Abstract
Background
Marchantia polymorpha is a model organism for research on evolutionary molecular-microbe interactions. Filamentous fungi pathogenic to M. polymorpha have been reported and a necrotrophic-like infection process has been proposed according to experiments conducted in the laboratory, but many aspects of M. polymorpha and microbes interaction remain unknown.
Objective
Field observations have been done and indicated that several kinds of fungi had ability to infect M. polymorpha and among of them, Bjerkandera adusta and Irpex lacteus infect M. polymorpha, and laboratory experiments have revealed that these fungi have a strong ability to infect M. polymorpha. Therefore, it was hypothesized that these fungi may produce specialized metabolites that lead to effectively invade into the liverwort. The aim of this research was to determine the chemical structures of the specialized metabolites and evaluate the biological activities of the compounds.
Materials and Methods
B. adusta and I. lacteus were obtained from GenBank Project, NARO. Male accessions of M. polymorpha, Takaragaike-1, were used for bioassay. The fungi were grown in potato dextrose broth medium. The culture filtrates were filtered and concentrated, and EtOAc (3 L) and H2O (1 L) were added to the crude material. Crude extracts obtained from the EtOAc were subjected to several kinds of purification steps to afford methyl vanillin from B. adusta and tremutin E from I. lacteus.
Results
Methyl vanillin and tremutin E were isolated from B. adusta and I. lacteus, respectively, according to a guidance of growth inhibitory activity-guided fractionations and identified using mass spectroscopic data together with 1D and 2D NMR techniques.
Conclusion
Methyl vanillin and tremutin E inhibited the growth of M. polymorpha with almost 100-fold stronger activity than that of acetic acid, a pivotal active ingredient of commercial anti-liverwort agent.
Keywords
Introduction
The thalloid liverwort Marchantia polymorpha is widely distributed in temperate regions and has been increasingly utilized as a model bryophyte since it is easy to grow in the laboratory, has a short life cycle, possesses the ability to transform, and is compatible with several gene editing methods.1–2 Similar to other bryophytes, gametophyte generation dominates the life cycle of M. polymorpha and the main plant body (thallus) is produced for line maintenance; furthermore, the costs of maintaining M. polymorpha cultures are lower than those for flowering plants. Moreover, M. polymorpha is a representative of an ancient lineage of land plants that colonized our planet more than 450 million years ago.3–4
M. polymorpha is reported to interact with different types of microorganisms such as viruses, bacteria, fungi, and oomycetes. 5 In this study, the hemibiotrophic oomycete pathogen Phytophthora palmivora colonized M. polymorpha, enabling the study of liverwort–pathogen interactions at the molecular level. 6 Furthermore, artificial inoculation with Pseudomonas syringae pv. tomato DC3000 leads to activation of the typical hallmarks of plant innate immunity. 7 Regarding the system establishing defense system in M. polymorpha, Monte et al reported that dinor-iso-12-oxo-phytodienoic acid (dinor-iso-OPDA) is a pivotal ingredient, which interacts with MpCOI1. 8 Additionally, the isolation of pathogenic fungi samples from wild M. polymorpha was performed by Matsui et al, 9 and four fungal strains were isolated: Bjerkandera adusta MI4 and MI3, Irpex lacteus MI1, and Phanerochaete peniophoroides MI2. Among these strains, I. lacteus demonstrated the strongest pathogenic effect on M. polymorpha, followed by B. adusta.
B. adusta, a wood-rotting white-rot basidiomycete, belongs to the Polyporaceae family and parasitizes particular wood types.10–11 This fungus secretes lignin peroxidase, manganese peroxidase, and other versatile peroxidases to degrade lignin in wood.12–14 In addition, the pathogenic wood-decaying fungus I. lacteus also belongs to the family Polyporaceae and has been widely used in Chinese folk medicine for decades to treat inflammation, bacterial/fungal infections, and urinary retention.15–17 A chemical investigation of this fungus revealed the presence of eight sesquiterpenoids, tremutins A-H, and three other known compounds. 18 Previously, Wang et al 18 investigated the toxicities of all of these compounds to five human cancer cell lines: breast cancer (MCF-7), hepatocellular carcinoma (SMMC-7721), human myeloid leukemia (HL-60), colon cancer (SW480), and lung cancer (A-549).
Numerous studies have reported that plant pathogenic bacteria and fungi can invade higher plants and produce specialized compounds such as fusicoccin, 19 alternaric acid, 20 phleichrome, 21 victorin, 22 tabtoxin, 23 and syringomycin. 24 These compounds are useful because they can facilitate investigations of plant-microbe interactions. Previous studies have supported possibility that identifying biologically active compounds, including those produced by infectious fungi that inhibit growth of the liverwort, may potentially help to elucidate the interactions between M. polymorpha and infectious microbes. Therefore, this study aimed to isolate and purify specialized compounds from B. adusta and I. lacteus that may facilitate invasion of M. polymorpha.
Materials and Methods
General Experimental Procedures
The optical rotations were determined using a JASCO P-2200 polarimeter. Nuclear magnetic resonance (NMR) spectra were recorded in CDCl3 using a JNM-ECZ 400 NMR spectrometer (1H NMR: 400 MHz, JEOL Ltd) and an AMX 500 Bruker system (1H NMR: 500 MHz, 13C NMR: 126 MHz). Assignments of proton and carbon signals were performed by obtaining 1H NMR, 13C NMR (referenced for CDCl3 at δH 7.24 and δC 77.23, respectively), COSY, HSQC, HMBC, and NOESY spectra. The high-resolution fast atom bombardment mass spectrometry (HRFDMS) analysis was performed using a JMS-T100GCV instrument (JEOL, Ltd). High-performance liquid chromatography (HPLC) was performed on a HITACHI CHROMASTER HPLC system equipped with a 5410 UV detector, 5310 column oven, 5110 pump, and YMC−Pack FA column (250 × 6.0 mm). Fractions were monitored using thin−layer chromatography on silica gel 60 F254 plates, and the spots were visualized by spraying the plates with a detection reagent (anisaldehyde solution prepared according to the general method) with heating. All chemicals used in this study were of analytical grade and were purchased from Sigma‒Aldrich (Tokyo, Japan) and Kanto Chemical Co., Inc. (Tokyo, Japan).
Fungal and Plant Material
The filamentous fungi B. adusta (MAFF 420156) and I. lacteus (MAFF 420244) were obtained from GenBank Project, NARO (National Agriculture and Food Research Organization), Japan. Male accessions of M. polymorpha, Takaragaike-1, were propagated in half-strength Gamborg's B5 medium (pH 5.5).
Extraction and Isolation of Methyl Vanillin (1 )
The fungus B. adusta was grown in potato dextrose broth medium (48 L) for 50 days in the dark at 25 °C. The culture was filtered and concentrated using a rotary evaporator. Ethyl acetate (EtOAc, 3 L) and H2O (1 L) were added to the crude material, and the mixture was allowed to stand for 48 h. The separated EtOAc layer was dried over anhydrous sodium sulfate (Na2SO4). The volatile components of the separated EtOAc layer were removed under reduced pressure to obtain a crude extract, which was subjected to column chromatography (CC) using silica gel and eluted using a solvent system composed of methanol (MeOH)/CHCl3 (5:95) to obtain four fractions (A-D). The fraction showing inhibitory activity (24.2 mg) was further separated using the same solvent (CC with MeOH/CHCl3 [5:95]) to yield compound
Methyl vanillin (
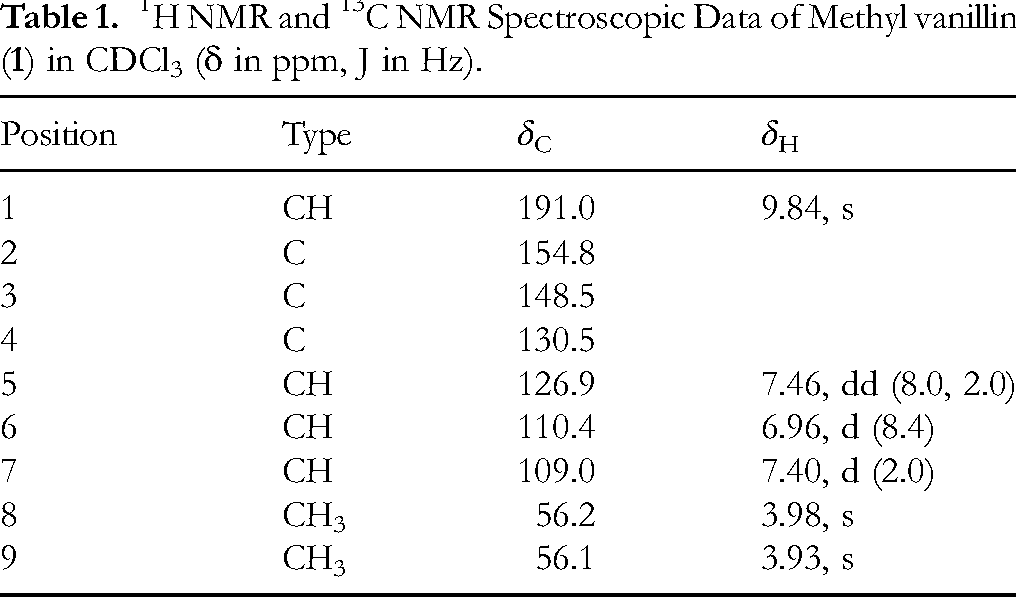
1H NMR and 13C NMR Spectroscopic Data of Methyl vanillin (
Extraction and Isolation of Tremutin E (2 )
The fungus I. lacteus was grown in potato dextrose broth (48 L) for 50 days in the dark at 25 °C. The culture was filtered and concentrated using a rotary evaporator. EtOAc (3 L) and H2O (1 L) were added to the crude extract, and the mixture was left to stand for 48 h. The separated EtOAc layer was dried over Na2SO4. After removing volatiles under reduced pressure, the residue was subjected to silica gel CC and eluted with a solvent system composed of MeOH/CHCl3 (5:95) to yield 40 fractions. The fraction showing inhibitory activity (275.6 mg) was further purified by HPLC (YMC-Pack FA, f 6.0 × 250 mm, MeOH:H2O:acetic acid (AcOH) = 58:42:0.1, 1 mL/min, A210 nm) to give compound
Tremutin E (
1H NMR and 13C NMR Spectroscopic Data of Tremutin E (

Diffusion Assay
M. polymorpha grown on half-strength Gamborg's B5 medium for 14 days was used to analyze the inhibitory effects of the crude extracts from B. adusta and I. lacteus (Figure 1). The crude extracts were diluted with ethanol (EtOH, 1000 µL), and these solutions were applied to a paper disk for duplicate antibiotic assays (ϕ: 8 mm, 1.5 mm thickness; ADVANTEC, Taipei City, Taiwan). Each paper disk received 500 µL of solution, and residual solvent was removed by air-drying on a stainless-steel needle point holder for 15 min. Each paper disk was then placed in the middle of a medium containing four 14-day-old M. polymorpha individuals. Paper disks treated with EtOH (1000 µL) served as mock controls. Each treatment was replicated thrice. The growth area was measured using the ImageJ software (National Institutes of Health, Bethesda, MD, USA).

Schematic Illustration of the Diffusion Assay.
Biological Activity Test of Methyl Vanillin (1 ) and Tremutin E (2 ) Using a Dilution Assay
To evaluate the biological activities of methyl vanillin (

Schematic Illustration of the Dilution Assay.
Statistical Analysis
The data were subjected to analysis using Tukey's test. Statistical significance was defined as P value less than 05. Values were presented as mean ± standard deviation (SD).
Result
Isolation and Structural Determination of Growth Inhibitory Compounds
First, we established a bioassay to evaluate M. polymorpha growth inhibition. A schematic illustration of the bioassay used to isolate low-molecular-weight bioactive compounds is presented in Figure 1, whose method is named as diffusion assay in this report. Procedural details of the assay are provided in the Experimental Section. Biological-activity-guided fractionation was performed using the diffusion assay to obtain compounds
Compound

Chemical Structures of Methyl Vanillin (
Compound
Growth Inhibitory Activity of Compounds 1 and 2 on M. polymorpha
Since compounds

Growth Inhibitory Test Using the Isolated Compounds. Each Value is Represented as the Mean Value ± SD. The Number of the Independent Replicates were Five for Mock and Ten for the Treatmets of AcOH, Methyl Vanillin, and Tremutin E. Different Letters Indicate Statistically Significant Differences (P < .05, Tukey's Test).
Discussion
It was reported that several kinds of fungi had ability to infect M. polymorpha and among of them, B. adusta and I. lacteus showed ability to infect M. polymorpha.
9
In this study, methyl vanillin (
Conclusion
This project was initiated by the isolation and purification of specialized compounds produced by infectious fungi in M. polymorpha, such as those that inhibit liverwort growth. In this study, two bioactive compounds, methyl vanillin (
Footnotes
Acknowledgements
We acknowledge the assistance of Dr Eri Fukushi and Mr Yusuke Takata (GC‒MS & NMR Laboratory, Research Faculty of Agriculture, Hokkaido University) for obtaining the spectroscopic data, and Dr Hirofumi Nakagami (Plant Proteomics Research Unit, RIKEN CSRS, Yokohama, Kanagawa, 230-0045 Japan) for providing information on fungal strains. We also acknowledge a scholarship for S.I. from Hitachi Corporative Education and Research Support (Hitachi Ltd). The authors also thank the Program for Forming Japan's Peak Research Universities (J-PEAKS) of the Japan Society for the Promotion of Science (JSPS) for financial support.
Ethical Considerations
Ethical Approval is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
The experiments described in this article were conducted in compliance with laws that protect Human and Animal Rights.
