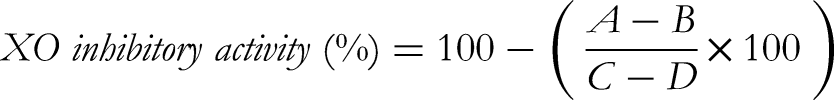Abstract
Objectives
Cosmos sulphureus Cav. has a rich history of traditional medicinal use in Southeast Asia, particularly for the treatment of diabetes, high blood pressure, inflammation and infections. This study aimed to optimize flavonoid extraction and evaluate the hypouricemic effects for the gout treatment of the extracts from the flowers of CS.
Methods
Extraction optimization was conducted using response surface methodology with a factorial design that included two variables – ethanol concentration (25%–70%) and solvent-to-material ratio (10-20 mL/g) - and three responses: extraction efficiency, total flavonoid content (TFC), and IC50 values for xanthine oxidase inhibition. Toxicity studies followed the Guidelines for Preclinical and Clinical Trials of Traditional Medicine and Herbal Medicines issued by the Vietnam Ministry of Health as well as the OECD (2001) with minor modifications. The pharmacological activity was assessed in acute and chronic potassium oxonate–induced hyperuricemia models.
Results
The optimization CS extraction conditions were identified as percolation using 70% ethanol with a solvent-to-material ratio of 20 mL/g. The extraction process was successfully scaled up to 1 kg of raw material per batch, demonstrating its stability and applicability for further pharmacological studies. Chemical characterization confirmed the extract was rich in flavonoids, with coreopsin and luteolin identified as major constituents. In a model of acute and chronic hyperuricemia induced by potassium oxonate (i.p), the 250 mg/kg CS flower extract demonstrated uric acid-lowering effects similar to the reference drug allopurinol at 10 mg/kg.
Conclusions
These findings highlight the novelty of using Cosmos sulphureus Cav. as a potent natural source for anti-hyperuricemic agents, supporting its potential for pharmaceutical development.
Introduction
Gout, a purine metabolism disorder characterized by hyperuricemia and oxidative stress, is an increasingly prevalent condition worldwide. 1 Current pharmacological interventions for gout treatment, including colchicine, anti-inflammatory drugs, and anti-hyperuricemic agents, often present adverse effects.2–4 Allopurinol, a first-line treatment, is associated with hepatotoxicity, nephrotoxicity, and hypersensitivity reactions, particularly in individuals with the HLA-B*5801 allele, which comprises approximately 18.6% of the Vietnamese population. 5
Flavonoids are naturally occurring phytochemicals with diverse biological activities and increasing pharmacological relevance. Recent studies have emphasized the biological effects of flavonoid-rich fractions, reporting pronounced antioxidant, anti-inflammatory, enzyme-inhibitory, and cytoprotective properties.6–8 With respect to xanthine oxidase (XO) inhibition, flavonoids have been reported to exhibit considerable inhibitory potential, contributing to their antioxidant and anti-hyperuricemic effects.7,9–14 These inhibitory activities are mediated through multiple mechanisms, including competitive interaction at the molybdenum cofactor site of XO and direct scavenging of reactive oxygen species.7,9–14
Cosmos sulphureus Cav. (CS, C. sulphureus) It is an ornamental plant widely cultivated across diverse geographical regions, including Vietnam and globally 10–15. It is an erect, branching herb that reaches 1–1.5 m in height, with a pubescent covering throughout. Inflorescences are bright yellow to orange-yellow capitula, borne on 10–20 cm long peduncles, terminal or axillary. Each capitulum comprises 7–8 ray florets and numerous disc florets. Ray florets are ligulate, 2–3 × 1–1.5 cm, orange-yellow or metallic yellow, with a 5 mm long orange-yellow corolla tube. Disc florets are tubular, 5–6 mm long, and orange-yellow. Anthers are black with acute or slightly obtuse apices. Style branches are elongated and pubescent (Figure 1). C. sulphureus Cav. has been traditionally used in China, Thailand, and Indonesia for its antimicrobial and anti-inflammatory activities.15–17 Studies indicate that its bioactive compounds inhibit microbial growth by disrupting membrane integrity and modulating inflammatory mediators. Additionally, the plant shows antidiabetic potential through α-glucosidase inhibition and anti-obesity effects via lipase suppression, collectively supporting its role in metabolic disorder management.15–17 The therapeutic potential of C. sulphureus Cav. is largely attributed to its diverse phytochemical profile. Previous studies have isolated several constituents from its flowers, including flavonoids such as quercetin, luteolin, and coreopsin, as well as carotenoids, which are known to inhibit the enzyme xanthine oxidase to reduce uric acid levels.18–21 These findings underscore the promise of C. sulphureus Cav. as a valuable source of xanthine oxidase inhibitors for the management of gout and related conditions.

Morphological characteristics of Cosmos sulphureus Cav. (A) Whole CS plant; (B) CS leaves; (C) CS flowers; (D) Shape of ray and disc florets.
To the best of our knowledge, this is the first study to investigate xanthine oxidase inhibitory and anti-hyperuricemic effects of Cosmos sulphureous Cav., which is also the first species in the Cosmos genus examined for these activities Our study addresses this critical knowledge gap through a multifaceted approach. Firstly, the extraction from CS flowers was optimized using RSM to analyze relationships between extraction parameters with the extraction yields and flavonoid contents. 22 Secondly, the xanthine oxidase inhibitory activity of the optimized extracts was evaluated using a standardized in vitro assay. Finally, and most importantly, its anti-hyperuricemic activity potential was assessed on a well-established potassium oxonate-induced hyperuricemia animal model. This research provides crucial insights for the initial development of pharmaceutical formulations containing C. sulphureus Cav. extracts for gout management. The results contribute to the scientific validation of traditional uses of C. sulphureus Cav. and promote its potential integration into modern therapeutic applications.
Although several bioactive flavonoids have been identified in this species, a comprehensive understanding of their synergistic interactions and the precise mechanisms underlying their anti-gout effects remains insufficiently understood. In particular, the interplay between flavonoid-mediated xanthine oxidase inhibition and other potential mechanisms, such as anti-inflammatory or renal protective effects, has yet to be clarified. The paucity of in vivo studies on these bioactive compounds hinders the phytopharmaceutical development and underscores the urgent need for comprehensive research.
Materials and Methods
Chemicals and Reagents
Pharmaceutical-grade ethanol and ether as the extraction solvent were bought from OPC company (Vietnam). Coreposin was provided by Herbest (China). Xanthine oxidase (20 U/mL), luteolin and allopurinol were procured from Sigma Aldrich (USA). Acetonitrile was obtained from Fisher Chemical (USA).
Sample Preparation
The flowers were collected in Dong Nai province, Vietnam. The specimens were air-dried to achieve constant weight, vacuum-sealed in polyethylene bags, and labeled as CS-DL-2022-1. They were genetically decoded for identification as Cosmos sulphureus. The dried samples were stored at 0–4 °C until further extraction procedures.
Animal Models
All procedures involving animals were strictly followed by the Institute for Laboratory Animal Research (ILAR) Guidelines for the Care and Use of Laboratory Animals. The experiments were conducted in the Laboratory Animal Unit, Department of Pharmacology, University of Medicine and Pharmacy, Ho Chi Minh City. Institute of Cancer Research (ICR) mice, 5–6 week old and weighing approximately 22 ± 2 grams, were purchased from the Institute of Vaccines and Medical Biologicals (IVAC) in Nha Trang. Prior to the initiation of the experimental procedures, the animals were randomized and acclimatized to laboratory conditions for a period of five days. During this time, they were provided with a standard diet and water ad libitum.
Chemical Characterization of Cosmos sulphureus Flower Extracts
Total flavonoids, alkaloids, coumarins, saponins and tannins were determined to obtain a comprehensive phytochemical screening profile of the CS flower. 20 grams of dried flower powder (after sieving 2 mm mesh) were mixed with 100 mL of ether, shaken for 30 min, and filtered to obtain the ether extract. It was then extracted with 100 mL of ethanol 96% under reflux for 30 min, cooled, and filtered to get the ethanol extract. This process was repeated with 100 mL of water to obtain the aqueous extract. These extracts were chemically determined with our routine analytical methods.
The Extraction Optimization
Flavonoid Extraction from Cosmos sulphureus Flower
The extraction solvent significantly impacts in vitro biological activities. 23 Additionally, flavonoids are susceptible to temperature effects. Percolation extraction not only mitigates temperature-related issues but is also applicable on an industrial scale. Therefore, this method was chosen and the maceration time (12, 24, 48 h) was evaluated. A total of 100 grams of powdered CS flowers were subjected to extraction using ethanol through a percolation method after 12-h percolation. Main extraction parameters, including ethanol concentration (25, 45, 70, 96%) and solvent-to-material ratio (10, 15, 20 mL/g), were systematically optimized to maximize extraction efficiency, total flavonoid content (TFC), and xanthine oxidase inhibition potential (measured by IC50). The resulting extracts were concentrated and dried using a rotary evaporator, and the dried extracts were subsequently stored for further analysis and investigations.
Extraction Efficiency Assessment
The extraction efficiency (%) was determined by comparing the dry weight of the extract to the initial mass of the medicinal herb, as follows:
Quantification of Total Flavonoid Contents
TFC in CS flower extract was determined spectrophotometrically using quercetin as a standard. A quercetin stock solution (0.5 mg/ml) was prepared and diluted to create a standard curve (1-16 μg/mL). The extract sample (100 mg) was dissolved in water, diluted with methanol to 100 mL, and filtered. 1 mL of sample or standard was mixed with 8 mL of methanol and 1 mL of 2% AlCl3 and allowed to react for 10 min. The absorbance was measured at 450 nm. Measurements were performed in triplicate. TFC was calculated using the formula:
Where C: flavonoid concentration from the standard curve (mg/mL); K: dilution factor;p: quercetin purity (%); m: sample weight (g); W: moisture content of the extract (%).
Extraction Optimization by Response Surface Methodology
The extraction parameters were optimized using Design Expert v13.0 (Stat-Ease, USA) software. The design incorporated two independent variables of extraction parameters including (X1) ethanol concentration (in range of 25-70%), (X2) solvent-to-material ratio (in range of 10-20 mL/g); three response outcomes including (Y1) TFC extraction yield (%), (Y2) TFC (mg/g), (Y3) IC50 value for xanthine oxidase inhibition (µg/mL), with a resolution IV factorial design. The experimental design comprised 12 individual trials. Each assay was conducted sequentially, and the extracts were analyzed in triplicate to ensure accuracy and reproducibility. The data were input in the software with various transformation algorithms, including No Transform, Square Root, Natural Log, Inverse Square Root, Inverse, and Power. A one-way analysis of variance (ANOVA) was performed to assess the statistical significance of the model, with a 95% confidence interval (CI). Response surface plots were generated to visually represent the interactions between independent and dependent variables. A p-value of less than 0.05 was considered statistically significant, confirming the reliability of the model.
Toxicity Studies
Toxicity tests follow the Guidelines for Preclinical and Clinical Trials of Traditional Medicine and Herbal Medicines issued by the Vietnam Ministry of Health (V.M.o.H 2015) as well as the OECD (2001) 24 with minor modifications.
Acute Toxicity Study
The CS extract sample was dissolved in distilled water to determine the maximum dose of CS extract that could be administered through the oral syringe (Dmax). Mice were fasted for 12 h before the experiment and divided into 2 groups (n = 10; 5 male - 5 female each) including control (distilled water, p.o.) and CS extract group (Dmax, p.o.). Activities, behaviors, fur condition, urination and death of mice were recorded during the next 72 h. If no death or abnormal behaviors were found, the observation would be continued for more than 14 days.
Sub-Chronic Toxicity Study
15 male and 15 female mice were classified into 3 groups each (n = 5 per group) and received oral treatment with distilled water (control group), 250 mg/kg of CS flower extract 25 and 500 mg/kg of CS flower extract, respectively. On the first day (t0), 30th day (t30) and 60th (t60) day of the experiment, mice were weighed and their blood samples were collected by snipping mice's tails. The whole blood samples were used for evaluating hematological parameters. Serum samples obtained by centrifuging the blood samples at 3000 rpm in 10 min at 25 °C were used to evaluate biochemical parameters. At the end of the experiment, mice were dissected to collect hearts, livers and kidneys for macroscopic and microscopic observation.
Investigation of Xanthine Oxidase Inhibitory Activity of Cosmos sulphureus Flower Extracts
The procedure follows the study of Nessa et al (2010) 26 with minor amendments. CS extract was prepared into a stock solution of 10 mg/ml, which was then diluted with phosphate buffer solution to working solutions. The assay mixture comprised of 0.1 mL CS extract from stock or working solutions, 0.475 mL phosphate buffer pH 7.5, 0.025 mL XO enzyme 0.2 U/mL (incubated for 10 min at room temperature) and 0.4 mL xanthine 2 mM. The mixture was then incubated for 30 min at room temperature. The reaction was stopped by the addition of 0.5 mL HCl 1 M and 0.5 mL phosphate buffer pH 7.5. The absorbance of the final solution was measured at 295 nm. The control mixture was prepared in the same way as described, but without 0.1 mL of CS extract. The blank mixtures were prepared as the sample and control mixture, but without 0.025 mL of enzyme XO. Lastly, the blank solution containing 0.025 mL phosphate buffer pH 7.5 was prepared.
XO inhibitory activity (expressed as the XO inhibitory percentage) was calculated as follows:
Where A, B, C and D: the average absorbance of assay samples: blank - assay sample, control sample, blank - control sample, respectively.
IC50 values were calculated based on the inhibitory percentage of each concentration and linear regression analysis, then compared with the IC50 value of Allopurinol.
Investigation of Anti-Hyperuricemic Effects of Cosmos sulphureus Flower Extracts
The male ICR mice were randomly divided into 5 groups (n = 8). Each group was treated as follows:
Control group: distilled water (p.o.). PO 300 group: potassium oxonate (300 mg/kg, i.p.) CS 250 group: potassium oxonate (300 mg/kg, i.p.) + CS flowers extract (250 mg/kg, p.o.)
25
CS 500 group: potassium oxonate (300 mg/kg, i.p) + CS flowers extract (500 mg/kg, p.o) Allopurinol group: potassium oxonate (300 mg/kg, i.p) + Allopurinol (10 mg/kg, p.o)
Anti-Hyperuricemic Effects for the Acute Hyperuricemia
Treatment protocol: The oral administration was conducted 1 h after the injection of potassium oxonate. Two hours after the injection, blood samples were collected into K2EDTA tubes to measure uric acid level
Prevention protocol: Mice from all groups received oral treatment for 4 consecutive days prior to the injection. On the fifth day, the control group was injected with NaCl 0.9% (i.p.) while the other groups were injected with potassium oxonate (300 mg/kg, i.p.). After 1 h, mice received oral treatment and uric acid levels were measured as described in the first experiment.
Anti-Hyperuricemic Effects for sub-Chronic Hyperuricemia
The injection was performed daily for 7 consecutive days, followed by oral treatment after 1 h. On the seventh day, blood samples were collected to measure uric acid level.
Quantification of Uric Acid Level in Plasma
Blood samples were centrifuged at 3000 rpm for 10 min at room temperature. Plasma was then drawn for uric acid quantification with Uric Acid Liquicolor Reagent Kit (Human Diagnostic Ltd Co, Germany) containing 3,5-dichroro-2-hydroxybezensulfonic acid (DCHBS) and 4-aminophenazon (PAP). 500 µl of the reagent was added into three separate tubes containing 10 µl of control (distilled water), standard (uric acid) and plasma sample. The tubes were shaken and incubated at room temperature in 10 min. Then, the absorbance was measured at 546 nm on semi-automatic biochemistry analyzer Screen Master 3000 (Biochemical Systems Ltd Co, Italy).
27
The level of uric acid was calculated by the formula:
Where, C and n: the level of uric acid in the plasma and standard samples, respectively (mg/dL),ODs and ODstd: the absorbance of the plasma and standard samples, respectively.
Hematoxylin and Eosin (H&E) Staining
The organs were collected and soaked in formalin 10% solution for 24 h at room temperature. paraffin-embedded specimens were prepared, and 5 µm sections were stained with hematoxylin and eosin (Sigma-Aldrich, St. Louis, MO, USA) 28 and observed by Olympus BX40 microscope.
Statistical Analysis
Data were presented as mean ± SEM (standard error of mean). The statistical analysis was carried out by one-way or two-way analysis of variance (ANOVA), followed by a Tukey post-hoc test using GraphPad Prism 9.5 (GraphPad Software Inc., USA). The differences were considered to be statistically significant at p < 0.05.
Results
Chemical Composition Profile of Cosmos sulphureus Flowers
The phytochemical screening of CS flowers revealed a diverse array of bioactive compounds, including: flavonoids, lipids, carotenoids, polyphenols, tannins, organic acids, and reducing compounds (Table 1). 29
Chemical Constituent Results of Cosmo sulphurus Flower.

(−) Negative result, (+) Weakly positive, (++) Moderately positive, (+++) Strongly positive.
Optimization of Extraction Process from Cosmos sulphureus flowers
There is no significant difference in extraction efficiency or TFC across the three maceration time points (12, 24, 48 h) (p > 0.05, one-way ANOVA). However, the shortest maceration time of 12 h was selected for the extraction process so that the procedure can be applied to various scales of production, from laboratory experiments to industrial applications.
As seen in the extraction results (Table 2), 70% ethanol was the optimal solvent in the percolation extraction method as it gives the highest flavonoid content, and in vitro biological activity.
Extraction Parameters and Results.

The RSM analysis shows that the experimental data fit accurately to a quadratic model. The Square Root approach effectively represented the correlation between independent variables and extraction efficiency, while the power algorithm was suitable for modeling TFC and xanthine oxidase inhibition. The extraction parameters, including ethanol concentration and solvent-to-material ratio, significantly impacted extraction efficiency, TFC, and xanthine oxidase inhibition (Table 3
Analysis of the Influence of Independent Variables on Dependent Variables.

S: significant (p < 0.05); NS: non-significant (-).
Ethanol concentration (X1) exhibited a statistically significant effect on extraction efficiency (Y1) at both first and second orders (Figure 2A). However, no statistically significant interaction was observed between ethanol concentration and solvent-to-material ratio (X1X2) on extraction efficiency (Y1). The extraction yields varied positively with both the solvent types and the ratio of raw material to solvent. Ethanol concentrations from 45% to 75% provided the highest extraction yields. These variables significantly influenced TFC (Figure 2A). Extraction temperature and ethanol concentration exhibited both first- and second-order effects on TFC, while solvent-to-material ratio predominantly affected at a first-order level. Importantly, no statistically significant interaction was also observed between the variables in relation to flavonoid content. Analysis of the dependent variable's range shows that the extraction yield varies positively with both the type of solvent and the ratio of raw material to solvent. Ethanol concentrations ranging from 60% to 75%, combined with a solvent ratio of 1:20, yield the highest TFC. Similarly, these extraction conditions significantly influenced xanthine oxidase inhibition (Figure 2C). Ethanol concentration exerted both first- and second-order effects, with high concentrations (70% and 96%) positively correlating with inhibition potential. Extracts with high-concentration ethanol (70% and 96%) exhibited more potent xanthine oxidase inhibition and anti-inflammatory activities compared to those obtained using ethanol 45% and water. However, the products extracted with 45% and 70% ethanol had higher antioxidant activity than those extracted with 96% ethanol, possibly due to DPPH model preferences for polar compounds. Water extract demonstrated the lowest biological activities and flavonoid content, leading to its exclusion from further experiments. No statistically significant interactions between variables were observed in relation to xanthine oxidase inhibition.

3D response surface plots showed the interactions between the variables. (A) Effect on extraction yield; (B) Effect on TFCl; (C) Effect on xanthine oxidase inhibition.
The experimental conditions were proposed based on the optimization result to achieve the target design objectives for the extracts, as detailed in Table 4. Statistical analysis, including ANOVA, indicated no significant differences between the observed Yi values and the predicted Yi values, confirming the model's accuracy. As a result, the optimization of the CS extraction process produced the following parameters: the extraction method is percolation, an ethanol concentration of 70%, and a solvent-to-material ratio of 20 mL/g.
Optimization of Extraction Process Conditions Using Design-Expert Software.

Toxicity Evaluation
Acute Toxicity Evaluation
A total of 5 grams of CS total extract was diluted with 3.9 mL distilled water to obtain Dmax of 25.64 g/kg body weight. After the oral administration of CS extract at that dose, no death and abnormal behavior in comparison with the control group were recorded in the next 72 h and 14 days afterwards. Hence, CS extract at the Dmax showed no acute toxicity in mice. The result was consistent with Megnigueu et al 30 In this study, no mortality in mice was observed at 2000 mg/kg of the CS extract. As CS flowers extract showed no acute toxicity at the dose 25.64 g/kg, no death was recorded LD50 value of this extract could not be identified. Based on the Globally Harmonized System of Classification and Labelling of Chemicals (GHS), CS extract at Dmax would fall into Category 5. This category is for substances with relatively low acute toxicity, typically having an oral LD50 between 2000 and 5000 mg/kg. Therefore, Cosmos sulphureus flowers extract can be classified as “relatively low acute toxicity” in Category 5.
Sub-Chronic Toxicity Evaluation
Hematological parameters: Red blood cell (RBC) and haemoglobin (Hb) values of CS 250 and CS 500 groups of both sexes showed no significant differences from the control groups at measurement time (p > 0.05). There was also no significant change after 30–60 days (p > 0.05). However, hematocrit (Hct value) at t60 of the female mice in the CS 500 group experienced an increase as compared to the same group at t0 (*p < 0.05), as well as to the same-sex control group at the same time (#p < 0.001) (Table 5). Regarding mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV) and red cell distribution width (RDW), there were no significant differences between CS 250 and CS 500 groups of both sexes from the control groups (p > 0.05), even after 30 to 60 days (p > 0.05) except for the CS 250 group (MCHC, $p < 0.05), CS 250 and CS 500 groups (MCV, $p < 0.001) of female mice, in which statistical declined at t60 in comparison to its value at t30. In terms of white blood cells (WBC) and platelets (PLT), there were no significant differences between all groups with the control group at the measurement time (p > 0.05). An increase in PLT value from t30 to t60 of the CS 500 group of female mice was noticed (p < 0.05).
RBC, Hct, Hb, MCH, MCHC, MCV, RDW, WBC and PLT Values of Different Groups in the Sub-Chronic Toxicity Assessment.

Data were expressed as Mean ± SEM, n = 5 mice each group. Comparisons between the groups were made by two-way analysis of variance (ANOVA) followed by Tukey post-hoc test. p < 0.05: difference from the same group at t0; p < 0.001: difference from the same-sex control group at the time of measurement.
Biochemical parameters: no significant difference was observed between the control and treated groups (p > 0.05), nor within each group after 3060 days (p > 0.05) (Table 6). According to the previous sub-chronic toxicity evaluation for 28 days, 30 CS extract at the doses of 250, 500, 750 mg/kg did not cause any hepatotoxic effect as the AST and ALT enzymes showed no appreciable increase in the treated animals. The extract at investigated doses did not cause renal impairment or kidney damage, because there was no significant change in the renal and hepatic function of mice after 30–60 days.
Biochemical Parameters of Different Groups in the Sub-Chronic Toxicity Assessment.
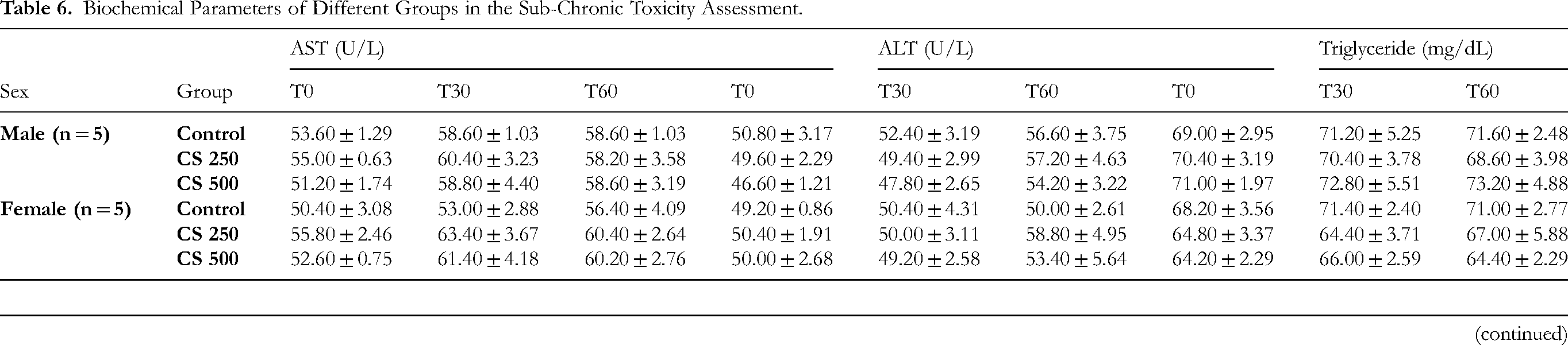
Data expressed as Mean ± SEM, n = 5 mice each group. Comparisons between the groups were made by two-way ANOVA and Tukey post-hoc test.
Histology: No macroscopic differences between CS 250, CS 500 and the control groups were found after 60 days. Heart tissues were within normal limits, with no endothelial cell damage in the blood vessels, no myocardial interstitial fibrosis, and no congestion observed (Figure 3A). Liver tissues in both physiological and test groups showed no fibrosis or bile duct calcification; however, severe inflammation was observed in cells of female mice (Figure 3C). Kidney tissues in both control and test groups showed interstitial nephritis, with normal tubular cells observed (Figure 3B).

Photomicrographs at 400x magnification of (A) heart; (B) kidney; (C) liver of mice in control, CS 250 and CS 500 groups. *Hematoxylin and eosin (H&E).
Xanthine Oxidase Inhibitory Activity Investigation
With an IC50 value of approximately 404.653 ± 2.26 μg/mL (Table 4), CS extract is shown to be less effective than allopurinol, a highly potent xanthine oxidase inhibitor (IC50 = 2.36 ± 0.04 μg/mL(equivalent to 13.9 µM).
Anti-Hyperuricemic Activity Investigation
Acute Hyperuricemia
Treatment protocol: According to the results (Figure 4A), mice in the PO 300 group had a 37.63% increase in uric acid level, which was significantly different from the control group (p < 0.0001). The allopurinol group significantly decreased uric acid level as compared to the PO 300 group (30.09%; p < 0.0001), and had no significant difference from the control group (p = 0.9636). The decrease in uric acid level of the CS 250 and CS 500 groups in comparison with the PO 300 group was 18.35% and 13.67% (p = 0.0008; p = 0.0178, respectively). Both groups showed anti-hyperuricemia effects, yet did not exert a dose-dependent manner. CS 250 and 500 groups did not significantly differ from the allopurinol group (p > 0.05). However, the uric acid level of the CS 500 group remained higher than the control group (p = 0.0178).
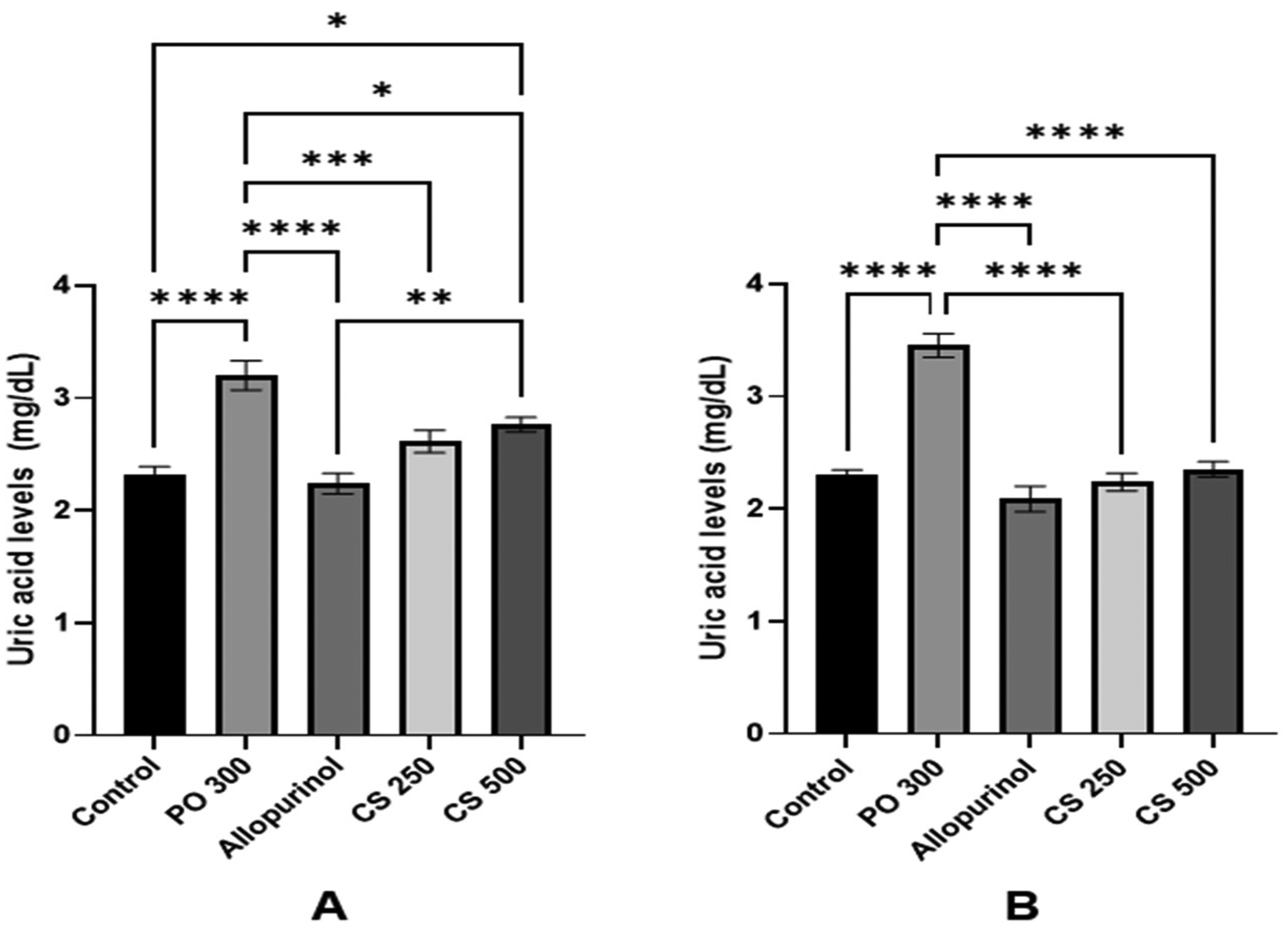
Effect of Cosmos sulphureus flower on the acute hyperuricemia. (A) Treatment of acute hyperuricemia; (B) Prevention of acute hyperuricemia. Data were expressed as Mean ± S.E.M, n = 8 mice each group. Comparisons between the groups were made by one-way analysis of variance (ANOVA) followed by Tukey post-hoc test (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001).
Prevention protocol: Results (Figure 4B), suggested that the PO 300 group experienced a significant increase in uric acid level by 50.00% as compared to the control group (p < 0.0001). The allopurinol group reduced uric acid production by 39.49% when compared to the PO 300 group (p < 0.0001); meanwhile, it did not differ from the control group (p = 0.4236). The decrease in uric acid level caused by CS 250 and CS 500 in comparison with the PO 300 group was 35.15% and 31.88% (p < 0.0001). Both groups showed anti-hyperuricemia effects, yet did not exert a dose-dependent manner. In this experiment, no significant difference between the CS 250, CS 500 and allopurinol groups was recorded (p > 0.05).
Sub-Chronic Hyperuricemia
As can be seen in Figure 5, the PO 300 group increased its uric acid level by 79.53%, which is statistically different from the control group (p = 0.0002). The allopurinol group showed a significant uric acid-lowering effect (39.09%) when compared to the PO 300 group (p = 0.0012), while it did not differ from the control group (p = 0.9784). The effects of CS 250 and CS 500 groups on decreasing uric acid level were 36.16% (p = 0.003) and 38.43% (p = 0.0015), respectively, in comparison with the PO 300 group. Dose-dependent manner between these two groups was not recorded. There was no significant difference between CS 250, CS 500 and the allopurinol group (p > 0.05).

Effect of Cosmos sulphureus flower on the sub-chronic hyperuricemia. Data were expressed as Mean ± S.E.M, n = 8 mice each group. Comparisons between the groups were made by one-way analysis of variance (ANOVA) followed by Tukey post-hoc test. **p < 0.01, ***p < 0.001 versus control group.
Discussions
The phytochemical screening of Cosmos sulphureus flowers revealed a diverse spectrum of bioactive constituents, including flavonoids, lipids, carotenoids, polyphenols, tannins, organic acids, and reducing compounds. These results are consistent with the findings of Jadav and Gowda, thereby reinforcing the established chemical profile of this species. 29
The percolation extraction technique ensures a continuous supply of fresh solvent to the plant matrix and enables exhaustive recovery of active constituents without thermal degradation—an essential advantage for preserving thermolabile phytochemicals. The absence of time-dependent variation in extraction efficiency and TFC is likely attributable to the porous floral structure, which facilitates rapid solvent penetration and efficient mass transfer. Based on these findings, and in accordance with the Vietnamese Pharmacopoeia V, a 12-h maceration period was selected as the minimum effective duration for optimizing extraction performance. Furthermore, percolation is suitable for a wide range of production scales, from laboratory research to industrial manufacturing, owing to its simple equipment requirements and relatively low operational cost.
Among the solvents tested, 70% ethanol demonstrated optimal performance in the percolation process, yielding the highest flavonoid content and the most pronounced in vitro biological activity. These observations are consistent with earlier studies by Megnigueu, Respatie, and Ortega-Medrano, who also employed 70% ethanol to extract CS for evaluating its biological properties.30–32 The superiority of 70% ethanol can be attributed to several factors: (i) its intermediate polarity, which enables efficient extraction of both polar (eg, flavonoid glycosides) and moderately non-polar constituents (eg, flavonoid aglycones); (ii) the presence of water, which enhances cellular swelling and improves solvent diffusion into intracellular compartments; and (iii) the safety, volatility, and cost-effectiveness of ethanol relative to other organic solvents, making it particularly suitable for large-scale pharmaceutical and nutraceutical applications.
Optimization of the CS extraction procedure yielded the following conditions: percolation as the extraction method, 70% ethanol as the solvent, and a solvent-to-material ratio of 20 mL/g. Under these optimized parameters, CS flowers exhibited a notably high extraction yield of 40%, which is comparable to or higher than yields reported for other Asteraceae species (eg, 38% for marigold flowers and 65.4% for Chrysanthemum morifolium).33,34 Variability in extraction yields across species can be attributed to differences in botanical characteristics, cultivation conditions, harvest timing, and methodological parameters. It is also noteworthy that both domestic and international studies on CS predominantly employ non-thermal extraction methods such as cold maceration.30,35
Despite histological evidence of severe inflammation, serum transaminase levels (ALT and AST) remained within normal ranges and did not differ significantly from those of the control group or male mice. It is well recognized that patients with compensated or decompensated cirrhosis may present with normal or low-normal aminotransferase levels, which can lead to misleading clinical reassurance. 36 Likewise, in hepatic fibrosis, activated Kupffer cells release cytokines that mediate inflammation and fibrogenesis without necessarily inducing hepatocellular necrosis or enzyme leakage. 37 Therefore, hepatic abnormalities cannot be reliably assessed using transaminase levels alone, and the observed biochemical–histological dissociation warrants further investigation.
To the best of our knowledge, no prior studies have reported haematological parameters in the context of sub-chronic toxicity evaluation of CS extract. In the present study, minor alterations were observed, including slight increases in hematocrit (Hct) and platelet count (PLT), accompanied by decreases in mean corpuscular haemoglobin concentration (MCHC) and mean corpuscular volume (MCV) in female mice. Sex-related variations in behavior, exposure, biochemistry, anatomy, physiology, and genetics are known to influence differential responses to environmental factors and pharmaceuticals. 38 The observed changes may therefore reflect a sex-specific metabolic response to CS extract in female mice. Further investigation is required not only to elucidate the underlying mechanisms and assess potential long-term implications but also examine relevant molecular pathways and evaluate whether similar effects may occur in humans.
Although CS extract exhibited lower antihyperuricemic activity compared with allopurinol, it was found to be rich in phenolic constituents—particularly flavonoids—which have been widely recognized for their biological activities, including xanthine oxidase inhibitory potential. 32 A previous study evaluated the in vitro xanthine oxidase inhibitory activity of several flavonoids at concentrations of 0, 10, 30, and 100 µM. Aglycones such as quercetin, luteolin, and apigenin exhibited dose-dependent inhibition, with IC50 values of 11.0 µM, 1.0 µM, and 3.5 µM, respectively. In contrast, their glycosylated derivatives, such as orientin and isoorientin, showed no inhibitory effect on uric acid production even at concentrations up to 100 µM. These findings indicate that the presence or absence of a glycoside moiety significantly influences the inhibitory potential of flavonoid molecules. 39 Additional studies have shown that glycosylation generally attenuates antioxidant and other biological activities, although certain specific bioactivities may be enhanced under particular conditions. 34 Because extraction with 70% ethanol at a 1:20 solvent ratio preferentially yields polar compounds, the CS extract is expected to contain substantial amounts of flavonoid glycosides. This compositional profile may therefore contribute to the higher IC50 values observed relative to allopurinol, the standard pharmaceutical reference agent.
Allopurinol was likely to possess higher efficacy than CS extracts in the treatment of acute gout. This result is usually encountered for herbal medicines, as they need to be used over long periods for the natural compounds to accumulate and display activities. Nevertheless, CS extracts were as effective as allopurinol in the acute prevention and sub-chronic treatment despite less potent activity in in vitro xanthine oxidase inhibitory activity assessment. Previous studies have shown that some flavonoids might not be potent in vitro, but display great in vivo effects. For example, rutin is not a potent xanthine oxidase inhibitor in vitro with an IC50 value of 168 μM, but in a gout treatment model on mice, rutin showed the ability to lower uric acid level by 33%–47% at the dose 25–100 mg/kg.40,41 Flavonoid glycosides are poorly absorbed in the small intestine after oral intake owing to their high hydrophilicity. Instead, they are converted to aglycones by β-glucosidase by the microflora in the intestinal epithelium and large intestine, allowing these aglycones to be absorbed more efficiently by the epithelial cells. 42 As mentioned, the presence or absence of the glycoside moiety significantly influences the inhibitory potential of flavonoid molecules. Therefore, the metabolic conversion of flavonoid glycosides into aglycones may contribute to the greater in vivo efficacy of CS extract. Antioxidants also play important roles in decreasing uric acid levels in the body. 43 CS extract was proved to have various effects, including free radical scavenging activity, ferric-reducing antioxidant power, lipid peroxidation inhibitory ability, and oxygen radical antioxidant capacity. 31 These activities are proposed to act synergistically, thereby enhancing the in vivo efficacy in gout treatment.
Two distinctive flavonoids were found in CS extract with high concentrations were coreopsin and luteolin. The effects of coreopsin on lowering uric acid levels have not been fully investigated yet, whereas the mechanism of action of luteolin was reported to diminish mURAT1 levels and suppress xanthine oxidase activity, both of which improve renal dysfunction caused by hyperuricemia and increase uric acid excretion. 44 In addition, luteolin also reduced the inflammation and paw swelling induced by monosodium urate crystals. 44 Further research suggested that luteolin reduced IL-1β and TNF-α levels, thereby alleviating inflammatory symptoms. 44 Thus, these could be possible mechanisms of action of the CS extract on lowering uric acid levels.
Both CS 250 and CS 500 groups throughout three experiments showed no difference in their results, in other words, increasing the dose did not lead to a greater effect. Such a result might be due to the “ceiling effect” in which treatment effects can be limited despite the increased dosage. 45 The receptors (eg, mRAT1) or enzymes (eg, xanthine oxidase) that interact with the drug become saturated at higher doses. Once saturation is reached, additional increases in the drug dose do not produce a greater effect because there are no more available receptors or enzymes for the drug to bind to or interact with. Therefore, CS extract 250 mg/kg in this study was defined to be the minimum dose for maximized therapeutic effect.
Before clinical translation, the experimental dose should be converted to a human equivalent dose (HED) based on body surface area. The estimated HED for the 250 mg/kg dose in mice is approximately 20 mg/kg for a 60 kg adult. This value is substantially lower than the average HED derived from the no-observed-adverse-effect level (NOAEL) in male and female mice (102.67 ± 134.9 mg/kg and 107.17 ± 151.2 mg/kg, respectively), indicating a favorable safety margin. However, it remains higher than the estimated maximum recommended starting dose (MRSD) values of 10.3 ± 13.5 mg/kg and 10.7 ± 15.1 mg/kg for male and female mice. 46 Therefore, further studies are warranted to optimize dosage and refine the translational relevance of the findings.
Most cases of gout are characterized by a primary overproduction of uric acid. Less commonly, uric acid is produced at normal rates, and hyperuricemia occurs because of decreased renal excretion of urate. This state subsequently results in the precipitation of urate crystals in joints. 37 Hence, it is the principal biochemical marker and a direct risk factor associated with gout pathogenesis. With the significantly reduced serum uric acid levels, this study has confirmed CS extract's potential for gout treatment. The encouraging results from this work provide a solid foundation for future studies to investigate additional biochemical and molecular parameters to elucidate the underlying mechanisms of action.
This study has several limitations. Firstly, the lack of gold standard HPLC/LCMS for identification and quantification of individual flavonoids like coreopsin and luteolin. Secondly, the exclusion of female mice in the efficacy study may limit the generalizability of the findings. Thirdly, the absence of a graded dose–response restricts PK/PD modeling and precise dose selection. Additionally, while linear regression was used in this study, a non-linear fitting approach would provide a more precise representation of dose-dependent trends. Finally, potential synergistic or antagonistic interactions among the extract's components may contribute to the observed effects, requiring further investigation.
Conclusions
This study successfully established the optimal conditions for flavonoid extraction from Cosmo sulphureus Cav. flowers, particularly ethanol concentration and solvent-to-material ratio. The robust extraction protocol developed in this study provides a solid foundation for future research and potential drug development. The standardized CS extracts demonstrated potent anti-hyperuricemic activity in mice, indicating that Cosmos sulphureus Cav. is a promising and viable source for novel anti-gout therapeutics, warranting further clinical investigation.
Footnotes
Acknowledgment
The authors would like to express our sincere gratitude to Department of Pharmaceutical Technology, Department of Pharmacology, School of Pharmacy, and UMP Science and Technology Center, University of Medicine and Pharmacy at Ho Chi Minh City for their support during this research.
ORCID iDs
Ethical Approval
This study was approved by the Animal Research Ethics Committee of the University of Medicine and Pharmacy, Ho Chi Minh City, Vietnam (Decision No. 596/GCN-HĐĐĐNCTĐV) on June 12, 2023.
Author Contributions
Le Minh Quan: Conceptualization, Methodology, Writing - Review & Editing, Supervision; Dinh Truong Son: Investigation, Data Curation, Formal analysis; Nguyen Thi Anh Thu: Validation, Writing - Original Draft; Do Hoang Han: Validation, Visualization, Writing - Review & Editing; Ta Quang Vuong: Resources, Methodology, Writing - Review & Editing; Nguyen Cat Tram: Formal analysis, Writing - Original Draft; Mai Huynh Nhu: Methodology, Writing - Review & Editing; Vo Thi Kim Khuyen: Writing - Original Draft, Visualization; Nguyen Duc Tuan: Methodology, Writing - Review & Editing, Supervision; Ngo Kien Duc: Conceptualization, Resources, Funding acquisition, Writing - Review & Editing;
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ho Chi Minh City Foundation for Science and Technology Development, (grant number No. 09/2022/HĐ-QKHCN).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.