Abstract
Objective
Calcium spirulan (CaSP) has been shown to prevent host cell penetration by several enveloped viruses, including Herpes simplex virus type 1 (HSV-1), in in vitro experiments. We investigated how CaSP inhibits HSV-1 penetration into human keratinocyte cells by studying the interactions among CaSP, HSV-1, and keratinocytes in vitro.
Methods
After chemical characterization of the CaSP, we conducted two separate experiments. First, we added different HSV-1 viral loads to HaCaT cells that had been incubated with CaSP for 3 h. Second, different viral loads were incubated with CaSP for 3 h and then the resulting mixtures were added to the cells. The incubation period lasted 48 h. The HSV-1 median tissue culture infectious dose (TCID50) was determined by measuring the cytopathic effect (CPE) index and using a colorimetric immunohistochemical (IHC) staining. CaSP degradation and penetration into keratinocytes were determined using an HLPC method developed in house.
Results
CaSP was detected in the supernatant but not in the cells. After pre-incubating the cells with CaSP, a viral load of 4.56 TCID50 was inhibited by concentrations of ≥50 µg/mL of CaSP after 48 h of incubation (CPE ≤2). An activity against the highest HSV-1 TCID50 (114) was observed only with 800 µg/mL of CaSP. Similar effects were observed after 48 h pre-incubation of HSV-1 with CaSP when ≥50 µg/mL of CaSP was added to a viral suspension of 4.56 TCID50. The higher viral load (TCID50 114) was not inhibited. Sustained inhibition of all HSV-1 titers used (from 0 to 1000 TCID50) was observed only at a CaSP concentration of 800 µg/mL, independent from the pre-incubation method.
Conclusion
CaSP does not enter keratinocytes and seems to interact with both cells and virus as viral inhibition was independent from the preincubation method.
This is a visual representation of the abstract.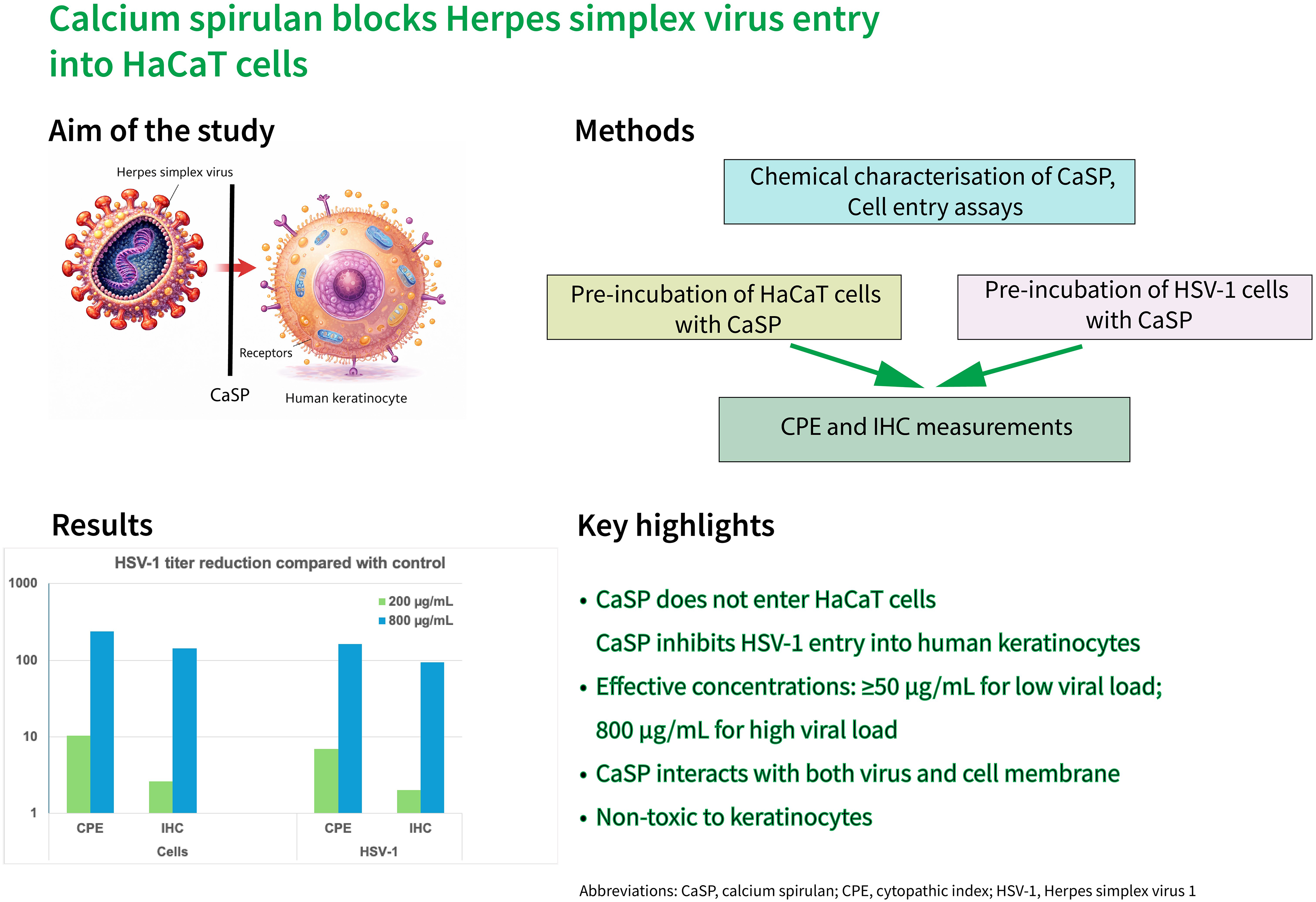
Keywords
Introduction
Herpes simplex virus type 1 (HSV-1) often causes mild, uncomplicated, and often recurrent mucocutaneous infections such as herpes labialis, also known as “cold sores” or “fever blisters”, 1 by entering keratinocytes via a nectin-1-dependent, rapid plasma membrane fusion pathway. 2 After the initial infection, HSV-1 resides in the neurons of the peripheral nervous system of its host in a latent state, persisting lifelong and often triggering recurrent disease. 3 It is estimated that worldwide approx. 3.7 billion people under the age of 50 are latently infected by HSV-1. 4
HSV-1 can also cause life-threatening diseases, 5 for which nucleoside analogues such as acyclovir, valaciclovir, penciclovir, and famciclovir are available as oral treatments.6–9 Herpes labialis management, on the other hand, is mainly symptomatic. Zinc oxide or zinc sulfate creams, as well as antiviral creams containing mainly acyclovir may reduce symptoms duration. Prophylactic treatment with oral or topical antiviral medicines before exposure to sunlight–one of the main triggering factors–may also provide some protection. 10
A water-soluble extract of the cyanobacterium Arthrospira platensis (synonym: Spirulina platensis) has been shown to inhibit HSV-1 replication in HeLa cells in a dose-dependent manner at concentrations of 0.08–50 mg/mL. 11 The extract inhibited viral, but not host cell protein synthesis. 11 In addition to providing viral protection, A. platensis extracts are known to possess antibacterial properties, 12 to help manage skin health, and to protect the skin barrier. 13 Calcium spirulan (CaSP), a sulfated, calcium-chelated polysaccharide found in A. platensis extracts, 14 is considered to be one of the most important active pharmacological ingredients in the extracts. Experimental studies, for instance, have suggested that CaSP may reduce tumor invasion and metastasis. CaSP inhibits HSV-1 invasion in the basal membranes of B16-BL6 melanoma cells, Colon 26 M3.1 carcinoma cells, and HT-1080 fibrosarcoma cells. This indicates that inhibition probably occurs by preventing viral adhesion to cell receptors. 15 In vitro, CaSP inhibits blood clot formation and assists in dissolving existing clots by cleaving fibrinogen or fibrin. CaSP acts as a potent enhancer of tissue-type plasminogen activator (t-PA) synthesis and exhibits anticoagulant properties by activating the heparin cofactor II. 16 It has also been shown to have antiviral activity in vitro against several enveloped viruses, including HSV-1, human cytomegalovirus, measles, mumps, influenza, and the human immunodeficiency virus (HIV),14,17 and topical formulations have been used as a safe treatment option for children with molluscum contagiosum, an infection caused by a poxvirus. 18
The main components of CaSP are 1,3-linked rhamnose and 1,2-linked 3-O-methyl rhamnose (acarbose) units in a ratio of about 5:3, 19 but the molecule also contains ribose, mannose, galactose, xylose, glucose, and galacturonic acid. 14 Replacing calcium with sodium or potassium does not decrease the anti-HSV-1 activity of CaSP, which, is reduced, when calcium is replaced by divalent and trivalent metal cations. 20 Depolymerization with hydrogen peroxide also decreases the antiviral potential of CaSP. 20 In HaCaT cells (human adult, low calcium, high temperature keratinocytes), CaSP exhibits an anti-HSV-1 activity similar to acyclovir. However, unlike acyclovir, CaSP apparently blocks viral attachment and penetration. 21
The antiviral activity of a cream containing CaSP has been confirmed in an observational clinical trial on volunteers with recurrent herpes labialis, in which a topical CaSP cream was superior to topical acyclovir in preventing recurrence, 21 but the mode of action of CaSP against HSV-1 has not been conclusively elucidated. This study aimed at investigating how CaSP inhibits HSV-1 penetration into human keratinocytes by examining the interactions among CaSP, viral particles, and keratinocytes in vitro.
Material and Methods
CaSP Extract, Chemicals, and Reagents
CaSP was provided as a sterile, 1% aqueous solution by Ocean Pharma GmbH (Reinbek, Germany). Acyclovir (ACV), carbamazepine, propranolol, trypan blue solution, and dimethyl sulfoxide (DMSO) were purchased from Sigma-Aldrich (Taufkirchen, Germany). Heparin and H2O2 were provided by Carl Roth (Karlsruhe, Germany), bovine serum albumin (BSA) by PAN Biotech (Aidenbach, Germany), and 3,3′-Diaminobenzidine (DAB) by Roche Diagnostics GmbH (Mannheim, Germany). Dulbecco's Modified Eagle Medium (DMEM), fetal bovine serum (FBS), trypsin-EDTA (0.25%), and Geneticin (G418) were supplied by Gibco (Darmstadt, Germany). Methanol and phosphate-buffered saline (PBS) were obtained from Th. Geyer GmbH & Co. KG (Höxter, Germany), and penicillin-streptomycin, Triton X-100, and sodium dodecyl sulfate (SDS) from Life Technologies/ThermoFisher Diagnostics GmbH (Darmstadt, Germany). Water for injection was purchased from B. Braun AG (Melsungen, Germany) and the XTT assay from Applichem GmbH (Darmstadt, Germany). All analytical-grade solvents were purchased from Merck (Darmstadt, Germany), except for the 99% MS-grade formic acid that was obtained from Biosolve (Valkenswaard, The Netherlands). All other solvents, as well as tris-(hydroxymethyl) aminomethane (Tris) (≥99.9%), NaCl, and sodium deoxycholate, were provided by Carl Roth (Karlsruhe, Germany).
The HaCaT KeratinoSense cell line (RE242, HaCaT cells transfected with an Nrf-2 luciferase reporter plasmid) used for the infection assays was obtained from acCELLerate GmbH (Hamburg, Germany). HSV-1 (2.34 × 106 TCID50 [median tissue culture infectious dose]/mL, catalog number 1806153v, lot number 1555) was provided by Public Health England (National Collection of Pathogenic Viruses [NCPV], Porton Down, Salisbury, England). Rabbit anti-goat IgG H&L (HRP) and anti-HSV-1 goat polyclonal antibody (ab20536) were purchased from Abcam plc (Cambridge, England).
Preparation of CaSP for Analytical and Antiviral Studies
The CaSP solution was freeze-dried in an aluminum bowl with an initial filling height of approximately 0.8 cm. There was no loss in quality or quantity, as confirmed by high-performance liquid chromatography (HPLC) coupled with a refractive index detector (HPLC-RI, VWR Hitachi, Darmstadt, Germany). The samples were frozen at −80 °C prior to freeze drying (FD) in an Alpha 1–4 freeze dryer (Christ, Osterode, Germany) that was run for 12 h at 37 PS. FD samples were stored at −20 °C until further use.
Characterization and Quantitative Measurements of CaSP
We carried out the chemical characterization of CaSP, determined its molecular weight, and checked for impurities present in the sample using an ultra-high-performance liquid chromatography (UPLC) on an Acquity Arc System (Waters Corp., Milford, MA, USA) coupled with a UV/VIS detector (Waters 2998 PDA Detector) and a mass spectrometer (Waters Acquity QDa Detector), using a C18 column (Waters Cortecs C18 2.7 μm, 90 Å, 3.0 × 100 mm,) operated at 40 °C. The injection volume was 5 μL. A H2O and acetonitrile gradient (0 min: 70% H2O; 4 min.: 10% H2O; 9 min: 0% H2O; 11.5 min: 70% H2O until 15 min) was used, and the flow velocity was set to 0.5 mL/min. Formic acid (0.1%) was added to both solvents. Optical spectra were recorded within the range 200–800 nm. The mass spectrometer with electrospray ionization was operated in positive mode with a cone voltage of 15 V and a probe temperature of 600 °C at 150–1250 m/z. Mass spectra were evaluated with MassLynx V4.1.
CaSP concentrations were determined using a HPLC-RI Hitachi Primaide System (Hitachi, Krefeld, Germany) equipped with a 1410 UV detector and Chromaster 5450 RI detector module on a Macherey-Nagel VA 300/7.8 Nucleogel Sugar 810 H column (sulfonated polystyrene/divinylbenzene resin, 8-10 μm, 100 Å, 300 mm×7.8 mm, Macherey-Nagel, Düren, Germany) with H2SO4 as the solvent at a flow rate of 0.6 mL/min and 35 °C. Spectra were evaluated using the Primaide System Manager HPLC Software provided with the Primaide System.
A calibration curve was established using the peak areas in the range of 0.1–9 mg/mL CaSP (w/v). Cell supernatant and lysate samples were injected directly into the HPLC-RI system, and peak areas were determined to calculate concentrations using the calibration curve.
Quantification of Heparin, Carbamazepine, ACV, and Propranolol
The concentrations of ACV, carbamazepine, and propranolol were measured using a Hitachi Primaide 1430 Diode Array Detector (HPLC-DAD, Hitachi, Krefeld, Germany) on an ACE® Equivalence 3 System (C18, 3 µm, 110 Å, 150 mm×4.6 mm, VWR Avantor, Darmstadt, Germany). A gradient solvent from 5% to 90% acetonitrile and acidic water (pH 2.2, formic acid) was used at a flow rate of 1.25 mL/min and a temperature of 40 °C. The injection volume was 5 µL. All compounds were quantified at 275 nm. Calibration curves were prepared for each compound and used to determine the concentrations of the compounds in cell lysates and supernatants. For heparin, calibration curves were prepared under the same conditions described for CaSP, but measurements were carried out using an HPLC-RI system.
Recovery of CaSP from Growth media and HaCaT Cells
HaCaT cells (5 × 105 cells/mL), which were pre-adapted to a serum-free medium, were maintained in DMEM supplemented with 10% human platelet lysate (HPL; PL BioScience, Aachen, Germany). The cells were passaged once a week when confluency approached 80%. Cell viability was assessed using a CASY cell counter (OLS Omni Life Science, Bremen, Germany). Once confluency reached approx. 80%, 5, 10, 40, and 100 µg/mL CaSP as well as 100 µg/mL of the controls ACV, carbamazepine, propranolol, and heparin (which is known not to penetrate the cell lumen 22 ) were added. The cells were then incubated for 1, 2, 3, and 24 h at 37 °C. 23 After incubation, the DMEM in which the cells had been cultivated was collected and vacuum-dried for analysis. Fresh DMEM was also dried and used as a control. The cells attached to the flask were lysed using a radioimmunoprecipitation assay lysis buffer containing Tris-HCl 1 M, Triton X-100, NaCl 5 M, 10% sodium deoxycholate, and 10% SDS (all p.A. grade, Carl Roth, Karlsruhe, Germany).
The concentrations of the test substances were determined at each time point in the supernatant and in the washed lysed cells. If the sum of the concentrations in the supernatant and the lysed cells concentrations did not equal 100%, the difference was considered to be caused by the degradation of the test substance. In all experiments, untreated cells served as a control. Cell viability was measured for all conditions, and all conditions were tested in 3 replicates.
Determination of Cell Viability
Experiments were carried out with HaCaT cells (105 cells/mL, or 2 × 104 cells/well) and cytotoxicity was determined by the XTT assay. 24 All measurements were carried out using a Biotek Cytation 5 (Agilent Technologies; Software Gen 5, version 3.14). The maximum concentrations of the compounds used in the experiments were chosen based on viability and cytotoxicity testing results.
Cell viability was also scored by determining the Cytopathic Effect (CPE) index 25 using a Zeiss Primovert light microscope, according to the scoring system described in the DIN EN ISO 10993-5_2009-10 documentation. 26 CPE scores range from 0 (no cell lysis evident and typical cell morphology) to 4 (complete monolayer disintegration with all the hallmarks of a dying or dead culture). Any other changes in cell morphology, cellular detachment, and signs of viral infection were documented as well.
Minimum Viral Load to be Used in the Experiments
To determine the optimal viral load for the cell entry studies, the cells were incubated with HSV-1 at 7 concentrations ranging from 10−3 to 103 TCID50/mL for 11 days. The experiment was carried out in 8 replicates. Cell viability was scored as described above, and the CPE was determined daily for 11 days.
CaSP Concentrations to be Used in the Infection Studies
The HaCaT cell density was adjusted to 1 × 105 cells/mL. Two hundred µL of the cell suspension were seeded into each well of two 96-well plates, with a separate plate used for each test compound. The plates were pre-incubated without test compounds for about 24 h, washed with DMEM without FBS, and then the test compounds were added. All experiments were carried out at 37 °C under 5% CO2. 200 µg/mL ACV and growth medium served as controls.
In a first experiment, 100 µL of CaSP, at concentrations ranging from 12.5 to 800 µg/mL, were added to the cells. The plates were incubated for approx. 3 h before 100 µL of three HSV-1 dilutions (0, 4.56, and 114 TCID50/mL) were added to the wells.
In a second experiment, CaSP concentrations ranging from 12.5 to 800 µg/mL were mixed with the three HSV-1 dilutions, incubated for 3 h, and the CaSP/HSV-1 mixture was then added to the cells.
Both experiments were carried out in duplicate. CPE, as well as changes in cell morphology, cellular detachment, and other signs of viral infection, were determined after three days.
HSV-1 Inhibition by CaSP
The cells were seeded at a density of 105 cells/mL, resulting in 2 × 104 cells per well, and incubated for 24 h at 37 °C and 5% CO2.
In a first experiment the cells were pre-incubated with CaSP, after which different viral loads (serial dilutions ranging from 1000 to 0.017 TCID50/mL) were added to the cells. In a second experiment, the virus was incubated with CaSP for three hours, after which the mixtures of virus and CaSP were added to the cells.
The incubation period for both experiments lasted 48 h. Both experiments were carried out in 8 replicates.
Immunohistochemical Staining
In addition to the CPE index, we evaluated the anti-HSV-1 activity of CaSP using a colorimetric immunohistochemical (IHC) staining with 3,3′-diaminobenzidine (DAB) substrate (Roche Diagnostics GmbH). Briefly, cell monolayers were washed twice with 100 µL of PBS, and 90 µL of cold methanol were added to each well. The plates were then incubated for approx. 15 min. at room temperature (RT). The reaction was blocked by adding 10 µL of 3% H2O2 and the plates were then incubated for an additional 15 min. at RT. The monolayer was washed twice more with 100 µL of PBS, after which the anti-HSV-1 primary antibody was added. After 60 min. incubation at RT, the monolayer was washed three times with 100 µL of PBS, the Horseradish peroxidase (HRP)-conjugated secondary antibody was added, and the plates were incubated for 60 min at RT. The monolayer was washed again three times with 100 µL of PBS, 100 µL of the DAB substrate were added to each well, and the plates incubated for 15 min. The substrate was discarded, the cells were washed with 100 µL of distilled water, and the plates dried and analyzed using a Zeiss Primovert light microscope. Readings were taken on days 2 and 3 after inoculation.
Data Analysis
The collected data are non-parametric; therefore, no means or standard deviations were computed. When there are enough repetitions, the medians and the 25th and 75th percentiles are computed, otherwise the minimum and maximum values are presented. The results were analyzed only descriptively, as there are not enough data points to allow for formal confirmatory statistical analyses. HSV-1 inhibition is presented graphically as medians ± percentiles (or minimum-maximum) of the concentrations. A CPE of 2 or less was arbitrarily set as the cutoff for HSV-1 inhibiting effects. We present graphical displays of the results, including lowess smoothing curves, resulting from a non-parametric regression technique used to visualize trends in data. 27 An analysis of variance (ANOVA) using pretreatment (cells or HSV-1), HSV-1 titer, and CaSP concentrations as covariates, was carried out as an hypothesis-generating analysis with no confirmatory value. All statistical parameters and graphical displays were generated using Stata, version 19.5 (StataCorp., College Station, Texas, USA).
Results
Characterization and Quantitative Measurements of CaSP
UPLC-UV/VIS detected a nonspecific absorption between 200 and 280 nm. HPLC-RI revealed a specific peak (Figure 1A) that was used for calibration. The lowest detection limit was 0.15 µg/mL. Mass spectrometry (Figure 1B) revealed a wide mass variation between 400 and 1200 kDa, with a maximum at 700 kDa.

Intracellular and Extracellular Distribution of CaSP
CaSP at a concentration of 100 µg/mL was only detected in the supernatant but not in the HaCaT cells. Similar results were obtained with incubation times of 1, 2, 3, or 24 h (Table 1) and with CaSP concentrations of 5, 10, or 40 µg/mL (data not shown). No penetration into the cells or degradation was observed.
Proportion (as Percentage of the Baseline [0 h]) Inside and Outside HaCaT Cells After Incubation for 1, 2, 3, and 24 h at 37 °C. The Data Refer to an Initial Concentration of 100 µg/mL for all Compounds. Figures are Means (Standard Deviation) of the Percentages of Three Measurements (n = 3). CaSp, Calcium Spirulan; E, Extracellular; I, Intracellular; D, Degradation.

As expected, heparin did not penetrate the cell lumina. In contrast, propranolol, ACV, and carbamazepine were detected both intra- and extracellularly (Table 1). Significant degradation was observed only for propranolol and carbamazepine. Cell viability was above 80% in all tested conditions.
Minimum Viral Load to be Used in the Experiments
Visual detection of HIV-1 infection occurred within 3 days after infection already at an HSV-1 titer of 1 TCID50/mL (data not shown). Therefore, we tested the HSV-1 inhibitory activity of CaSP against three viral loads: a control with no viral particles), 4.56 TCID50, and 114 TCID50.
CaSP Concentrations to be Used in the Infection Studies
Pre-Incubation of the HaCaT Cells with CaSP
CaSP inhibited a viral load of 4.56 TCID50 at a concentration ≥50 µg/mL after 48 h of incubation (CPE ≤2). An anti-HSV-1 activity against the highest TCID50 (114) was observed only at a concentration of 800 µg/mL CaSP. After 72 h, anti-HSV-1 activity was observed at concentrations of 400 and 800 µg/mL for 4.56 TCID50. The highest TCID50 level (114) was inhibited only by a concentration of 800 µg/mL CaSP (Figures 2A and 2B). CaSP was not toxic to the cells at any of the concentrations investigated (Figures 2A and 2B, controls). A dose-dependent activity was seen only with an HSV-1 TCID50 of 4.56 at both time points. However, after a 72-h incubation period, only the two highest concentrations (400 and 800 µg/mL) remained active.
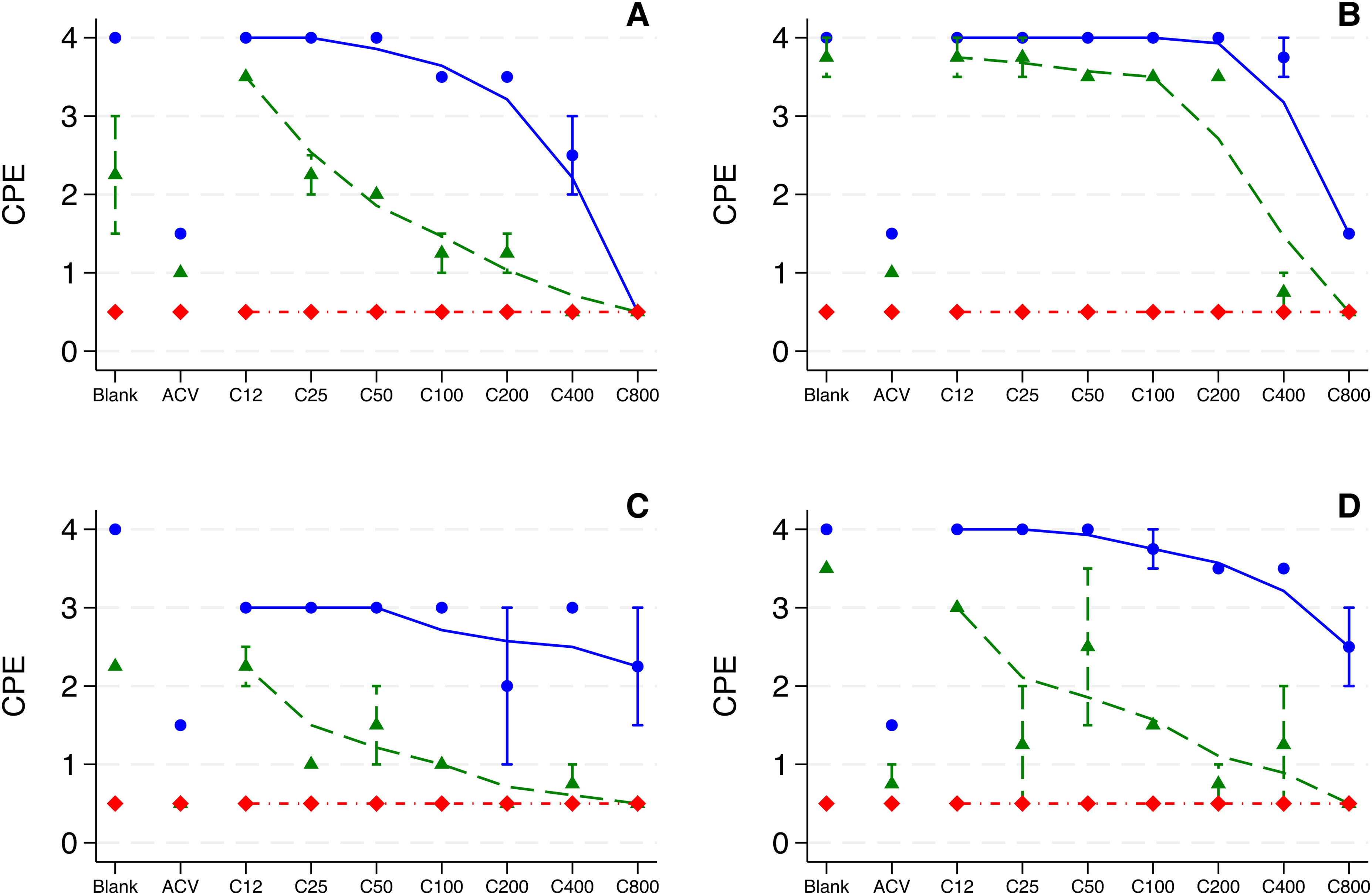
Cytopathic effect (CPE) index as a measure of HSV-1 inactivation by different calcium spirulan (CaSP) concentrations. Higher CPE values indicate lower anti-HSV-1 activity by CaSP. CPE values are the medians of two independent measurements (n = 2).
Pre-Incubation of HSV-1 with CaSP
Similarly, pre-incubating HSV-1 for 3 h with ≥50 µg/mL CaSP led to an inhibition of HSV-1 infection (CPE ≤2) at 48 h when CaSP was added to a viral suspension of 4.56 TCID50. This activity was still present at 72 h with a concentration of >50 µg/mL CaSP. CaSP was not active on the 114 TCID50 at any time (Figure 2C and 2D) and it was not toxic to the cells at any of the investigated concentrations (Figures 2C and 2D, controls). The dose-dependent activity was similar to that observed after preincubating the cells with CaSP.
The results of these two experiments guided us in selecting 200 and 800 µg/mL of CaSP and 48 h incubation time for the HSV-1 inhibition studies.
HSV-1 Inhibition by CaSP
After 48 h of incubation, a CPE ≤2 (ie inhibition of HSV-1 infection) was observed in HaCaT cells with a 12.4 TCID50 HSV-1 titer, when no CaSP was added (Figure 3A). Adding 200 µg/mL CaSP increased cell resistance to HSV-1 penetration to 37.1 TCID50 (Figure 3B). However, sustained inhibition of all HSV-1 titers was only seen at a CaSP concentration of 800 µg/mL (Figure 3C). Pre-treatment of the cells or viral particles with CaSP did not influence the outcomes. In fact, the one-way ANOVA revealed that CaSP concentration (p = 0.0003), and HSV-1 titer (p = 0.0000), were statistically significant factors for HSV-1 inhibition, while the type of pretreatment (p = 0.2511) was not.
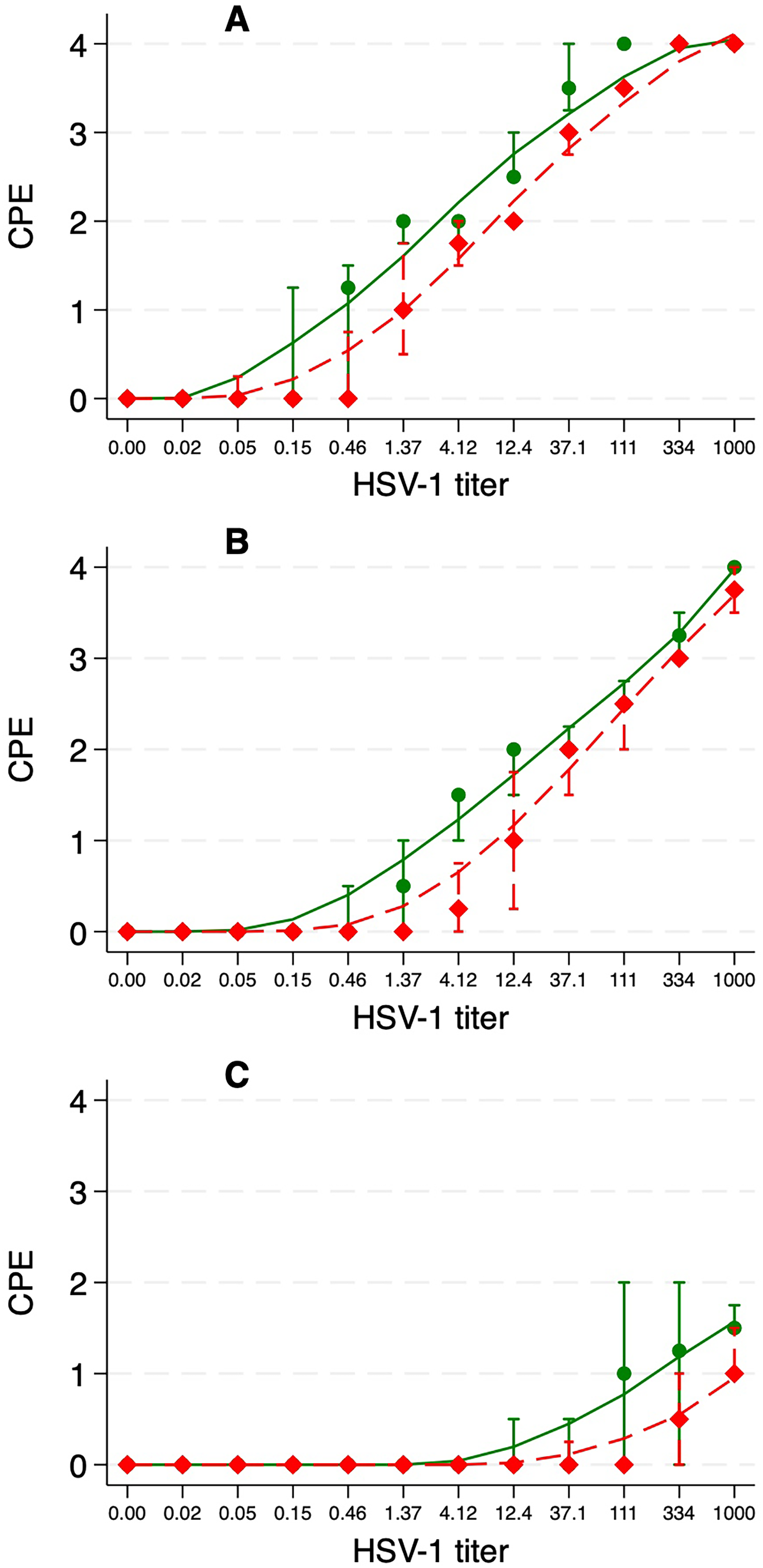
Cytopathic effect (CPE) index versus viral load as a measure of anti-HSV-1 activity by CaSP at concentrations 0 (
We observed a clear reduction in the HSV-1 titer after CaSP treatment as measured by both CPE and IHC measurements, with the concentration of 800 µg/mL CaSP being more active than 200 µg/mL (Table 2).
Reduction of HSV-1 Titers Compared to Control After pre-Incubation of HaCaT Cells or HSV-1 with CaSP. The Reduction Factor has Been Computed Using the Formula R = TCID50(Control)/TCID50(Treatment), Using the Means of Two Measurements (n = 2). CPE, Cytopathic Effect Ivndex; Cells, pre-Incubation of Cells with CaSP; HSV-1, pre-Incubation of HSV-1 with CaSP.

Discussion
The chemical analysis of the CaSP extract used in this study corroborates previous data suggesting that spirulan is a polysaccharide with chains of various lengths. 28 Mass spectrometry revealed significant variation in the CaSP mass, ranging from 400 to 1200 kDa, with a peak at 700 kDa. Molecules of this size are unlikely to penetrate the cell membrane, as shown in a previous study on a different model of transmembrane transport, in which only molecules with a maximum size of approx. 150 kDa could diffuse passively through the cell membrane. 29
CaSP reduces HSV-1-induced cytopathic effects on HaCaT cells, without affecting cell viability. Pretreating human keratinocytes with CaSP or preincubating HSV-1 with CaSP inhibits HSV-1 penetration. These results align with previous observations that the antiviral activity of CaSP depends on the timing of addition during infection: reduced activity is observed when CaSP is added post-infection, while optimal antiviral effects are seen when it is added during infection or shortly thereafter. 20
The efficacy demonstrated by pre-incubating cells or HSV-1 particles with CaSP indicates that CaSP is effective at preventing cell infection. HSV-1 penetration in the cells was inhibited during the first 48 h, indicating that CaSP primarily hinders virus entry, as suggested by previous studies. 30 We observed a statistically significant, dose-dependent activity of CaSP regardless of the pre-incubation method used (p = 0.2511). A dose-dependent relationship was observed at a low (4.56 TCID50) HSV-1 titer, whereas only 400 and 800 µg/ml CaSP were active against the high (114) TCID50 titer.
At a concentration of 800 µg/mL, the efficacy of CaSP against a viral TICD50 of approx. 5 is comparable to that of 200 µg/mL of acyclovir. CaSP's antiviral activity decreases over time, a trait also observed with acyclovir. Acyclovir exhibits significant antiviral activity within the first 48 h, but its effectiveness diminishes over time, especially compared to drugs with a longer intracellular half-life.31,32
However, while the virustatic action of acyclovir has been extensively studied (eg,25,29) the mode of action of CaSP and other sulfated polysaccharides remains unclear. In our cell penetration study, we used heparin as a negative control, because it is a highly sulfated polysaccharide with a strong negative charge due to numerous sulfate groups. Similar to CaSP, heparin does not penetrate cells, 33 yet it exhibits significant antiviral activity against several viruses, most notably SARS-CoV-2 (COVID-19). 34 While heparin is primarily known as an anticoagulant, it is also considered a pleiotropic drug with anti-inflammatory and antiviral properties. 35 As an antiviral agent, heparin binds directly to the viral surface proteins, probably acting as a coating agent. 36 Our results suggest that CaSP, also a sulfated polysaccharide, may act similarly to heparin by inhibiting viral attachment to cell surfaces. 37 The antiviral efficacy of heparin against HSV-1 is directly linked to its sulfation moiety 38 a feature reported, at least indirectly, for CaSP. 20 Mader et al 21 showed that CaSP blocks viral attachment and penetration into host cells. CaSP has also been shown to inhibit cell penetration of a wide variety of enveloped viruses, 17 including HSV-1, human cytomegalovirus, measles, influenza, and HIV-1, with half-maximal effective concentrations (EC50) values in the 0.92–23 mg/mL range. 39 As with other polysaccharides, it is unlikely that CaSP acts via specific receptor binding activities.
This study has some limitations. First, the penetration study was not carried out for a long period of time. Four time points up to 24 h were studied, which may not reflect long-term cellular interactions or degradation. Additionally, the CaSP/HSV-1 interaction has not been studied using molecular assays. Binding assays and ligand-receptor interaction studies similar to those carried out with other compounds,40,41 would help elucidate the mode of action of CaSP further. Finally, this study design does not allow drawing conclusions about the clinical significance of the observed interactions between CaSP and HSV-1. However, the results of Mader et al 21 confirm the efficacy of CaSP in the symptomatic treatment of herpes labialis.
Conclusions
Marine sulfate polysaccharides derived from seaweed and other marine organisms have garnered interest as potential antiviral agents. Their natural abundance, minimal toxicity, and potential as a renewable resource make them important assets that open the doors to safe, innovative antiviral therapies. Our study demonstrates that CaSP does not enter keratinocytes, thus not interfering with the cell metabolic activities, and it provides further evidence of a non-toxic interaction between CaSP and human keratinocytes, which prevents HSV-1 infection.
Taken together, our results support the use of CaSP in the topical prophylaxis and treatment of recurrent HSV-1 infections. 42
Footnotes
Abbreviations
Acknowledgements
Orlando and Liliane Petrini (Breganzona, Switzerland) critically reviewed the manuscript.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
CZ had the idea for this study and supervised its implementation together with NG. AL and MJM planned and supervised the scientific aspects of the study. NA, DE, LN-P, SP, and MJ performed and analyzed the experiments with keratinocytes. AL, NA, SP, NG, and CZ prepared the manuscript and reviewed the results. All authors were involved in the preparation and review of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Cesra Arzneimittel GmbH & Co. KG, Flugstrasse 11, 76532 Baden-Baden, Germany.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christian Zimmermann and Nils Günnewich are employees of Cesra Arzneimittel GmbH. Deniz Elyorgun, Nuril Azizah and Antje Labes received a project-specific grant. Silvia Pezer, Larissa Nkenmei-Pietsch and Marina Jorge Miranda work for Tentamus Pharma & Med Deutschland GmbH, a contract research organization that specializes in preclinical research.
Data Availability Statement
Data from the study are available from the corresponding author upon justified request.
