Abstract
Objective
Herbal medicinal products (HMPs) are commonly used to treat symptoms of mild functional gastrointestinal disorders (FGID) such as functional dyspepsia (FD) and irritable bowel syndrome (IBS). Gasteo®, an HMP containing a combination of 6 liquid plant extracts, has been on the market for over 60 years to treat FD, but little is known about its mode of action. This in vitro study aimed to investigate the effects of the HMP on pharmacological targets relevant for FD.
Methods
We investigated the anti-inflammatory properties of the HMP by measuring the release of the interleukins IL-1ß, IL-6, IL-8, the monocyte chemoattractant protein-1 (MCP-1), the tumor necrosis factor-α (TNF-α), and the prostaglandin E2 (PGE2) release in a human monocyte cell model. We also assessed its effects on colonic epithelial barrier dysfunction by measuring the transepithelial electrical resistance (TEER) in Caco-2 cells, and carried out GPR55, TGR5 and TRPV1 functional assays, as well as muscarinic M3 and serotonin–type 3 (5-HT3) receptor binding assays.
Results
The HMP inhibited the lipopolysaccharide-induced release of all investigated inflammatory mediators in human monocytes and prevented colonic epithelial barrier dysfunction in Caco-2 cells. The HMP had no effects on GPR55 or TGR5 functions, but it antagonized TRPV1. It also inhibited the binding of [3H]-N-methylscopolamine to the M3 receptor and showed a weak inhibition of the binding of [3H]-GR65630 to 5-HT3.
Conclusion
The HMP studied exhibited several in vitro pharmacological properties that could explain the effects observed on FD.
Introduction
Functional gastrointestinal disorders (FGIDs), also known as disorders of the gut–brain–interaction, are a group of chronic digestive problems that cause abdominal pain, bloating, or altered bowel habits. They include functional dyspepsia (FD) and irritable bowel syndrome (IBS).1,2 FGIDs affect multiple parts of the digestive tract, including the esophagus, the stomach, the small and the large intestine, and the anus, whereas the symptoms and diagnoses overlap. 3
FD and IBS are the most prominent disorders, with a combined prevalence of 40.3%. 3 FD alone affects approximately 685 million people world-wide, with a prevalence of 8.4%. 4 FD can manifest itself as the postprandial distress syndrome (PDS), which is usually triggered by meals and leads to symptoms such as fullness and early satiation. Alternatively, it can manifest as epigastric pain syndrome (EPS), which is related to burning symptoms caused by gastroesophageal reflux.5,6
The pathogenesis of FD involves, among others, duodenal low-grade inflammation and epithelial barrier dysfunction.5,6
Chronic duodenal low-grade inflammation may lead to motor and sensory abnormalities in the gastrointestinal-neural system, although the exact cause of the inflammation is unknown. 7 An altered secretion of cytokines has been linked to epigastric symptoms, 8 an up-regulated expression of gastric interleukin IL-6 has been associated with epigastric burning, IL-8 with epigastric pain, bloating and belching, and the monocyte chemoattractant protein 1 (MCP-1) with nausea. 8 IBS patients typically have higher peripheral concentrations of tumor necrosis factor-α (TNF-α) and IL-1β. 9 High gastric IL-1β and IL-8 secretion is positively correlated with depression, a disorder that often affects FD patients. 8
A leaky gut syndrome (LGS) indicates an impaired duodenal epithelial barrier function. It is prevalent among IBS patients 10 and is characterized by increased permeability of the intestine, caused by changes in the function and/or expression levels of tight junction (TJ) proteins. 11 The increased intestinal permeability may allow microorganisms, toxins, or undigested food particles to enter the bloodstream and affect the hormonal, immune, nervous, respiratory, or reproductive systems.10,11 There is a significant negative correlation between the expression levels of several cell-to-cell adhesion proteins and the extent of increased permeability and the severity of low-grade inflammation, as demonstrated by increased infiltration of mucosal mast cells and eosinophils. 12
Several gastrointestinal cell receptors are involved in digestive physiology, and thus in FGID. The bile acid receptor TGR5 (Takeda G protein–coupled receptor 5) is expressed by enteric neurons and endocrine cells. It is a mediator of colonic motility and a potential treatment target of constipation and diarrhea. 13 GPR55 (G protein-coupled receptor 55) regulates gut motility and colon relaxation, as observed in a mice model, and has pro-inflammatory properties. 14 TRPV1 (transient receptor potential cation channel subfamily V member 1) is upregulated in FD patients and is involved in the onset of inflammation and/or pain in pancreatitis, colon diseases and IBS. 8 Most FD patients have visceral chemohypersensitivity (eg, impaired or altered perception of acidity) involving TRPV-1 pathways. A subgroup of these patients also has somatic hypersensitivity. 15 Gastric secretion pathways include the activation of the muscarinic acetylcholine receptor M3 by acetylcholine, histamine H2 receptors by histamine, and the cholecystokinin 2 receptor (CCK2) by gastrin. Activation of these receptors increases cyclic adenosine monophosphate (cAMP) concentrations in parietal cells and activates H+/K + ATPases. This increases the concentration of hydrochloric acid in gastric fluids, which often leads to gastric mucosal damage or gastritis. 16 The serotonin type 3 receptor 5–HT3 (hydroxytryptamine), delays colonic transit and decreases the contractile and tonic responses to meal ingestion. 17
Evidence-based herbal FD and FGID therapies in Europe include the herbal combination STW-5 and peppermint oil. Rikkunshito (Japanese) and DA-9701 (Korean) are evidence-based traditional Asian medicines, but proof of their efficacy in Europe is not yet robust or widely established. 18
Gasteo® (Cesra Arzneimittel GmbH & Co. KG, Baden-Baden, Germany), a liquid herbal medicinal product (HMP) containing a combination of six plant extracts has been on the market for over 60 years as a traditional remedy for mild FGID. In a non-interventional trial this herbal remedy improved FD-related symptoms such as vomiting, retching, nausea and loss of appetite. 19 However, the mode of action of this combination has not yet been studied.
The goal of this study was to gather preliminary information on the in vitro pharmacological effects of this HMP on inflammation, epithelial barrier dysfunction, and selected gastrointestinal (GI) receptors.
Material and Methods
Study Conduct
The monocyte assay was conducted at VivaCell in Germany and the 5-HT3 and M3 receptor binding were carried out at rent-a-lab in Germany. The functional assays (TRPV1, GPR55 and TGR5) and the TEER assay were performed at Maimonides Biomedical Research in Spain. All investigations were performed in 2023.
Herbal Remedy
Ten mL of the HMP contain 3.5 mL Potentilla anserina L., 2.0 mL Matricaria recutita L., 1.5 mL Glycyrrhiza glabra L., 0.5 mL Angelica archangelica L., 0.5 mL Artemisia absinthium L., and 0.5 mL Centaurea benedicta L. extract. Monographs for each plant have been published by either the European Committee on Herbal Medicinal Products (HMPC) and/or the European Scientific Cooperative on Phytotherapy (ESCOP). The plant raw materials are from Heinrich Klenk GmbH & Co. KG (Schwebheim, Germany) and Christof Peter GmbH & Co. KG (Schwebheim, Germany). All plants materials were extracted with 40% (V/V) ethanol except for M. recutita, which was extracted with 45% (V/V) ethanol. The drug extract ratio was 1:1.7–2.2 for all extracts. Batch analyses and releases were performed according to the European Pharmacopoeia Europea (Ph. Eur.) and internal methods and standard operating procedures.
Monocyte Cell Cultures
Human primary monocytes were obtained from buffy coats of three healthy, 18–68-year-old human blood donors. In Germany, the use of buffy coats derived from blood of donors is covered by the consent given by the donors at the time of blood donation and before donating blood, the donors gave their written consent for their blood to be used for research purposes by signing a blood donor questionnaire at the blood donation center at the University Medical Center of Freiburg, Germany. Cell cultures were prepared using an endotoxin-free culture method. 20 Briefly, 25 mL of lymphocyte separation medium (anprotec, Bruckberg, Germany) were mixed with 25 mL of buffy coat. A gradient was established by centrifugation at 1800 rpm and 20 °C for 40 min. The peripheral blood mononuclear cell interphase was removed, re-suspended in 50 mL of prewarmed phosphate-buffered saline (PBS; PAN-Biotech, Aidenbach, Germany), and centrifuged. The resulting pellets were washed in 50 mL PBS and gradient-centrifuged for 10 min at 1600 rpm and 20 °C. The resulting pellets were resuspended in low-endotoxin RPMI-1640 medium (Roswell Park Memorial Institute 1640) supplemented with 10% human serum (HexCell GmbH, Berlin, Germany). The cells obtained were seeded and incubated at 37 °C with 5% CO2 for 24 h. After incubation, the medium and the non-adherent cells (mostly lymphocytes) were removed and fresh RPMI-1640 medium containing 1% human serum was added. Quality controls to confirm the monocyte identity were carried out according to published protocols.20,21
Viability of Human Monocyte Cells
The viability of basal human monocytes from a healthy blood donor was tested by exposing the cells to increasing concentrations of the HMP dissolved in 0.1% DMSO (PanReac AppliChem, Darmstadt, Germany). The cells were seeded in quadruplicate in 96-well plates at a density of 220 000 cells/well. One µL of the HMP stock solution was added to four wells and the same number of replicates were performed for its dilutions (0.01%, 0.05%, 0.1%, 0.25%, 0.5%, 1%, 2.5%, 5%, and 10%). 250 µg/mL NaF was used as the positive, and the cell growth medium as the negative control. After incubating for 24 h at 37 °C and 5% CO2, 10 µL of AlamarBlue® (resazurin; Biosource, USA) were added to each well. Fluorescence, an indicator of viability, was measured after a two-hour incubation at 37 °C and 5% CO2 using a fluorescence spectrophotometer with an excitation wavelength of 544 nm and an emission wavelength of 590 nm.20,22
Anti-Inflammatory Assay in Human Monocytes
The human monocyte model has been shown to reliably detect the anti-inflammatory effects of herbal extracts. 23 Monocytes of 3 donors were seeded separately in duplicates in 24-well plates at a density of 2,200,000 cells/well. The cells were then incubated at 37 °C and 5% CO2. Thirty minutes prior to treatment with 10 ng/mL of lipopolysaccharide (LPS; from Salmonella enterica serotype typhimurium [Sigma]), the monocytes were incubated with five non-cytotoxic concentrations of the test items (0.001% to 1.0% V/V). After a 24 h incubation period, the supernatants were removed and centrifuged. The concentrations of IL-1ß, IL-6, IL-8, MCP-1, TNF-α, and PGE2 were determined using ELISA (R&D Sysatems, Cayman Chemical) according to the manufacturer's protocols.
Cell Lines Used
We used Caco-2 cells (ATCC HTB-37) from the American Type Culture Collection (ATCC; Manassas, VA, USA) for intestinal barrier function studies. These cells are commonly utilized in gastrointestinal models, 24 also because they form a dense, differentiated and polarized epithelial monolayer in cell cultures, with measurable tight junctions (TJ). 24 The cells were maintained at 37 °C with 95% relative humidity and a 5% CO2 atmosphere in Dulbecco's Modified Eagle's Minimum Essential Medium (DMEM; Pan Biotech, Aidenbach, Germany) supplemented with 10% fetal bovine serum, 1% (V/V) penicillin/streptomycin (both Thermo Fisher Scientific, Karlsruhe, Germany) and 2 mM L-glutamine (Sigma, Taufkirchen, Germany).
The following cells were used in the reporter-receptor expression models: human embryonic kidney cells (ATCC CRL-3216) from the HEK293T-GPR55 and HEK293T-TRPV-1 cell lines (Human Fetus; Beros Bioderma Technology, Córdoba, Spain), which were transfected in-house; Chinese hamster ovary cells (ATCC CCL-61); and transfected CHO-TGR5-CRE-Luc cells (Beros Bioderma Technology). The cells were cultured in DMEM supplemented with 400 µg/mL geneticin (G418 sulfate; Merck KGaA, Darmstadt, Germany).
Cytotoxicity in Caco-2 Cells
We used the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; #N6876, Sigma) assay to evaluate the metabolic activity of Caco-2 cells and, consequently, the HMP cytotoxicityof the HMP. We seeded the cells at a density of 10 000 cells/well in 96-well plates and preincubated them with HMP concentrations ranging from 0.05% to 20% for 24 h. Then we added 5 mg/mL of MTT to each well and incubated the plates for 4 h at 37 °C in the dark. The reaction was stopped by removing the supernatant and adding 100 µL of dimethyl sulfoxide (DMSO), and the absorbance was measured at 550 nm using a TriStar LB 941 (Berthold Technologies GmbH & Co. KG, Bad Wildbad, Germany). 25
Transepithelial Electrical Resistance Assay
Epithelial integrity was evaluated using the transepithelial electrical resistance (TEER) method, 26 expressed as Ω/cm2, with a Millicell® ERS-2 (Electrical Resistance System, Millipore, MA, USA) as previously described. 27 Caco-2 cells were seeded at a density of 500 000 cells/well onto 6.5 mm polycarbonate inserts with 0.4 µm pore size (Corning). The inserts were placed into 12-well plates containing 0.2 mL of DMEM in the apical chamber and 0.6 mL DMEM in the basolateral chamber. The cells were treated with non-cytotoxic concentrations (0.01%, 0.1% and 1%) of HMP and incubated for 24 h. After incubation, 200 μL of PBMC supernatant, which had been activated with phytohemagglutinin-M (PHA-M; Roche) for 48 h were added. PHA-M is a mitogen that triggers massive cell proliferation, cytokine release, and metabolic changes. The cells were then incubated for 30 min, and TEER was measured as described by Prados et al 27 TEER values were calculated by subtracting the blank resistance (the background electrical resistance from an insert without cells, including filter and medium) and multiplying the resulting values by the effective growth area of the membrane (0.6 cm2). The results are expressed as a percentage of epithelial disruption; 0% represents the degree of disruption in untreated cells.
GPR55-, TGR5-, TRPV-1-Functional Assays, and M3-, and 5-HT3-Receptor Binding
GPR55 activity was determined using HEK293Tcells stably transfected with the human GPR55 cDNA. The HEK293T-GPR55 cells were then transiently transfected with 0.2 µg of the CRE-Luc reporter plasmid, which contains six consensus cAMP-responsive elements (CREs) linked to the firefly luciferase reporter gene (Roti® Fect, Carl Roth, Karlsruhe, Germany). The cells were treated with 0.01%, 0.1%, or 1% HMP. Five µM lysophosphatidylinositol (LPI; Sigma), a known endogenous GPR55 agonist, was used as a positive control and in combination with the HMP. After 6 h, the cells were lysed in 100 µL of lysis buffer (pH 7.8) containing 25 mM Tris-phosphate, 7% glycerol (both PanReac AppliChem), 8 mM MgCl2, 1 mM DTT, and 1% Triton X-100 (all from Sigma). Luciferase activity was measured using the Luciferase Assay System (#E1500, Promega, Madison, WI, USA), according to the manufacturer's instructions, with a TriStar2 LB942 multimode reader (Berthold Technologies GmbH & Co. KG, Bad Wildbad, Germany).
The TGR5 agonistic/antagonistic activity was determined using the CHO-TGR5-CRE–Luc cell line (Beros Bioderma Technology, Spain). This cell line was generated by stable transfection of pcDNA3-TGR5 and CRE-Luc plasmids in CHO cells (ATCC CCL.61), followed by selection with G418. Selected clones were tested for their response to 10 µM lithocholic acid (LCA; Sigma) a secondary bile acid and TGR5 agonist. 28 The cells were incubated in 24-well plates (1 × 105 cell/mL) for 24 h. Then, the cells were incubated with 0.01%, 0.1%, or 1% HMP; 20 µM LCA to achieve 100% activation; and in combination with the HMP for 6 h. The cells were then lysed, and luciferase activity was measured as described for the GPR55 activity.
HEK293T-TRPV-1 cells (2 × 104/well) were cultured in 200 µL of DMEM (cf) using 96-well plates. This cell line was generated by the stable transfection of the pcDNA3-hTRPV-1 plasmid followed by selection with G418. The cells were incubated in triplicate with 0.01%, 0.1%, and 1% HMP for 30 min. Then, 1 µM of the agonist capsaicin (Sigma) was added and the cells were incubated again for 6 h. Ten µM capsazepine (Sigma), a TRPV-1 antagonist, were used as a positive control. Capsaicin-induced cytotoxicity was measured using the MTT assay. 25 Antagonistic activity was measured quantitatively as the inhibition of capsaicin-induced apoptosis. 29
Binding experiments were performed according to the data sheet provided by the receptor membrane supplier (PerkinElmer) with minor modifications.
Muscarinic M3 Receptor
Membranes expressing the human muscarinic M3 receptor (code RBHM3 M, lot 2275) were incubated in a buffer solution of 50 mM Tris-HCl, 2.5 mM MgCl2 and 1 mM EDTA at a pH of 7.4. The solution contained increasing concentrations (0.000316%, 0.001%, 0.00316%, 0.01%, 0.0316%, 0.1%, 0.316%, 1%, 3.16%, and 10%) of HMP and 0.3 nM 1-[N-methyl-3H] scopolamine methyl chloride ([3H]-NMS; code TRK 666, specific activity 80 Ci/mmol; from GE Healthcare, Germany). The incubation was performed for 90 min at 25 °C. Membranes were collected by filtration over GF/C filter plates pre-soaked with assay buffer. The filter plates were washed four times with 200 µl of ice-cold 50 mM Tris-HCl pH 7.4 and the filter-bound radioactivity was determined using a microplate reader (Microbeta, Wallac, Finnland). Bound radioactivity in the presence of 1 µM 4-DAMP (4-diphenylacetoxy-1,1-dimethylpiperidine iodide; Sigma) was defined as the non-specific radioligand binding.
Serotonin 5-HT3 Receptor
Membranes expressing the human serotonin 5-HT3 receptor (Code: RBHS3M44UA, Lot: 520-872-A) were incubated in a buffer solution of 50 mM Tris-HCl, 5 mM MgCl2 and 1 mM EDTA buffer pH 7.4 with the same HMP concentrations used for the M3 assay and 0.25 nM [N-methyl-3H]GR65630 (code: NET-1011, specific activity: 83.8 Ci/mmol; PerkinElmer, Rodgau, Germany) for 60 min at room temperature. Membranes were collected by filtration over GF/C filter plates that had been pre-soaked with 0.5% polyethyleneimine. The filter plates were then washed four times with 200 µl of ice-cold 50 mM Tris-HCl pH 7.4, and the filter-bound radioactivity was subsequently determined using a microplate reader (Microbeta, Wallac, Finland). The bound radioactivity in the presence of 10 µM MDL 72 222 (Bemesetron; Biotrend, Cologne, Germany) was defined as the radioligand non-specific binding.
Data Analysis
Results of the cytokine release assay are expressed as a percentage of the control (untreated LPS-activated monocytes) for each donor. 30 Measurements were conducted in duplicate for each buffy coat donors. For the binding assays each data point was normalized to 100% specific binding (total binding minus non-specific binding). Data from all experiments, except those involving muscarinic M3 and serotonin 5-HT3, are presented as means and 95% confidence intervals (95%CI) of the data normalized as percentages. As the sample sizes for all experiments are small, no confirmatory statistical analyses were carried out. IC50 values (the concentrations causing half-maximal inhibition of control-specific binding) were determined by a non-linear regression analysis of the competition curves according to Cheng-Prusoff, 31 with Prism 9 (GraphPad, San Diego, USA).
Results
Anti-Inflammatory Effects in Human Monocytes
Concentrations up to 1% (V/V) of the HMP had no effect on the monocyte viability (Fig. S1, Supporting Information). The herbal remedy inhibited the release of IL-1β, IL-6, IL-8, MCP-1, PGE2 and TNF-α in a dose dependent manner. Generally, reduction to levels lower than 80% began at an HMP concentration of 0.05%. At a concentration of 1%, cytokine release was decreased to 10–30% (Figure 1).
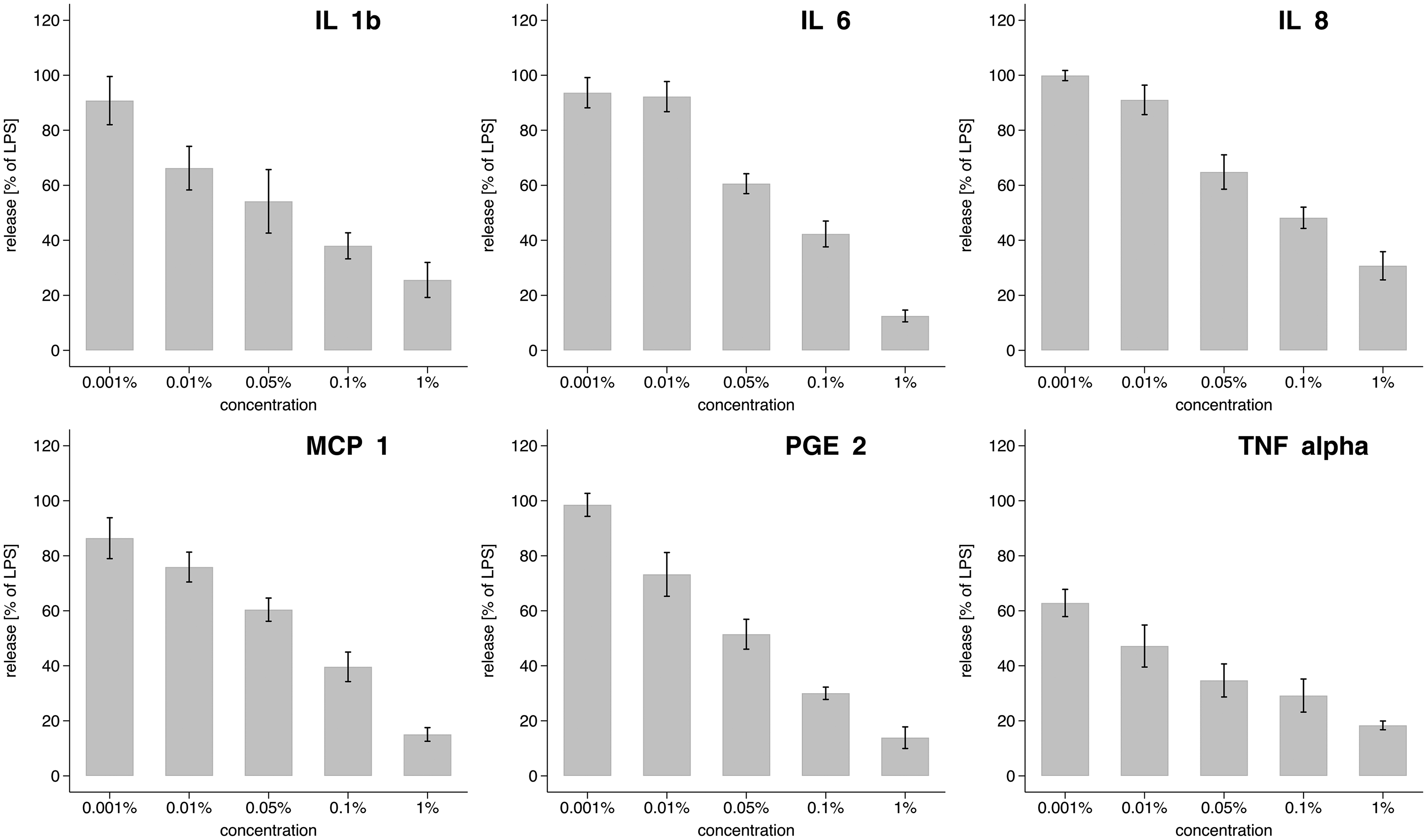
Effects of the herbal remedy mixture on lipopolysaccharide (LPS) induced cytokine release. Human monocytes of three different donors were stimulated separately with LPS (10 ng/mL) and incubated at increasing concentrations of the herbal remedy. The experiment was performed in duplicate (total n: 6). Results are expressed as % of LPS-induced cytokine release with no treatment. Concentrations of the HMP used: 0.001, 0.01, 0.05, 0.1, and 1%. Bars: mean cytokine release (%); range plot with capped spikes: 95% confidence intervals of the means.
Effects on Intestinal Barrier in Caco-2 Cells
The herbal remedy was cytotoxic in Caco-2 cells at concentrations ≥5% (see Fig. S2, Supporting Information). Therefore, the maximum concentration used in the TEER assay to measure PBMC-induced epithelial disruption was 1%. The herbal remedy restored colonic epithelial barrier function in a concentration-dependent manner (Figure 2). At concentrations of 0.1% and 1% the epithelial integrity was comparable to that of the control cells.

Effect of the herbal remedy mixture on PBMCs-induced epithelial disruption in Caco-2 cells. Epithelial integrity was assessed by measuring the transepithelial electrical resistance (TEER, W/cm2) as described in the Methods section. The experiment was performed in triplicate (total n: 3). Concentrations of the HMP used: 0.01, 0.1, and 1%. Bars: mean epithelial disruption (%); range plot with capped spikes: 95% confidence intervals of the means.
Effects on GPR55, TGR5 and TRPV1 Function
At all concentrations tested, the HMP did not affect GPR55 and TGR5 function (See Figures S3 and S4, Supporting Information). However, it was effective in antagonizing TRPV1 function, as demonstrated by its ability to prevent the capsaicin-induced cytotoxicity in 293T-TRPV1 cells (Figure 3). The control capsazepine clearly inhibited TRPV1 function.
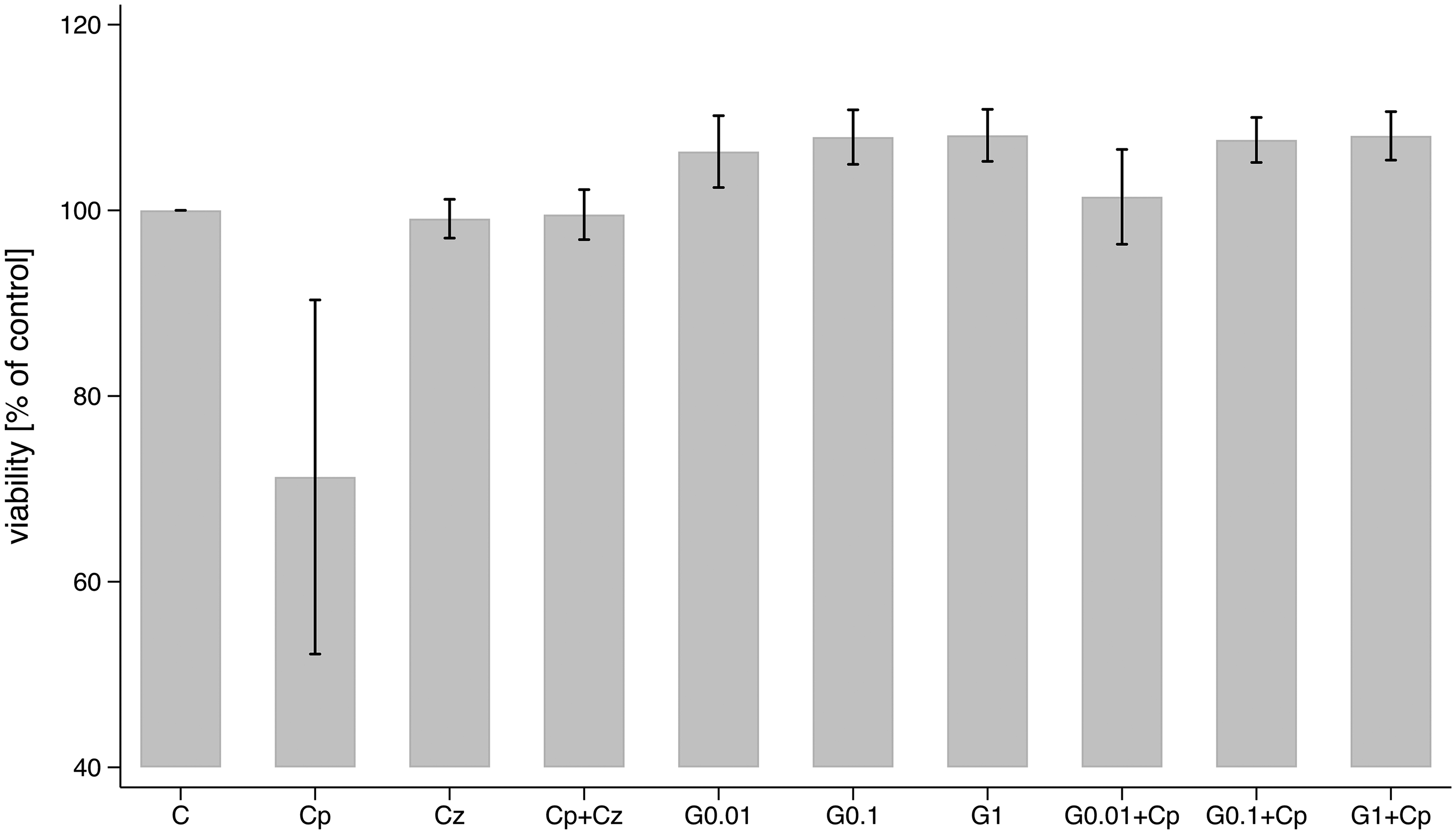
Effect of the herbal remedy on TRPV-1 receptor functionality. 293T-TRPV-1 cells were treated with capsaicin in the presence of capsazepine or increasing concentrations of the herbal remedy for 6 h. Cell viability was measured usin the MTT assay. The experiment was performed in triplicate (total n: 3). C, untreated cells; Cp, Capsaicin; Cz, Capsazepine; G, herbal remedy mixture; 0.01, 0.1, 1: percentage of herbal remedy mixture used. Bars: mean viability (%); range plot with capped spikes: 95% confidence intervals of the means.
Effects on Muscarinic M3 and Serotonin 5-HT3 Receptor Binding
The specific muscarinic M3 receptor antagonist 4-DAMP competed with [3H]-NMS for binding to the membranes of CHO cells expressing the human muscarinic M3 receptor with an IC50 of 2 nM (data not shown). Similarly, the specific serotonin 5-HT3 receptor antagonist MDL 72 222 competed for the binding of [3H]-GR65630 to HEK293 cells expressing on their membranes the human serotonin 5-HT3 receptor with an IC50 of 6.7 nM (data not shown). Therefore, both assays were considered suitable to detect competition binding.
The HMP inhibited the binding of [3H]-N-methylscopolamine to the M3-receptor in a concentration-dependent manner, with an IC50 of 0.63% (Figure 4) but only weakly affected the 5-HT3 receptor. Inhibition of binding of [3H]-GR65630 was evident only at the highest concentration of 10% (IC50: 9.8%; Figure 5).

Competitive binding of the herbal remedy with [3H]-N-methylscopolamine to membranes of CHO cells expressing the human muscarinic M3 receptor. Data represent the mean specific binding ± standard error of the mean of two experiments performed in duplicate (total n = 4). IC50: 0.63%. For additional details see text.

Competitive binding of the herbal remedy with [3H]-GR65630 to membranes of HEK293 cells expressing the human serotonin 5-HT3 receptor. Data represent the mean specific binding ± stanard error of the mean of two experiments performed in duplicate (total n = 4). IC50: 9.8%.
Discussion
In this in vitro study, Gasteo, a combination of 6 active herbal ingredients, decreased the release of inflammatory cytokines, chemokines, and prostaglandins, and restored the epithelial integrity in Caco-2 cells after inflammatory disruption. A similar mechanism has also been observed with a Prunus cerasus L. anthocyanin extract. 32 These results also align with those made with another commercially available plant combination (STW 5-II) that is used to treat FGID symptoms and contains M. recutita, G. glabra, and other ingredients. 33
The HMP tested did not influence the bile acid receptors TGR5 and GPR55 and we observed an antagonistic effect on the TRPV1 receptor.8,15 These observations are interesting, but we cannot conclusively link them to the efficacy of the HMP in vivo. The extract combination bound to human M3 and 5-HT3 receptors with IC50 values of 0.63% and 9.8%, respectively. According to literature, targeted modulation of M3 and 5-HT3 receptors could help relieve symptoms of gastritis and FD.16,17 This is valuable supportive evidence of the role played by the HMP in the symptomatic treatment of FGID, but our observations do not allow us to establish the functional relevance of these results in clinical settings.
Our observations are supported by existing data on the efficacy of the ingredients present in the HMP in alleviating gastrointestinal symptoms. P. anserina is traditionally used for the treatment of inflammation, diarrhea and gastric spasms, dysmenorrhea and urogenital tract disorders.34,35 It contains, among other compounds, ellagic acid, 34 which has been shown in an animal model to protect the ileum, treat diarrhea, and reduce IL-1β, IL-6 and TNF-α levels. 36 Ellagitannins are also present in P. anserina and are metabolized by the gut microbiota to urolithins, which purportedly exert anti-inflammatory and protective effects on the gastrointestinal tract. 34 The protective effects of urolithins have been demonstrated in rodent models of chronic IBS or acute colitis, in which they reduced markers of colonic inflammation and improved mucosal integrity. 37 M. recutita is traditionally used to treat abdominal colic, gastrointestinal pain and ulcers, digestive discomfort, and diarrhea. M. recutita is also considered to exert anti-inflammatory effects. 38 In a randomized, double-blind, placebo-controlled clinical trial a G. glabra extract alleviated FD symptoms. 39 In other clinical studies it reduced IBS-induced diarrhea. 17 G. glabra contains non-competitive and competitive 5-HT3 receptor antagonists that are involved in the reduction of visceral sensation, retard colonic transit, and provide contractile and tonic responses to meal ingestion. 39 In rodent models, liquiritin, a compound present in G. glabra, inhibits TRPV1. 40 A. absinthium is traditionally used as appetizer and to reduce gastrointestinal ailments. It has also been shown to have antimicrobial and diuretic properties. 41 Carvone, a constituent of A. absinthium, acts as a biphasic agonist toward TRPV1: it is stimulatory at low and inhibitory at high concentrations. 42 A. archangelica is used as a carminative and diuretic. It is also utilized to treat gastric ulcers and stimulate gastric and pancreatic secretion. 43 Imperatorin and other furanocoumarins isolated from Angelica species43,44 are partial human TRPV1 agonists. They desensitize the capsaicin-stimulated receptor, thereby reducing the nociceptive response and acting as an analgesic. 44 C. benedicta is traditionally used to regulate menstrual irregularities and stimulate lactation; it is also effective against infectious diseases and has been shown to reduce IL-6 and TNF-α levels in vitro. 45 It is described as having anti-inflammatory properties, as stimulating appetite and digestion, enhancing bile secretion, and attenuating jaundice and flatulence. 46
Our study has some limitations. Plants and plant extracts are complex mixtures of active ingredients that exhibit agonistic, antagonistic, synergistic, toxic, and complementary effects.47,48 The lack of a comprehensive, chemical characterization of the extracts limit reproducibility and the translational interpretation of the observed effects. It is difficult to determine which components of the extracts are responsible for a given activity and the amounts in which they are present. The lack of bioavailability data further hinders the interpretation of these in vitro results, because it is not possible to determine the relevant concentration of active ingredients. All these are also reasons why it is nearly impossible to conduct pharmacodynamic and pharmacokinetic studies in animals or humans with herbal medicinal products. Transepithelial electrical resistance measurements were carried out using the Caco-2 model, which reflects the colonic rather than duodenal epithelium, but a leaky gut can also be found in the colon of IBS patients. 10 Finally, the small sample size prevents the reliable use of inferential statistical tests. Therefore, well-designed efficacy studies are needed to reproduce these in vitro observations in clinical settings.
Conclusion
Our study has shown that the herbal blend contained in Gasteo® reduces the release of inflammatory cytokines, chemokines, and prostaglandins, and restores the epithelial integrity in Caco-2 cells following inflammatory disruption. It also exerts an antagonistic effect on the TRPV1 receptor, which is involved in inflammation and pain. It acts on the M3 and 5-HT3 receptors, which play an important role in gastritis and FD. These preliminary in vitro findings contribute to our understanding of the potential mechanisms of action of the HMP studied, laying the foundation for future in vivo and clinical studies that aim to explain its efficacy in the symptomatic treatment of functional gastrointestinal disorders.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261431441 - Supplemental material for An Herbal Remedy for the Treatment of Functional Dyspepsia Shows Multi-Target Pharmacological Effects in vitro
Supplemental material, sj-docx-1-npx-10.1177_1934578X261431441 for An Herbal Remedy for the Treatment of Functional Dyspepsia Shows Multi-Target Pharmacological Effects in vitro by Nils Günnewich, Kurt Appel, Thorsten Rose, Juan A. Collado Rojas, Eduardo Muñoz, Carsten Tober and Christian Zimmermann in Natural Product Communications
Footnotes
Acknowledgements
Orlando and Liliane Petrini (Breganzona, Switzerland) and Alexandra Sink (Cesra Arzneimittel) critically reviewed the manuscript.
Statement of Ethics
Before blood donation, donors agree (or disagree) to the use of their blood for research purposes by signing a questionnaire (Supplemental data) of the blood donation center of the University Medical Center of Freiburg (Germany). The Ethics Committee (EC) of the Albert Ludwig University (Freiburg, Germany) has provided a written statement stating that provision of blood products for research purposed by the blood donation center does not constitute a procedure requiring ethics committee approval or EC review. All other human or animal cell lines used are commercially available.
Author Contributions
Conceptualization: NG and CZ. Study performance: KA and TR performed all anti-inflammatory tests; EM and JAC conducted the functional receptor assays and TEER study; CT carried out the receptor binding. Manuscript writing: KA, NG, and CZ. All authors contributed to data analysis, drafting and critical review of the paper, gave their final approval of the manuscript and are accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from Cesra Arzneimittel.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christian Zimmermann and Nils Günnewich are employees of Cesra Arzneimittel. All other authors work for CROs conducting preclinical research and report no other conflicts of interests.
Data Availability Statement
The raw data can be made available by Cesra Arzneimittel to scientists upon reasonable request using the e-mail address: medwiss@cesra.de.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
