Abstract
Objective
Different extraction vehicles are traditionaly used for herbal extraction. Despite its frequent use in modern cosmetic and pharmaceutical formulations, there is a lack of scientific data to support safety of propilene glycol (PG) plant extract for topical use.
Methods
To estimate skin safety profile of PG as extraction solvent in herbal extracts for topical use regarding the skin irritation potential, using Alchemilla vulgaris PG extract as an example. In vitro cytotoxicity alongside in vivo effects on human skin employing biophysical measurements (TEWL, erythema index and pH) were assessed.
Results
In vivo/in vitro testing showed a satisfactory safety profile of both PG per se and tested PG extract.
Conclusion
PG was shown to be a suitable solvent for obtaining bioactive plant extracts without causing serious skin irritation and suitable for topical formulations correspondent to the therapeutic need.
Introduction
Propylene glycol (PG), propane-1,2-diol, is a clear, viscous hygroscopic liquid, an organic compound used by the chemical, food, and pharmaceutical industries. It is very common in dermopharmaceutics and cosmetics as a skin conditioning and viscosity decreasing agent. The European Medicines Agency (EMA) reported that PG could be used as a humectant in topicals, a preservative in solutions or as a co-solvent. 1
Since PG is highly presented, the route of introduction into the body is multiple and the public concern about its safety is understandable. EMA states that PG is not acutely harmful after ingestion or skin contact and that dermal absorption through the intact skin is expected to be very limited. 1 The Cosmetic Ingredient Review (CIR) Expert Panel concluded that PG is safe for use in cosmetic products at concentrations up to 50%. 2
On the other hand, PG poses an intriguing scientific and regulatory conundrum with respect to allergic contact dermatitis (ACD). 3 Uncertainty in PG potential to induce skin irritation and sensitisation contributes its bad reputation as an excipient in topicals, particularly cosmetics. Nowdays, in order to emphasize a specific benefit of cosmetic it is often claimed that it is “propylene glycol free”, together with „paraben free“ or „mineral oil free“ claims.
Plants offer wide ranges of therapeutic applications and plant extracts are being used extensively in topicals. The quality of herbal products is greatly affected by the extraction procedure. The choice of solvent, co-solvent and method of extraction is crucial for achieving the desired therapeutic goal. PG and glycerin are solvents employed in herbal industry for their additional benefits 4 - bacteriostatic and fungistatic properties, and solubility improvement of some ingredients. 5 PG has been also used as penetration enhancer and humectant in semi-solids including dermocosmetics. When used as extraction solvent, PG improves anti-inflammatory, antibacterial, and antioxidant properties of some herbal extracts. Our previous study pointed that classical maceration procedure with 80% PG led to extraction of significant amounts of valuable phenolic compounds. 6 Despite its obvious synergism with herbal ingredients in the treatment of common skin issues, there have been safety concerns regarding the PG skin irritation potential which could limit the use of plant PG extracts. 2
Lady's Mantle (Alchemilla vulgaris L. syn. Alchemilla xanthochlora Rothm., Rosaceae) is used in folk medicine to heal skin disorders. Recent in vitro and in vivo studies provided data about Alchemilla species broad spectrum of biological activities. 7 We previously reported the wound healing potential of A. vulgaris extracts obtained with different extraction solvents, 6 and presumed that PG could show some additional benefits on skin. In our knowledge, there were no studies investigating the irritation potential of PG in vivo/in vitro, nor investigating the same aspect of herbal extracts commonly used for topical application. Lady's Mantle (A. vulgaris L.) was used as an example due to its traditional use in treating skin disorders and its rich phytochemical composition. Therefore, the study objectives were to determine the suitability of PG as an extraction solvent for herbal extracts intended for topical application regarding skin irritation potential, by: (1) evaluating the in vitro cytotoxicity of A. vulgaris PG extract using L929 fibroblast cells; and (2) assessing the in vivo effects of PG and PG extract on human skin by measuring biophysical parameters (TEWL, erythema index, and pH) under occlusive conditions.
Materials and Methods
Plant Material and Preparation of the Extracts
Commercial source of the Alchemilla vulgaris L. sample (Alchemillae herba) (ID 302041) was the Institute of Medicinal Plants Research “Dr Josif Pančić”, Belgrade, Serbia. The plant was identified at the Faculty of Pharmacy, University of Belgrade, Department of Botany, by prof Branislava Lakušić. The voucher specimen was deposited (No 02290117). Dry plant material was reduced to a fine powder and extracted with PG (pharmaceutical grade) by maceration, as described in the European Pharmacopoeia 11.0. It was used as liquid macerate (DER 1:5, 80% PG, m/m) and marked as AP.
In Vitro Viability and Cytotoxicity Assays
For in vitro examination a mouse fibroblastscell line (L929, ATCC (Washington DC, USA) was used. All cell culture reagents were purchased from Capricorn Scientific GmbH, Germany.
Viability assay was performed as described in our study. 6 The study was conducted in October 2017. at the Department for Cell and Tissue Engineering, Scientific Research Center for Biomedicine, Faculty of Medicine, University of Niš. Briefly, cells were seeded in 96 well plates (Greiner Bio-One, Germany) at density 2 × 104 cells per well where extract was added in five different concentrations per tested sample 24 h after cultivation of cells. Propylene glycol A. vulgaris extract (AP) diluted in complete DMEM was tested in concentration range from 0.3125 to 50 mg/mL. Control (C) contained control cells grown only in culture medium. Propylene glycol was tested alone as PG 80% and PG 45% extraction solvent. MTT test was performed after the incubation period of 24 h.
The percentage of viable cells was calculated according to the following formula:
In Vivo Skin Performance—EI, pH and TEWL Measurements
In vivo skin effects were assessed via parameters – TEWL (often used in support of claims of product mildness), erythema index (EI) and pH.
8
Short-term study was conducted at the Pharmacy department, University of Niš-Faculty of Medicine, Niš, Serbia over a 24
For a safety assessment, parameters were measured prior to (baseline values-first day) and 60 min upon cessation of 24 h occlusive treatment (second day). 12 healthy female volunteers (23.4 ± 1.1 years) were recruited. Two spaces on the flexor side (3 × 3 cm) of the left forearm were treated, one with 10% PG water solution (10%PG), and the other with PG extract of A. vulgaris (10% water solution, AP). Additional site on the right forearm was left as a non-treated control under occlusion (UCO). Samples were applied using a graduated plastic syringe in quantities of 0.016 g/cm2 (approximately 0.16 mL for the treated area) and spread vigorously, skin sites were immediately covered with Parafilm® and then with cotton adhesive tapes.
In vivo measurements were performed in accordance with the Declaration of Helsinki, and the volunteers signed a written consent. They were informed of the study and instructed. All subjects had healthy skin without allergies. Before any measurements were taken, the subjects were asked to acclimatize for 30 min under controlled conditions (21 ± 1 °C and 50 ± 5% RH). TEWL was measured using Tewameter®TM 210, EI using Mexameter®MX 18, pH using pHmeter®900 (all Courage + Khazaka, Germany).
This double-blind study was performed using non-invansive method, approved by the local Ethical Committee on Human Research of the Faculty of Medicine in Niš (Approval number 12-8818-2/3).
Statistics
All results are presented as mean values ± standard deviation.
For in vitro study each concentration of sample was examined in four to eight replicates.
The results for each of investigated samples in vivo were compared mutually and related to UCO. The values of measured parameters after occlusion removal were compared to appropriate baseline values using paired sample t-test. Data obtained from skin sites treated with different samples were compared mutually and to UCO using t-test for unpaired data.
Software SPSS version 18.0 was used. The differences were accepted as statistically significant at P < .05.
Results
In Vitro Viability and Cytotoxicity
The results of MTT test are shown in Figure 1. Only high examined concentrations of the PG extract (5-50 mg/mL for AP, Figure 1b) were cytotoxic for cells, with a statistically significant reduction of cell viability. Concentration-dependent cell viability was observed. It was shown on Figure 1a that when PG alone was examined as an extraction solvent, PG80 (final PG conc. 4%) reduced cell viability to 55.53 ± 11.2%, while the same dilution of PG45 (final PG conc. 2.8%) reduced cell viability to 74.44 ± 7.67%. The dilution of tested PG alone corresponds to the concentration of PG in the plant extract of 5 mg/mL and 2.5 mg/mL. No significant cytotoxic effects were observed at concentrations below 2.5 mg/mL of extract (Figure 1b).
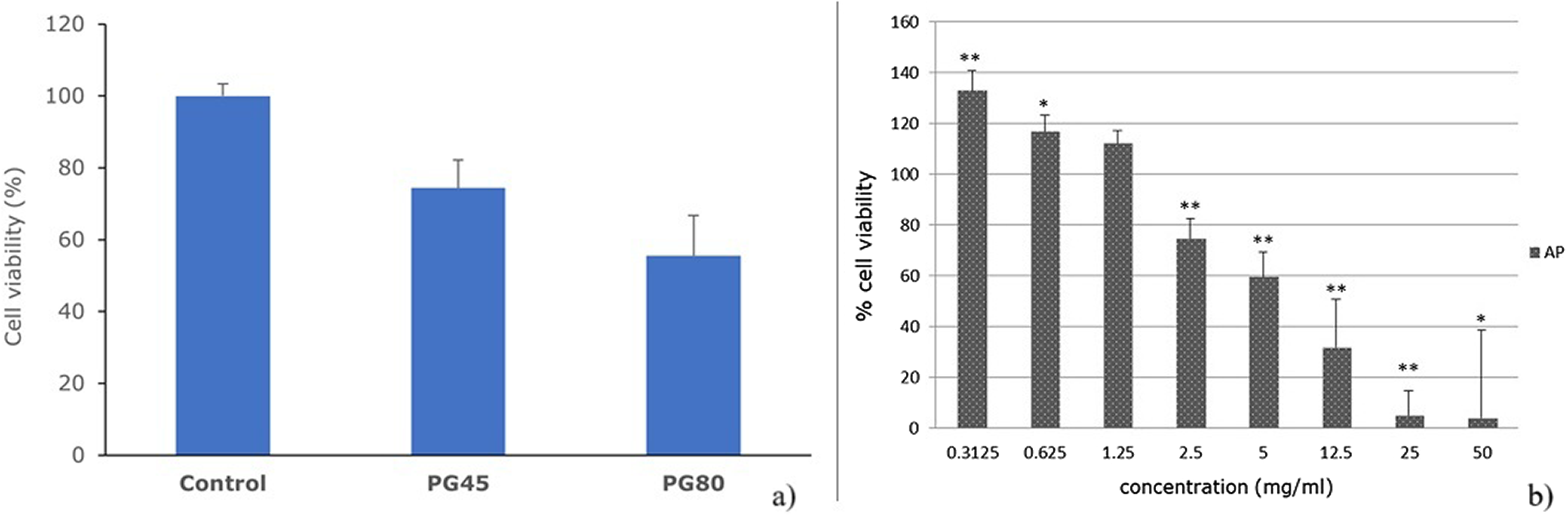
The effects of (a) PG solutions per se and (b) AP on the viability of L929 cells after 24 h of incubation, evaluated by MTT test; **P < 0.001, *P < 0.01.
In Vivo Skin Performance
The relative changes of the examined biophysical parameters (TEWL, EI, and pH) compared to their corresponding baseline values are presented in Table 1. Investigated samples showed changes for all parameters after 24 h occlusion, but without statistically significant changes compared to the controls. The results indicate that a 24-h occlusion with both the 10% PG solution and the A. vulgaris PG extract (AP) led to minor fluctuations in skin barrier function and redness. Specifically, a slight increase in TEWL and EI values was observed for both treated sites compared to the untreated control under occlusion (UCO), likely due to the occlusive effect itself. However, statistical analysis revealed that these changes were not significantly different from the control site (UCO) or between the treated sites (P > .05). Similarly, the pH values showed a decrease across all sites, consistent with the occlusion effect, but no statistically significant acidification was induced by the PG samples compared to the control. These findings demonstrate that neither the solvent vehicle nor the herbal extract caused measurable skin irritation or disruption of the skin barrier integrity under the test conditions.
Relative (%) Changes of Parameters Measured Before and After 24 h Occlusion Study.

Discussion
Adverse drug reactions may also be due to an excipient. 9 The potential of PG for skin irritation was especially of concern under occlusive conditions, and it could be concentration dependent. Patch testing in humans already pointed that PG is a weak allergen under certain conditions, and an uncommon cause of ACD. On the other hand, PG is negative in predictive toxicology tests for skin sensitisation, which could present an intriguing inconsistency. 3 The focus of this study was on the investigation of PG as an extraction solvent for topical formulations. A. vulgaris L. extract was used as an example because of its rich phytochemical composition and because various species of Alchemilla L. (Rosaceae) are used in traditional medicine for wound healing. 10
In the in vitro assay, the lack of significant cytotoxic effects below 2.5 mg/mL suggests that the extract is safe at concentrations usual for topical use. Furthermore, other extracts examined in the same study 6 (where ethanol and water are used for extraction) were significantly cytotoxic even in much lower examined concentrations, which suggests that PG could be used as a solvent for biologically active plant extracts for topical use, less cytotoxic than other organic solvents.
Regarding the in vivo study, the absence of statistically significant changes implies that the samples are not expected to cause skin irritation.8,11 The results for in vitro cytotoxicity (Figure 1), indicative parameter for skin irritation potential investigations, 8 were in line with the results for in vivo measurements of biophysical parameters that are directly related to the structure and function of the skin (Table 1).
Conclusion
In accordance with the stated study objectives, this research successfully determined the suitability of propylene glycol (PG) as an extraction solvent for herbal extracts regarding skin irritation potential. First, by evaluating the in vitro cytotoxicity, we demonstrated that the Alchemilla vulgaris PG extract exhibits a satisfactory safety profile on L929 fibroblast cells, maintaining cell viability at concentrations relevant for topical application. This suggests that PG extracts can be safe alternatives to more cytotoxic organic solvents. Second, by assessing the in vivo effects on human skin, we confirmed that neither PG per se nor the PG extract induced significant alterations in crucial biophysical skin parameters—TEWL, erythema index, and pH—even under rigorous occlusive conditions. Consequently, based on these complementary findings, we can conclude that PG is a highly suitable extraction solvent. It allows the formulation of dermopharmaceutics and cosmetics containing bioactive plant extracts safe and effective in topical application, minimizing the risk of skin irritation while meeting therapeutic needs. This is the first study, to our knowledge, to investigate skin irritation potential of PG as an extraction solvent and PG herbal extract using both in vivo/in vitro methods.
Footnotes
Acknowledgments
Authors would like to thank the Ministry of Education, Science and Technological Development of the Republic of Serbia Ministry of Science, Technological Development and Innovation of the Republic of Serbia (No. 451-03-136/2025-03/200113 and 451-03-137/2025-03/200113).
Ethical Approval
This study was approved by the local Ethical Committee on Human Research of the Faculty of Medicine in Niš (Approval number 12-8818-2/3).
Statement of Informed Consent
The volunteers signed a written consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja, (grant number No. 451-03-136/2025-03/200113, No. 451-03-137/2025-03/200113).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

