Abstract
Background
Bauhinia rufescens (B. rufescens) and Ocimum basilicum (O. basilicum) are widely used in African traditional medicine for treating infectious and oxidative stress–related conditions. However, the specific phytochemicals responsible for these effects and their safety profiles remain insufficiently characterized. This study addresses this gap by isolating, identifying, and biologically evaluating individual bioactive compounds from both plants.
Objective
To isolate, characterize, and evaluate the antibacterial, antioxidant, and safety-related cytotoxicity profiles of purified compounds obtained from B. rufescens and O. basilicum. In addition, this study establishes novelty by reporting several compounds for the first time in B. rufescens.
Methods
Ethyl acetate fractions were purified using High-Performance Liquid Chromatography (HPLC), and compound structures were elucidated by Proton Nuclear Magnetic Resonance (1H-NMR) and Mass Spectrometry (MS). Antibacterial activity was evaluated by the broth microdilution method against Escherichia coli (E. coli) (KCTC 2593), Staphylococcus aureus (S. aureus) (KCTC 1916), and Salmonella typhi (S. typhi) (ATCC 14028). Antioxidant activity was assessed using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical scavenging assays, while cell viability was determined via the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay on C2C12 myoblast cells to ensure accurate assessment of safety.
Results
Six compounds were isolated: epicatechin, hydroxyphenylacetic acid, nepetoidin B, rosmarinic acid, rutin and taxiphyllin. Epicatechin, hydroxyphenylacetic acid, and taxiphyllin were identified in B. rufescens for the first time, expanding its known phytochemical profile. Rosmarinic acid and rutin exhibited the strongest antibacterial activity, with Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) values of 0.62 µg/mL against all tested strains. These two compounds also demonstrated the highest antioxidant capacity, displaying the lowest half-maximal inhibitory concentration (IC₅₀) values in both radical scavenging assays. Cytotoxicity testing indicated that all isolated compounds displayed only moderate, dose-dependent reductions in cell viability, with IC₅₀ values ranging from 6.82 ± 0.05 µg/mL to 18.71 ± 0.00 µg/mL, supporting their preliminary safety for potential therapeutic applications.
Conclusion
The isolated compounds from B. rufescens and O. basilicum exhibit notable antibacterial and antioxidant activities coupled with favorable safety-related cytotoxicity profiles. The first-time identification of three compounds in B. rufescens underscores its chemical novelty and therapeutic potential. These findings justify further pharmacological and mechanistic investigations.
Keywords
Introduction
Natural products have long served as a cornerstone of drug discovery and development, contributing significantly to the discovery of antibiotics, anticancer agents, and other pharmacologically active compounds.1,2 Their structural diversity and broad spectrum of biological activities make them particularly valuable in addressing contemporary global health challenges, especially infectious diseases and oxidative stress–related disorders.3,4 Plant-derived secondary metabolites such as phenolic acids, flavonoids, and terpenoids are of particular interest due to their well-documented antibacterial, antioxidant, anti-inflammatory, and cytoprotective properties.5,6 The increasing interest in isolating individual bioactive molecules from medicinal plants reflects the need for new therapeutic candidates with both efficacy and safety. 7
Infectious diseases continue to pose a major global health burden, with conditions such as lower respiratory infections, diarrheal diseases, malaria, tuberculosis, and urinary tract infections accounting for a substantial proportion of worldwide morbidity and mortality.8,9 The alarming rise of antimicrobial resistance further complicates treatment outcomes, increasing the urgency to identify new antibacterial agents with novel mechanisms of action. 10 Bioactive plant compounds are increasingly recognized as promising alternatives or adjuncts to conventional antibiotics due to their ability to inhibit bacterial growth through diverse biochemical pathways.
Oxidative stress is another key contributor to human disease and results from an imbalance between reactive oxygen species (ROS) generation and the body's endogenous antioxidant defenses. Excess ROS can damage lipids, proteins, and nucleic acids, contributing to the pathogenesis of chronic conditions such as cardiovascular diseases, diabetes, neurodegenerative disorders, and aging-related diseases.11,12 The search for potent natural antioxidants is therefore essential, particularly those capable of neutralizing free radicals and preventing oxidative damage in biological systems.13,14
B. rufescens (Fabaceae) and O. basilicum (Lamiaceae) are widely used in Chadian and broader African ethnomedicine for managing microbial infections, inflammation, and oxidative stress–related ailments. Previous studies have demonstrated that these plants are rich in phenolic compounds, flavonoids, and other secondary metabolites associated with antimicrobial and antioxidant effects.15,16 However, despite their long traditional use, the specific bioactive compounds responsible for these therapeutic properties remain insufficiently characterized. A recent increase in phytochemical investigations on B. rufescens and O. basilicum highlights the need for studies that go beyond crude extracts to isolate, identify, and evaluate individual compounds.
The selection of B. rufescens and O. basilicum for this study was based on three major criteria: (i) their long-standing ethnopharmacological use in treating infectious and inflammatory conditions, (ii) their high content of phenolic metabolites known for strong antibacterial and antioxidant properties, and (iii) preliminary phytochemical evidence suggesting the presence of compounds that had not yet been chemically characterized from these species
Furthermore, ethanol was selected as the extraction solvent because it is a safe, food-grade solvent capable of extracting a wide polarity range of phenolic compounds, flavonoids, and glycosides. Ethanol provides high extraction efficiency while preserving thermolabile constituents, and it is less toxic than methanol.19,20 Its ability to extract both hydrophilic and moderately lipophilic molecules makes it particularly suitable for isolating phenolic acids and flavonoids, which were the primary chemical targets of this study. Ethanol has also been widely used in previous phytochemical studies of B. rufescens and O. basilicum, allowing methodological consistency with the literature
This study also addresses a key gap in the literature by reporting, for the first time, the presence of hydroxyphenylacetic acid, epicatechin, and taxiphyllin in B. rufescens. The discovery of these compounds expands the known phytochemical profile of the species and provides important new insights into its chemical diversity and potential pharmacological activities.
Given the traditional use of these plants and their demonstrated biological relevance, this study aimed to isolate and characterize secondary metabolites from the leaves of B. rufescens and O. basilicum, and to evaluate their antibacterial, antioxidant, and safety-related cytotoxicity properties. By combining phytochemical analysis with biological assays, this work provides a foundational understanding of the therapeutic potential of these compounds and supports further exploration of African medicinal plants as sources of novel pharmacologically active agents.
Materials and Methods
Plant Materials
Leaves of B. rufescens and O. basilicum were collected in September 2022 in N’Djari, N’Djamena, Chad (12.12704° N; 15.07897° E). The collected samples were placed in polyethylene bags and transported to the laboratory for processing. Identification of both plant species was performed at the Department of Plant Science, University of N’Djamena, and voucher specimens were deposited under the numbers BR-TCH-2022-09 and OB-TCH-2022-09 for future reference. Approximately 5 kg of fresh leaves of each species yielded 1 kg of dried powder after air-drying, corresponding to a fresh-to-dry conversion ratio of 5:1.
Extraction and Isolation
The dried leaves of B. rufescens and O. basilicum were washed under running water, cut into small pieces, and ground into a fine powder using a mechanical grinder. The plants were dried prior to grinding; no fresh material was ground. One kilogram of powdered material from each plant was macerated in 3 L of 70% ethanol for five days at room temperature with occasional stirring. The resulting extracts were filtered and concentrated using a rotary evaporator at 70 °C, yielding 70 g of crude extract.
Fifty grams of each ethanolic extract were subjected to liquid–liquid fractionation with ethyl acetate, producing approximately 20 g of ethyl acetate fractions. Fifteen grams of these fractions were further purified using high-performance liquid chromatography with methanol as the eluent, resulting in 65 fractions for each plant. Preparative HPLC purification yielded four compounds from B. rufescens hydroxyphenylacetic acid (fraction 10, 4.0 mg), taxiphyllin (fraction 11, 3.1 mg), epicatechin (fraction 14, 3.9 mg), and rutin (fraction 16, 4.2 mg) and two compounds from O. basilicum: rosmarinic acid (fractions 21/22, 4.0 mg) and nepetoidin B (fraction 28, 3.0 mg).
Structural characterization was performed using 1H-NMR and MS, and compound identities were confirmed by comparison with published data.
The complete 1H-NMR and MS spectra for all isolated compounds:
Epicatechin: 1H NMR (500 MHz, MeOD) δ 6.99 (d, J = 2.0 Hz, 1H), 6.82 (dd, J = 8.3, 1.9 Hz, 1H), 6.78 (d, J = 8.1 Hz, 1H), 5.96 (s, 1H), 5.94 (d, J = 1.8 Hz, 0H), 4.84 (s, 1H), 4.22–4.18 (m, 1H), 2.88 (dd, J = 16.8, 4.6 Hz, 1H), 2.76 (dd, J = 16.8, 3.0 Hz, 1H); HR-MS 291.0858 for [M + H]+.
Hydroxyphenylacetic acid: 1H NMR (500 MHz, MeOD) δ 7.05 (d, J = 8.5 Hz, 2H), 6.70 (d, J = 8.5 Hz, 2H), 3.47 (s, 2H); HR-MS 153.0546 for [M + H]+.
Nepetoidin B: 1H NMR (500 MHz, MeOD) δ 7.75 (d, J = 15.8 Hz, 1H), 7.31 (d, J = 2.0 Hz, 1H), 7.26 (d, J = 7.2 Hz, 1H), 7.15 (d, J = 2.1 Hz, 1H), 7.07 (dd, J = 8.1, 2.1 Hz, 1H), 6.93 (dd, J = 8.3, 2.0 Hz, 1H), 6.84 (d, J = 8.2 Hz, 1H), 6.77 (d, J = 8.2 Hz, 1H), 6.48 (d, J = 15.9 Hz, 1H), 5.65 (d, J = 7.3 Hz, 1H); HR-MS 315.0856 for [M + H]+.
Rosmarinic acid: 1H NMR (500 MHz, MeOD) δ 7.57 (d, J = 15.9 Hz, 1H), 7.06 (d, J = 2.0 Hz, 1H), 6.97 (dd, J = 8.2, 2.1 Hz, 1H), 6.80 (d, J = 8.1 Hz, 1H), 6.77 (d, J = 2.0 Hz, 1H), 6.72 (d, J = 8.0 Hz, 1H), 6.63 (dd, J = 8.1, 2.1 Hz, 1H), 6.28 (d, J = 15.9 Hz, 1H), 5.20 (dd, J = 8.5, 4.3 Hz, 1H), 3.17 (dd, J = 14.3, 4.3 Hz, 1H), 3.03 (dd, J = 14.3, 8.4 Hz, 1H); HR-MS 361.0909 for [M + H]+.
Rutin: 1H NMR (500 MHz, MeOD) δ 7.69 (d, J = 2.1 Hz, 1H), 7.65 (dd, J = 8.5, 2.2 Hz, 1H), 6.91–6.88 (m, 2H), 6.43 (d, J = 2.0 Hz, 1H), 6.24 (d, J = 2.1 Hz, 1H), 5.13 (d, J = 7.7 Hz, 1H), 4.54 (d, J = 1.8 Hz, 1H), 3.83 (dd, J = 10.1, 5.7 Hz, 2H), 3.65 (dd, J = 3.5, 1.6 Hz, 1H), 3.56 (dd, J = 9.4, 3.3 Hz, 2H), 3.49 (ddd, J = 13.8, 7.0, 2.7 Hz, 2H), 3.44 (d, J = 9.2 Hz, 1H), 3.41 (d, J = 4.2 Hz, 0H), 3.31–3.28 (m, 1H), 1.14 (d, J = 6.3 Hz, 3H); HR-MS 611.1600 for [M + H]+.
Taxiphyllin: 1H NMR (500 MHz, MeOD) δ 7.41 (d, J = 8.5 Hz, 1H), 6.90–6.84 (m, 1H), 6.81 (d, J = 8.0 Hz, 1H), 5.81 (s, 1H), 4.21–4.17 (m, 1H), 3.93 (dd, J = 11.9, 2.3 Hz, 1H), 3.71 (dd, J = 11.8, 6.2 Hz, 1H); HR-MS 329.1337 for [M + NH4]+.
Antibacterial Assay
Bacteria and Growth Conditions
The antibacterial activity of the isolated compounds was evaluated against three bacterial strains: E. coli (KCTC 2593), S. aureus (KCTC 1916), and S. typhi (ATCC 14028). These strains were selected because they represent pathogenic Gram-negative and Gram-positive bacteria commonly implicated in food-borne and clinical infections. All bacterial strains were cultured at 37 °C on Luria-Bertani agar under aerobic conditions. Bacterial growth procedures followed standard guidelines and are supported by published protocols.17,21
Inoculum Preparation
Bacterial inocula were prepared by streaking each strain onto Luria-Bertani agar and incubating at 37 °C for 24 h to obtain isolated colonies. A few colonies were transferred into test tubes containing 10 mL Luria-Bertani broth and incubated until mid-log phase. The optical density was adjusted to 0.09 at 625 nm (≈108 CFU/mL) and diluted 1:100 to obtain a working inoculum of 106 CFU/mL, following the 0.5 McFarland standard.18–22
MIC and MBC
MIC and MBC values were determined using the broth microdilution method following the Clinical and Laboratory Standards Institute (CLSI) guidelines. 21 In sterile 96-well microplates, 10 µL Luria-Bertani broth was added to each well except the first column. Twenty microliters of each compound (100 µg/mL) were added to the first column and serially diluted two-fold. Ten microliters of inoculum (106 CFU/mL) were added to all wells except blank controls. Tetracycline served as a reference antibiotic. MIC was recorded as the lowest concentration without visible growth. MBC was determined by plating 10 µL aliquots from wells without visible growth onto agar plates and counting colonies after incubation.
All experiments were performed in triplicate.
Antioxidant Assays
DPPH Radical Reduction Test
The free radical scavenging activity was determined colorimetrically using the DPPH decolorization method, with slight modifications to the procedure previously described. In a 96-well microplate, 90 µL of a freshly prepared methanolic solution of DPPH (0.1 mM) was mixed with 10 µL of the sample solutions, which were prepared at concentrations ranging from 100 µg/mL to 5 µg/mL. The mixture was incubated in the dark at room temperature for 10 min. Following incubation, the absorbance was measured at 515 nm using a SpectraMax ABSPlus multi-detection microplate reader. The percentage of DPPH radical inhibition was calculated using the following formula:
Determination of the ABTS Antioxidant Activity
To prepare the antioxidant reagent, a 14 mM solution of ABTS was dissolved in water and reacted with a 4.9 mM potassium persulfate solution. The mixture was incubated overnight at 4°C in the dark to generate ABTS free radicals. The antioxidant assay was performed following the protocol with minor modifications.
25
Briefly, 10 µL of sample solutions, prepared at concentrations ranging from 100 µg/mL to 5 µg/mL, were added to a 96-well microplate and reacted with 90 µL of the ABTS+ reagent. The plates were then incubated in the dark for 10
Cytotoxicity Assay
Cell Culture and Reagents
C2C12 myoblast cells were cultured in Dulbecco's modified Eagle's medium supplemented with 15% fetal bovine serum and 1% penicillin–streptomycin. Cultures were maintained at 37 °C in a humidified incubator with 5% CO₂.
MTT Cell Viability Assay
Cells were seeded at 8000 cells/well in 96-well plates and incubated for 24 h. The next day, they were treated with isolated compounds (1.5, 2.5, 5, 10, and 20 µg/mL). Three controls were included: (i) untreated control, (ii) vehicle control, and (iii) doxorubicin (10 µg/mL) as a positive cytotoxicity control. After 24 h exposure, 100 µL of MTT solution (10% in culture medium) was added and incubated for 2–3 h. Formazan crystals were dissolved in 10% Sodium Dodecyl Sulfate (SDS) in 0.01 M Hydrochloric acid (HCl) and incubated overnight. Absorbance was measured at 450 nm.
Assessment of Cytotoxicity and Safety
Cell viability (%) was calculated relative to the untreated control using the following formula:
Cell viability (%) = (Absorbance of sample / Absorbance of control) × 100
Cytotoxicity was evaluated as a preliminary safety-screening parameter to identify concentration-dependent effects of isolated compounds on normal mammalian cells. The half-maximal inhibitory concentration (IC₅₀) values were calculated and used as comparative indicators of relative cytotoxic potency, rather than definitive toxicity thresholds.
For interpretation purposes, compounds were classified based on their dose–response behavior and comparison with the positive control (doxorubicin), as commonly applied in natural product cytotoxicity screening studies:
IC₅₀ ≤ 5 µg/mL: high cytotoxicity IC₅₀ between 5–20 µg/mL: moderate cytotoxicity IC₅₀ > 20 µg/mL: low cytotoxicity
Compounds exhibiting IC₅₀ values substantially higher than the positive control and maintaining high cell viability at lower tested concentrations were considered to have acceptable preliminary safety profiles for further pharmacological investigation. Untreated and vehicle controls were included to confirm baseline cell viability and exclude solvent-related effects.
Statistical Analysis
All experiments were conducted in triplicate, and results were expressed as mean ± standard deviation.
Statistical significance was determined using one-way Analysis Of Variance (ANOVA) followed by Tukey's post-hoc test. All analyses were performed using GraphPad Prism version 9 (GraphPad Software, San Diego, CA, USA). Values were considered statistically significant when P < .05.
Results
Structure Elucidation of Isolated Compounds
The ethyl acetate fractions from B. rufescens and O. basilicum yielded six purified compounds: hydroxyphenylacetic acid, epicatechin, taxiphyllin, rutin, rosmarinic acid, and nepetoidin B. Structural characterization using 1H-NMR and MS confirmed the identities of all isolates. Notably, hydroxyphenylacetic acid, epicatechin, and taxiphyllin were identified in B. rufescens for the first time, expanding the known phytochemical profile of this species (Figure 1).

Isolated compounds from B. rufescens (1, 2, 5, 6) and O. basilicum (3, 4).
Antibacterial Properties of the Isolated Compounds
The antibacterial activity of the isolated compounds was evaluated against E. coli (KCTC 2593), S. aureus (KCTC 1916), and S. typhi (ATCC 14028). All tested compounds exhibited inhibitory effects with varying intensities, and rosmarinic acid and rutin demonstrated the strongest activity with MIC and MBC values of 0.62 µg/mL across all strains (Table 1).
Antibacterial Activities of Isolated Compounds from B. rufescens and O. basilicum (MIC and MBC Values in µg /ml).

MIC: minimum inhibitory concentration; MBC: minimum bactericidal concentration.
*Antibacterial activity of rosmarinic acid and rutin indicating respectively highest antibacterial potentials with MIC and MBC of 0.62 µg/ml.
Antioxidant Activity (DPPH and ABTS Assays)
All isolated compounds exhibited DPPH and ABTS radical scavenging activity. Rosmarinic acid and rutin demonstrated the strongest antioxidant capacity, with the lowest IC₅₀ values (Figure 2).
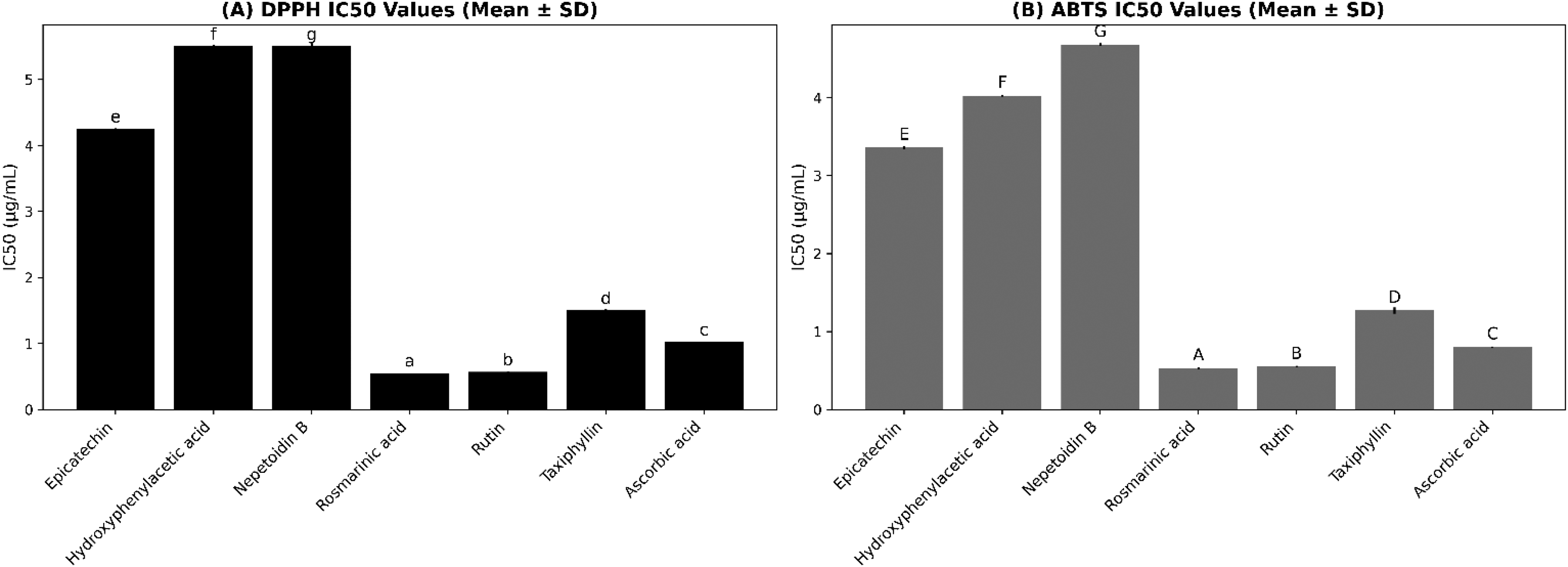
IC50 values (mean ± SD) of isolated compounds determined using DPPH (A) and ABTS (B) radical scavenging assays. Small lowercase letters represent statistically significant differences in the DPPH assay, whereas uppercase letters represent statistically significant differences in the ABTS assay (one-way ANOVA, Tukey post hoc; P < .05). Data are expressed as mean ± SD (n = 3).
Cytotoxicity and Safety Evaluation (MTT Assay)
The effects of isolated compounds on C2C12 myoblast cell viability were assessed using the MTT assay.
All compounds showed dose-dependent but moderate reductions in cell viability, with hydroxyphenylacetic acid and rutin having the lowest IC₅₀ values, while taxiphyllin showed the highest, indicating the lowest toxicity (Table 2).
IC₅₀ Values of Isolated Compounds on C2C12 Cells (with Controls).

IC₅₀ values (µg/mL) are presented as mean ± SD from three replicates. Lowercase letters indicate significant differences among treatments according to Tukey's post-hoc test (P < .05).
All isolated compounds showed low to moderate cytotoxicity, confirming acceptable preliminary safety.
Discussion
This study provides new insights into the phytochemical composition and biological activities of secondary metabolites isolated from B. rufescens and O. basilicum. Six compounds were purified and structurally characterized, and their antibacterial, antioxidant, and safety-related cytotoxicity effects were evaluated. A key finding of this work is the first-time identification of epicatechin, hydroxyphenylacetic acid, and taxiphyllin in B. rufescens, as these compounds have not been reported in previous phytochemical studies of this species.17,18,26 This discovery adds considerable value to the existing literature, which until now had focused predominantly on crude extracts rather than individual pure compounds.
The antibacterial findings demonstrated strong inhibitory and bactericidal effects for rosmarinic acid and rutin, with MICs as low as 0.62 µg/mL. These results confirm the high antimicrobial potential of these phenolic compounds and are in line with established pharmacological reports showing their ability to disrupt bacterial membranes, chelate essential ions, inhibit bacterial enzymes, and destabilize cell-wall integrity.27,28 The activity observed against both Gram-positive and Gram-negative bacteria is particularly important, as such broad-spectrum activity supports their potential application against common pathogens affecting resource-limited communities. 28 In contrast, other compounds such as hydroxyphenylacetic acid and nepetoidin B exhibited only moderate antibacterial activity, although their effects remain noteworthy given that they were tested as isolated phytochemicals rather than enriched mixtures.
The antioxidant results further emphasized the potency of rosmarinic acid and rutin, which recorded the lowest IC₅₀ values in both DPPH and ABTS assays. Their strong radical-scavenging properties reflect their well-known structures, rich in hydroxyl groups capable of donating hydrogen atoms and stabilizing free radicals. Antioxidants of this nature can mitigate oxidative stress, which plays a major role in aging, inflammation, cardiovascular disorders, and several chronic diseases. 13 The superior antioxidant activity observed for these compounds suggests that B. rufescens and O. basilicum may offer protective benefits for populations commonly exposed to oxidative stress from environmental pollutants, malnutrition, or chronic infections. 12
The cytotoxicity results indicated that all isolated compounds exhibited low to moderate toxicity toward C2C12 myoblast cells. IC₅₀ values ranged from 6.82 to 18.71 µg/mL, with hydroxyphenylacetic acid and rutin showing the lowest toxicity thresholds among the group. These results, combined with the antibacterial and antioxidant findings, support the preliminary safety of these compounds and support their potential for future therapeutic applications. 26 Importantly, the inclusion of untreated, vehicle, and positive controls (doxorubicin) ensures robust comparison and aligns with recommended in vitro cytotoxicity standards.
In comparison to previous studies, this work provides a deeper characterization of individual compounds instead of crude extracts, which was a limitation in much of the earlier research on these plants. Earlier studies frequently reported broad biological activities without identifying the precise molecules responsible. Crude extract studies, while informative, often lack specificity and may mask the contribution of individual compounds. 17 Our study overcomes this gap by isolating and characterizing pure compounds, permitting a more accurate evaluation of their pharmacological potential.
Although advanced extraction technologies such as radiation-assisted extraction or ultrasound methods exist, the classical ethanol extraction used here was selected due to its proven ability to preserve thermolabile compounds, its food-grade safety profile, and its high efficiency in extracting phenolics. This solvent was therefore appropriate for the targeted chemical classes and consistent with the methodological framework applied in earlier studies. 19
From a mechanistic perspective, the newly identified compound taxiphyllin a cyanogenic glycoside presents an intriguing finding. Cyanogenic glycosides have dual implications in pharmacology; at low concentrations, they may modulate inflammatory pathways and exhibit bioactivity beneficial for therapeutic development. 7 The role of taxiphyllin in contributing to anti-inflammatory or antioxidant pathways in these plants warrants further investigation, and its first-time identification in B. rufescens opens new research opportunities.
Despite the strengths of this work, several limitations should be acknowledged. First, all experiments were conducted in vitro, which may not fully represent biological effects in vivo. Second, only three bacterial strains were tested, which does not cover the full spectrum of clinically relevant pathogens. Third, antioxidant assays relied solely on chemical radical scavenging methods rather than cellular antioxidant systems. Fourth, mechanistic studies such as membrane disruption assays, enzyme inhibition, or molecular docking were not included and will be necessary to confirm the pathways underlying the observed bioactivities. Finally, the yields of some isolated compounds were low, limiting further biological testing. These limitations highlight the need for expanded investigations using additional pathogens, in vivo models, mechanistic assays, and higher-yield isolation approaches.
Overall, the present study demonstrates that B. rufescens and O. basilicum are valuable sources of bioactive compounds with strong antibacterial and antioxidant potential and favorable cytotoxicity profiles. By identifying novel compounds in B. rufescens and confirming the potent bioactivity of known phenolics, this research reinforces the ethnomedicinal importance of these plants and supports their continued exploration in drug discovery. The results justify further pharmacological studies, including mechanism elucidation, synergistic testing with conventional antibiotics, and evaluation in animal models.
Conclusion
This study provides important new insights into the phytochemical composition and biological activities of compounds isolated from B. rufescens and O. basilicum. Six pure compounds were successfully isolated, and three of them epicatechin, hydroxyphenylacetic acid, and taxiphyllin were identified for the first time in B. rufescens, expanding the known chemical profile of this medicinal plant. The isolated compounds demonstrated notable antibacterial and antioxidant activities, with rutin and rosmarinic acid exhibiting the strongest effects. Cytotoxicity evaluation showed that all compounds possessed acceptable safety-related profiles in in vitro assays, reinforcing their potential applications in therapeutic development.
These findings support the traditional use of B. rufescens and O. basilicum and highlight their relevance as valuable sources of bioactive natural products with promising antibacterial, antioxidant, and pharmacological properties. The study underscores the importance of isolating and characterizing individual compounds rather than relying solely on crude extracts, thereby enabling clearer mechanistic understanding and more targeted therapeutic exploration.
Future research should focus on in vivo evaluations of these compounds, deeper mechanistic studies including membrane integrity assays, enzyme inhibition tests, and molecular docking and potential synergistic interactions with existing antibiotics. It is also recommended to explore other medicinal plants from similar ecological regions using comparable isolation methods, as this may reveal additional novel compounds with therapeutic value. Expanding this research across more species could contribute significantly to drug discovery efforts and support the development of natural-product-based interventions for infectious and oxidative stress–related diseases.
Footnotes
Acknowledgments
The authors express their gratitude for the support provided by the Korea Institute of Science and Technology (KIST), Gangneung Institute of Natural Products, the Partnership for Skills in Applied Sciences, Engineering and Technology (PASET), and the SACIDS Foundation for One Health. Their institutional, technical, and logistical contributions were essential to the successful completion of this study.
Ethical Considerations
This study did not require ethical approval because it involved no human participants, no human data, and no animal experimentation. All experimental procedures were limited to in vitro assays using bacterial cultures and the C2C12 myoblast cell line. According to the ethical guidelines of the Korea Institute of Science and Technology (KIST), Gangneung Institute of Natural Products, Gangneung, Republic of Korea, in vitro studies using established cell lines do not require review or approval by an Institutional Review Board (IRB). Therefore, ethical approval was waived.
Author Contributions
Conceptualization, H.H.A.-r. and J.-S.P.; methodology, H.H.A.-r., J.-S.P., A.S.H., G.G.B., and C.-H.P.; software, H.H.A.-r. and J.-S.P.; validation, J.-S.P., A.S.H., G.G.B., and C.-H.P.; formal analysis, H.H.A.-r.; investigation, H.H.A.-r.; resources, H.H.A.-r.; data curation, H.H.A.-r.; writing original draft preparation, H.H.A.-r.; writing review and editing, H.H.A.-r., J.-S.P., A.S.H., G.G.B., and C.-H.P.; visualization, A.S.H. and J.-S.P.; supervision, J.-S.P., A.S.H., G.G.B., and C.-H.P.; funding acquisition, H.H.A.-r. and J.-S.P.
All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Korea Institute of Science and Technology (KIST, Gangneung Institute of Natural Products) and the Partnership for Skills in Applied Sciences, Engineering and Technology (PASET).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article. No additional datasets were generated or analyzed.
This study was not preregistered on the Open Science Framework (OSF). All experiments followed predefined protocols, and no post hoc changes were made to the study design or outcome measures.


