Abstract
Objective
Toxicodendron succedaneum (Vietnamese lacquer tree) and T. vernicifluum (Korean lacquer tree) have attracted scientific interest due to their rich polyphenolic content and therapeutic potential. This study aimed to isolate methyl gallate (MG) from T. succedaneum and quantify MG, gallic acid (GA), and fisetin in both species. Additionally, the antioxidant, anti-inflammatory, and hepatoprotective activities of MG and GA were assessed to explore the pharmacological potential of T. succedaneum as a source of bioactive phenolics.
Methods
MG was isolated from the bark of T. succedaneum and structurally confirmed via NMR and mass spectrometry. Quantitative analysis of MG, GA, and fisetin in various plant parts of both T. succedaneum and T. vernicifluum were performed using a validated HPLC/PDA method. Antioxidant activity was evaluated using DPPH and MDA assays; anti-inflammatory activity was assessed via nitric oxide (NO) inhibition in LPS-stimulated RAW 264.7 cells; and hepatoprotective effects were examined using an APAP-induced HepG2 cell injury model.
Results
MG was isolated from T. succedaneum bark with a yield of 0.2% (w/w). Both MG and GA were detected in all tested plant parts, whereas fisetin was confined to the stem, bark, and branch of T. vernicifluum and the stem of T. succedaneum. MG content was highest in the stem of T. succedaneum (5.82 mg/g). MG exhibited stronger lipid peroxidation inhibition than GA in antioxidant assays (corresponding IC50 values of 40.12 µM and 230.55 µM), while GA was more effective in NO inhibition. Both compounds showed dose-dependent hepatoprotective effects, with MG offering greater efficacy at lower concentrations. At 100 µM, a reversal of protective activity was observed–consistent with the hormesis phenomenon.
Conclusion
T. succedaneum, traditionally exploited for lacquer but rarely studied for its phytochemicals, was identified as a rich and underutilized source of bioactive phenolics, particularly MG. The compound demonstrated promising antioxidant, anti-inflammatory, and hepatoprotective effects.
Introduction
Lacquer trees belonging to the Anacardiaceae family, particularly Toxicodendron vernicifluum (Stokes) F.A. Barkley and T. succedaneum, have long been valued in East and Southeast Asia, not only for their sap used in traditional lacquerware, but also for their ethnopharmacological applications.1–3 T. vernicifluum, distributed in Korea, China, and Japan, has been widely studied for its diverse phytochemical profile, including polyphenolic and flavonoid compounds such as gallic acid (GA), fisetin, fustin, butein, quercetin, kaempferol, and sulfuretin. 4 These compounds have been associated with antioxidant, anti-inflammatory, hepatoprotective, and anticancer activities.4–7 In addition to its role in lacquer production, T. vernicifluum has been used in traditional Korean medicine for centuries. The bark has been prescribed for gastrointestinal disorders (eg, dyspepsia, abdominal pain, and infections) and inflammatory conditions such as bronchitis, arthritis, and neuralgia.8–10 Notably, it has been employed since the fifteenth century in the treatment of gastric and uterine cancers in Korea. Despite its therapeutic potential, the use of T. vernicifluum has been limited by the presence of urushiol, a potent allergenic compound responsible for severe contact dermatitis. To mitigate this, the bark is traditionally boiled with poultry such as chicken or duck to reduce toxicity. The resulting dish, consumed as a medicinal food, is believed to deliver systemic benefits. 9
Although T. succedaneum and T. vernicifluum have been investigated for their biological activities and chemical constituents, several important research gaps remain. To date, no comprehensive comparative analysis has directly evaluated their major bioactive compounds and pharmacological activities, which is essential for clarifying the chemical and therapeutic distinctions between species of different geographical origins. In addition, while the phytochemical profile of T. vernicifluum has been extensively characterized, the composition of T. succedaneum–particularly the presence and quantification of marker compounds such as methyl gallate (MG)–remains poorly defined. Moreover, previous studies have focused mainly on the resin, whereas other plant parts, including the stem bark, may also contain valuable bioactive constituents that are yet to be systematically explored. Furthermore, lacquer trees in Vietnam are often discarded after 8–10 years due to declining sap quality, leading to substantial biomass waste. This presents a unique opportunity to valorize T. succedaneum as a novel source of bioactive metabolites.
Methyl gallate (MG), a naturally occurring ester of GA, is a known antioxidant and anti-inflammatory agent. 7 However, its comparative distribution or bioactivity across lacquer tree species is not fully understood. Moreover, the presence of fisetin in T. succedaneum has not yet been documented. Therefore, the present study aimed to address these research gaps by conducting a comprehensive comparative analysis.
To address these research gaps, the present study conducted a comprehensive comparative analysis. The study focused on first isolating and structurally elucidating MG from the bark of T. succedaneum. We then performed fingerprinting analysis by TLC, HPLC/PDA, and LC-QTOF/MS to compare the chemical profiles of different parts (bark, stem, branches, and leaves) from both T. succedaneum and T. vernicifluum. Based on these qualitative results, we quantitatively analyzed and compared the levels of MG, GA, and fisetin in the samples using HPLC/PDA, and finally evaluated and compared the antioxidant, anti-inflammatory, and hepatoprotective activities of MG and GA using established in vitro models.
Materials and Methods
All experiments were conducted from May 2024 to June 2025 at the UMP Science and Technology Center and School of Pharmacy, University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam.
Plant Materials
Samples of T. vernicifluum were obtained in May 2024 from the medicinal plant garden at Seoul National University, Korea and authenticated by Prof. J.H. Park (College of Pharmacy, Seoul National University). These samples including bark, stem, branches, and leaves were harvested from small stems and branches to avoid felling the trees. Due to the small quantity, they were dried in an oven at 50 °C until the moisture content was below 10% and then shipped to Vietnam. T. succedaneum samples were harvested in June 2024 from a farm in Phu Tho province, Vietnam (21.242206°N, 105.264003°E) and authenticated by one of the authors, Thi Hong Van Le, in collaboration with the farm manager. As whole trees were felled and the bark was peeled, the larger quantity of T. succedaneum samples was first air-dried in the shade before being finished in an oven at 50 °C until the moisture content was less than 10%. All samples were authenticated via morphological characteristics and ITS gene sequencing (detailed in Supplemental material 1) prior to studies. A voucher specimen of the samples has been deposited at the UMP Science and Technology Center, University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam (UMPTV202405 for T. vernicifluum and UMPTS202406 for T. succedaneum).
Chemicals, Reagents, and Research Equipment
Extraction and Isolation
Dried T. succedaneum bark (3.0 kg) was reflux extracted three times with 15 L of methanol, 4 h each time, at 80 °C. The solvent was then evaporated under reduced pressure to obtain 700 g of total extract (23.33% yield). This extract was dissolved in 700 mL water and partitioned with n-hexane (2.1 L, three times) to remove lacquer resin. The aqueous phase was then partitioned with ethyl acetate (2.1 L, three times) to collect the ethyl acetate phase, which was then concentrated by evaporation under reduced pressure at 40 °C. The concentrate was stored at 4–8 °C for 24 h, forming a yellow precipitate, which was then filtered and washed with cold ethyl acetate to obtain 11.4 g of precipitate (1.63% yield).
The precipitate (11.4 g) was purified using flash column chromatography (11 cm in height and 7 cm in diameter) with 342 g silica gel (230-400 mesh) and eluted sequentially with dichloromethane, dichloromethane/ethyl acetate (1:1, v/v), ethyl acetate, and methanol. The fraction obtained by eluting with dichloromethane/ethyl acetate (1:1, v/v) was evaporated under reduced pressure to remove the solvent, resulting in light yellow crystals.
For recrystallization, 7 g of the crude crystals were dissolved in 50 mL of ethyl acetate with the assistance of an ultrasonic bath to ensure complete dissolution. The solution was stored at 4–8 °C for 24 h to induce crystallization. The resulting crystals were purified by repeating this recrystallization process three times to obtain 6 g of MG.
The structure of the isolated compound was determined using UV, MS, and NMR spectroscopy. The UV spectrum was acquired on a Shimadzu UV-1800 UV-VIS Spectrophotometer (Japan) using the sample dissolved in methanol (MeOH). Mass spectrometry was performed on a LC-MS QTOF instrument (Waters, USA) with the sample dissolved in MeOH. NMR spectra were recorded on a Bruker AvanceNEO 600 MHz spectrometer (Germany) with the sample dissolved in DMSO.
This extraction and isolation procedure had been standardized in our laboratory.
Fingerprinting Analysis
The specific analytical conditions for TLC, HPLC/PDA, and LC-MS analyses were developed and standardized in our laboratory.
TLC Analysis
Standard solutions of MG, GA, and fisetin were prepared at 25 μg/mL in 50% methanol. 100 mg of the dry herbal material (bark, stem, branches, and leaves of T. vernicifluum and T. succedaneum) was extracted using sonication with 1 mL of the solvent for 30 min at 30 °C. TLC was performed on silica gel 60 F254 plates. Each sample solution (20 µL) was applied as a 0.8 mm band, 1 cm from the bottom edge of the plate, with each band separated by 0.5 cm. The developing chamber was prepared with the mobile phase, a solvent system of CHCl3–MeOH–H2O–HCOOH (14:6:2:1, v/v/v/v, lower phase). A filter paper was placed inside the chamber and allowed to saturate with the mobile phase until the filter paper was completely wet. The plate was then placed into the chamber and developed until the solvent front reached a distance of 10 cm. The spots were visualized under UV light (254 or 356 nm) or by staining with 1% FeCl3 in 96% ethanol, then dried using a hair dryer, and the results were observed under visible light.
HPLC/PDA Analysis
Standard solutions of MG, GA, and fisetin were prepared at 25 μg/mL in 50% methanol. The bark, stem, branches, and leaves of T. vernicifluum and T. succedaneum were ground into a fine powder. 100 mg of each powder was extracted using sonication with 20 mL of 50% methanol for 30 min at 30 °C. This extraction step was performed twice. All samples were filtered through a 0.22 µm filter prior to injection. The chromatographic separation of the compounds was achieved using a Phenomenex C18 column (150 mm × 4.6 mm, 3 µm) with column oven temperature maintained at 35 °C. The mobile phase consisted of acetonitrile (A) and 1% phosphoric acid (B). The mobile phase flow rate was 1 mL/min with gradient elution as follows: 0–5 min, 4%; 5–12 min, 9%; 12–25 min, 22%; 25–35 min, 22%; 35–36 min, 95%; 36–42 min, 95%; 42–43 min, 4%; 43–50 min, 4% of solvent A. The injection volume was 10 μL, and the PDA detector was set at 210 nm.
LC-QTOF/MS Analysis
Standard and sample solutions were prepared under the same conditions as for the HPLC/PDA analysis. The chromatographic column and column temperature were also identical. The mobile phase consisted of the same solvent system as used in the HPLC/PDA analysis, but with a flow rate of 0.5 mL/min. The gradient elution program was as follows: 0–10 min, 5%–10%; 10–15 min, 10%–30%; 15–20 min, 40–50%; 20–25 min, 50%–95%; 25–40 min, 95%; 40–42 min, 5%; 42–45 min, 5% of solvent A. The injection volume was 2 μL. Samples were analyzed in negative Electrospray Ionization (-ESI) mode, with a gas temperature of 120 °C and a flow rate of 120 L/h. The mass range was from 100 to 1500 m/z, with collision energies of 6, 20, and 30 eV.
Quantitation Analysis
The quantitative analysis of marker compounds (MG, GA and fisetin) was conducted using the methodology described in the Fingerprinting Analysis - HPLC/PDA Analysis section. The method was validated in accordance with the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines.
Biological Activity
DPPH Radical Scavenging Assay
DPPH radical scavenging activity was determined as described previously with modifications. 11 MG, GA and quercetin (positive control) were prepared in methanol (at the range of cocentrations from 0.5 to 10 µM). The amount of 50 μL each sample or quercetin solution, 50 μL of 0.08 mM DPPH in methanol, and 100 μL of methanol were added to a 96-well microplate. The resulting mixture was mixed vigorously and incubated at 25 °C for 30 min in the dark. DPPH decoloration was measured at 517 nm. The scavenging percentage was calculated using the formula (1): (1–ODsample/ODcontrol) × 100. The IC50 values were determined by plotting the inhibition percentage against the concentration of the samples using GraphPad Prism 8.0.2 software.
MDA Assay
Malondialdehyde (MDA) assay was performed with modifications from a previously reported method. 11 Samples (MG and GA) and positive control (quercetin) were initially dissolved in DMSO and diluted in phosphate buffer (pH 7.4), to ensure a final DMSO concentration of ≤ 0.5%. Mouse liver was homogenized in ice-cold 1.15% KCl at 1:10 (w/v) ratio. Various concentrations of samples and quercetin, ranging from 5 to 500 µM, were prepared for analysis. The assay mixture contained 50 μL sample, 250 μL homogenate, and phosphate buffer pH 7.4 to bring the final volume of 1 mL, and was incubated at 37 °C for 60 min. To stop the reaction, 500 μl of 10% trichloroacetic acid was added, followed by centrifugation. Subsequently, 500 μl of the supernatant was mixed with 500 μL of 0.8% thiobarbituric acid and heated at 100 °C for 15 min. Absorbance was measured at 532 nm. Antioxidant activity was calculated using the following formula (2): (1–Abssample/Abscontrol) × 100.
NO Inhibition Assay
Nitric oxide (NO) inhibition and cell viability assays were performed as previously described.12–14 MG, GA, and dexamethasone (positive control) were dissolved in DMSO and diluted in culture medium, ensuring final DMSO concentration of ≤ 0.5%. RAW 264.7 cells were incubated in DMEM supplemented with 2 mM L-glutamine, 10 mM HEPES, 1 mM sodium pyruvate, and 10% FBS at 37 °C in 5% CO2. Cells were seeded in a 96-well microplate at 2 × 106 cells/well for 24 h. Subsequently, they were treated with various concentrations of the test sample (at the range of cocentrations from 5 to 500 µM) or dexamethasone (at the range of cocentrations from 5 to 100 µM) for 2 h. Following this, the cells were either treated with 1 μg/mL LPS or left untreated and incubated for 24 h. After 24 h, 100 uL of culture medium was transferred to a new 96-well microplate, and 100 uL of Griess reagent (1% sulfanilamide in 5% H3PO4 and 0.1% N-(1-Naphthyl)ethylenediamine dihydrochloride in water) was added. This mixture was incubated at room temperature for 10 min, and the nitrite concentration was subsequently measured using a microplate reader at 540 nm. The amount of nitrite was obtained by extrapolation from a standard curve with NaNO2. The percentage of NO inhibition was calculated using the formula (3): % Inhibition = (1−NOsample/NOLPS) × 100. Following the NO inhibition assay and supernatant collection for NO content determination, 100 µL of medium containing MTT (0.5 mg/mL) was added for an additional 4 h. The resulting formazan was dissolved in DMSO and measured on a microplate reader at a wavelength of 570 nm. Cell viability was calculated using the following formula (4): ODsample/ODnormal × 100.
Hepatoprotective Assay
The hepatoprotective assay was determined as described previously with modifications.15,16 MG, GA, and silymarin (positive control) were prepared by dissolving in DMSO and diluting in culture medium, ensuring final DMSO concentration of ≤ 0.5%. HepG2 cells were incubated in DMEM supplemented with 10% FBS, streptomycin (100 mg/mL), and penicillin (100 U/mL) at 37 °C in 5% CO2. HepG2 cells were harvested and seeded in a 96-well microplate at 1 × 104 cells/well for 24 h. For the cell viability assay, cells were then treated with various concentrations of test samples or 100 µM silymarin for 24 h. For the hepatoprotective assay, cells were pre-treated with various concentrations of test samples (at the range of concentrations from 6.25 to 100 µM) or 100 µM silymarin for 1 h, followed by the addition of 20 mM acetaminophen (APAP) and further incubated for 24 h. After incubation, the cells were incubated in 100 µL of medium containing MTT (0.5 mg/mL) for an additional 4 h. Cell viability was determined using the MTT assay, as described in the NO inhibition assay. Hepatoprotective activity was calculated based on cell viability using the following formula (5): (sample–disease control) × 100/ (negative control–disease control).
Statistical Analysis
All experiments were performed in triplicate, and the results are represented as the mean ± standard deviation (SD). IC50 values were determined using GraphPad Prism 8.0.2 software. Statistical significance was determined using the analysis of variance (ANOVA), followed by a Dunnett's test for pairwise comparisons with p < 0.05 considered statistically significant.
Results
Isolation and Identification of MG
MG (Figure 1) was isolated and purified as white crystals from the ethyl acetate fraction of T. succedaneum bark methanol extract. Its structure was determined using UV, HR-MS, and NMR spectroscopy. The spectroscopic data for MG are as follows: UV (methanol) λ: 215, 272 nm; 1H NMR [600 MHz, dimethyl sulfoxide (DMSO)-d6]: δ 6.95 (2H, s, H–2, −6), 3.74 (3H, s, –COOC

Structures of Methyl gallate (MG), Acid Gallic (GA), and fisetin.
Fingerprinting Analysis
TLC analysis provided a rapid qualitative assessment of MG, GA, and fisetin across different plant parts of Toxicodendron succedaneum and T. vernicifluum (Figure 2). Under UV 254 nm, MG and GA displayed strong UV-absorbing bands with retention factors corresponding to the reference standards (lanes 9 and 10) and were consistently observed in nearly all extracts (lanes 1-8), indicating their widespread distribution in both species. When visualized under UV 365 nm, fisetin exhibited a characteristic green fluorescence (lane 11), confirming its flavonol structure. This fluorescent band was prominently detected in the stem and bark extracts of T. vernicifluum (lanes 1 and 2), and faintly in the stem of T. succedaneum (lane 6), but was absent in leaf and branch samples of both species. Upon staining with FeCl3, GA and MG produced dark bluish-black spots, consistent with the presence of phenolic hydroxyl groups. The TLC findings support the presence of key polyphenolic compounds GA and MG, and highlighting the selective part localization of fisetin in lacquer trees.

TLC chromatograms of T. vernicifluum methanol extracts, including (1) bark, (2) stem, (3) branch, and (4) leaves; T. succedaneum methanol extracts, including (5) bark, (6) stem, (7) branch, and (8) leaves; (9) MG, (10) GA, and (11) fisetin standards.
The HPLC chromatograms (Figure 3, Supplemental material 3) clearly demonstrate the retention times and distribution patterns of GA, MG, and fisetin in both T. succedaneum and T. vernicifluum. The standard mixture (lane 1) confirmed the retention times of GA (∼5.0 min), MG (∼15.6 min), and fisetin (∼32.6 min), providing reference peaks for compound identification. GA and MG were clearly detected in all plant parts of both species (lanes 2-9), as evidenced by distinct peaks aligning with the respective standards. The most prominent MG peak was observed in the stem of T. succedaneum (lane 7), indicating the highest concentration of this compound in that part, consistent with quantitative data. Similarly, GA exhibited strong signals in the leaves of T. succedaneum (lane 9) and the bark of T. vernicifluum (lane 2), reflecting part-specific accumulation. Fisetin, a flavonol with known bioactivity, appeared as a distinct peak near 32.6 min but was only present in selected samples–specifically, the stem and bark of T. vernicifluum (lanes 2-3) and and in a lower amount at the stem of T. succedaneum (lane 7). This selective presence reinforces the species- and organ-specific localization of fisetin. The chromatographic profiles also revealed complex polyphenolic backgrounds in all samples.
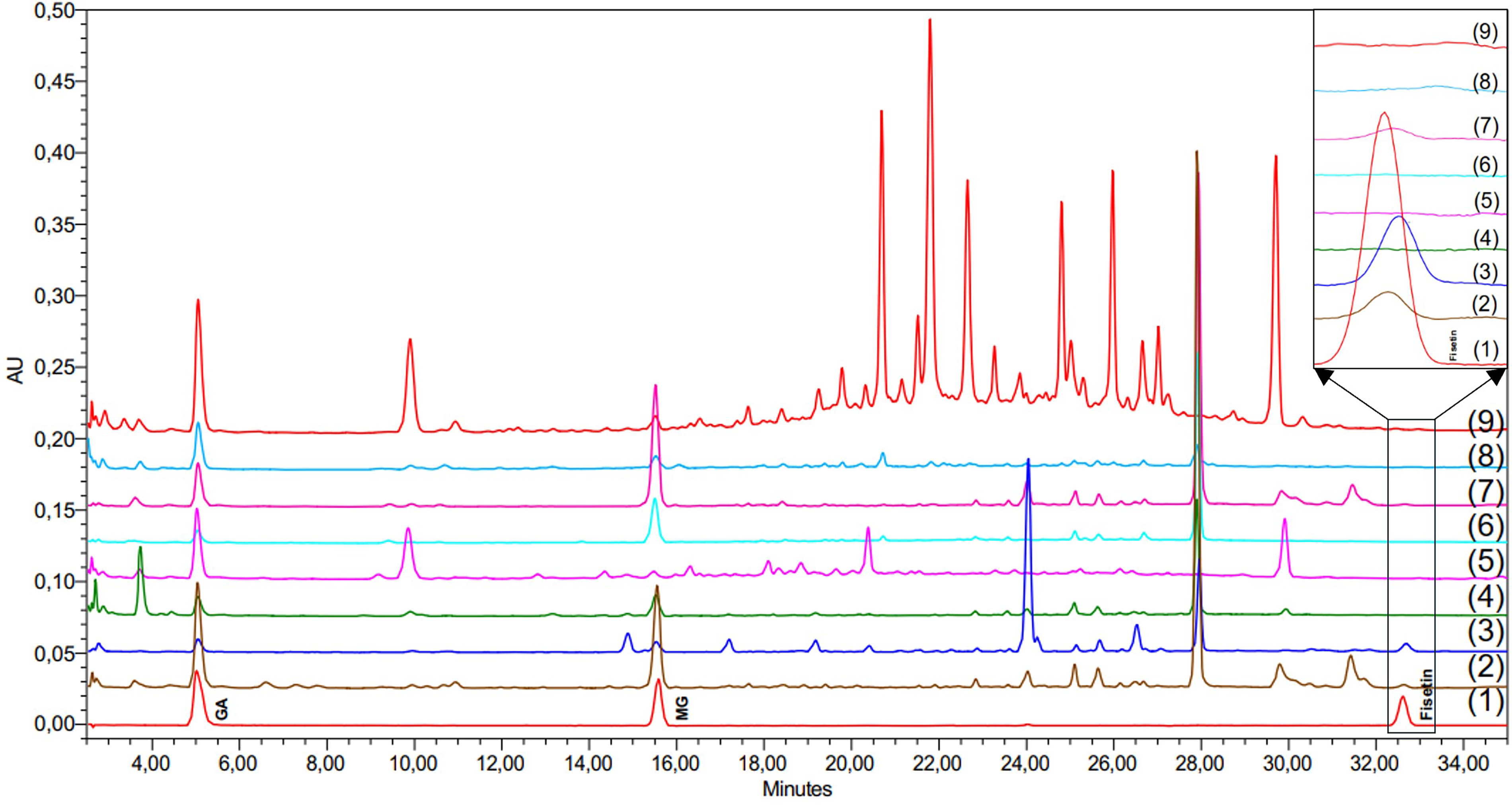
HPLC chromatograms at 210 nm: (1) a standard mixture at 5 μg/ml; methanol extracts of T. vernicifluum samples, including (2) bark, (3) stem, (4) branch, and (5) leaves; methanol extracts of T. succedaneum samples, including (6) bark, (7) stem, (8) branch, and (9) leaves.
A major compound eluting at 28 min was detected in all analyzed parts of both T. vernicifluum and T. succedaneum. This compound exhibited characteristic UV absorption maxima at 217 nm and 279 nm, spectral features highly similar to those of GA and MG, suggesting its probable identity as a GA derivative. The bark of T. vernicifluum (lane 2) and T. succedaneum (lane 6) were qualitatively similar regarding their principal peaks, though T. vernicifluum generally contained a greater number of minor constituents. Conversely, the stem of T. vernicifluum (lane 3) was marked by the presence of a substantial peak at 24 min. This compound, also spectroscopically similar to GA, was detected in the corresponding T. succedaneum sample (lane 7) but at a significantly lower concentration. The branch extracts of both species (lanes 4, 8) showed minimal differences in their major components and generally yielded a lower complexity of peaks detectable by HPLC/PDA. A clear difference was seen in the leaf extracts. The leaves of T. succedaneum (lane 9) showed many strong peaks in the 20–28 min range, while these peaks were absent or only present at trace levels in the leaves of T. vernicifluum (lane 5). This clear difference suggests that T. succedaneum, especially its leaves, may be a rich source of polyphenolic compounds, as they showed the highest number of peaks within the species. Further analysis of the leaf components of T. succedaneum is needed and may provide important insights. Collectively, the HPLC results validated the successful isolation and quantification of MG, GA, and fisetin and confirmed their differential distribution across species and plant parts.
Additionally, Table 1 summarizes the qualitative detection of MG, GA, and fisetin in various parts of Toxicodendron succedaneum and T. vernicifluum using three complementary analytical techniques as TLC, HPLC/PDA, and LC-QTOF/MS (LC-QTOF/MS data were presented in Supplemental material 4). Both MG and GA were consistently detected in all plant parts (bark, stem, branch, and leaves) of the two species across all analytical platforms, indicating their widespread presence and chemical stability. The congruence of detection via TLC, HPLC/PDA, and LC-QTOF/MS confirms the reliability of the profiling results. This also supports the quantitative data showing high levels of MG and GA in the stem and leaves of T. succedaneum and in the bark of T. vernicifluum. In contrast, fisetin exhibited a more restricted distribution. It was only detected in the stem of T. succedaneum and in the stem and bark of T. vernicifluum, while absent from branches and leaves of both species by TLC and HPLC/PDA. Notably, fisetin was also detected in the branch of T. vernicifluum, but exclusively by the LC-QTOF/MS technique. This finding underscores the superior sensitivity and specificity of LC-QTOF/MS compared to TLC and HPLC/PDA, highlighting its capacity to detect trace amounts of compounds that may be below the detection limits of conventional methods. These fingerprinting results reinforce the species- and part-specific accumulation patterns of polyphenolic compounds in lacquer trees and highlight the potential of T. succedaneum as a source of bioactive MG, with complementary chemical profiles compared to the more extensively studied T. vernicifluum.
Fingerprinting Analysis of MG, GA and Fisetin in T. succedaneum and T. vernicifluum.
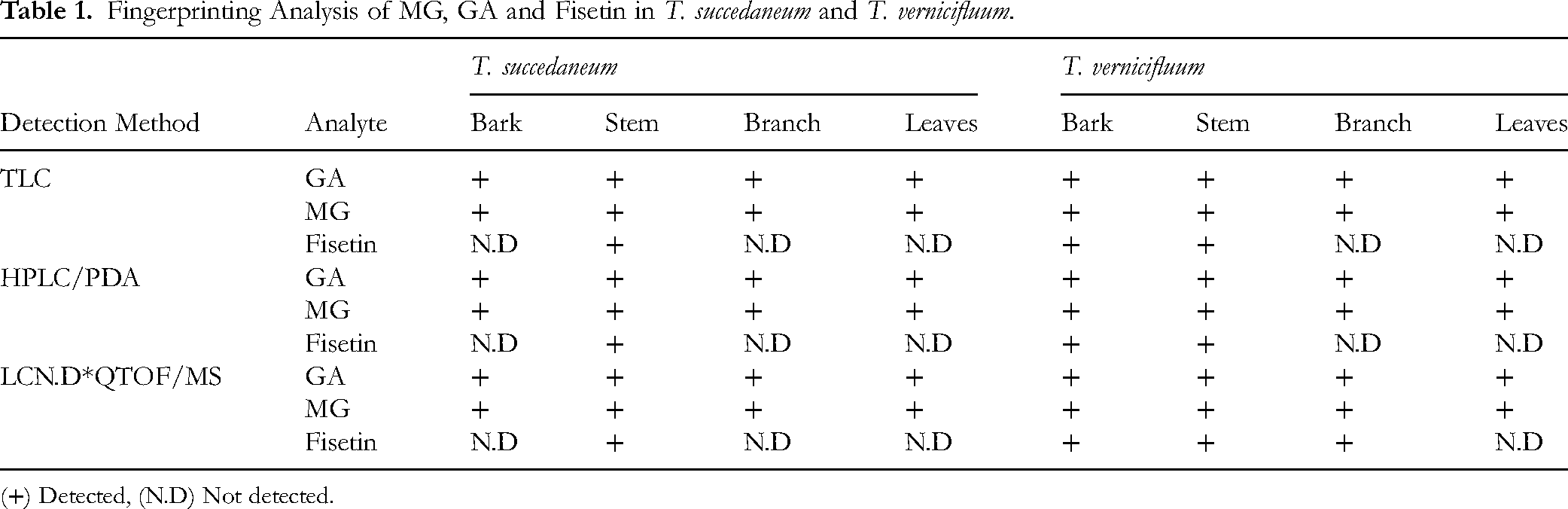
(+) Detected, (N.D) Not detected.
LC-QTOF/MS analysis (Figure 4, Table 2) identified eight additional major polyphenols, including gluconic acid (No. 3), fustin (No. 9), sulfuretin (No. 11), naringenin (No. 12), and four urushiol congeners (No. 14-17). These findings further highlight the chemical differences between the two species, especially in their flavonoid and urushiol composition.
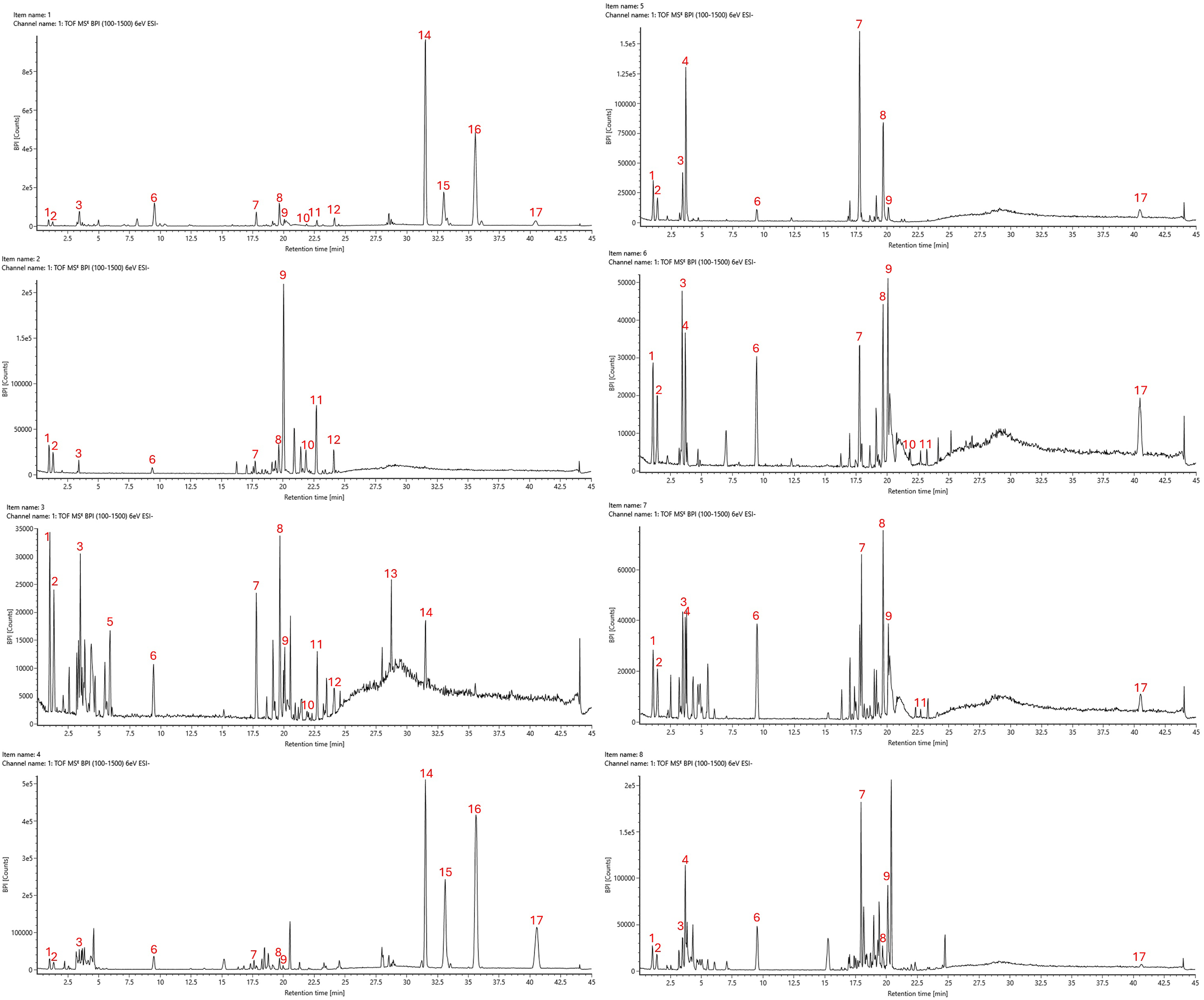
LC-QTOF/MS chromatograms: methanol extracts of T. vernicifluum samples, including (1) bark, (2) stem, (3) branch, and (4) leaves; methanol extracts of T. succedaneum samples, including (5) bark, (6) stem, (7) branch, and (8) leaves.
LC-QTOF/MS Data and Compound Identification of Metabolites from T. succedaneum and T. vernicifluum Methanol Extracts.

Ba: bark, S: Stem, Br: branch, L: leaves, N.D: Not detected.
Among the detected compounds, three unknowns (No. 1, 2, 8), together with gluconic acid (No. 3) and fustin (No. 9), appeared consistently in every plant part of both T. vernicifluum and T. succedaneum. Some metabolites showed clear species-specific patterns: unknown No. 4 was found only in T. succedaneum, while unknowns No. 5 and No. 13 were unique to T. vernicifluum. Sulfuretin (No. 11) was more widely distributed in T. vernicifluum, and naringenin (No. 12) showed a narrow distribution, occurring only in the bark, stem, and branches of this species. Overall, T. vernicifluum displayed a more diverse flavonoid profile.
For the urushiols, the unsaturated C15 forms (C15:3, C15:2, C15:1) were detected almost exclusively in T. vernicifluum, with the highest levels in the bark and leaves. In contrast, the saturated congener C15:0 appeared in the bark and leaves of T. vernicifluum but was also consistently present across all parts of T. succedaneum.
HPLC Analysis
A quantitative method was developed and validated for the simultaneous determination of MG, GA, and fisetin in T. succedaneum and T. vernicifluum (detailed Supplemental material 5). Table 3 presents the calibration parameters for the quantitative determination of GA, MG, and fisetin using a validated HPLC/PDA method. All three analytes exhibited excellent linearity within the tested concentration range of 0.25–100 µg/mL, as reflected by their high correlation coefficients (R2 = 0.9997), indicating strong linear relationships between peak area and analyte concentration. The limit of detection (LOD) for GA and MG was 0.025 µg/mL, while for fisetin it was slightly higher at 0.050 µg/mL. Similarly, the limit of quantification (LOQ) values were 0.083 µg/mL for GA and MG, and 0.167 µg/mL for fisetin. These results demonstrate the method's high sensitivity, particularly for GA and MG, allowing for reliable detection and quantification even at low concentrations in plant extracts. The strong linearity, broad dynamic range, and low LOD/LOQ values confirm that the developed HPLC/PDA method is robust and suitable for simultaneous quantification of MG, GA, and fisetin in complex herbal matrices derived from T. succedaneum and T. vernicifluum.
Calibration Curve Data for MG, GA, and Fisetin.

The quantitative analysis of MG, GA, and fisetin were in perfomed under the optimized HPLC/PDA conditions. Table 4 presents the quantified concentrations of GA, MG, and fisetin in various parts (bark, stem, branch, and leaves) of T. succedaneum and T. vernicifluum, expressed as mg/g of dried plant material. MG exhibited the highest accumulation in the stem of T. succedaneum (5.82 ± 0.11 mg/g), followed by the bark of T. vernicifluum (4.83 ± 0.09 mg/g). MG was present in all tested parts of both species, though at lower levels in the leaves and branches. Similarly, GA was ubiquitously distributed, with the leaves of T. succedaneum containing the highest GA concentration (3.86 ± 0.07 mg/g), whereas the bark of T. vernicifluum showed substantial GA content (3.30 ± 0.03 mg/g). These patterns suggest differential part localization of MG and GA between the two species, possibly reflecting differences in biosynthetic regulation or physiological roles. In contrast, fisetin was detected only in specific organs: the stem of T. succedaneum (0.10 ± 0.00 mg/g), and both the stem (0.58 ± 0.01 mg/g) and bark (0.23 ± 0.00 mg/g) of T. vernicifluum. It was not detected (N.D.) in any leaf or branch samples of either species (See Supplemental material 3). The notably higher fisetin content in T. vernicifluum aligns with previous reports on its flavonol profile and highlights its phytochemical distinctiveness from T. succedaneum. These findings demonstrate the organ-specific and species-dependent distribution of major phenolic compounds and emphasize the chemical richness of T. succedaneum, particularly its stem, as a novel source of MG.
MG, GA and Fisetin Contents in T. succedaneum and T. vernicifluum.

*Not detected.
Biological Activity
The in vitro activities of MG and GA are illustrated in Figure 5, which presents their antioxidant and anti-inflammatory effects, and in Figure 6, which shows their hepatoprotective properties.
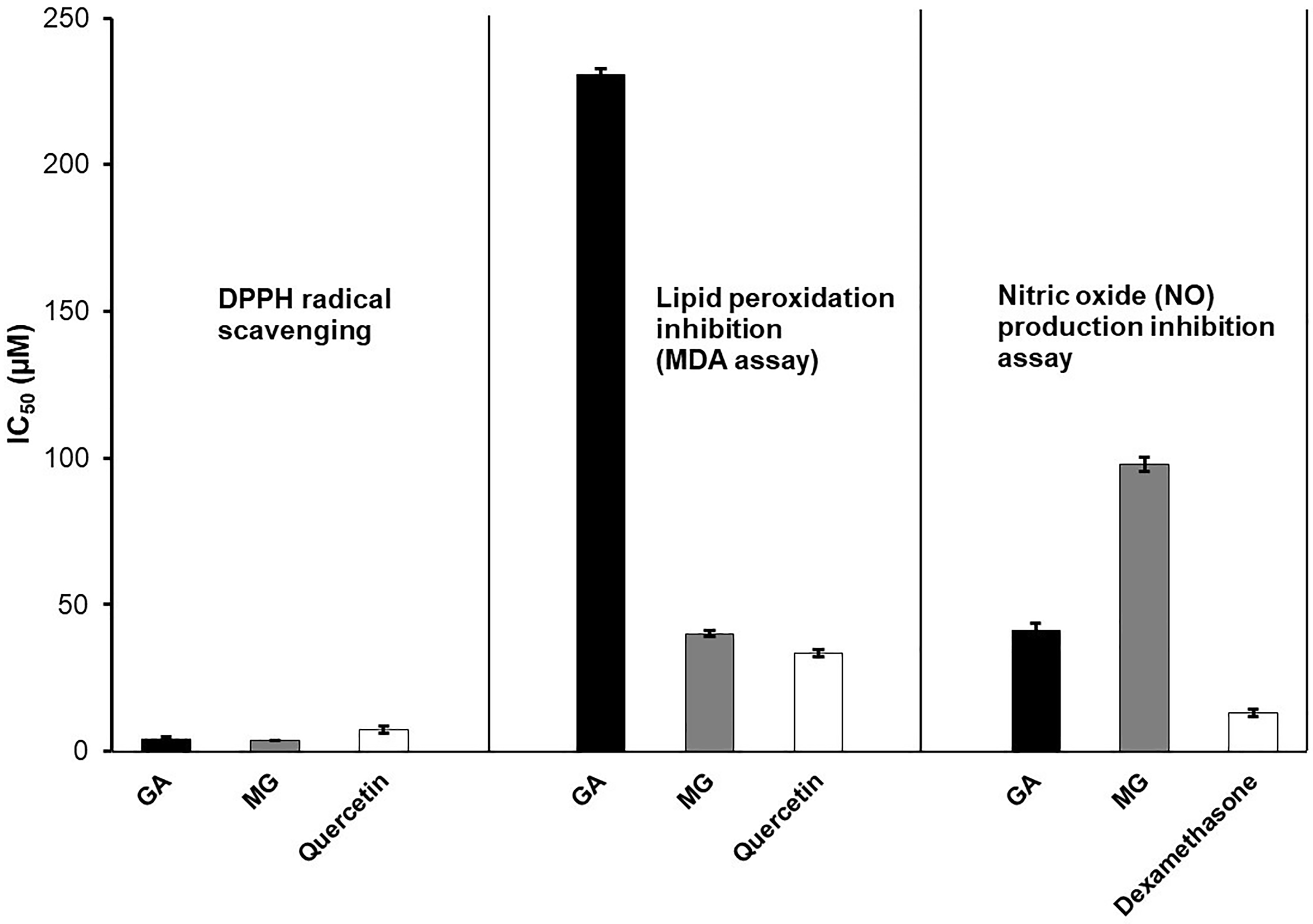
IC50 values of GA, MG, and positive controls in DPPH radical scavenging, lipid peroxidation inhibition (MDA assay), and nitric oxide (NO) production inhibition assay.

Hepatoprotective effect of GA, MG, and silymarin on HepG2 cells damaged by Acetaminophen (APAP) (* p < 0.05 and ** p < 0.01 compared with APAP-alone treated cells).
Antioxidant Activity by DPPH Assay
In the DPPH assay, both MG and GA exhibited potent antioxidant activity, with MG showing slightly superior radical scavenging capacity (IC50 = 3.89 µM) compared to GA (IC50 = 4.30 µM), and both outperforming the positive control quercetin (IC50 = 7.39 µM). These results highlight the strong free-radical scavenging potential of both compounds.
Antioxidant Activity by MDA Assay
In the MDA assay, which measures inhibition of lipid peroxidation, MG displayed markedly stronger activity (IC50 = 40.12 µM) than GA (IC50 = 230.55 µM), and also surpassed quercetin (IC50 = 33.55 µM). This suggests that MG, due to its increased lipophilicity from the methyl ester group, may have greater affinity for lipid environments, enabling more effective suppression of oxidative membrane damage.
Anti-Inflammatory Activity by NO Inhibition Assay
The NO inhibition assay revealed that GA was more effective than MG in suppressing nitric oxide production in LPS-stimulated RAW 264.7 macrophages, with IC50 values of 41.16 µM and 97.96 µM, respectively. However, both compounds were less potent than the reference anti-inflammatory agent dexamethasone (IC50 = 13.24 µM). The higher polarity of GA may facilitate better interaction with aqueous-phase targets such as inducible nitric oxide synthase (iNOS), thereby enhancing its inhibitory effect. Together, these findings indicate that both GA and MG possess notable antioxidant and anti-inflammatory properties, with MG demonstrating greater efficacy in lipid-phase oxidative protection, while GA is more potent in aqueous-phase NO suppression.
Hepatoprotective Activity
Figure 6 illustrates the hepatoprotective effects of GA, MG, and the reference drug silymarin on APAP-induced cytotoxicity in HepG2 cells at varying concentrations (6.25-100 µM). Both MG and GA exhibited a dose-dependent protective effect at low to moderate concentrations. Notably, MG consistently showed higher protective efficacy than GA across all doses from 6.25 to 50 µM, with a peak effect approaching that of silymarin at 100 µM. At 100 µM, however, MG's protective effect plateaued near zero, while GA demonstrated a striking reversal of activity, displaying negative protection values. In this stuty, silymarin, used as the positive control, maintained robust protective activity throughout the entire concentration range, with maximal efficacy at 100 µM. It is posible to note that, the biphasic response, particularly the loss and reversal of protection by GA at high doses, suggests the occurrence of a hormetic effect a well-documented phenomenon in which certain polyphenols exhibit beneficial effects at low doses but become cytotoxic at higher concentrations. The absence of overt cytotoxicity in preliminary MTT assays at these concentrations further implies a context-dependent modulation of cellular redox homeostasis, rather than direct cell killing. Collectively, these findings indicate that MG possesses superior hepatoprotective potential compared to GA at pharmacologically relevant doses, while silymarin remains the most effective compound overall. The data support the therapeutic relevance of MG as a candidate for liver-protective interventions and highlight the importance of dose optimization for phenolic compounds in cytoprotective applications.
Discussion
The lacquer trees Toxicodendron vernicifluum and T. succedaneum have long been culturally and economically important across East and Southeast Asia. Among them, T. vernicifluum, commonly found in Korea, Japan, and China with another names as Rhus verniciflua, and Rhus vernicifera, has been extensively studied for its diverse phytochemical profile and biological activities. 4 Notably, the bark is also used as a functional food ingredient, particularly in Korea, by boiling it with poultry (chicken or duck) to reduce its urushiol content while retaining its therapeutic benefits.4,7,17,18 This culinary medicinal overlap exemplifies the unique ethnobotanical relevance of T. vernicifluum, which has led to increasing research interest in its polyphenol-rich chemical constituents.19,20 By contrast, T. succedaneum, although widely cultivated in Vietnam for lacquer production, remains largely unexplored in terms of its non-resin constituents and medicinal potential.
In this study, we selected two Toxicodendron sp, one from Korea, and one from Vietnam to compare the main phenolic compounds using HPLC/PDA, and to evaluate and compare the antioxidant, anti-inflammatory, and hepatoprotective activities of MG and GA using established in vitro models. MG was first isolated from the bark of T. succedaneum with a high yield (0.2% w/w). Fingerprinting analysis using TLC, HPLC/PDA, and LC-QTOF/MS revealed clear chemical distinctions between T. succedaneum and T. vernicifluum. The complementary strengths of these methods yielded a comprehensive phytochemical profile, and their consistent results confirmed the reliability of our analysis. This study represents the first comprehensive comparison of key polyphenolic compounds-GA, MG, and fisetin-across different anatomical parts (bark, stem, branch, and leaves) of T. succedaneum and T. vernicifluum. MG, which was successfully isolated from T. succedaneum for the first time, was found in all plant parts of both species, with the highest concentration detected in the stem of T. succedaneum (5.82 mg/g). This finding is consistent with previous reports of methyl gallate's presence in the wood, lignum, and leaves of T. vernicifluum. 7 GA was also present throughout both species, with peak accumulation in T. succedaneum leaves (3.86 mg/g) and T. vernicifluum bark (3.30 mg/g). Interestingly, fisetin, an established flavonol marker of T. vernicifluum, was absent in most parts of T. succedaneum, detected only in the stem at low levels (0.10 mg/g), while it was more abundant in T. vernicifluum bark and stem (0.23 and 0.58 mg/g, respectively). Notably, a trace amount of fisetin was also found in the branch of T. vernicifluum. This detection was achieved exclusively by the highly sensitive LC-QTOF/MS technique, as it was below the detection limits of conventional HPLC/PDA. This finding underscores that LC-QTOF/MS is a powerful tool for detecting compounds present in very low concentrations, complementing traditional analytical techniques. The highly sensitive LC-QTOF/MS technique was affirmed not only by detecting trace levels of fisetin (No. 3) in T. vernicifluum branch but also by allowing the detection of eight additional major constituents and 6 unknown (Table 2). These compounds, specifically flavonoids, served as crucial markers to distinguish the chemical between the two species.
The most critical finding from the LC-QTOF/MS profiling relates to the allergenic alkyl catechols (urushiols), which define the safety profile of Toxicodendron species. Our analysis focused on the C15 urushiol isomers (No. 14-17, alkyl chain C15) commonly found in T. vernicifluum. Extracts from T. vernicifluum demonstrated extremely high detector counts for unsaturated C15 urushiols, which is consistent with the recognized high allergenicity of the species and the traditional need for toxicity mitigation. In stark contrast, the non-resin biomass of T. succedaneum analyzed showed a near absence of these C15 urushiol isomers, with only a trace amount of saturated urushiol 15:0 detected in the T. succedaneum bark. This pronounced chemical difference in the C15 alkyl catechol profile of the biomass is highly significant. This result aligns with the established composition of lacquer sap: the resin from T. vernicifluum (Korean lacquer) primarily consists of urushiol (C15 chain), whereas the sap from T. succedaneum (Vietnamese lacquer) mainly contains laccol (3-heptadecylcatechol, C17 chain).
These findings indicate that T. succedaneum shares a comparable polyphenolic backbone with T. vernicifluum, but lacks high levels of flavonol accumulation, suggesting species-specific regulation of flavonoid biosynthesis. The high yield of MG in T. succedaneum also underscores its potential as a sustainable source of this bioactive phenolic ester, particularly given that the biomass from older lacquer trees is typically discarded after resin harvest.
Regarding biological activity, both GA and MG showed antioxidant properties. In the DPPH assay, MG exhibited slightly greater efficacy than GA, with both compounds stronger than quercetin, the positive control. For anti-lipid peroxidation activity (MDA assay), MG proved significantly more effective than GA. While GA showed stronger anti-inflammatory activity in the NO production inhibition assay compared to MG, both were less potent than dexamethasone. A structure-activity relationship was observed between GA and its ester derivative, MG. MG exhibited significantly superior anti-lipid peroxidation activity in the MDA assay. This can be attributed to its increased lipophilicity due to the methyl ester group, potentially facilitating better distribution into the lipid-rich environment of the liver homogenate where peroxidation occurs. Conversely, the more polar GA proved to be a more potent inhibitor of nitric oxide production in an aqueous cell culture medium. In the hepatoprotective assay using APAP-induced HepG2 cell damage, both GA and MG displayed dose-dependent protective effects at 6.25 to 50 µM with MG higher than GA. However, at 100 µM, an unexpected reversal to exacerbated cellular damage occurred, causing cellular damage instead of protection. This finding is consistent with the hormesis phenomenon widely reported for polyphenols. Hormesis describes a biphasic dose-response: beneficial at low doses, toxic at high doses. Further mechanistic studies are needed to fully understand these complex interactions.
Taken together, this study provides a comprehensive phytochemical and pharmacological profile of T. succedaneum, a traditionally underutilized lacquer tree species. The successful isolation of MG in high yield, its widespread distribution in plant parts, and its strong biological activity—particularly antioxidant and hepatoprotective, demonstrate its promise as a bioactive lead compound. While T. vernicifluum remains a rich source of fisetin and other flavonoids, the findings here suggest that T. succedaneum biomass, often discarded after lacquer production, represents a valuable and sustainable resource for developing health-promoting natural products. This work contributes to bridging the knowledge gap between two chemically related but unequally studied lacquer tree species and lays the groundwork for further mechanistic and translational research on methyl gallate and its derivatives.
While this study provides valuable insights, certain limitations should be acknowledged. All bioactivity assays were performed in vitro, and additional in vivo studies are needed to validate the therapeutic relevance. Moreover, the analysis targeted only a subset of compounds, even though the phytochemical profiles are inherently complex, indicating that other bioactive metabolites may remain unidentified. In addition, the conclusions regarding compound distribution and bioactivity could be constrained by the use of samples from a single geographical origin, and the hormetic effects observed in the hepatoprotective assay warrant further mechanistic investigation to clarify the dose-dependent reversal of protection.
Conclusions
This study provides the first comprehensive phytochemical and biological evaluation of T. succedaneum in comparison with T. vernicifluum. Methyl gallate was successfully isolated from the bark of T. succedaneum for the first time, and its quantification, alongside GA and fisetin, revealed distinct species- and part-specific distribution patterns. We performed fingerprinting analysis by TLC, HPLC/PDA, and LC-QTOF/MS to compare the chemical profiles of different parts from both species. Based on these qualitative results, we quantitatively analyzed and compared the levels of MG, GA, and fisetin in the samples. Our findings confirmed that MG and GA were ubiquitously present in both species, while fisetin was predominantly confined to T. vernicifluum. LC-QTOF/MS profiling showed that T. vernicifluum contains much higher levels of specialized flavonoids, matching its known flavonoid-rich nature. In contrast, the chemical profile of T. succedaneum is largely dominated by methyl gallate. A clear difference was also seen in the allergenic components: the non-resin parts of T. vernicifluum had high amounts of C15 urushiols, whereas T. succedaneum contained only trace levels of C15:0. This pattern is consistent with reports that Vietnamese lacquer sap is mainly composed of C17 laccol.
In vitro assays demonstrated that MG exhibited superior lipid peroxidation inhibition and hepatoprotective activity, whereas GA was more effective in nitric oxide suppression. Notably, both compounds showed dose-dependent hepatoprotective effects, with a hormetic response observed at higher concentrations. These findings highlight T. succedaneum as a promising and underutilized source of bioactive phenolic compounds, particularly MG, and support its potential development as a functional phytopharmaceutical ingredient. Further mechanistic studies and in vivo validation are warranted to advance its therapeutic application.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251414828 - Supplemental material for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum
Supplemental material, sj-docx-1-npx-10.1177_1934578X251414828 for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum by Thuong Thi-Thuong Le, Pham Thi Thu, Thao Thi Thu Nguyen, Quoc Vu Do, Thi Hong Van Le, Tran Ngoc Dang and Thanh Dung Phan in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251414828 - Supplemental material for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum
Supplemental material, sj-docx-2-npx-10.1177_1934578X251414828 for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum by Thuong Thi-Thuong Le, Pham Thi Thu, Thao Thi Thu Nguyen, Quoc Vu Do, Thi Hong Van Le, Tran Ngoc Dang and Thanh Dung Phan in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251414828 - Supplemental material for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum
Supplemental material, sj-docx-3-npx-10.1177_1934578X251414828 for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum by Thuong Thi-Thuong Le, Pham Thi Thu, Thao Thi Thu Nguyen, Quoc Vu Do, Thi Hong Van Le, Tran Ngoc Dang and Thanh Dung Phan in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X251414828 - Supplemental material for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum
Supplemental material, sj-docx-4-npx-10.1177_1934578X251414828 for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum by Thuong Thi-Thuong Le, Pham Thi Thu, Thao Thi Thu Nguyen, Quoc Vu Do, Thi Hong Van Le, Tran Ngoc Dang and Thanh Dung Phan in Natural Product Communications
Supplemental Material
sj-docx-5-npx-10.1177_1934578X251414828 - Supplemental material for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum
Supplemental material, sj-docx-5-npx-10.1177_1934578X251414828 for Comparative Quantitative and Bioactivity Analysis of Methyl Gallate and Related Polyphenols from Toxicodendron Succedaneum and Toxicodendron Vernicifluum by Thuong Thi-Thuong Le, Pham Thi Thu, Thao Thi Thu Nguyen, Quoc Vu Do, Thi Hong Van Le, Tran Ngoc Dang and Thanh Dung Phan in Natural Product Communications
Footnotes
Acknowledgments
This work received financial assistance from the Korea International Cooperation Agency (KOICA). The University of Medicine and Pharmacy at Ho Chi Minh City also provided essential support and facilities for the execution of this research. We would also like to extend our sincere thanks to Professor J.H. Park from Seoul National University for his generous provision of the T. vernicifluum samples, which were instrumental to our research.
ORCID iDs
Ethical Approval
Ethical Approval is not applicable for this article.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received support from the Korea International Cooperation Agency (KOICA) under the project entitled “Education and Research Capacity Building Project at the University of Medicine and Pharmacy at Ho Chi Minh City,” conducted from 2024 to 2025 (Project No. 2021-00020-3).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect tothe research, authorship, and/or publication of this article and all supplementary files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
