), for antimicrobial activity testing on SR: vanillin, vanillic acid, and p-coumaric acid. The methods utilized in the broth antimicrobial susceptibility testing were developed in our laboratory, however broth dilution testing finds its origins in Alexander Fleming's initial report of a broth serial dilution technique from 1929. 18 Briefly, stock solutions of the three compounds were made in 200 proof ethanol, as water solubility was poor, at the following concentrations: 100 mg mL−1 vanillin, 50 mg mL−1 vanillic acid, and 25 mg mL−1 p-coumaric acid. Antimicrobial activity was determined using 4-fold serial dilutions in Hungate tubes pre-filled with BM and sealed under 95% CO2/5% H2, as described above. Briefly, the first tube in the dilution series was amended 1% v/v from the stock solution(s) and serially diluted 4x at 10% v/v for working concentrations of vanillin, vanillic acid, or p-coumaric acid between: 1/0.5/0.25 mg mL−1, and 1 × 10−4/5 × 10−5/2.5 × 10−5 mg mL−1. Tubes were then inoculated with stationary phase SR (1% v/v). Unamended and EtOH (1% v/v amendment) growth controls of SR were used as well as several sterility controls: 1 × 10−4/5 × 10−5/2.5 × 10−5 mg mL−1 vanillin/vanillic acid/p-coumaric acid controls, and an EtOH (1% v/v) control. Tubes were left to incubate in a standard microbiological incubator at 39 °C for ∼ 24 h.
Lignin-Derived Compounds Identified from Ground Cannabis sativa L. Hurd Tissue.
Statistical Analysis
GC-MS analysis was performed on each composite sample per cultivar (12 individual samples, including the female/male and combined MS-77 hurd biomass), ie, the hurd tissue from each cultivar plot was collected, processed, and combined as a single composite sample (one composite per cultivar) for analysis. Therefore, statistical analysis was not performed on these data. Additionally, the antimicrobial susceptibility experiments were performed in triplicate without statistical analysis as the results were qualitative, i.e., growth or no growth.

Representative chromatogram of ethanosolv-treated hemp hurd biomass – Anka.
Results
Ethanosolv Treatment of Hemp Cultivars
Ten hemp cultivars were subjected to ethanosolv treatment and analyzed via GC-MS to evaluate their potential as sources of antimicrobial and value-added compounds. The resulting GC-MS chromatograms, presented in Figure 1 and Figures S1-11, correspond to the ethanosolv-treated hemp cultivars. A few peaks were identified as ethylated derivatives, likely formed through reaction with the ethanosolv reagent, e.g., ethyl vanillate (Table 2). Several organic acids were also detected, likely released during the torrefaction step prior to the ethanosolv treatment, e.g., lactic and glycolic acids. The majority of the peaks in the chromatograms, however, were dominated by monomeric compounds, which is not surprising since lignin monomers are primarily connected through β-O-4′ bonds (Figure 2). The method's ability to cleave these β-O-4′ bonds is evident from the prevalence of monomers. In contrast, dimers produced by ethanosolv treatment, which are typically linked by bonds such as β-β’, β-5′, and 5-5′, were less abundant. Only one lignin-derived dimer, syringaresinol, was observed in all chromatograms, although in different amounts (Figure 1, Figures S1–11 and Table 2). Syringaresinol is formed by the β-β’ linkage of two S- type monomeric units. S units, one of the three major types of lignin monomers, have two methoxy groups attached to the 3- and 5-positions of a 4-hydroxybenzene ring, whereas G-type units have only one methoxy group (Figure 3). Another dimer, α-peltatin, was detected in the chromatograms of the Fibranova and H-51 cultivars (Figures S4 and S5, respectively), although it has not been previously reported in hemp hurd biomass (Figure 3). The predominance of monomers over dimers suggests that the ethanosolv method effectively cleaved the β-O-4′ bonds but was less effective at breaking the bonds within dimers.

A representative lignin polymer with β-O-4′, β-′, and β-5′ linkages that demonstrates how cleavage at these bond types generates either monomers or dimers, respectively. Where S, G, and H represent syringyl (S), guaiacyl (G), and p-hydroxyphenyl (H) units.

List of monomers and dimers identified in ground hemp hurd biomass – (1) 4-hydroxybenzaldehyde, (
Antimicrobial Susceptibility of A. sticklandii to Ground Hemp Hurd and its Constituents
The plate bioassays investigating the antimicrobial activity of ground hemp hurd tissue on the growth of SR demonstrated no apparent inhibition after either 24 h at 39 °C or 48 h at 21 °C for any of the 10 cultivars under the testing conditions of this study (Figure 4).

Representative of ground (2 mm) hemp hurd tissue (MS-77 cultivar) plate bioassay in basal medium agar inoculated with A. sticklandii SR (left) compared to an A. sticklandii SR growth control plate (right).
While the hemp hurd tissue itself did not display inhibitory activity on SR using the agar bioassay method employed here, the ethanosolv treatment revealed ample candidate compounds associated with the lignin (as described above; Table 2). Due to availability and laboratory capabilities (safe-handling and method compatibility) – vanillin, vanillic acid, and p-coumaric acid – were selected for antimicrobial susceptibility testing on SR using broth serial dilutions. Of the compounds tested, only vanillin displayed antimicrobial activity (full growth inhibition) at a concentration of 1 mg mL−1, while the other compounds did not appear antimicrobial at the concentrations and under the conditions tested (data not shown).
Discussion
Hemp is a versatile crop plant used for its fiber, seed, cannabinoids, and bioactive compounds. 1 The crop possesses many bioactive molecules such as the terpenes, phenolics, and cannabinoids all of which are largely relegated to the flowers, leaves, and seeds. However, a large portion of the above-ground biomass belongs to lignin structures, which has been largely understudied. The objective of this study was to characterize lignin-derived compounds from the hurd of 10 fiber/grain Cannabis sativa L. cultivars and investigate the antimicrobial activity of select compounds on the wasteful ruminant bacterium, Acetoanaerobium sticklandii SR.
A mild ethanol-based organic solvent method (ethanosolv) was selected for hemp hurd biomass treatment due to its ability to yield high-purity cellulose with minimal degradation. 19 In this context, “mild” refers to operating at relatively low temperatures and little to no external pressure. Polar protic ethanol serves two main purposes: first, it disrupts the internal linkages between lignin and hemicellulose, as well as the ether and glycosidic bonds in lignin and hemicellulose, respectively; second, it prevents repolymerization by dissolving the oligomers and oligosaccharides released. 20 Thus, allowing for precipitation of the cellulosic component of the biomass and retention of the lignin in the solvent fraction. Water facilitates α-O-4′ bond cleavage and preserves the reactive hydroxy groups. 21 In the reaction, HCl was used as the catalyst to break down the β-O-4′ bonds which are the most prevalent bond type in lignin (∼ 50%). 22 A short heat pretreatment step (torrefaction) before the ethanosolv process was incorporated into the protocol to enhance biomass degradation into smaller monomeric and dimeric products.
Thus, the lignin monomers identified in this study consisted of approximately equal amounts of G- and S-type units, with a small fraction of H-type units (lack methoxy groups; Figure 3), which aligns with previous reports in hemp – an herbaceous angiosperm with a woody core. For example, a study observing the changes in lignin monomers present in hypocotyl hemp tissue (including bast and hurd) over time demonstrated a decrease in H-type units from d6 to d20, no statistical change to G-type units, and a dramatic increase in S-units, resulting in an increased S/G ratio. 23 Another study reported the effect(s) of harvest and field retting time on the chemical composition of mature hemp fiber and found that the lignin content of bast fibers steadily increased as retting duration increased (0-70d) and was much more apparent from early to late harvest times. 24 While the study did not report on the quantities of H, G, or S-type units specifically, they did confirm the presence of both syringyl (S) and guaiacyl (G) units in the stem samples. Gandolfi et al also reported lignin monomer content of mature hemp cultivars and found a large presence of S and G-type units with a notably smaller presence of H-type units, which was consistent with the data reported here.25,26 In agreement with this, Fehrmann et al concluded that two cultivars (Frog1 and Han NE) had low abundance of H-units with generally greater abundance of S and G-units. 27 However, they also reported changes in S and G-unit abundance with crop maturity: Frog1 saw reduced abundance of G-units with increased S-units, while Han NE saw increased abundance of both S and G-units indicating maturity and cultivar-mediated differences in monomeric composition. Unfortunately, as hurd is generally the less desirable fiber source in industrial hemp (compared to bast fibers for textiles), there are precious few studies reporting detailed analysis of its lignin content at maturity.
p-Coumaric acid, an H-type monomer identified in our analysis (Table 2), is known to be present in the cell walls of herbaceous plants, where it forms cross-linkages between lignin and hemicellulose. 28 In fact, Schroyen et al identified p-coumaric acid and vanillic acid, a G-type unit, as predominant monomers in hemp tissue. 29 In hurd tissue, specifically, Gandolfi et al identified the major components of Carmagnola, an Italian grain/CBD variety, as vanillin (45.1%) and syringaldehyde (35.1%) with several minor components also identified: 4-hydroxybenzaldehyde (p-hydroxybenzaldehyde; 8.5%), vanillic acid (0.9%), syringic acid (6.3%), and p-coumaric acid (2.9%). 26 Interestingly, these compounds were identified in all 10 cultivars tested here with the following exceptions: vanillic acid was present in all but the female biomass of MS-77, and p-coumaric acid in all but MS-77, including the separated male and female biomass (Table 2). The seemingly prominent monomers across the 10 cultivars in the present data set include: syringaldehyde, vanillin, and syringic acid.
Several of the identified peaks were present across chromatograms, indicating some similarity in the lignin profiles of each of the hemp cultivars. However, the abundance of several of these peaks appeared variable (e.g., syringaldehyde, syringaresinol; Table 2). As reported in Hammami et al, this variability could be due more to spatiotemporal reasons than cultivar-dependent reasons. 11 Alternatively, other works have noted differences in phenolic compound concentrations in bast fibers between male and female dioecious plants. 30 In agreement with this observation, the male composite biomass of MS-77 (HR 2021 MS-77 Male) analyzed in the present study possessed detectible vanillic acid whereas the female composite biomass (HR 2021 MS-77 Female) did not. However, further study including additional cultivar replicates would need to be performed to reach definitive conclusions of spatiotemporal and/or cultivar-mediated differences in lignin-derivative variability.
The antimicrobial plate bioassays investigating the antimicrobial activity of the hurd material prior to lignin-isolation and derivatization demonstrated a lack of antimicrobial activity on SR under the testing conditions (Figure 4). In agreement with these findings, Tyagi et al also explored the antimicrobial activity of hemp hurds on E. coli (strain TOP10) by spreading a lawn on agar and found that the hurd powder (1 mm; incorporated into a diffusion disk) yielded no antimicrobial activity. 31 However, Khan et al reported growth inhibition of Escherichia coli strain 25922 by ground hemp hurds of different size (0.2-0.02 μm), retting status (retted or not), and heat-treatment. 32 There are fundamental differences in the methodologies between our study, Tyagi et al, and Khan et al that may account for the differing results.31,32 For example, in Khan et al 32 the hemp hurd powder was mixed with inoculated media and spread onto the surface of an agar plate, where reduction in colony formation, compared to unamended controls, was then measured for growth inhibitory effects. Similarly, Tyagi et al spread a lawn of E. coli inoculum on the surface of an agar plate, but instead of mixing the inoculum with the hurd powder itself, placed a hurd powder-infused disk into the center of the plate and measured a zone of inhibition. 31 Whereas in the present study, the agar itself was inoculated with stationary phase culture and larger particle size (2 mm) hemp hurd was added to the center of the plate where the development of a zone of inhibition at the hurd-agar interface (similar to disk diffusion) would indicate inhibitory activity. It's possible that the more homogenous exposure to the antimicrobial substance, via mixing the hurd particulate into the inoculated broth (as in Khan et al), 32 allowed for greater diffusion and subsequent contact than allowable in an agar well or disk and subsequently demonstrated greater susceptibility. Therefore, it is feasible that the differing methodologies could explain the opposing results. However further study exploring these methodologies may be necessary to discern the true antimicrobial activity of ground hemp hurd on various bacteria of interest.
The hemp hurd tissue itself was not found to possess antimicrobial activity using the bioassay methods employed presently, however the ethanosolv treatment revealed ample candidate compounds (Table 2). Of the compounds tested using the broth dilution (vanillin, vanillic acid, and p-coumaric acid), vanillin was discovered to possess antimicrobial activity against SR at 1 mg mL−1. These data agree with previous reports investigating antimicrobial activity of major lignin constituents (intermediates) on other genus and species of bacteria. For example, vanillin was found to be antimicrobial against a toxigenic strain of E. coli (strain 35150) at a concentration of 2 mg mL−1 but saw significant reductions in colony forming units starting at 0.5 mg mL−1. 33 Vanillin was also tested on spoilage organisms of stored cut mangoes, which demonstrated antibacterial (10-13 mM) and antifungal (12-20 mM) activity that was particularly effective against yeasts (< 7 mM). 34 SR appears to be more sensitive to the activity of vanillin (1 mg mL−1 or 6.6 mM) under the conditions used here than those methods used in previous reports. However, unlike the sensitivity displayed to vanillin, SR was not more sensitive to either vanillic acid nor p-coumaric acid when compared with previous reports by other groups. For example, vanillic acid was found non-antimicrobial against SR at 0.5 mg mL−1, which was consistent with a previous report showing antimicrobial activity on Staphylococcus aureus (and other pathogens) at concentrations ≥ 0.6 mg mL−1. 35 The data presented here showed that growth of SR was not inhibited by ≤ 0.25 mg mL−1 p-coumaric acid, consistent with previous work that reported a minimum inhibitory concentration (MIC) of 2 mg mL−1 on both Gram (+) and (-) bacteria. 9
While this work explored the inhibitory activity of just three of the identifiable lignin-derived compounds within hemp hurd on one bacterial species, there are many other compounds present that could prove to be antimicrobial to many other organisms. For example, the hydroxycinnamaldehyde and hydroxycinnamic acid intermediates of the lignin specific pathways, several of which were identified in our own analysis, e.g., coniferyl aldehyde and sinapaldehyde, had growth inhibitory characteristics between 8–2 mg mL−1. 9 Yet more lignin-derived compounds identified in this study have had their antimicrobial activity investigated by other groups. Syringaldehyde revealed broad spectrum activity against both Bacillus subtilis and Klebsiella pneumonia. 36 Stearic acid was tested on B. subtilis, B. cereus, and Mycobacterium fortuitum and had a MIC of 0.01 mg mL−1. 37 Syringic acid was tested on several strains of Cronobacter sakazakii, a common opportunistic pathogen present in powdered infant formula and had a reliable MIC of 5 mg mL−1. 38 Regardless, it is evident that many of the lignin-derived compounds identified in this study from the hemp hurds of fiber/grain cultivars have antimicrobial potential.
Of particular relevance, previous work has demonstrated the potential of hemp hurds/dust as an industrial source of monomeric compounds via a pilot study. Candy et al attempted the extraction of ferulic and p-coumaric acids from both hemp hurds (potentials of 0.3 and 3.5 g kg−1 dry matter, respectively) and hemp dust (potentials of 0.1 and 0.8 5 g kg−1 dry matter, respectively). 39 From the hemp hurds, they were able to recover 50% and 33% of the potential free and bound p-coumaric and ferulic acids; in the hemp dust, all the potential p-coumaric acid and 60% of the potential ferulic acid was recovered. Another group investigated the potential extraction capabilities of acid-catalyzed hydrolysis on commercial sodium lignosulfonate conifer black liquor for value added compounds. 40 They were able to extract 1.729 g kg−1 isovanillic acid, 0.088 g kg−1 vanillin, and 0.007 g kg−1 syringaldehyde, among others. These works demonstrate the possibility that the lignin in hemp hurds could be a viable source of the antimicrobial lignin-derivatives identified here and in previous works.
Additionally, some reports demonstrate the antimicrobial activity of lignin fractions rather than specific derivatives. For example, Yun et al fractionated lignin from bamboo and discovered that the low molecular weight lignin had the highest phenolic hydroxyl content and the broadest antimicrobial activity against both Gram (-) and (+) pathogenic species. 41 Similarly, Sholahuddin et al reported that the low molecular weight fraction of steam-exploded palm oil mill lignocellulosic biomass had high antimicrobial activity against pathogenic bacteria regardless of Gram status. 42 In this manner, it appears that if the extraction of specific lignin-derivatives is not practical, fractionating the lignin and collecting those that are antimicrobial may prove a valuable alternative. However, given the cultivar/maturity-mediated variability in the monomeric content of lignin in hemp, as reported in Fehrmann et al, 27 it may be prudent to determine if these differences impact antimicrobial effect prior to the exploration of industrial applications.
The present study identifies several monomeric and dimeric lignin-derivatives across 10 fiber and fiber/grain industrial hemp cultivars where the data indicated that the abundance of the derivates appeared variable which could be cultivar or spatiotemporally mediated. However, the study is limited by replication and cultivar representation (10), while there are many other dioecious and monoecious cultivars that should be investigated to elucidate these observed differences. Additionally, the study is limited by the extraction methodology used as the method is particularly efficient at cleavage along β-O-4′ bonds, but not as efficient at β-β’, β-5′, and 5-5′ bonding sites, where dimers are typically linked. Furthermore, the present study investigated the antimicrobial activity of just three of the identified lignin derivatives on one, agriculturally significant bacterium (A. sticklandii strain SR) and determined that vanillin possessed antimicrobial activity. Further investigations into other lignin-derived monomers and/or dimers against a broader variety of microorganisms should be undertaken to more fully understand hemp hurd lignin-derivatives as a potentially valuable source of novel antimicrobials. Additionally, studies investigating the extraction efficiency (yield), using a variety of methodologies, of the identified antimicrobial lignin-derivatives should be undertaken to determine the viability of hemp hurd lignin as a source of these potentially valuable compounds.
Conclusions
Ground hurd samples from 10 cultivars of hemp (C. sativa L.) were processed using an ethanosolv method to identify and quantify the lignin-derived compounds present in the material using GC-MS. Of the identified monomeric peaks, there were approximately equal portions of G- and S-units with substantially fewer H-units identified. Syringaldehyde (S), vanillin (G), and syringic acid (S) were the most prominent lignin-derivatives present across the cultivars. Vanillic acid (G) and p-coumaric acid (H) were less dominant monomeric derivatives with vanillic acid absent from the female-separated biomass of MS-77 (HR 2021 MS-77 Female) and p-coumaric acid absent from the MS-77 cultivar altogether. Neither vanillic acid nor p-coumaric acid demonstrated antimicrobial activity on A. sticklandii SR, however vanillin was antimicrobial at 1 mg mL−1. While several of these identified derivatives were present across the chromatograms of each cultivar, their abundance appeared variable, this coupled with previous reports in literature indicate possible cultivar-based (dioecious or monecious) or spatiotemporally mediated differences. However, further replication and research is required to elucidate these observed differences. This work not only aids in the characterization/identification of lignin-derivatives in hemp fiber and fiber/grain cultivars but also contributes to the growing body of literature identifying hurd materials/compounds as a potential source of antimicrobials for amino-acid fermenting bacteria.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261436515 - Supplemental material for Characterization and Antimicrobial Activity of Lignin-Derived Compounds in Ground Hurd Tissue of Cannabis sativa L. (Hemp) Cultivars
Supplemental material, sj-docx-1-npx-10.1177_1934578X261436515 for Characterization and Antimicrobial Activity of Lignin-Derived Compounds in Ground Hurd Tissue of Cannabis sativa L. (Hemp) Cultivars by Jourdan E. Lakes, Anjam Sadik, Robert C. Pearce, Barbara L. Knutson, Stephen E. Rankin, Bert C. Lynn and Michael D. Flythe in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to acknowledge Dr. Angela M. Gutierrez of the Sustainability and Analytics Equipment Facility (SAEF) in the Pigman College of Engineering at the University of Kentucky for the use of their gas chromatograph – mass spectrometer.
ORCID iDs
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Conceptualization and methodology: JEL, AS, MDF, BCL, BLK; Formal analysis and investigation: JEL, AS, RCP; Writing – original draft: JEL, AS, RCP; Writing – editing/review: BLK, SER, BCL, MDF; Funding acquisition: MDF, BCL, BLK, SER; Resources: MDF, BCL, BLK, SER
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AS was funded by the SunGrant initiative grant # 3200005447. JEL was supported as an Administrator-Funded Post Doctoral Fellow by the Agricultural Research Service (ARS) National Program NP-101, Food Animal Production (5042-32630-003-000D). The research and MDF were supported by the USDA, ARS National Program NP-215, Grass, Forage and Rangeland Agroecosystems (5042-21500-001-00D).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
