Abstract
Background
The development of antimicrobial resistance has become a global health concern, leading to significant challenges to the effective management of infectious diseases caused by pathogenic microorganisms. Mangifera zeylanica is an endemic plant in Sri Lanka, traditionally used in ayurveda for its antimicrobial benefits, holds promise as an alternative therapeutic agent. This study investigates the antimicrobial activity of M. zeylanica leaf extract and its potential incorporation into a topical cream formulation.
Methodology
The antimicrobial activity of ethyl acetate and ethanol leaf extracts of M. zeylanica were investigated against five test microorganisms: Staphylococcus aureus, Escherichia coli, Candida albicans, Pseudomonas aeruginosa, and Staphylococcus epidermidis using agar well diffusion assay. A topical cream formulation incorporating ethanol leaf extract at a concentration of the 25 mg/mL was developed and assessed for its physiochemical properties.
Results
Both leaf extracts exhibited antimicrobial activity with ethanolic leaf extract showing a higher ZOI against all test microorganisms than ethyl acetate leaf extract. For the ethanolic leaf extract, S. aureus had a significantly lower ZOI at each concentration, compared to the positive control, ampicillin (37.7 ± 0.3 mm at 0.01 mg/mL), while C. albicans showed a significantly higher ZOI at each concentration than its positive control, gentamycin (16.1 ± 0.3 mm at 0.01 mg/mL). The ethanolic leaf extract had MIC and MBC of 12.5 mg/mL. GC-MS and FTIR analysis of the ethanolic leaf extract identified 1,2,3-benzenetriol (86.75%) as the major bio-active compound contributing to the antimicrobial activity. The formulated cream showed ideal spreadability (10.17 g.cm/s), smooth texture and stability, with no phase separation observed during the storage for 14 days and 90 days.
Conclusion
The ethanolic leaf extract of M. zeylanica demonstrated promising antimicrobial activity against tested bacterial strains and fungal strains, indicating its potential as a therapeutic agent for topical applications.
Introduction
The rapid spread of antibiotic resistance has become a critical global health concern, significantly hindering the effective management of infectious diseases caused by pathogenic bacteria and fungi. 1 Microbial infections are responsible for millions of deaths every year, with recent data indicating that 7.7 million deaths worldwide, which accounts 13.6% or one in eight of all global deaths, were associated with bacterial infection. 2 Thus, the development of novel antimicrobial dosage forms has become a major focus of pharmaceutical research to combat multidrug-resistant bacterial, viral, and fungal strains. 3 Plant-derived natural products, demonstrated a pivotal role in treatment and prevention of diseases since ancient times. However, the extensive use of synthetic antimicrobial agents led to several adverse effects, including emergence of antimicrobial resistance, 4 toxicity 5 and environmental contamination, 6 disrupting ecosystems and making infections harder to treat. Even though synthetic antimicrobial agents have been widely approved and used in many countries, there is a growing interest in scientific research towards the natural compounds derived from microorganisms, animals or plants. 7 Theses bio-active compounds have shown promising results in combating antibiotic resistance against bacterial pathogens. 8 Plants produce a wide array of secondary metabolites that exhibit beneficial biological activities, including antioxidant, antibacterial, and antifungal properties.9,10 Many of these compounds can restore the clinical application of present antibiotics by enhancing their potency and overcoming the mechanism of resistant development. 10 According to the World Health Organization, plant extracts or their active constituents are used in traditional medicine by 80% of the global population and over 50% of all modern clinical drugs are derived from natural products. 11
Mangifera zeylanica (known as “Etamba” and “Sri Lankan wild mango”), belongs to the Anacardiaceae family and genus Mangifera, is an evergreen tree endemic to Sri Lanka. 12 The dark green leaves are stiffly coriaceous, glabrous, and they emit a distinct mango aroma when damaged. 13 The leaves are dispersed while partly aggregating at the end of twigs. 14 The ripe fruit is yellowish in color with a reddish flush. The plant mainly grows in forests of the intermediate and wet zones up to elevations of 800 meters; however, it can also be found in the dry zone along waterways and moist valley areas. 15 Based on the 2012 National Red List of Sri Lanka, 16 M. zeylanica is categorized in the national conservation status ‘Least Concerned’, and it holds global conservation status as ‘Vulnerable’. 17
It has been reported that different parts of this plant have been traditionally used in various cultures to treat a wide range of issues. 18 The traditional Sri Lankan medicine utilizes the bark of M. zeylanica to treat a variety of maladies, including bladder infections, diarrhea, oral diseases, hiccups, vomiting and cancer, present in the uterus and bladder. 19 M. zeylanica leaf juice was utilized in the Ayurveda treatment for infections caused by bacteria and fungi, and acne. 20 The bark of M. zeylanica produced several compounds. 21 Additionally, two novel halogenated compounds were isolated: bromomangiferic acid and chloromangiferamide. 22 Two previously recognized compounds, quercetin and catechin, were also found. 22 Therapeutic activities of different parts of the tree have been investigated: ie, anticancer activity of bark, 22 cytotoxicity against cancer cell lines, 22 antioxidant and apoptotic properties of fruit peel, 23 anti-bacterial activity of stem bark, 24 sun protection activity of stem bark. 25
M. indica, another species within the same family, has been extensively studied for its therapeutic applications. 26 In contrast, M. zeylanica remains relatively unexplored despite its longstanding use in Ayurveda treatments in Sri Lanka. The gap in research is notable, as M. zeylanica could consist of distinct medicinal properties that have not yet been discovered by modern science. Thus, exploring the antimicrobial activity of M. zeylanica leaves offers an opportunity to expand the scope of medicinal plant research and new therapeutic agents. The pre-clinical studies have well-documented the potential anti-acne agent, 27 and antibacterial properties 28 of the ethanol extract from M. indica leaves. Moreover, the potential use of M. indica leaf extract in topical formulation preparations targeting of antibacterial and anti-acne actions also has been studied. 28 However, according to our knowledge, no studies have evaluated M. zeylanica leaves for antimicrobial properties or investigated their potential for use in antibacterial cream formulation. Therefore, this study was designed to investigate the antimicrobial activity of leaf extracts of M. zeylanica and the preparation of a topical formulation to treat skin infections caused by microbes.
Material and Methods
Chemicals and Bacterial Cultures
Chemicals
The hexane 99%, and ethyl acetate 99.5%, purchased from Sisco Research Laboratories PVT. Ltd, Mumbai, India, and ethanol 99%, purchased from Supelco, Darmstadt, Germany were used for the extraction process. Barium chloride anhydrous (Loba chemie (PVT) Ltd, Mumbai, India) and sulphuric acid 96% (Supelco, Darmstadt, Germany) were used for the preparation of McFarland standard. Dimethyl sulfoxide (DMSO) and 99.9% propylene glycol (USP) purchased from Heiltropfen, London was used to prepare a concentration series of ethyl acetate leaf extract.
The culture media: Mueller-Hinton agar, Bacteriological agar and Mueller–Hinton broth were obtained from HiMedia, Kennett Square, USA. Ampicillin, gentamycin, nystatin, and clindamycin purchased from Bioanalyse, Ankara, Turkey were employed as standard antibiotics in the study.
Bacterial Cultures
Pathogenic reference strains of Staphylococcus aureus (ATCC 25923), Escherichia coli (ATCC 25932), and Candida albicans (ATCC 10231) were obtained from the Department of Pharmacy and Pharmaceutical Sciences, Faculty of Health Sciences, CINEC Campus, Malabe, Sri Lanka. Pseudomonas aeruginosa (ATCC 27953) was obtained from Microbiology Laboratory, Bureau Veritas, Rathmalana, Sri Lanka. Staphylococcus epidermidis (ATCC 12228) was obtained from the Industrial Technology Institute (ITI), Colombo 07, Sri Lanka.
Collection and Preparation of Plant Material
Leaves of the plant M. zeylanica were collected from Yakkala area in Gampaha District, Sri Lanka (7°06'03.5"N 80°02'22.4"E https://maps.app.goo.gl/79SFQ1qJVffKVhsDA?g_st = iw (Figure 1). The plant was identified and authenticated by the National Herbarium, Department of National Botanic Gardens, Sri Lanka. The fresh leaves of M. zeylanica were thoroughly washed with tap water to remove surface impurities, then air-dried until a constant weight was reached. The dried leaves were ground into powder using a kitchen-scale blender and stored in airtight containers at room temperature until use.

Leaves of the Plant M. zeylanica.
Preparation of Plant Extract
Plant extracts were prepared using a reflux extraction method based on the procedure described by Chua, Latiff and Mohamad, 2016,
29
with slight modifications. Ten grams of leaf powder was sequentially reflux extracted using three different solvents: i) hexane at 60 °C for 30 min, ii) ethyl acetate at 65 °C for 30 min, and iii) ethanol at 70 °C for 30 min, in a round-bottom flask. The resulting solvent extracts: i) hexane extract (HE) ii) ethyl acetate extract (EAE) and iii) ethanol extract (EE), were suction filtered, and the obtained filtrates were concentrated to dryness using rotary evaporation. The solid crude extracts of HE, EAE and EE yielded 0.5%, 2.1%, and 15.4% of the weight of dry leaf powder, respectively.
The leaf extracts, EAE and EE, which yielded in quantifiable amounts were selected for further study. Stock solutions of EE and EAE were prepared at 200 mg/mL concentration by dissolving EE in sterile distilled water and EAE in 1% DMSO in sterile distilled water. Subsequent dilutions were prepared using appropriate culture media.
Antimicrobial Assay
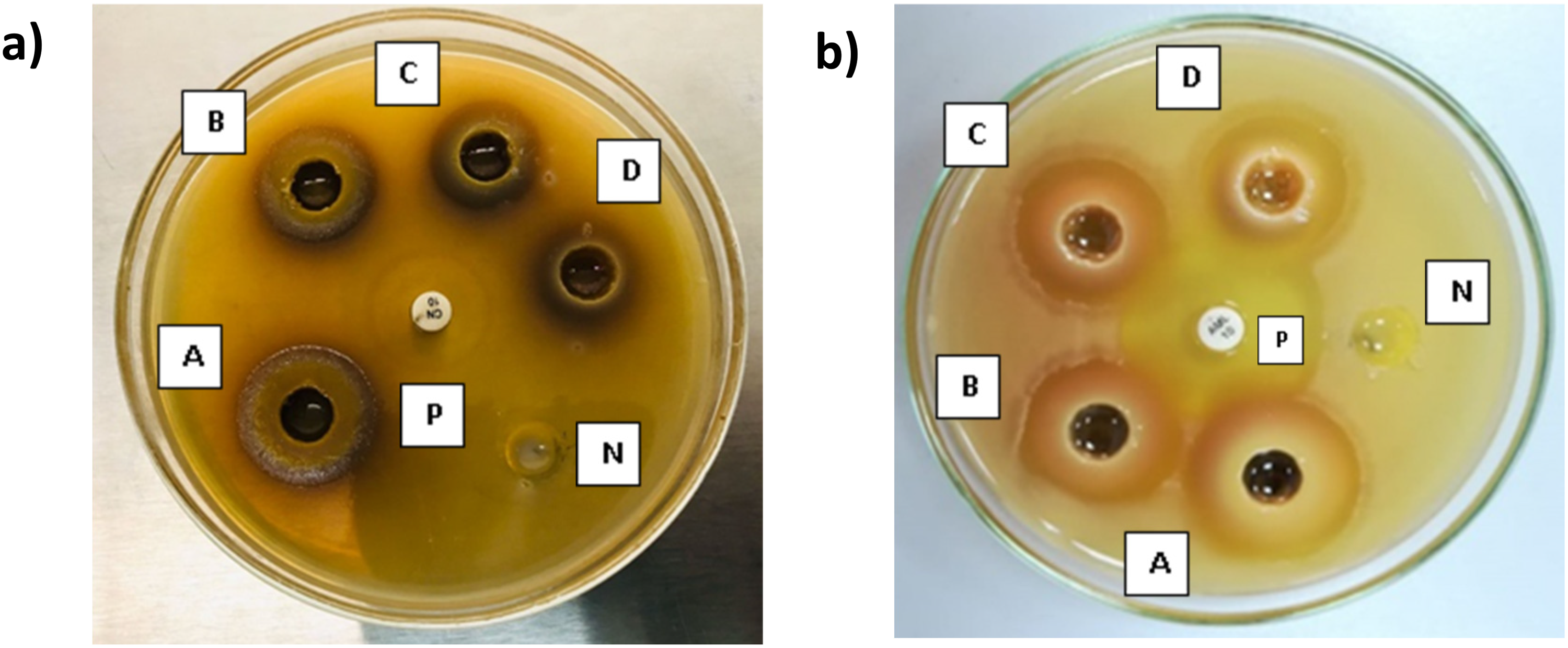
Ethanol extract (EE) and Ethylacetate extract (EAE) were screened for the antimicrobial activity against S. aureus, P. aeruginosa, E. coli, C. albicans and S. epidermidis by agar well diffusion assay following the methods of Valgas et al, 2007 and Magaldi et al, 2004 with slight modifications.30,31 The bacterial suspensions equal to 0.5 McFarland turbidity standard were prepared in 0.9% (w/v) sterile saline using 18 h-old pre-cultures and inoculated on to Muller Hinton agar (MHA) plates. Six wells (6 mm in diameter) were made in each MHA plate using a sterile cork borer, and the bottom of the wells were sealed with a drop of MHA. Each plate was treated with 200 µL of four concentrations of the plant extract (200, 100, 50, 25 mg/mL), a positive control (standard antimicrobial compounds) and a negative control (sterile distilled water for EE and 1% DMSO for EAE). The positive controls were ampicillin (0.01 mg/mL), gentamycin (0.01 mg/mL), nystatin (0.1 mg/mL), gentamycin (0.01 mg/mL), clindamycin (0.002 mg/mL) respectively for S. aureus, P. aeruginosa, E. coli, C. albicans and S. epidermidis culture plates. The plates were incubated at 37 °C for 24 h and then the zone of inhibition (ZOI) was measured. The assays were performed in triplicate (Figure 2).
Determination of minimum Inhibitory Concentration
The minimum inhibitory concentration (MIC) was determined according to the method described by of Elshikh et al, 2016. 32 A stock solution of the plant extract was prepared at 25 mg/mL concentration in MHB and a two-fold serial dilution was performed to obtain a concentration range of 12.5 mg/mL – 0.02 mg/mL. Then 100 µL from each dilution was added to columns 1 to 10 of a 96-well microtiter plate in triplicate. The inoculum of each test microorganism was prepared by adjusting freshly cultured cells to match 0.5 McFarland turbidity standard (1.5 × 108 CFU/mL) and then diluted by 1:100 in MHB to achieve approximately 5 × 105 CFU/mL in a well. A 100 µL of the diluted inoculum was added to each well from column 1–10 and mixed well. Column 11 was served as the positive control (containing diluted bacterial suspension without plant extract), while column 12 served as the negative control (containing uninoculated MHB). Upon completion of 24 h incubation at 37 °C, 30 µL of resazurin (0.015%) was added to each well to detect microbial growth. The plates were further incubated for an additional 2 h at 37 °C. MIC was determined by the lowest concentration of the extract that prevented a color change of resazurin from blue to pink.
Minimum Bactericidal Concentration
The contents in the wells that indicated the change in resazurin color from blue to pink (under the section 2.5) were inoculated on MHA plates and incubated for 24 h at 37 °C. Minimum bactericidal concentration (MBC) was defined as the lowest concentration of the extract at which no visible microbial colony growth was observed on the agar surface.
Procedure for GC-MS Analysis
The Gas chromatography-mass spectrometry (GC-MS) analysis was determined according to the method described by of Ezhilan and Neelamegam, 2012 with slight modifications. 33 The ethanol extract which demonstrated the highest ZOI was analyzed by GC-MS using a HP-5 MS capillary column (5% Phenyl methyl Siloxane) (30 m × 250 μm, i.d., 0.25 μm film thickness) in an Agilent 6890N gas chromatograph, with electron ionization (EI). Helium was employed as the carrier gas at a constant flow rate of 1.0 mL/min. The oven temperature was initially maintained at 60 °C for 10 min and then ramped to 230 °C at a rate of 3 °C/min. A1.0 μL aliquot of the sample, prepared as a 10 fold dilution of a 25 mg/mL ethanol extract in ethanol, was injected in splitless mode. The total run time of the sample analysis was 67 min. The compounds were identified by comparison of the resulted mass spectra with those in the NIST 14 L mass spectral library. The compounds with Match factor (MF) and the Reverse Match Factor (RMF) values above 800 were considered reliably identified.
Procedure of FTIR Analysis
The Fourier-transform infrared (FTIR) analysis was performed according to the method described by Durak and Depciuch, 2020. 34 An ATR-FTIR spectrometer (Thermo Scientific – Nicolet iS10) was used to analyze the crude ethanol and ethyl acetate leaf extracts in solid form. The recording range of the spectrum was 525–4000 cm−1 at 16 scans per minute with a resolution of 4 cm−1 in transmission mode. The solid crude extracts were analyzed without any additional processing and were directly placed on the sample holder for measurement.
Development of Skin Cream Formulation
Development of skin cream formulation was designed following methods of Chen, Alexander and Baki, 2016, with modifications. 35 Primary emulsion was prepared by mixing oil, distilled water, and Tween 80 at randomly selected five different ratios and their stability was monitored at room temperature for 15 days to observe phase separation. The most stable ratio was employed in the preparation of secondary formulation. The secondary emulsion was developed by melting the two emulsified agents at 70 °C followed by adding Tween 80 and oil to prepare the oil phase and stirred well by the homogenizer at 500 rpm. 25 mg/mL of ethanolic plant extract, dissolved in water, and other ingredients including glycerin, preservatives, and fragrance, were added dropwise while stirring and homogenized at 1000 rpm for 1 h. Then the mixture was cooled to room temperature, it was stirred at 500 rpm, until the creamy texture was obtained. Developed cream formulation (10 g) was composed of emulsifying agent 1 (1.5 g), emulsifying agent 2 (0.5 g), Tween 80 (0.5 mL, virgin coconut oil (2.0 mL), distilled water (5.5 mL), glycerin (1 drop), preservative (1 drop), lavender oil (1 drop).
Evaluation of Physical Properties
The prepared formulation was tested for physical appearance, color, texture, phase separation, and homogeneity by visual observation. Phase separation was tested by keeping and observing preparation for 14 days at room temperature. Homogeneity and texture were determined by pressing a small quantity of the formulated cream between the thumb and index finger. A prepared blank formulation without plant extract was tested for comparison. 36
pH Value
The pH value of the cream formulation was measured by pH meter (Hanna HI5222) at 27 ± 2 °C in triplicate according to the SLS standard for skin creams: SLS: 743:2014.
Spreadability
The spreadability was determined by applying pressure on the cream sample placed between two glass plates. The cream preparation (1 g) was placed on the center of the lower plate, and the standard weight of 500 g was applied to the upper plate. The spreading diameter of the sample between two horizontal glass plates was measured after five minutes. The average diameter was taken after triplicate measurements.
The spreadability (S) can be calculated using the following equation,
Where; S = Spreadability (cm2); A = Area of the formed circumference (cm2); π = Constant (3.1416); r = Radius of the circumference formed by the emulsion (cm). 37
Viscosity Measurement
Viscosity was measured as per ASTM D 2196-05 using a Brookfield viscometer (DVE) by measuring the 81.1% torque on a 28-spindle rotating at a 0.6 constant speed. 38
Centrifugation Test
A cream sample of 5 g was placed in a centrifugal tube and centrifuged at 4000 rpm for five minutes. After centrifugation, phase separation in the cream formulation was observed after five minutes to evaluate the formulation's stability. 39
Thermal Stability
Thermal stability was done following methods of Chandrasekar et al, 2018, with modifications. 40 The cream formulation was kept in the hot air oven at 45 °C for 07 days and observed for any changes to evaluate the stability of the formulation. For long term stability testing, the developed cream formulation was stored at a normal room temperature for 90 days.
In Vitro Antimicrobial Activity of the Formulation
The antimicrobial activity of the cream formulation was evaluated against five test microorganisms: S. aureus, P. aeruginosa, E. coli, C. albicans and S. epidermidis using the agar well diffusion assay.30,33 The wells were created on MHA plates using a sterile cork borer, and the base of each well was sealed with MHA. The cream formulation of 0.9 g was loaded into each well. Four wells were prepared per plate: two wells containing antimicrobial cream formulation with preservatives and two containing antimicrobial cream formulation without preservatives. Standard discs of antibiotics: ampicillin (0.01 mg/mL), gentamycin (0.01 mg/mL), nystatin (0.1 mg/mL), gentamycin (0.01 mg/mL), clindamycin (0.002 mg/mL) were placed at the centre of the agar plates to serve as positive control. Test plates microorganisms were incubated at 37 °C for 24 h, and the average diameters of ZOI were measured.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism version 10.4.1 software. Data were presented as mean ± standard deviation (SD). Statistical comparisons between the average ZOI of test concentrations against test microorganisms and their positive controls were carried out using one-way analysis of variance (ANOVA) followed by Dunnett's multiple comparison test. A value of p < 0.05 was considered statistically significant.
Results
Antimicrobial Activity of Ethanol and Ethyl Acetate Leaf Extracts
The growth of all test microorganisms was inhibited by both leaf extracts (EE and EAE) as evidenced by the formation of ZOI on the agar plates across all the tested concentrations (25-200 mg/mL). However, ethanolic leaf extract has shown a greater ZOI against all test microorganisms compared to ethyl acetate leaf extract. Interestingly, for both extracts, the maximum antimicrobial activity was recorded at the highest tested concentration (200 mg/mL), while the lowest activity corresponded to the lowest concentration (25 mg/mL), as shown in Figure 2.
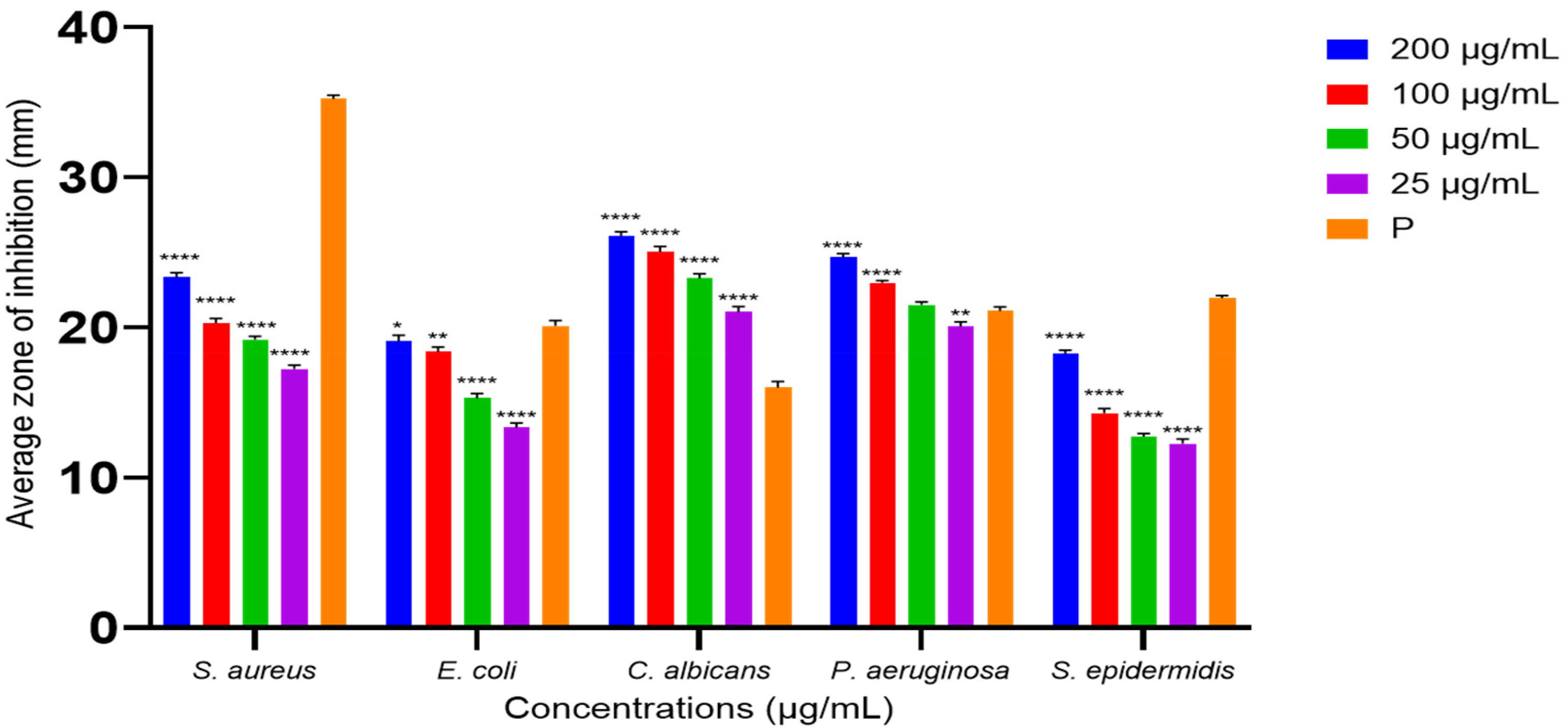
Figure 3 illustrates the ZOI of ethanolic leaf extract against test microorganisms. S. aureus showed significantly lower ZOI at each test concentration compared to its positive control; ampicillin disc with ZOI of 37.7 ± 0.3 mm (0.01 mg/mL), while C. albicans showed significantly higher ZOI at each concentration compared to positive control; Gentamycin (0.01 mg/mL) with ZOI of 16.1 ± 0.3 mm. The other test microorganisms responded differently at each concentration but showed significantly higher ZOI at the concentration of 200 mg/mL, with the average inhibition zones of 20.1 ± 0.4 mm for E. coli and 25.3 ± 0.5 mm for P. aeruginosa, compared to their positive control: gentamycin (0.01 mg/mL) with ZOI of 19.0 ± 0.4 mm and 20.2 ± 0.3 mm respectively. However, S. epidermidis exhibited a lower ZOI of 22.0 ± 0.4 mm at 200 mg/mL, compared to its positive control; clindamycin (0.002 mg/mL), with a ZOI of 21.3 ± 0.2 mm. Figure 4 indicated the ZOI of ethyl acetate leaf extract against test microorganisms. S. aureus, E. coli, and S. epidermidis exhibited significantly lower zones of inhibition in comparison to positive controls; ampicillin (0.01 mg/mL), gentamycin (0.01 mg/mL) and clindamycin (0.002 mg/mL), respectively. Whereas C. albicans and P. aeruginosa showed significantly higher ZOI in response to the ethyl acetate leaf extract compared to their positive controls; nystatin (0.1 mg/mL), and gentamycin (0.01 mg/mL), respectively.

Statistical Significance Between the Average ZOI of Each Concentration of Ethanolic Leaf Extract Against Test Microorganisms and Their Positive Controls was Analyzed. Positive Controls (P) Used for S. aureus; Ampicillin (0.01 mg/mL), E. coli; Gentamycin (0.01 mg/mL), C. albicans; Nystatin (0.1 mg/mL), P. aeruginosa; Gentamycin (0.01 mg/mL), S. epidermidis; Clindamycin (0.002 mg/mL). Data Were Shown as Mean ± Standard Deviations from Three Replicates (n = 3). * Denotes Statistical Significance (p < 0.05) as Determined by one -way ANOVA (** p < 0.01, *** p < 0.001, and **** for p < 0.0001).
Statistical Significance between the Average ZOI of Each Concentration of Ethyl acetate Leaf Extract Against Test Microorganisms and their Positive Controls was Analyzed. Positive Controls (P) Used for S. aureus; Ampicillin (0.01 mg/mL), E. coli; Gentamycin (0.01 mg/mL), C. albicans; Nystatin (0.1 mg/mL), P. aeruginosa; Gentamycin (0.01 mg/mL), S. epidermidis; Clindamycin (0.002 mg/mL). Data were Shown as Mean ± Standard Deviations from Three Replicates (n = 3). * Denotes Statistical Significance (p < 0.05) as Determined by One -Way ANOVA (** p < 0.01, *** p < 0.001, and **** for p < 0.0001).
Minimum inhibitory concentration (MIC) and minimum bacterial concentration (MBC) were tested against concentrations from 12.5 mg/mL – 0.0245 mg/mL of ethanolic leaf extract. The MIC and MBC value was observed as 12.5 mg/mL.
GC-MS Analysis
Gas Chromatography–Mass Spectrometry (GC-MS) analysis revealed three major volatile chemical constituents in the ethanolic leaf extract of M. zeylanica. Compound identification was based on comparison with the NIST 14.L mass spectral library, with only those exhibiting match factors (MF) and reverse match factor (RMF) greater than 800 considered for interpretation. The predominant compound identified was 1,2,3-benzenetriol (pyrogallol) eluted at 25.8 min, accounting for 86.75% of the total peak area in the crude extract. The characteristic peaks for mass fractions of the compound were given by the mass spectrum as mass to charge (m/z) ratio of 126, 108, 80 and 52 confirms the compound identification. At 34.9 min, co-elution of 2-(2,6-dimethylheptyl)-cyclobutanone and 6,10,14-trimethylpentadecan-2-ol, were observed and both compounds collectively contributed 9.38% of the total peak area. The top peaks for 2-(2,6-dimethylheptyl)-cyclobutanone were observed for m/z ratio of 112, 57, and 55. The m/z ratio of 112, 57, and 45 confirms the presence of 6,10,14-trimethylpentadecan-2-ol. Carboxylic acids eluted at 47.7 min represented 3.87% of the total peak area, and two compounds were identified as tetradecanoic acid (miristic acid) and undecanoic acid. The mass spectrum analysis of tetradecanoic acid showed its peaks for m/z ratios of 129 and other expected peaks (m/z ratios of 117, 132, 285 and 145) were not observed. However, the characteristic peaks for m/z ratios of 60, 73, 41, 43 were clearly reported in the mass spectrum supporting the identification of undecanoic acid (Figure 5, 6 and 7).

GC-MS Chromatogram of Ethanolic Extract of M. zeylanica. Where: 1,2,3-Benzenetriol Eluted at 25.766 min; 2-(2,6-Dimethylheptyl)-Cyclobutanone and 6,10,14-Trimethylpentadecan-2-ol Elueted at 34.955 min and Carboxylic Acids (Tetradeconoic Acid and Undecanoic Acid) Eluted at 47.747 min.

Mass Spectrum of the Ethanolic Leaf Extract of M. zelanica. The Spectrum Displays the Following Compounds:(a) 1,2,3-Benzenetriol with m/z ratios of 126, 108, 80, and 52; (b) 2-(2,6-dimethylheptyl) Cyclobutanone with m/z ratios of 112, 57, and 55; (c) 6,10,14 - Trimethyl-pentadecan-2-ol with m/z ratios of 112, 57, and 45; (d) Tetradeconoic Acid with an m/z ratio of 129; and (e) Undecanoic Acid with m/z Ratios of 60, 73, 41, and 43.
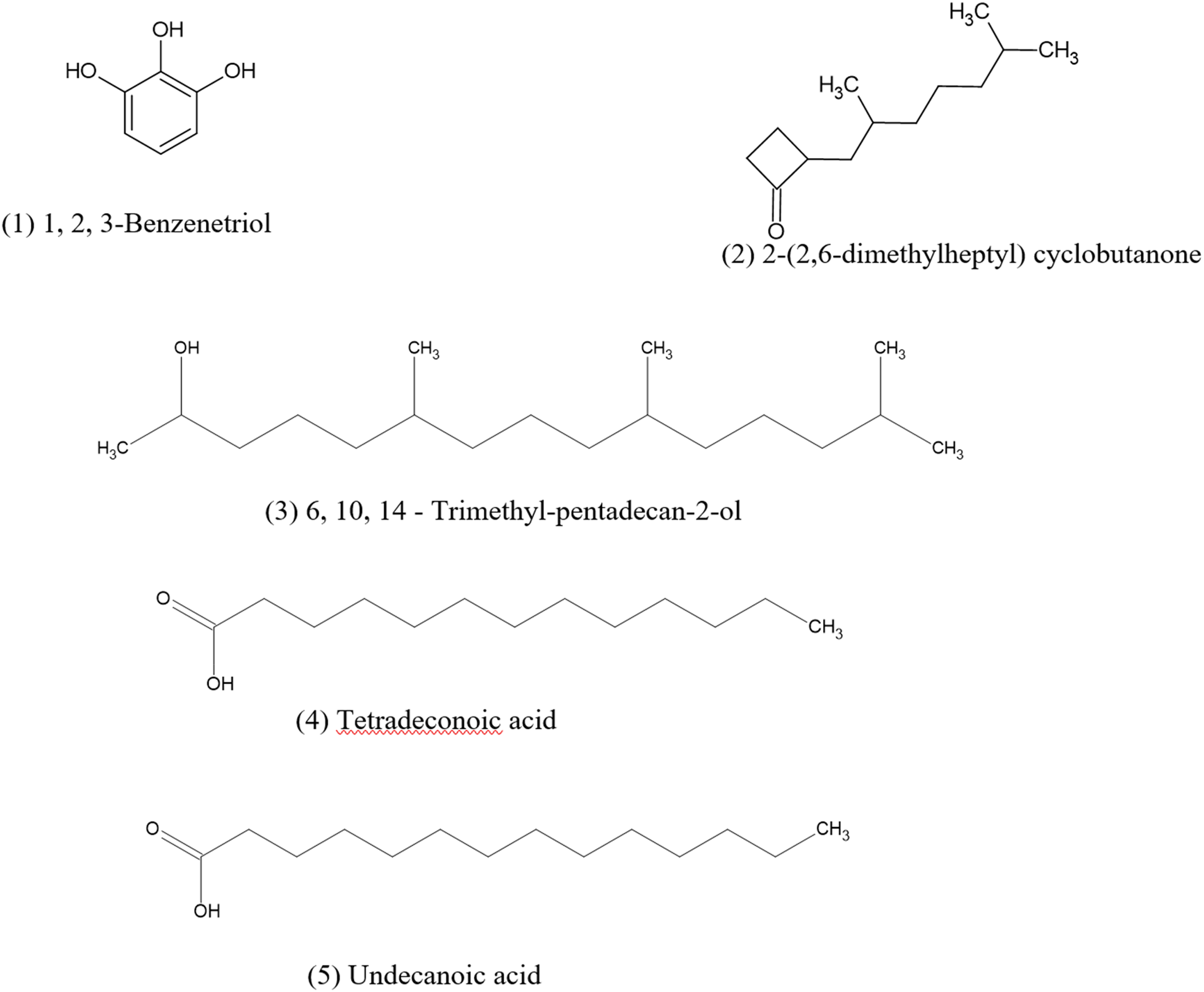
Structures of Phytochemicals Identified from the Ethanolic Leaf Extract of M. zeylanica Using GC-MS Analysis;(1) 1,2,3-Benzenetriol, (2) 2-(2,6-Dimethylheptyl)Cyclobutanone (3), 6,10,14-Trimethyl-Pentadecan-2-ol (4), Tetradeconoic Acid (5), Undecanoic Acid.
FTIR Analysis
FTIR data was interpreted following the method of Nandiyanto et al, 2019. 41 Figure 8 presents the FTIR spectrum of the ethyl acetate leaf extract. A broad peak at 3317.52 cm−1 suggests the presence of O-H stretching vibrations, commonly found in alcohols, phenols, or carboxylic acids. Narrow peaks observed at 2917 cm−1, 2849.04 cm−1, and 1444.64 cm−1 correspond to C-H vibrations characteristic of aliphatic hydrocarbon chains. A prominent peak at 1701.97 cm−1 corresponds to a carbonyl group such as a ketone, aldehyde, or ester. Peaks at 1606.53 cm−1 and 1534.31 cm−1 may indicate the presence of C = C stretching vibrations of aromatic rings. The peaks at 1313.45 cm−1, 1192.55 cm−1, and 1080.68 cm−1 may suggest C-O vibrations, which are prevalent in ethers, alcohols, or esters. 41

FTIR Spectra of Ethyl Acetate Leaf Extract of M. zeylanica. Where Peaks of : 3317.52 cm-1 (O-H group), 2917 cm−1, 2849.04 cm−1, and 1444.64 cm−1 (C-H group), 1701.97 cm−1 (C = O group), 1606.53 cm−1, 1534.31 cm−1 (C = C group), 1313.45 cm−1, 1192.55 cm−1, and 1080.68 cm−1 (C-O group).
Figure 9 shows the FTIR spectrum of the ethanolic leaf extract sample. The peak at 3233.64 cm-1 generally indicates the presence of O-H (hydroxyl) stretching vibrations, commonly found in alcohols, phenols, or carboxylic acids such as 1,2,3-benzenetriol, 12,4-benzenetriol, 6,10,14-trimethyl-pentadecan-2-ol, tetradeconoic acid, and undecanoic acid. The peak at 1602.67 cm−1 indicates the presence of C = C stretching vibrations, commonly observed in aromatic rings and alkenes, which can be attributed to 1,2,3-benzenetriol. The peak at 1315.86 cm−1 corresponds to C-H bending vibrations, potentially related to aliphatic chains or other hydrocarbon-containing structures, such as 6,10,14-trimethyl-pentadecan-2-ol, and undecanoic acid. Additionally, peaks at 1208.85 cm−1 and 1030.74 cm−1 correspond to C-O stretching vibrations, commonly found in ethers, alcohols, or esters, such as those present in 6,10,14-trimethyl-pentadecan-2-ol. 41

FTIR Spectra of Ethanolic Leaf Extract of M. zeylanica. Where Peaks of : 3233.64 cm-1 (O-H Group), 1602.67 cm-1 (C = C Group), 1315.86 cm-1, 1208.85 cm-1 and 1030.74 cm-1 (C-O Group).
Physical Evaluation of Antimicrobial Cream
The final cream formulation appeared brown-green in color, slightly greasy and smooth in texture with no grittiness and no visible signs of phase separation. The spreadability of the formulation was measured at 10.17 g.cm/s, falling within the ideal spreadability range (5-15 g.cm/s) for a topical cream. 42 The viscosity of the formulation was recorded at 675800 cP. The pH value of the cream was 5.81 ± 0.12, aligning with acceptable range for skin compatibility. Stability evaluation of the formulation showed no phase separation under various stress conditions: centrifugation at 4000 rpm for five minutes, storage at 45 °C for one week and ambient storage for 90 days. Long-term storage (90 days) conditions did not show significant changes in pH, texture or viscosity.
In-Vitro Antimicrobial Activity of the Formulation
The topical cream formulated with EE at a concentration of 25 mg/mL demonstrated ZOI against all the tested organisms. However, the ZOI were smaller than those produced by the direct application of the ethanolic extract at the same concentration. S. aureus and S. epidermidis demonstrated relatively low susceptibility to the cream formulation, with inhibition zones smaller than those produced by the corresponding positive controls. Remarkably, C. albicans was shown a greater susceptibility to the antimicrobial cream formulation (Table 1).
Average Zone of Inhibition for Tested Bacterial Strains and Fungal Strain at Developed Antimicrobial Cream.

Data are presented as Mean diameter of the inhibition zone ± standard deviation
Discussion
The Present study is among the first to evaluate the antimicrobial activity of M. zeylanica leaf extract against a range of microorganisms, including S. aureus, E. coli, P. aeruginosa, S. epidermidis, and C. albicans. Hexane, ethyl acetate and ethanol were selected as extraction solvents to ensure the extraction of a range of phytochemicals with varying polarities as previous studies have not investigated the antimicrobial potential of M. zeylanica leaves. The ethanolic extract yielded the highest extractive percentage, followed by the ethyl acetate extract. The hexane extract produced a negligible yield and was therefore excluded from further analysis. Among the two solvent extractions tested for antimicrobial properties, the ethanolic leaf extract demonstrated superior activity against all test microorganisms relative to the ethyl acetate extract. Consequently, the ethanol leaf extract was incorporated in the topical cream formulation. A cream formulation was selected for topical delivery of plant extract due to its ability to provide localized treatment directly at the infection site, targeting pathogens. And cream formulation allows for effective absorption, moisturization, and a soothing effect on irritated skin, making them ideal for conditions like cellulitis, folliculitis, and candidiasis.43,44 Additionally, they are non-greasy, easy to apply, and help to minimize systemic absorption, reducing potential side effects. 43 Although the leaf extracts of M. zeylanica have not been previously explored for antimicrobial properties, the stem bark was studied previously from two climatic zones in Sri Lanka. 24 The study revealed that none of the extracts (aqueous or methanol) demonstrated antimicrobial action against E. coli, while methanol extracts of inner bark shown antibacterial activity against S. aureus irrespective of the climate zone. These findings highlight the effects of geographical factors and extraction techniques on biological activity of the plant-derived compounds.
The present study assessed the minimum inhibitory concentration (MIC) and minimum bacterial concentration (MBC) of ethanolic leaf extract of M. zeylanica across a concentration range from 12.5 mg/mL to 0.024 mg/mL. At every concentration tested, microbial proliferation was observed. All the test microorganisms (S. aureus, E. coli, P. aeruginosa, S. epidermidis, and C. albicans) were not susceptible to the antimicrobial activity at the concentrations below 12.5 mg/mL. The ethanolic leaf extract demonstrated antimicrobial activity within 12.5 and 200 mg/mL concentration range, with the lowest MIC identified at 12.5 mg/mL. This aligns with findings from Doughari and Manzara., 2008, who reported MIC and MBC of similar extracts ranging from 12.5 -75 mg/mL and 25 -175 mg/mL, respectively. 45 Additionally, Bbosa et al, 2007 reported MIC values for ethanolic extracts of M. indica ranging from 5481.0 to 43750.0 µg/mL, identifying it as one of the most bioactive leaf extracts studied. 46 Physical evaluations revealed that all cream formulations were opaque, uniformly mixed, and odorless and had a smooth texture. The pH measurements showed that the cream had a stable, slightly acidic pH range of 4.5 to 6 over a 28-day storage period which is acceptable for skin care products, 47 over a 28-day storage period. Additionally, spreadability and viscosity values were found to be within the optimal limits for safe and effective application to the skin. The observed decrease in antimicrobial activity in cream formulation may be due to reduced diffusion of active compounds, possible interactions with formulation excipients. However further studies need to be conducted to evaluate the possible reasons, and formulation can be optimized by adjusting extracted concentration, selecting suitable excipients, and ensuring proper release of compounds to improve performance.
In the current study, the cream formulation incorporating ethanolic leaf extract of M. zeylanica exhibited limited inhibition against S. aureus and S. epidermidis compared to their respective positive controls. However, C. albicans showed greater susceptibility to the antimicrobial cream formulation surpassing the antifungal activity observed with the standard positive control. This finding contrasts with the study by Amgad A. Awad El-Gied et al, 2015, where the cream formulation containing ethanolic seed extract of M. indica showed the lowest inhibition activity against C. albicans but highest against E. coli. 28 This indicates variability in antimicrobial efficacy among plant species, plant parts used for extraction, and the microbial species tested, highlighting the need for further research to understand these complex interactions and optimize the formulations for specific antimicrobial applications.
GC-MS analysis of the ethanolic leaf extract of M. zeylanica identified 1,2,3-benzenetriol as the most abundant compound, comprising 86.75% of the extract. 48 Next most abundant compound class is 2-(2,6-dimethylheptyl) cyclobutanone and 6,10,14-trimethyl-pentadecan-2-ol (34.955 min). Furthermore, carboxylic acids were identified at the retention of 47.747 min, and there are more specifically tetradeconoic acid and undecanoic acid. Observed co-elution of compounds can be attributed to their structural similarities such as isomeric forms and long chain branched structures. Chromatographic co-elution occurs when compounds exhibit similar retention behaviors, resulting in overlapping peaks that cannot be resolved within the resolution of the method. 49 Although the separation was limited, mass spectrometry enabled their identification through distinct fragmentation patterns. 50 Separation can be improved by modifying factors such as mobile phase, stationary phase, column length or operating temperature. 49 n-Benzenetriol, an aromatic compound featuring a benzene ring with three hydroxyl groups, includes derivatives, such as 12,3-benzenetriol (pyrogallic acid) and 12,4-benzenetriol, are known for their antimicrobial properties. 51 Specifically, 1,2,3-benzenetriol, a major component of Quercus cortex, is used in European folk medicine to treat microbial infections due to its significant antibacterial activity. 52 Further, 1,2,3-benzenetriol was present in the methanol extract of Phyllanthus emblica fruits, reported to act as an active antibacterial agent, altering microbial cell membrane permeability and inducing the generation of reactive oxygen species. 53 The compound, 2-(2,6-dimethylheptyl)cyclobutanone, is a four-membered cyclic ketone and isosteric with β-lactam. It is identified as the first family of inhibitors showing activity across all class of β-lactam. 54 Moreover, cyclobutanone derivatives have been studied as potential new antibiotic candidate by inhibiting diaminopimelate desuccinylase, an enzyme necessary for bacterial growth. 55 6,10,14-Trimethylpentadecan-2-ol was identified in GC-MS analysis of antimicrobial assessment of Hibiscus cannabinus L leaves at retention time of 14.1 min, further supports the findings of present study. 56 Moreover, naturally occurring carboxylic acids have been found to possess strong antimicrobial properties. Previous studies revealed that tetradeconoic acid identified through GC-MS and NMR analysis of the heartwood and stembark of Albizia adianthifolia and Pterocarpus angolensis possessed antimicrobial activity. 57 Interestingly, the synergistic effect of quinic acid-derived from Syzygium cumini and undecanoic acid against Candida spp. in inhibiting biofilm formation and their virulence were investigated and antifungal activity of undecanoic acid was reported. 58 Thus, these findings highlight the potential of M. zeylanica leaf extracts, particularly the ethanolic extract, as a source of antimicrobial agents.
FTIR analysis of ethanolic leaf extract supports the GC-MS findings from by confirming the key functional groups of detected compounds. The strong O-H stretching peak and C = C stretching peak suggest the presence of hydroxyl groups and aromatic rings, aligning with dominant compound, 1,2,3-benzenetriol. In addition, C-H bending and C-O stretching support the presence of aliphatic alcohol and carboxylic acids, correlating with 6,10,14-trimethyl-pentadecan-2-ol, tetradeconoic acid and undecanoic acid. These findings suggest that phenolic compounds are the major constituent of ethanolic leaf extract, which may contribute to its biological activity. However, the presence of cyclobutanone cannot be explained directly with FTIR data. Antimicrobial activity of ethanolic leaf extract of M. zeylanica can be attributed to 1,2,3-benzenetriol, the primary compound identified from GC-MS analysis. 1,2,3-Benzenetriol generates reactive oxidative species, causing oxidative damage to microbial membrane, protein and DNA, leading to cell membrane disruption, leakage of essential cellular components, and microbial death.53,54 Additionally, undecanoic acid exerts anti-fungal activity by disrupting fungal cell membrane, inhibiting ergosterol biosynthesis and preventing biofilm formation, 58 highlighting natural potential the extract as an antimicrobial agent.
The present study demonstrates the antimicrobial potential of M. zeylanica leaf extracts, particularly the ethanolic leaf extract. However, the study was limited to in-vitro testing against five types of microorganisms, and the effectiveness against other pathogens, such as multi-drug resistant and clinical strains, remained unexplored. The concentrations of standard antibiotic disc used were selected at their effective concentration referring to similar studies and reports. There was no necessity of testing the leaf extract at the same concentration as the antibiotic dics, since our primary goal was to determine an effective concentration of the leaf extract which functions well against a range of microorganisms. However, further studies with equal concentrations or dose-response experiments will be needed for any additional development of the study. The apparent abundance of benzenetriols in the GC-MS analysis likely reflects the bias of the technique toward volatile and thermally stable compounds, rather than the true phytochemical profile of the ethanolic extract. More comprehensive profiling using LC-MS or NMR is recommended to identify polar, non-volatile constituents that may not be detected by GC-MS. Anti-fungal studies can be conducted to further validate the antimicrobial activity of developed cream formulation. This study used MHA medium for antifungal activity, which is primarily optimized for bacterial growth, this may affect the consistency and reliability of fungal strain results. Since this study only incorporated organic extracts in formulation, future research could compare the effects of water and organic extracts to enhance the formulation development. Future studies should address these limitations to further explore the therapeutic potential of M. zeylanica in antimicrobial applications with toxicological and in-vivo experiments. The developed topical cream formulation requires further analysis with appropriate control experiments, to confirm that the observed antimicrobial activity is specifically attributed to plant extract.
Conclusion
The antimicrobial effectiveness of ethyl acetate and ethanol leaf extracts from M. zeylanica demonstrated inhibitory effects on test microorganisms, with the ethanolic extract exhibiting the most potent antimicrobial activity. GC-MS analysis identified 1,2,3-benzenetriol (86.75%), as the major compounds. These findings hold the promising implications to investigate and use these compounds as novel antimicrobial agents. Antimicrobial cream could serve as an effective treatment for microbial infections; however, further studies are recommended to assess the toxicological evaluations and test the effectiveness against a broader spectrum of microorganisms, to validate its clinical applicability.
Footnotes
Acknowledgements
The authors are grateful to CINEC Campus, Malabe, Sri Lanka, for funding this research, and for providing access to the laboratories to develop the formulation.
ORCID iDs
Ethical Considerations
The study did not require ethical approval as it did not involve human or animal subjects.
Author Contribution
S.M.R.N. Senarathne1, conceptualized the study, designed the methodology, conducted investigations, curated data and drafted the initial manuscript. O.P.C. Deshan1, contributed to method development, performed formal analysis and participated in manuscript writing. S. N. Warnakulasuriya2, provided resources, supervised the project, conceptualized the study, validated findings and contributed to manuscript review and editing. S.L.A. Gunawardana1*, created visualizations, conceptualized the study, acquired funding, validated findings, and contributed to manuscript review and editing. All authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statements of Human or Animal Rights
This study did not involve any experiments on humans or animals, therefore, no statement of compliance with human or animal rights is applicable.
Statement of Informed Consent
As no human participants were involved in this study, obtaining informed consent was not necessary.


