Abstract
Objective
To analyze the chemical composition of tea tree oil (TTO) and elucidate the antibacterial mechanisms of its major component, terpinen-4-ol (T-4-O), against Staphylococcus aureus.
Methods
The chemical constituents of TTO were determined using GC MS. The antibacterial mechanism of T-4-O was investigated from multiple perspectives, including its in vitro antibacterial activity, effects on bacterial growth, morphology, cell membrane and cell wall integrity, as well as leakage of cell lysates and proteins, cell cycle progression, and transcriptional responses.
Results
GC-MS analysis revealed that TTO contained 15 compounds, primarily alcohols, alkenes, alkanes, and esters, among which T-4-O was the most abundant component, accounting for 31.09%. T-4-O exhibited strong antibacterial activity against S. aureus, with a minimum inhibitory concentration (MIC) of 2.12 mg/mL and a minimum bactericidal concentration (MBC) of 8.47 mg/mL. At 2 × MIC, T-4-O effectively suppressed bacterial growth. Furthermore, T-4-O disrupted metabolic pathways associated with biofilm formation, leading to approximately 50% inhibition of S. aureus biofilm development. It also induced structural disorganization of the cell membrane and impaired cell wall synthesis, resulting in leakage of cellular lysates, proteins, and other intracellular components. As a consequence, normal cell division and growth were inhibited, and the bacterial cell cycle was arrested at the R phase. Ultimately, these cumulative effects caused cell membrane rupture and bacterial death.
Conclusion
T-4-O exerts significant antibacterial effects against S. aureus primarily by disrupting cell membrane integrity, inhibiting biofilm formation, and inducing cell cycle arrest, collectively leading to bacterial death.
Introduction
Plant essential oils are secondary metabolites in plants with very complex compositions, mainly including terpenes and their derivatives. 1 Owing to their intricate chemical compositions, these oils exhibit a broad spectrum of biological activities, such as antibacterial, anti-inflammatory, and antioxidant effects. They are also known to promote animal feed intake, enhance productive performance, and boost immune function. Additionally, due to their non-toxic nature and absence of drug residues, plant essential oils are considered promising alternatives to growth-promoting antibiotics and have become a focus of research both domestically and internationally in recent years.2,3 Melaleuca ahemifolia belongs to myrtaceae melaleuca plants, with about 230 species, and is widely distributed in Fujian, Sichuan, Yunnan, Guangdong, Guangxi and other provinces in China. 4 Tea tree oil (TTO), also known as the oil from Melaleuca alternefolia, is a colorless to light-yellow liquid, extracted from the shoots and leaves of myrtaceae melaleuca plants in Australia by steam extraction, and contains the aroma of camphor and peppermint. 5 Because of its high economic value, unique efficacy, and safety profile, TTO is widely used in spices, food, agriculture, and cosmetics, and has been recognized by many European and American countries. It is also one of the few essential oils recorded in the seventh edition of the European Pharmacopoeia. 6
TTO is a complex mixture containing over a hundred chemical components, primarily consisting of monoterpenes, sesquiterpenes, and alcohols. 7 The major components in TTO can be categorized into six chemical types: turpentine, turpentine-4-ol (T-4-O), and four kinds of 1,8-eucalyptus. 8 Notably, T-4-O and 1,8-eucalyptus (1,8-cineole) collectively constitute more than 50% of the total oil content, which is primarily responsible for TTO's potent antimicrobial activity. 9 As a plant extract, TTO has garnered substantial clinical evidence supporting its antimicrobial efficacy and other beneficial effects. For instance, Groppo. 10 employed a randomized controlled single-blind method to investigate the clinical efficacy of a 6% TTO gel for treating recurrent herpes labialis, revealing significant therapeutic outcomes. Similarly, Arweiler et al 11 examined the bactericidal effect of a 0.34% TTO mouthwash on oral bacteria and concluded that TTO could markedly reduce the number of oral bacteria. However, the chemical composition of TTO can vary considerably depending on the geographical origin of cultivation, leading to differences in its antibacterial effects. 12 Therefore, this study employed gas chromatography-mass spectrometry (GC-MS) to analyze the chemical composition of a commercially available tea tree essential oil. The in vitro antibacterial activity of its major component T-4-O and the underlying antibacterial mechanism against Staphylococcus aureus were thoroughly investigated, aiming to provide a theoretical basis for the further development and utilization of TTO and its major components.
Materials and Methods
Essential Oils
TTO (Product No. SMB00386) and T-4-O (Product No. 11584) were both purchased from Sigma-Aldrich (Shanghai, China) Trading Co., Ltd with purities of 98% and 95%, and densities of 0.908 and 0.910, respectively.
Bacterial Strains and Culture Media
Escherichia coli (ATCC44102), Staphylococcus aureus (ATCC26003), and Candida albicans (ATCC98001) were all purchased from the Shanghai Collection of Microorganisms in China. Bacterial culture was conducted using LB (LuriaBertani) medium, whereas fungal culture was performed using YPD (Yeast Peptone Dextrose) yeast medium.
Reagent Kits and Main Instruments
Total RNA extraction was performed using the Omege soil RNA Kit (Product number:R6825-02) from Shanghai Yuanmu Biotechnology Co., Ltd (China). The AKP assay kit (Product number A059-3-1) was purchased from Nanjing Jiancheng Bioengineering Institute(China). UV-5200 UV Visible spectrophotometer was purchased from Shanghai Yuanxi Instrument Co., Ltd, China. The GC-MS gas chromatography-mass spectrumetry instrument was purchased from Agilent Technologies (China) Co., Ltd The scanning electron microscope was purchased from Hitachi Group (Japan). The enzyme-linked immunosorbent assay (ELISA) reader and Fourier transform infrared spectrometer were both purchased from Thermo Fisher Scientific (USA). The freeze dryer was purchased from Ximeng International Group Co., Ltd (China). The flow cytometer was purchased from BD Corporation(USA).
Preparation of TTO and T-4-O
Based on preliminary experiments which confirmed that a 1:19 (v/v) mixture of TTO or T-4-O with anhydrous ethanol had no effect on bacterial growth, stock solutions were prepared by dissolving TTO and T-4-O in anhydrous ethanol to achieve mass concentrations of 880 mg/mL and 846.5 mg/mL, respectively.
Composition Analysis of TTO by GC-MS
Chromatographic conditions: A capillary column (30 m × 0.32 mm × 0.25 μm) was used for separation. High-purity helium (≥ 99.999%) served as the carrier gas at a constant flow rate of 1.2 mL/min. The injector temperature was set at 250 °C in splitless mode. The oven temperature program was as follows: initial temperature of 60 °C held for 1 min, then ramped to 230 °C at a rate of 15 °C/min.Mass spectrometry conditions: Analysis was performed using an electron ionization (EI) source. The ion source temperature, quadrupole temperature, and transfer line temperature were set at 230 °C, 150 °C, and 280 °C, respectively. The electron energy was 70 eV. Data acquisition was conducted in full scan mode, covering a mass range of 50 to 650 m/z.
Determination of in Vitro Antibacterial Activity of T-4-O Against Different Bacterial Strains
The antibacterial activity of T-4-O against Escherichia coli, Staphylococcus aureus, and Candida albicans was evaluated by measuring the inhibition zone using the Oxford cup method. Each test strain was first activated and cultured on its respective medium before the assay. Bacterial suspensions were prepared by transferring the strains into sterile saline tubes to achieve a concentration of 108 CFU/mL. A 0.1 mL aliquot of each bacterial suspension was spread evenly onto the corresponding agar plate. An Oxford cup (6 mm in diameter) was placed on each inoculated plate, and 150 μL of the prepared T-4-O solution (846.5 mg/mL) was added into the cup, while 150 μL of ampicillin solution (100 μg/mL, used for bacterial assays) or amphotericin B (10μg/mL, used for fungal assay) was added into another cup on the same plate as a positive control. The plates were incubated in a biochemical incubator (37 °C for bacteria and 30 °C for fungi) for 24–48 h. The diameter of the inhibition zone for each solution was measured. Anhydrous ethanol was used as the negative control. All experiments were performed in triplicate with three independent replicates to ensure the reliability of the results, and this applies to all subsequent experiments described herein.
The Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) of T-4-O were determined using the broth dilution method. A series of ten sterile test tubes were prepared and numbered 1 through 10. Each tube initially received 1 mL of LB medium. Subsequently, 1 mL of the diluted T-4-O solution (677.2 mg/mL, 1.25-fold dilution) was added to tube 1 and mixed thoroughly. A serial two-fold dilution was then performed by transferring 1 mL from tube 1 to tube 2, mixing, and continuing this process up to tube 7. After mixing tube 7, 1 mL of the mixture was discarded to maintain equal volumes. Then, 3.8 mL of LB medium was added to all ten tubes. Tubes 1 to 7 were inoculated with 0.2 mL of the bacterial suspension (108 CFU/mL). Tube 8 received 0.2 mL of T-4-O solution (sterility control), tube 9 received 0.2 mL of LB medium (medium control), and tube 10 received 0.2 mL of the bacterial suspension (growth control), the above experimental steps were shown in Table 1. All tubes were incubated in an orbital shaker at 37 °C and 120 rpm for 24 h. After incubation, the MIC was determined as the lowest concentration in tubes 1–7 that showed no visible turbidity, using tubes 8–10 as reference controls. For MBC determination, 50 μL aliquots from all clear tubes (showing no turbidity) were plated onto LB agar plates. These plates were incubated for an additional 24 h at 37 °C. The MBC was defined as the lowest concentration that resulted in no visible bacterial growth on the agar plates.
The Dilution Ratio and Concentration of T-4-O Solution in Different Test Tubes.

Effect of T-4-O on the Growth and Structure of S. aureus
The effect of T-4-O on the growth of S. aureus was determined as follows: An S. aureus suspension was cultured to the logarithmic phase and adjusted to 1 × 108 CFU/mL, this same protocol for strain activation and suspension preparation was followed in all subsequent procedures. Subsequently, T-4-O was added to the S. aureus suspension at three concentrations (1MIC 2.12 mg/mL,2MIC 4.24 mg/mL and 3MIC 6.36 mg/mL, the same below), using distilled water serving as the control. The mixed samples were then cultured in a shaker at 37 °C, and 1 mL of samples were taken at 0, 0.5, 1, 2, 3, and 4 h, respectively. The OD600 value of the bacterial suspension was measured using a spectrophotometer to evaluate the influence of T-4-O at different concentrations on the growth of S. aureus.
The overall structure of S. aureu was observed using scanning electron microscopy (SEM). S. aureu suspensions were prepared according to the method previously described. The prepared suspensions were then treated with different concentrations (1 MIC, 2 MIC, and 3 MIC) of T-4-O. These treated suspensions were then incubated in a shaker at a speed of 180 r/min, with distilled water serving as the control. The incubation temperature was maintained at 37 °C for 1 h. Subsequently, 2 mL of each bacterial suspension was centrifuged at 4000 r/min for 10 min. The collected cells were washed twice with a 50% ethanol/water solution. A 20 μL aliquot of the suspension was placed on a cover glass, fixed with 100 μL of 2.5% glutaraldehyde solution (diluted in phosphate buffer) for 2–3 h, and finally sputter-coated with gold before SEM examination.
The effect of T-4-O on the cell wall of S. aureus was determined as follows: Under normal physiological conditions, alkaline phosphatase (AKP) is confined within the cell wall of Gram-positive bacteria and is not released extracellularly; thus, its activity remains undetectable in the supernatant. Therefore, the detection of AKP activity in the extracellular environment serves as an indicator of cell wall integrity. The AKP assay kit enables the colorimetric detection of substances associated with the cell wall using a spectrophotometer. In this experiment, S. aureus was activated and bacterial suspensions were prepared as described previously. According to the manufacturer's instructions, AKP activity was measured to assess cell wall integrity. The resulting AKP data were further correlated with the corresponding SEM observations of cell wall morphology for comprehensive analysis. Specifically, the effect of T-4-O on the cell wall was evaluated by measuring AKP activity at 520 nm using a microplate reader after treatment with various concentrations of T-4-O (1 MIC, 2 MIC, and 3 MIC), with distilled water serving as the control.
Effect of T-4-O on Cell Membrane and Substance Leakage of S. aureus
The effect of T-4-O on the cell membrane of S. aureus was assessed using the following procedure: Bacterial activation and suspension preparation were performed as described previously. The bacterial suspension was mixed with T-4-O at different concentrations (1 MIC, 2 MIC, and 3 MIC) by blowing, and 300 μL of the mixture was taken and added into a 96-well plate, and incubated at 37 °C for 36 h with LB medium as a blank control, after which the liquid in the 96-well plate was aspirated, and 200 μL of methanol was added to each well to stand for 15 min. After removing the methanol, each well was cleaned three times by adding 30 μL of sterile phosphate buffered saline (PBS). Following the PBS wash, the wells were air-dried before adding 250 μL of 10 g/L crystal violet solution to each well, and allowed to stand for 15 min.
The crystal violet solution was discarded and rinsed with sterile PBS 3 times. Each well was dissolved with 250 μL of 30% acetic acid. After 30 min, the OD was measured at a wavelength of 570 nm. The effect of T-4-O on biofilm formation of S. aureus was evaluated by crystal violet staining. After bacterial solution was treated with T-4-O, the supernatant was discarded by centrifugation, frozen with glutaraldehyde solution for 4 h at low temperature, then dehydrated step by step with ethanol, and completely replaced with isoamyl acetate. After freezing step by step, it was sublimated and dried by freeze dryer. The tablet was subjected to Fourier transform infrared spectroscopy. The change of absorption peak intensity serves as an indicator of the change of component content in the specific information area, to explore the effect of T-4-O on cell membrane compositions.
Cell lysate leakage was tested using the following method: Bacterial activation and suspension preparation were performed as described previously. The bacterial suspension was centrifuged at 4000 r/min for 10 min, and the bacteria were collected, washed three times with sterile saline, and then resuspended in the initial volume of sterile saline. T-4-O at three concentrations (1 MIC, 2 MIC, and 3 MIC) was added to the equal portion of the bacterial suspension with a volume of 25 mL, and distilled water was used as the control. The samples were cultured at 37 °C in a constant temperature oscillator. The mixture after cultivation for 0, 1 and 2 h was collected and centrifuged at 4000 r/min for 10 min, respectively, and the absorbance of the supernatant was measured at 260 nm using a visible spectrophotometer. Substances absorbed at 260 nm include proteins and nucleic acids, and the OD of the supernatant at 260 nm was an indicator for cell membrane rupture and release of cellular components, and the change in OD260 was used for reflecting the leakage of cell lysates.
Intracellular protein leakage was tested as follows: S. aureus activation and suspension preparation were carried out as described above. Protein leakage was investigated by determining protein content in supernatants of bacterial suspension with the addition of T-4-O at three concentrations (1 MIC, 2 MIC, and 3 MIC). Protein contents in supernatants were determined by the Coomassie Brilliant Blue method and UV-visible spectrophotometer at 595 nm after 1 and 2 h incubation, respectively. The amount of protein released was calculated from the standard serum albumin curve. 13
Effect of T-4-O on Cell Cycle of S. aureus
Taking bacterial liquid of S. aureus at the logarithmic phase, according to the above test results, a final concentration of 2 MIC of T-4-O was added and thoroughly mixed. The mixture was then incubated at 37 °C with a shaking speed of 150 r/min for 1 h. Simultaneously, an equal volume of sterile water was added as a blank control. After incubation, the bacteria were collected and centrifuged at 4000 r/min for 10 min, the supernatant was discarded, and the cells were washed twice with PBS. Subsequently, 1 mL of pre-cooled 70% ethanol was added to fully resuspend the bacterial precipitates and placed in a refrigerator at 4 °C for fixation overnight. The next day, centrifugation at 4000 r/min was conducted for 10 min to remove the supernatant, and the cells were washed twice with PBS. Then, 450 μL of PBS was added to resuspend the cell precipitates of S. aureus, followed by the addition of 50 μL of 1 mg/ml RNase solution and 5 μL of 1 mg/mL propidium iodide (PI) solution, resulting in final concentrations of approximately 100 μg/mL and 10 μg/mL, respectively. The mixture was thoroughly mixed and incubated at 4 °C for 15 min under light-free conditions. The following controls were prepared in parallel to ensure data specificity and accuracy: unstained control (no dye), PI-only control and RNase-only control. The stained S. aureus cells were analyzed using a BD Accuri C6 flow cytometer. Data were acquired using the FL2 channel (wavelength 488 nm) for PI fluorescence. A threshold was set on forward scatter (FSC) to exclude debris, and 20,000 events within the gated bacterial population were collected for each sample. The gating strategy was as follows: the primary bacterial population was first identified on an FSC-A versus SSC-A dot plot, and the PI fluorescence of this gated population was displayed on a histogram. The data were analyzed using FlowJo software. 14
Effect of T-4-O on Transcriptomics of S. aureus
Following the pre-culture procedure described in the cell cycle analysis section, bacterial cells from the T-4-O treatment group and the blank control group (three biological replicates each) were collected for total RNA extraction using the Omega Soil RNA Kit. The extraction was performed according to the manufacturer's protocol, which included cell lysis at 70 °C, purification of RNA by binding to a silica matrix membrane, and final elution with RNase-free water. To enrich for mRNA, ribosomal RNA (rRNA) was removed from the total RNA samples. The qualified RNA samples were sent to Beijing Ovison Genetics Co., Ltd for library construction and sequencing on the Illumina HiSeq 4000 platform (PE150). On average, approximately 40 million raw reads were generated per sample. The raw sequencing data were first processed to ensure quality: Trimmomatic software (v0.33) was used to remove adapter sequences and low-quality reads. The resulting clean reads were then aligned to the S.aureus reference genome using TopHat 2 software, achieving an average mapping rate of 95%. Gene expression levels were quantified using HTSeq software (union mode) and expressed as FPKM (Fragments Per Kilobase of exon model per Million mapped fragments). Genes with an FPKM > 1 were considered expressed. 15 Differential gene expression analysis was performed using DESeq, with a significance threshold of q value < 0.05. 16 The obtained differentially expressed genes (DEGs) were subsequently subjected to functional annotation and enrichment analysis in the Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) databases. 17
Data Statistical Analysis
All data were presented as the mean ± standard deviation (SD) from at least three independent replicate experiments. Data processing and graph generation were performed using Microsoft Excel. Statistical analyses, including independent-samples t-tests and one-way analysis of variance (ANOVA), were conducted with SPSS software (version 21.0). P < 0.05 was considered statistically significant.
Results
Chemical Composition Analysis of TTO
The chemical composition of TTO was analyzed by GC-MS, with the total ion chromatogram presented in Figure 1 and Table 2. The analysis revealed 20 distinct peaks, among which 15 compounds were successfully identified (some peaks corresponded to column effluents or non-essential oil components, accounting for 15.46%). The identified compounds included alcohols (38.92%), alkenes (14.66%), alkanes (14.96%), and esters (16.00%). Notably, T-4-O constituted 31.09% of the total composition, while 1,8-cineole accounted for 2.12%. These results are consistent with the specifications of GBT 2651-2011 for Melaleuca alternifolia (tea tree) oil, terpinen-4-ol type, which requires T-4-O content to be ≥ 30% and 1,8-cineole ≤ 5%.
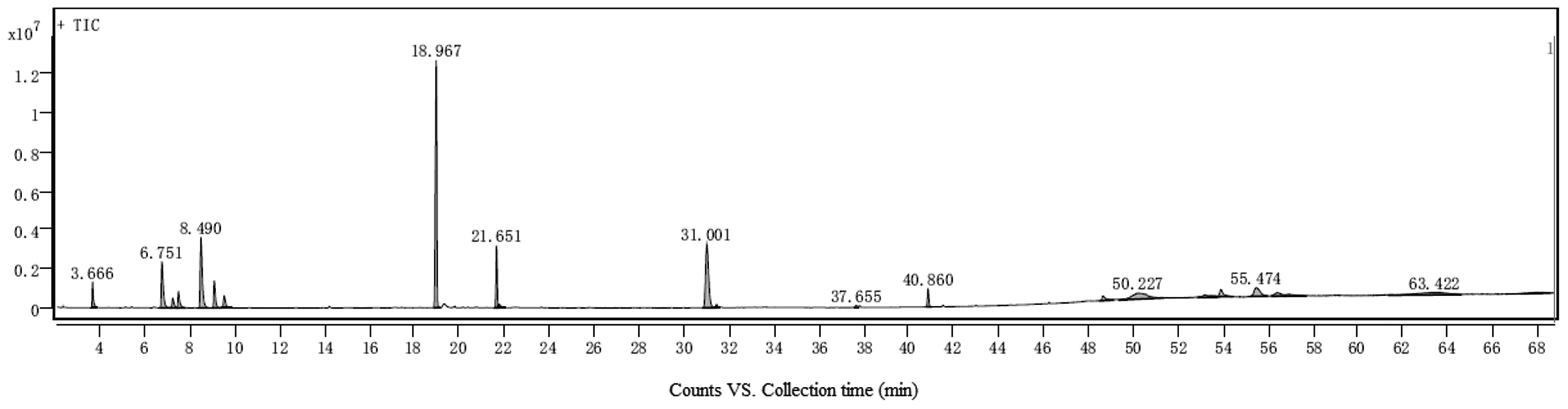
Total ion flow chart of TTO GC/MS analysis. 20 peaks appeared in the figure, and 15 compounds were identified, some of which were column effluent and non essential oil components.
The Analysis Results of the Representative Substance Components of Each Peak in TTO GC/MS Analysis.

Measured Results of Bacteriostatic Properties of T-4-O
The determined results of the bacteriostatic circle showed that T-4-O exhibited varying degrees of antibacterial activity against the tested strains, with the strongest effect observed against S. aureus (22 mm inhibition zone), followed by E.coli (16 mm) and C.albicans (10 mm) (Figure 2A–C). Notably, in comparison with the positive controls ampicillin and amphotericin B (Figure 2D–F), the inhibition zones of T-4-O were slightly smaller, T-4-O exhibited promising antibacterial efficacy, supporting its potential as an alternative or adjunctive agent to conventional antibiotics.

Antibacterial activity of T-4-O and ampicillin (positive control) against Escherichia coli (A, D), Staphylococcus aureus (B, E), and Candida albicans (C, F).
The measurement results of MIC and MBC are presented in Table 3, the MIC and MBC of T-4-O against different strains showed the strongest effect on S. aureus, with MIC of 2.12 mg/mL and MBC of 8.47 mg/mL, respectively.
MIC and MBC Determination of Different Strains by T-4-O.

The Effect of T-4-O on the Growth and Structure of the Strain
The impact of T-4-O on the growth of S. aureus was meticulously examined at varying concentrations. As shown in Figure 3a, T-4-O exhibited a pronounced inhibitory effect on S. aureus compared with the control group. Bacterial growth was increasingly inhibited with higher T-4-O concentration, although the inhibitory effect of T-4-O at the concentration of 3 MIC did not show a significant difference compared to 2 MIC.

The impact on the growth and structure of bacterial strains. (A) The effect of different concentrations of T-4-O on the growth of bacterial strains; (B) Scanning electron microscopy images of Staphylococcus aureus treated with different concentrations of T-4-O for 1 h; (C)AKP activity measured by different concentrations of T-4-O bacterial solution. For panels (a) and (c), values were expressed as mean ± standard deviation (sd) of three biological replicates (n = 3).
The overall structure of S. aureus was observed by SEM after co-culture with different concentrations of T-4-O. As shown in Figure 3b, the untreated S. aureus in the control group had intact cell structure with smooth and spherical surfaces. However, after treated with T-4-O at different concentrations for 1 h, the bacterial cells displayed varying degrees of membrane damage, including surface wrinkling and partial collapse. Moreover, higher concentrations of T-4-O led to more severe damage to the cell membrane, resulting in increased cellular collapse and rupture.
The cell wall integrity of S. aureus was evaluated by measuring extracellular AKP activity after treatment with T-4-O. As shown in Figure 3c, when different concentrations of T-4-O were added to the co culture with Staphylococcus aureus, the activity of AKP significantly increased compared to the control group, and AKP activity also increased with the increase of T-4-O concentration, indicating that the higher the concentration of T-4-O, the more obvious its damage to bacterial cell walls. This biochemical finding is consistent with the morphological alterations observed via SEM (Figure 3b). Specifically, as the T-4-O concentration increased, SEM images revealed correspondingly more severe structural damage to the bacterial surface, including wrinkling, collapse, and rupture. Together, the increased AKP leakage and the direct visual evidence of cell wall disruption confirm that T-4-O effectively compromises the cell wall integrity of S. aureus, contributing to its antibacterial action.
The Effect of T-4-O on the Cell Membrane and Substance Leakage of the Strain
The effect of T-4-O on the cell membrane of S. aureus was assessed through biofilm removal analysis and infrared spectroscopic characterization. As shown in Figure 4a, it had a significant removal effect on the biofilm of S. aureus when compared with the control group. The removal of biofilm was enhanced with increased T-4-O concentration. The inhibitory rate on the formation of S. aureus biofilm was approximately 50% when the addition amount of T-4-O was 2 MIC, but there was no difference when compared with 3 MIC (P > 0.05). The infrared spectra of S. aureus co-cultured with different concentrations of T-4-O were analyzed. As shown in Figure 4b, the peak of 1080−1 was the structure of peptidoglycan glycosidic bonds in glycosidic bonded polysaccharides, and the phosphorus-containing groups in phospholipids, nucleic acids, and lipids constituting cellular components; the peak of 1800–1500−1 was the amide information region for proteins and polysaccharides, and the peak of 3000–2800−1 was the information region for fatty acids. The results showed that with the increased concentrations of essential oil, the absorption peaks of the above groups in S. aureus were significantly increased, indicating that the cell membrane, peptidoglycan, proteins, lipids, and other components in cell wall were destroyed, and the cell membrane may be dislocated, thereby resulting in the formation of holes in cell membrane, and the entry of some macromolecules into cells, and causing the leakage of cell contents in bacteria, as well as ultimately leading to cell death of bacteria.

Effects on bacterial cell membrane and substance leakage. (A)The effect of different T-4-O on bacterial biofilm; (B) Infrared spectroscopic analysis of T-4-O bacterial structure with different concentrations added; (C) The effect of adding different concentrations of T-4-O on bacterial cell lysates; (D) The effect of adding different concentrations of T-4-O on bacterial cell protein leakage. All values were expressed as mean ± standard deviation (n = 3). Means labeled with different letters (a-c) were significantly different by Waller-Duncan's test (P < 0.05) after one-way ANOVA.
Cell lysate leakage was assessed by measuring the OD260 values of S. aureus treated with T-4-O at varying concentrations for 0, 1, and 2 h. As shown in Figure 4c, higher concentrations of T-4-O led to a significant increase in OD260 values, indicating enhanced cell lysate leakage. A statistically significant difference (P < 0.05) was observed between the 2 MIC and 1 MIC groups after both 1 and 2 h of treatment, whereas no significant difference (P > 0.05) was detected when comparing the 3 MIC group with the other concentrations.
Intracellular protein leakage was evaluated by measuring the OD595 values of S. aureus after treatment with T-4-O at different concentrations for 0, 1, and 2 h. As shown in Figure 4d, the addition of T-4-O induced the release of proteins from cells, with OD595 values increasing with both time and T-4-O concentration compared to the control group. This indicates that T-4-O promotes protein leakage in S. aureus, and the effect became more pronounced at higher concentrations. However, while a significant difference (P < 0.05) was observed between the 2 MIC and 1 MIC groups across different time points, no significant difference (P > 0.05) was found when comparing the 3 MIC group with the other concentrations.
Measurement of Cell Cycle
In prokaryotic cells, the cell cycle of bacteria is divided into 3 phases, including I, R and D phases. The R phase, which is equivalent to the S phase of the cell cycle in eukaryotic cells, is the bacterial DNA replication phase. 18 The results of flow cytometry are shown in Figure 5a and Figure 5b, the number of normal S. aureus cells at the R phase was 15.1%, and after the action of T-4-O at the dose of 2 MIC, the number of cells entering the R-phase increased to 26.67%, and the number of S. aureus at the R-phase in the T-4-O group was extremely significantly elevated (P < 0.01). This suggests that T-4-O can bind to the DNA in S. aureus, thereby causing more bacteria to remain in the R phase and affecting the replication of DNA in S. aureus.

Effects of T-4-O on the cell cycle of Staphylococcus aureus. (A) Flow cytometry of Staphylococcus aureus, the blue population represents the early stage of DNA synthesis, the green population corresponds to the DNA replication period, and the red population indicates the late stage of DNA synthesis; (B) Number of R-phase cells of Staphylococcus aureus, all values were expressed as mean ± standard deviation (n = 3), means labeled with an asterisk (*) are statistically significant (P < 0.05) as determined by the Student's t-test compared to the control.
Transcriptome Sequencing Analysis
The obtained transcriptome data were transformed and processed and then screened according to the screening principle. As shown in Figure 6a, compared with the control group, a total of 4768 DEGs were screened in the T-4-O-treated group, of which 2362 genes were up-regulated, accounting for 49.54% of total DEGs; 2406 genes were down-regulated, accounting for 50.46% of total DEGs. The GO enrichment analysis was performed on DEGs and 30 GO terms with the most significant enrichment were plotted (Figure 6b), with the involvement of biological processes including carbohydrate transport and cellular protein metabolic process as the most enriched differential genes; in the category of cellular component, with cellular component, cell part and cell; in the category of molecular function, with substrate-specific transporter activity, substrate-specific transmembrane activity, and molecular function. The 20 KEGG metabolic pathways with the most significantly enriched DEGs were analyzed (Figure 6c). Among them, the enrichment of DEGs in pyrimidine metabolism, nitrogen metabolism, organelle membrane, integral component of membrane and intrinsic component of membrane was significantly different (P < 0.05), and these metabolic pathways were closely related to bacterial biofilm formation. 19 Collectively, these transcriptomic data indicate that T-4-O treatment caused extensive disturbances to the cell membrane system (Cellular component, Membrane) and transmembrane transport function (Transmembrane transporter activity) of S. aureus. Furthermore, the significant impact on fundamental metabolism, particularly the pathways of “Pyrimidine metabolism” and “Nitrogen metabolism” which are closely linked to nucleic acid synthesis, likely disrupted the supply of materials and energy required for normal bacterial growth and division.
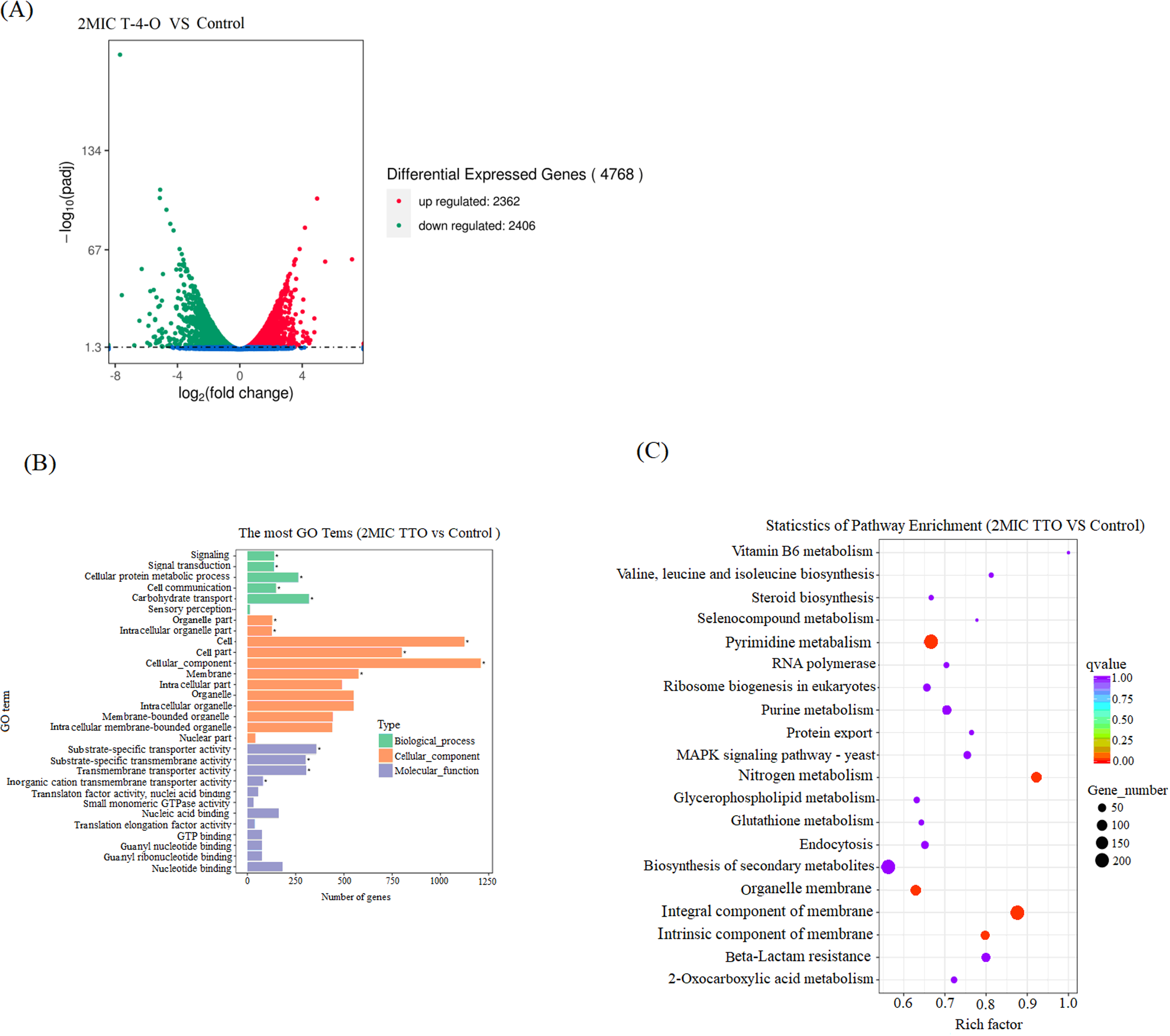
Volcano plot and GO and KEGG enrichment analysis of DEGs. (A) Volcano diagram; (B) GO; (C) KEGG.
Discussion
The antibacterial properties of plant essential oils, as valuable natural bioactive substances, have been widely documented.2,20 However, the variability in their botanical sources and chemical compositions leads to significant differences in their efficacy. Therefore, elucidating the specific antibacterial mechanisms of individual essential oils through compositional analysis is crucial for their development and application as natural therapeutic agents. In this study, we characterized the chemical profile of Tea Tree Oil (TTO) and identified 15 compounds, predominantly alcohols, alkenes, alkanes, and esters. Terpinen-4-ol (T-4-O) was the most abundant constituent, comprising 31.09% of the total. Notably, T-4-O, as a plant-derived compound, is frequently associated with a lower risk of inducing bacterial resistance, potentially due to its ability to disrupt multiple cellular targets (eg, membrane integrity, biofilm formation, and cell cycle progression) as observed in this study, and it has been reported to exhibit a favorable safety profile in certain applications, 21 highlighting its potential as an alternative to conventional antibiotics in specific contexts. Consequently, this study focused on T-4-O as a single constituent to enable a more precise and scientifically grounded investigation into the antibacterial mechanism of TTO's primary component.
Consistent with previous reports establishing T-4-O's activity against S. aureus,22,23 our study determined a MIC of 2.12 mg/mL, which aligns with the published range of 1.25–2.50 mg/mL. 24 We further demonstrated a concentration-dependent inhibition of S. aureus growth, with near-complete suppression achieved at 2× MIC. Our multi-faceted investigation provides a systematic elucidation of T-4-O's antibacterial mechanism. The primary action appears to be directed against the cell membrane structure. Transcriptome sequencing revealed that T-4-O treatment significantly enriched DEGs in pathways critical for biofilm formation and membrane integrity, including pyrimidine metabolism, nitrogen metabolism, organelle membrane, integral component of membrane, and intrinsic component of membrane (P < 0.05). These molecular findings are strongly corroborated by phenotypic evidence: Fourier-transform infrared spectroscopy (FTIR) indicated alterations in membrane composition, SEM visually confirmed membrane rupture and cellular collapse, and biochemical assays documented substantial leakage of cellular lysates and proteins. Collectively, these data suggest that the cell membrane is likely a principal target of T-4-O.
Furthermore, our results suggest a multi-faceted mechanism that extends beyond membrane disruption. The transcriptomic changes in “Pyrimidine metabolism” point to a potential disruption of nucleotide synthesis. This offers a plausible molecular explanation for the observed cell cycle arrest during the DNA replication (R) phase, as quantified by flow cytometry. The compromised membrane and cell wall integrity, caused by T-4-O, likely facilitates the leakage of essential intracellular components. This leakage, combined with the potential inhibition of DNA synthesis, ultimately disrupts normal cell division and growth, leading to bacterial death. Thus, T-4-O appears to exert its synergistic antibacterial effect by simultaneously compromising cell envelope integrity, interfering with fundamental metabolic pathways, and impeding the cell cycle.
When the T-4-O concentration was 2× MIC, it achieved approximately a 50% inhibition rate on S. aureus biofilm formation, indicating its effectiveness at this concentration. However, the inhibitory effect of T-4-O on the initial adhesion phase of biofilms at sub-MIC concentrations (eg, 0.5× MIC, 0.25× MIC) remains to be fully elucidated. Investigating this aspect would provide deeper insights into its early-stage anti-biofilm action and low-concentration efficacy, potentially informing new strategies for preventing biofilm-associated infections.
Finally, it is important to acknowledge the limitations of this study. Firstly, all experiments were conducted in vitro; therefore, the in vivo antibacterial efficacy and potential toxicity of T-4-O require further investigation. Secondly, the T-4-O used was a commercially purchased pure compound. The potential synergistic or antagonistic interactions between T-4-O and other components within the complete TTO mixture were not explored. Furthermore, while the transcriptomics analysis revealed significant changes in metabolic pathways, the precise causal relationships and underlying molecular interactions need validation through subsequent functional studies, such as gene knockout or overexpression. Addressing these limitations in future work will be essential for a more comprehensive understanding of T-4-O's antibacterial mechanisms and its full application potential.
Conclusion
GC-MS analysis indicates that TTO comprises a total of 15 compounds, including alcohols, alkenes, alkanes, and esters. Among these, T-4-O is the predominant component, accounting for 31.09% of the total composition. Additionally, T-4-O demonstrates a significant inhibitory effect on S. aureus, with an MIC of 2.12 mg/mL and an MBC of 8.47 mg/mL. When the concentration of T-4-O reaches 2 MIC, it effectively suppresses the growth of S. aureus and disrupts the metabolic pathways associated with bacterial biofilm formation, achieving an inhibitory rate of 50% for S. aureus biofilm formation. Furthermore, T-4-O can induce dislocation in the bacterial cell membrane, interfere with cell wall formation, and cause leakage of cellular lysates, proteins, and other substances. This leads to abnormal cell division and growth, maintaining the cell cycle at the R phase, and ultimately triggers cell membrane rupture and cell death, thereby exerting its bacteriostatic effect.
Footnotes
Acknowledgements
The authors would like to thankujian Zhongmu Bio-Pharmaceutical Co., Ltd for their valuable technical assistance and support in GC-MS analysis. We also appreciate the contributions of all the researchers and staff involved in this study.
Ethical Approval
Ethical Approval is not applicable for this article.
Author Contributions
Writing-original draft preparation B.-S.L. and H.-B.C.; data curation B.-S.L. and W.-J.W.; methodology W.-J.W. and C.-C.C.; writing-review and editing B.-S.L. and L.-X.Q. All authors have assented to the manuscript's published version.
Funding
This study was supported by the Fujian Provincial Department of Science and Technology University Industry University Research Cooperation Project under grant number2023N5009, the Doctoral Research Fund Project of Longyan College under grant number LB20202001, the Fujian Provincial Department of Science and Technology External Cooperation Project under grant number 2025I0029, the Industry University Research Technology Innovation Project in Xinluo District, Longyan City, Fujian Province under grant number 2022XLXYZ007,and the Natural Science Foundation of Fujian Provincial Department of Science and Technology under grant numbe2023J01993.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data referenced in the paper are all included in the paper.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
