Abstract
Introduction
S. terebinthifolia(ST) is a plant belonging to the cashew family Anacardiaceae, native to subtropical and tropical South America. ST is commonly called Brazil pepper and aroeira. Several reports have been made on the biological activities of ST, but studies on leaf extracts, especially lectins, have mainly been reported. ST leaves contained 14-kDa lectin, which showed antibacterial and anti-inflammatory effects.1,2 Matsuo et al has reported that ST contains compounds that induce apoptosis in melanoma, and block the progression of metastases. 3 In vitro, lectin also reduced tumor weight and induced the apoptosis of Sarcoma 180 cells (IC50 of 8.30 μg/mL) in sarcomas. 4
Some analytical studies on the essential oil of ST have also been reported. Essential oils collected from six sites in Brazil were reported to be rich in monoterpenes and sesquiterpenes, with α–pinene and limonene as the main constituents. Both essential oils showed anti-inflammatory effects, and the two components were presented as indicators of quality control. 5
The bioactive substances of the S. terebinthifolia leaf extract were analyzed using HPLC−ESI−MS/MS. Eleven phenolic substances in ST leaf extract (coumaric acid, (+)–catechin, myricetin–3–O–glycuronide, pentagalloyl, glucose kaempferol–3–O–glucoside, myricetin, methyl gallate, myricitrin, gallic acid, quercetin,and ethyl gallate) have been reported to have the potential for the prevention or treatment of diseases related to oxidative stress. 6 Compared to studies on the pharmacological activity or components of ST leaves, there have been few studies on ST bark. The acute toxicity of ST bark 70% ethanol extract has been reported, and no toxicity was found when extract was orally administered to Wistar rats for 45 days (d) at a dose of 1.5 g/kg/d. 7
The pharmacological activity of ST stem bark has been reported to have an antiviral effect against HSV−1 and gallocatechin catechin as an active ingredient Extract of ST stem bark has been used for the topical treatment of skin wounds, and for oral hygiene. 8
In the present study, we evaluated various biological activities of ST bark in addition to their traditional therapeutic uses, and investigated the utilization of ST bark as medical and cosmetic materials. As a result of the study, ST extract showed xanthine oxidase, elastase, and tyrosinase inhibitory activity, and HPLC and GCMS confirmed biomarkers related to enzyme inhibition. Through this study, it was predicted that the ST bark could be used as a source for inflammatory diseases related to gout, whitening, and wrinkle improvement.
Materials and Methods
Plant Material and Extract Preparation
S. terebinthifolia bark was supplied by Mr Sung-Sik, Yang and Mrs Hea-Kyung Kim for supplying raw materials from Missionario Evangelico Três Fronteira (Brazil). A voucher specimen (MNUCSS−ST−01) was deposited at Mokpo National University (Muan, Korea). Dried ST bark (20 g) was extracted twice with 100 mL of (20 to 100) % ethanol solution (EtOH) at RT for 48 h, or extracted with boiled water for 4 h. The resultant part was freeze dried.
DPPH Free Radical Assay
Antioxidant activity was determined following a DPPH radical scavenging assay. Briefly, sample was added to the DPPH solution (0.4 mM, Sigma Co.USA), and mixed for 10 min and measured at 517 nm by microplate reader (Perkin Elmer, Waltham, MA, USA). 9
Determination of Total Phenolic Content
The total phenolic content was determined by Folin reation. 10 The samples were mixed with Folin-Ciocalteu reagent (Sigma Co.USA) and Na2CO3 (Daejung Chem Co, Korea) solution for 10 min. Mixture was measured at 750 nm. Gallic acid was used as standard. Results were expressed as milligrams of gallic acid (Sigma Co.USA) equivalents per gram of sample.
Determination of Xanthine Oxidase (XO) Inhibitory Activity
Sample and xanthine (1 mM, Sigma Co.USA) were mixed in potassium phosphate buffer (0.1 M, pH 7.5), and reacted for 5 min. Second reaction was processed with xanthine oxidase (0.1 unit/mL, Sigma Co.USA) at 37 °C for 15 min. Then, 1 N HCl was added to stop the reaction. The supernatant was separated, and measured at 292 nm. 11
Determination of Elastase Inhibitory Activity
The assay was performed according to the protocols of Chiocchio et al. 12 Elastase (10 ug/mL) was mixed with Tris–HCl (0.2 M) of STANA (2.5 mM, N-Succinyl-Ala-Ala-Ala-p-nitroanilide, Sigma Co.USA) and of the sample at 37 °C for 30 min. After centrifugation, supernatant was measured at 405 nm
Determination of Tyrosinase Inhibitory Activity
Tyrosinase inhibition assay was performed according to previous reference. 13 Briefly, reaction mixtures with phosphate buffer (pH 6.8, 100 mM), and sample (100 µg/mL) were prepared. Secondly, mushroom tyrosinase (Sigma Co.USA) added to mixture for 10 min at 37 °C. Absorbance was measured using absorbance microplate Reader (Perkin Elmer, USA) at 475 nm.
Identification and Quantitication of Biomarkers Using GC-MS
The active constituents from ST bark using GC−MS were analyzed based on a moderately modified previous reference. 14 Agilent 7890 gas chromatography (GC) and Agilent 5975 quadrupole mass spectrometry (MS) system (Agilent Technologies, USA) were utilized to analyze molecular mass fragments of (50−550) amu of the ST extract. Agilent HP−5MS silica capillary column was used for analysis, the mass fragments. A GC oven was isothermally programmed at 65 °C for 10 min and 10 min−1 to 300 with helium as a carrier gas. All the data were analyzed with the NIST 2017. ST extract was silylated with N,O-bis(trimethylsilyl)trifluoroacetamide (SUPELCO, St. Louis, Mo, USA) to derivatize the constituents to their trimethylsilyl-derivatives (TMS-derivatives) before GC-MS analysis. 15
Constituent Profiling by High-Performance Liquid Chromatography (HPLC) Analysis
(+) −Catechin (Sigma Co, USA) analysis of ST extracts was performed with HPLC. All HPLC analyses were performed using the Alliance 2695 HPLC system (Waters, USA) equipped with a UV detector. Table 1 describes the analysis method, and Figure 1 shows the chromatographic profiles.

Content of catechin in extracts from S. terebinthifolia.
Analytical HPLC Conditions.

Acetonitrile 2 0.2% phosphoric acid
Statistical Analysis
All data were analyzed repeatedly three times, and the average and error were calculated. The data were analysised using Excel® software.
Results
Extraction Yields and Analysis of Active Compounds
Table 2 shows the calculated yield of ST extract, for which the hot water extract showed the lowest yield at 5.62%, while the 60% ethanol extract showed the highest extraction yield at 12.54%. Figure 1 contest of catechins of each extracts, which showed the highest content at 3.62% in 80% ethanol extract. Table 3 shows that analysis of the 80% ethanol extract by GCMS revealed that α–amyrin, β–amyrone, and 11-Oxo-α–amyrin were the main constituents. In particular, α–amyrin showed the highest proportion (29.74%).
Extraction Yields of S. Terebinthifolia Extracts.

Results of GCMS Analysis of 80% Ethanolic Extract.

Antioxidant Effect of ST Extracts
As a result of evaluating the electron donating ability of ST extract with ascorbic acid at the same concentration (25 µg/mL), the hot water extract (84.46%) showed higher antioxidant activity than ascorbic acid (79.75%) (Figure 2).
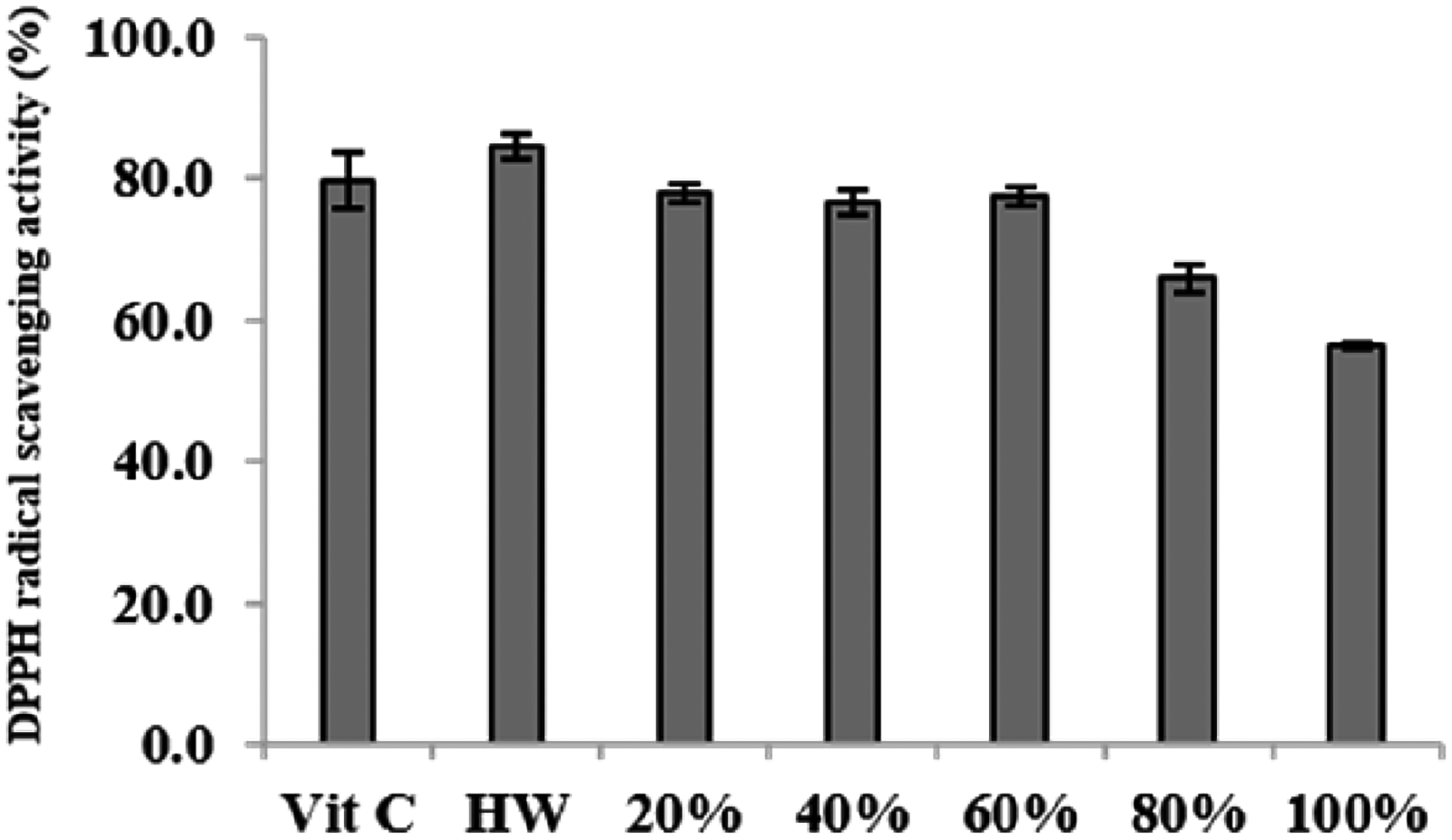
DPPH radical scavenging activity of water and ethanol extracts from S. terebinthifolia (extract cont, Asorbic acid : 25 µg/mL).
On the other hand, reducing power and total phenol contents showed the highest values at 109.61 as Ascorbic Acid eq. μg/100 μg Extract, and 263.48 as Gallic Acid eq. mg/g (Table 4).
Reducing Power and Total Phenolics of Extract from S. terebinthifolia.

Xanthine Oxidase Inhibitory Effects of ST Extract
Figure 3 shows that the ST extract displayed excellent xanthine oxidase activity. Allopurinol, a control, when treated with 200 ug/mL showed about 95.6% xanthine oxidase inhibitory activity. The 80% ethanol ST extract, when treated with 1 mg/mL, showed about 91.7% xanthine oxidase inhibitory activity, and it was the most effective among the extracts. Figure 3D shows that catechin, α–amyrin, and β–amyrone are substances that inhibit xanthine oxidase, and a mixture of catechin, α–amyrin, and β–amyrone equal to the content in the 80% ethanol extract was used to reduce xanthine oxidase. As a result of confirming the inhibitory effect, it showed activity similar to that of the 80% ethanol extract.

Xanthine oxidase inhibitory effect of ST extracts and related biomarkers. Xanthine oxidase activity in hot water and ethanolic extract from S. terebinthifolia (1 mg/mL), (A) Xanthine oxidase activity in hot water and ethanolic extract from S. terebinthifolia (1 mg/mL); (B) S. terebinthifolia 80% ethanolic extract by concentration.
(C) xanthine oxidase inhibitory activity of β-amyrone.
(D) Xanthine oxidase activity in individual markers and mix contained in Schinus terebinthifolia extract (catechin : 35 μg/mL, α-amyrin : 300 μg/mL, β-amyrone : 80 μg/mL) and 80% ethanolic extract (1 mg/ml).
(A), (B), (C), (D) The positive control was allopurinol (100 μg/mL).
Elastase Inhibitory Effects of ST Extract
Figure 4 shows that as a result of confirming the elastase inhibitory effect, phospharamidon (PPRM), a control group, when treated with 0.5 mg/mL showed an elastase inhibitory effect of about 84%, and at a concentration of 1 mg/mL, the extract showed an even elastase inhibitory effect (Figure 4A). In particular, the effect of 80% ethanol extract was the highest. Catechin, α–amyrin, and β–amyrone were made into standards and mixtures identical to the contents in the 80% ethanol extract to confirm the elastase inhibitory ability. As a result, the mixture was about 63%, and the ethanol extract showed an inhibition rate of 85% (Fig. 4B). Thus, biomarkers other than catechin, α–amyrin, and β–amyrone are thought to exist.

Elastase inhibitory effect of ST extracts and related biomarkers.
Tyrosinase Inhibitory Effect of ST Extract
Figure 5 shows that the ST extract had an excellent tyrosinase inhibitory effect. As a control, at the same concentration (1 mg/mL), arbutin and ascorbic acid showed (28.5 and 49.3) % of tyrosinase inhibitory activity, respectively. As for the ST extract (1 mg/mL), the hot water extract showed the best inhibitory activity of 67%.

Tyrosinase inhibitory effect of ST extracts and related biomarkers.
Discussion
In folk medicine, the stem bark of S. terebinthifolia (ST) is known to be used for inflammation, respiratory diseases, gout, and skin diseases. 16 A number of studies on ST leaves have been reported, but there are few studies on the usefulness of the bark. Therefore, in our study, ST bark extract was prepared; antioxidant, anti-hyperuricemia, and skin improvement-related effects were confirmed; and the basic analysis of bio-markers was performed.
For antioxidant evaluation, electron donating ability, reducing power, and total phenol analysis were used. As for the ST extract, hot water and ethanol extracts were prepared. Antioxidant effects are widely related to the suppression of oxidative stress-induced inflammatory and metabolic diseases. 17 Among the ST extracts, the hot water extract had the highest electron donating ability, but the 80% ethanol extract showed the highest reducing power and total phenol content (Fig. 2, Table 4).
The anti-hyperuricemic effect of ST has not been reported so far. Hyperuricemia (gout) is caused by high levels of uric acid in the blood, and gouty arthritis is caused by uric acid crystals in the blood. 18 Therefore, it was confirmed whether the ST extract regulates blood uric acid-inducing factor, xanthine oxidase. At a concentration of 1 mg/mL, the 80% ethanol extract showed more than 90% inhibitory activity in tube test. Based on the previous reports, in the case of showing high xanthine oxidase inhibitory activity at a concentration of 1 mg/mL, in vivo results also showed a decrease in blood uric acid and inhibition of xanthine oxidase in blood and liver. Yoon et al reported that Camellia japonica leaf extract when treated at a concentration of 1 mg/mL in vitro showed xanthine oxidase inhibitory activity in the 50% range, but in vivo showed significant anti-hyperuricemic efficacy. 19 Song et al also presented a similar report. Song et al showed that if the ethanol extract of Cudrania. tricuspidata showed xanthine oxidase inhibition at a concentration of 1 mg/mL in tube test, it was sufficiently effective in animal experiments. In fact, in in vivo, it was reported that C. tricuspidata extract decreased XO activity in plasma and liver through oral administration within 30 to 100 mg/kg. 20
Therefore, based on previous reports, ST extract can be expected to have a higher anti-hyperuriemic effect than Camellia japonica and Cudrania tricuspidata. A mixture of catechin (35 μg/mL), α–amyrin (300 μg/mL), and β–amyrone (80 μg/mL), which are components of the ST extract, was prepared at the same concentration contained in 80% ethanol, to confirm the xanthine oxidase inhibitory activity. Catechin, α–amyrin, and β–amyrone can be seen as the main biomarkers of the extract, as it showed 66% enzyme inhibition, while 80% ethanol extract showed 83% inhibition. Dhiraj et al reported that catechin and gallic acid had inhibitory activity by binding to xanthine oxidase in a docking simulation. However, the actual inhibition pattern of catechin was not demonstrated. 21 While no previous evaluation of the xanthine oxidase inhibitory ability of catechin has been reported, α–amyrin, and β–amyrone, this study reported that the three biomarkers had xanthine oxidase inhibitory ability under the condition of constant concentration.
Elastase proteolytic enzyme is related to skin wrinkling through the degradation of elastin, and causes loss of dermal retractility. Thus, as modulators of elastin destruction that causes skin aging, elastase inhibitors have found as agents in the cosmetic industry.22,23 Melanization is controlled by melanin biosynthesis. 1) Hydroxylation of L–tyrosine to L−DOPA 2) oxidation of L−DOPA to O–quinone. Thus, natural tyrosinase inhibitors are beneficial in cosmetics that inhibit excessive pigmentation with melanin, and improve skin whitening. 24 The elastase inhibitors could be developed as anti-wrinkle material to treat skin aging. 12
To investigate the possibility of developing cosmetic materials of ST, the improvement of skin wrinkles through elastase inhibition and whitening through tyrosinase inhibition were evaluated. ST extract showed about 85% elastase inhibitory ability in 80% ethanol extract. α–amyrin showed best elastase inhibitory activity at the same concentration as PPRM (84.4%), and was considered a major biomarker (60.4%). When a mixture of catechin, α–amyrin, and β–amyrone, components of the ST extract, was prepared at a concentration contained in 80% ethanol, the xanthine oxidase inhibitory activity was confirmed, 63.6% of the enzyme inhibitory activity was shown, and the 80% ethanol extract showed an inhibitory activity of 85.6%. The β–amyrone was the main biomarker of the extract, and catechin and α–amyrin were thought to be minor biomarkers. No previous report on the elastase inhibitory activity of catechin, α–amyrin, and β–amyrone has been made. ST extract showed better tyrosinase inhibitory activity (67.1%) than arbutin and ascorbic acid at (28.5 and 49.3) % in hot water extract. We measured the tyrosinase inhibitory ability of catechin, α–amyrin, and β–amyrone, and found weak inhibitory activity, so it is considered necessary to discover tyrosinase inhibitors of ST extracts in the future.
Although our report identified the basic components of ST extract and its antioxidant properties, elastase inhibition, and XO inhibition through in tube tests, it showed the limitations of developing sufficient anti-gout and wrinkle improvement source. There is a need to conduct animal experiments in the future to derive an effective dose for anti-gout or to specifically calculate the optimal treatment concentration for wrinkle improvement.
Conclusion
In the present study, the component analysis, antioxidant, xanthine oxidase, elastase, and tyrosinase inhibitory activities of S. terebinthifolia (ST) bark extract were evaluated. Hot water extracts showed the strongest electron donating ability and tyrosinase inhibitory activity. The 80% ethanol extract showed the highest reducing power, total phenolic, xanthine oxidase, and elastase inhibitory ability. Catechin, α-amyrin, and β–amyrone were identified through ST extract analysis, and 80% extract contained the highest amount of catechin. Catechin, α–amyrin, and β–amyrone appear to be the main xanthine oxidase inhibitors, while β–amyrone is considered to be the main inhibitor of xanthine oxidase and elastase. Since catechin, α–amyrin, and β–amyrone showed weak tyrosinase inhibition, the biomarkers of tyrosinase inhibition need to be further investigated. Through this study, we reported the basic information that S. terebinthifolia bark extract was used in folk medicine as an anti-inflammatory, anti-gout, and skin disease improvement material S. terebinthifolia bark extract could be used as an anti-gout natural drug or cosmetic material.
Footnotes
Acknowledgments
We thank Mr Sung-Sik, Yang and Mrs Hea-Kyung Kim for supplying raw materials
Author Contributions
Conceptualization, S.-S.C. and D.-H.P.; S.-H.B.; investigation, S.-Y.L. and S,-H.L.; data curation, J,-W.P.; writing—original draft preparation, S.-S.C. and D.-H.P.; All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
No data were used to support this study
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea, (grant number 2022R1A5A8033794).
Ethical Approval and Statement of Human and Animal Rights
Because we did not use animals or human-derived cells” or “no human or animals included in the study.
