Abstract
Objective
The polyol (sorbitol) pathway plays a central role in diabetic complications by promoting oxidative stress and the formation of advanced glycation end products (AGEs).
Method
CRRE was prepared using selective solvent partitioning and analyzed by LC-MS fingerprinting. Total phenolic content and Antioxidant activity were assessed using Folin Ciocalteu and β-carotene linoleic acid assay, respectively. Anti-glycation potential was evaluated in a Glucose-BSA (Bovine Serum Albumin) system. Molecular docking simulations were performed between identified key stilbenoids and the Human Aldose reductase enzyme (PDB ID: 1IEI), and their ADME was determined using SWISSADME.
Results
LC-MS analysis revealed the presence of piceatannol, resveratrol, scirpusin A, scirpusin B, and cyperusphenols A–D. CRRE showed high total phenolics (26.4 mg GAE/g), strong antioxidant inhibition (69.6%), and significant antiglycation activity (81.5%). Docking studies showed strong ARI binding for scirpusin B (−9.9 kcal/mol), scirpusin A (−8.8 kcal/mol), and piceatannol (−8.6 kcal/mol), comparable to epalrestat.
Conclusion
This study demonstrates that CRRE, rich in polyphenolic stilbenoids, exhibits promising antioxidant, antiglycation, and aldose reductase inhibitory activity, with favorable ADME profiles. Furthermore, CRRE, with its strong antiglycation and antioxidant properties, along with favorable pharmacokinetics, underscores its potential role in mitigating oxidative stress and diabetes-related complications.
Introduction
The growing interest in
Diabetes Mellitus, commonly referred to as Type 2 Diabetes, is a metabolic disorder characterized by chronic hyperglycemia, leading to dysregulation of glucose homeostasis. According to data from the CDC, approximately 38.4 million people in the United States—accounting for 11.4% of the population—are diagnosed with diabetes. Even more concerning is the prevalence of prediabetes, identified by elevated fasting glucose or A1C levels, with an estimated 97.6 million U.S. adults affected. 9
Diabetes is influenced by both modifiable (unhealthy diet, obesity, sedentary lifestyle, sleep disorders, and smoking) and non-modifiable risk factors (age, pre-existing medical conditions, ethnicity, and family history), which result in complications such as cardiovascular diseases, neuropathy, nephropathy, retinopathy, oxidative stress and even stroke.10,11 Several cohort studies have confirmed that diabetes is, in fact, a major risk factor for several cancers. 12
Under normal glycemic conditions, the glycolytic pathway is responsible for metabolizing approximately 80%–90% of the body's glucose. However, in hyperglycemic states, the glycolytic pathway becomes saturated, leading to the diversion of excess glucose into alternative metabolic pathways, such as the glycation pathway, hexosamine pathway, protein kinase C (PKC) pathway, and the polyol or sorbitol pathway. 13 The polyol pathway is a two-step process that converts glucose into sorbitol, which is subsequently converted to fructose. This pathway is catalyzed by two enzymes: aldose reductase, which reduces glucose to sorbitol, and sorbitol dehydrogenase, which converts sorbitol to fructose. Aldose reductase (AR) is the rate-limiting enzyme in this process. 13 AR belongs to the Aldo-keto reductase superfamily (Figure 1) and is found in most mammalian cells, though it has a higher distribution in specific tissues such as the eye lens, sciatic nerve, testes, and heart. 14 This pathway is critically involved in oxidative stress, tissue damage, cataract formation, inflammation, glycation, and various diabetic complications. 15 Research suggests that targeting aldose reductase (AR) may be a promising therapeutic strategy to prevent diabetic complications. Although several compounds have been investigated as potential aldose reductase inhibitors (ARIs), many have shown limited efficacy and bioavailability, with only a few, such as Epalrestat, successfully passing clinical trials. Therefore, there is a need for further exploration of potential ARIs with better safety and efficacy profiles. With the growing interest in natural products, there has been an increasing focus on phyto compounds like polyphenols for their potential therapeutic benefits in managing diabetes-related complications.

Crystal Structure of Human Aldose Reductase Enzyme (PDB ID 1IEI).
Nutraceuticals like polyphenols are now widely recognized for treating metabolic disorders like diabetes. Among the polyphenols, stilbene polyphenols are considered as biologically active compounds to ameliorate insulin resistance and hence play important role in managing diabetes. 16
This study aims to characterize the chemical composition of stilbenoids in the methanolic/ethyl acetate fraction of
To our knowledge, this is the first comprehensive study to combine LC–MS-based fingerprinting of stilbenoids in
2.1 Material
All solvents were purchased from Millipore-sigma (Merk. KGaA, Darmstadt, Germany), and the water used was from the Millipore Direct Q®3UV Filter system. The C18 column used in the study was Kinetex® (Phenomenex- Torrence, CA) with dimensions of 250 × 4.6 mm, 5 µ. Dry
2.2 Extract Preparation
LCMS Conditions
LCMS fingerprint was developed using Agilent 1200 (Agilent Technologies, Wald Bronn, Germany). The column used in the analysis was Kinetex® core-shell technology (250X4.6 mm, 5 µ). A photodiode array detector was used, and the sample was observed on multiple wavelengths, 280 nm, 220 nm, and 320 nm. The mobile phase included pump A (0.1% formic acid) and pump B (acetonitrile: water 98:2). A gradient program was followed to achieve maximum separation of components. The program was initiated with pump B concentration at 10%, which gradually changed to 40% in 20 min. In the next 20 min, it changed from 40% to 80%, reaching 92% in another 10 min. The column was washed for 5 min, and the pump B concentration returned to 10% in the next 5 min. The flow rate was maintained at 1 ml/min. ChemStation software version was C.01.10[314]. The mass spectrum was recorded by Agilent 6120 HPLC single Quad MS using ESI mode. The MS conditions were as follows: Drying gas flow was 12 ml/min, nebulizer pressure was 35 psig, drying gas temperature was 350 °C, and capillary voltage was kept at 4000 V for both positive and negative modes.
Antioxidant Activity and Phenolic Content
Total phenolic content (TPC) was measured using a slightly modified method by Ragazzi and Veronese. 17 100 mg CRRE was prepared previously and dissolved in 20 ml of methanol. In 1.0 ml of this extract, 1.0 ml of Folin's Reagent (1N) and 2.0 ml of Na2CO3 (20%) were added subsequently. The test mixture was then appropriately mixed using a sonicator and left at room temperature for 30 min, and the volume was maintained to 25 ml with methanol. The absorbance of the test mixture was measured at the λmax 725 nm on Shimadzu Cary 60 Spectrophotometer. Total phenolic content was expressed as gallic acid equivalent (GAE) mg/g on a dry weight basis. The quantity of phenolic content was calculated by comparison with the standard curve of gallic acid (Stock solution: 50 mg in 50 ml of methanol), drawn under identical experimental conditions.
The antioxidant activity (AOA) was measured using the auto-oxidation of β-carotene and linoleic acid as described by Emmons and Peterson. 18 100 mg of CRRE extract, prepared previously, was dissolved in 20 ml of methanol. 2.0 mg of Beta carotene was dissolved in 20 ml of chloroform. 3.0 ml of the chloroform solution was added to 40 mg of linoleic acid and 400 mg of Tween 40. Chloroform was removed from the mixture under reduced pressure. 100 ml of deionized water was further added to the mixture to obtain a stable emulsion. 3.0 ml aliquots of this emulsion were mixed with 40 microliters of previously prepared CRRE. The combined solution was incubated for 1 h at 50 °C in a water bath. The absorption of the mixture was recorded at 0 and 60 min of incubation at λmax of 470 nm. The antioxidant activity (AOA) was expressed as percent inhibition relative to control after 60 min. The absorption of the mixture was compared with the standard curve prepared with Butylated hydroxytoluene (BHT) over a concentration range of 0–1.4 µg in 40 µL of solution. BHT is generally used as a commercial antioxidant and a standard for measuring antioxidant activity.
Antiglycation Activity
Antiglycation activity for CRRE was assessed by monitoring the Glucose-BSA system using a Varian fluorescence spectrophotometer, as per the method described for measuring antiglycation activity in spice extracts. 19 100 mg of CRRE was weighed, and 1 ml of aq. ethanol was added. The resulting solution was sonicated for 1 h. at room temperature. The supernatant was collected, and the process was repeated 3 times. The pooled supernatant was dried under a vacuum and dissolved in 1 ml of phosphate buffer (pH 7.4). Separate solutions of BSA (10 mg/ml), Aminoguanidine (1 mol/L), and glucose (90 mg/ml) were prepared in phosphate buffer (pH 7.4) Test sample, positive control and blank samples were prepared with 1 ml of each of the solution of CRRE, Aminoguanidine (AG) and 1 ml phosphate buffer added in mixture of 1 ml of glucose and 1 ml of BSA in polypropylene test tube. The three solutions were incubated for 3 days (72 h) at 37 °C in a water bath. Advanced glycation end products were measured by measuring the excitation and emission intensity at 360 nm and 420 nm. Triplicate samples were used for measurement. Inhibition was calculated by the formula below
Inhibition (%) = 1- (FI of Extract/FI of blank)×100
Processing of Protein Target and Ligand
High resolution x-ray crystal structure of human AR (PDB ID 1IEI) was downloaded from a protein data bank with a resolution of 2.5 A. The downloaded structure was already complexed with Zenarestat (ARI). Since PDB ID: 1IEI already contains Zenarestat in its experimentally determined bound conformation, a separate re-docking validation was not performed. The protein was further processed on Biovia's Discovery Visualizer (ver v25.10), which included removing water molecules, removing Hetatm, and stabilizing the structure by adding polar hydrogen. The 3D structure of ligands (stilbenoids and Polyphenols) identified from LCMS of methanolic extract of
Molecular Docking
Molecular docking simulation was conducted using PyRx open-source software, including Open Babel and Auto Dock Vina program.
21
Previously prepared PDBQT files of both protein (PDB ID 1IEI) and ligands Piceatannol (
Center – X = -1.55; Y = 2.42; Z = 2.94
Dimensions (in Angstrom)- X = 48.31; Y = 55.14; Z = 59.14
The ligand binding site on Aldose Reductase protein was analyzed by Ligand Scout Ver 4.0. The binding pocket containing zenarestat (Aldose Reductase Inhibitor) comprised of the following residues – TYR 309, THR 113, LEU 300, PHE 122, VAL 47, TRP 111 (Figure 2).

Binding Pocket Residue in Aldose Reductase Protein (PDB ID: 1IEI) Containing Zenarestat.
Canonical SMILES (Simplified Molecular Input Line Entry System) of the ligands were obtained through the PubChem database, which was subsequently entered into the SWISSADME online tool to evaluate the pharmacokinetic properties, including absorption, distribution, metabolism, and excretion. The following parameters were considered for physicochemical properties – lipophilicity, represented as XLogP3, Molecular Weight (MW) in g/mol, polarity represented by topological polar surface area TPSA, solubility in Log S, saturation of molecule representing the fraction of carbon in Sp3 hybridization and no. of flexible bonds. We also created a BOILED-Egg (Brain Or Intestinal EstimateD permeation method) plot using lipophilicity and polarity of molecules using SWISSADME tools. BOILED-Egg plot is an intuitive model for simultaneous estimation of absorption of compound through blood brain barrier and Gastrointestinal pathway. 22
Statistical Analysis
Total phenolic content, antioxidant activity and antiglycation activity were determined in triplicates and results are expressed as mean ± standard deviation (SD).
Results
LCMS Analysis
The compounds in CRRE were identified based on their mass fragmentation and UV spectrum (Table 1). Mass spectrum of major compounds in CRRE is given in supplementary file (Figure 6-12). Mass error of identified compounds was less than 1 ppm. The HPLC Chromatogram and TIC & 3D plot are presented in Figure 3a, 3b & 3c.

(a) HPLC Chromatogram of CRRE at 320 nm Wavelength; (b) Total ion Chromatogram (TIC) of CRRE in Positive Mode; (c) 3D Plot of the CRRE.
The Mass Spectral Characteristic of CRRE Detected by LC- ESI- MS.

Piceatannol (3,4,3´´,5´-Tetrahydroxy-Trans-Stilbene)
Compound

(a) Structure of Piceatannol (Compound
Compound

(a) Structure of Resveratrol (Compound
pattern In -ESI showed ions m/z 185 (Figure 5b) and m/z 159 (Figure 5c), which were formed by breaking of phenol moiety and resorcinol moiety respectively of the parent structure. The observed fragmentation pattern matched the reported literature.23
Compound

Fragmentation Scheme of
Scirpusin B Compound

Fragmentation Scheme for Scirpusin B (Compound

(a), (b), (c), (d) and (e) - the major Fragments of Scirpusin b (Compound
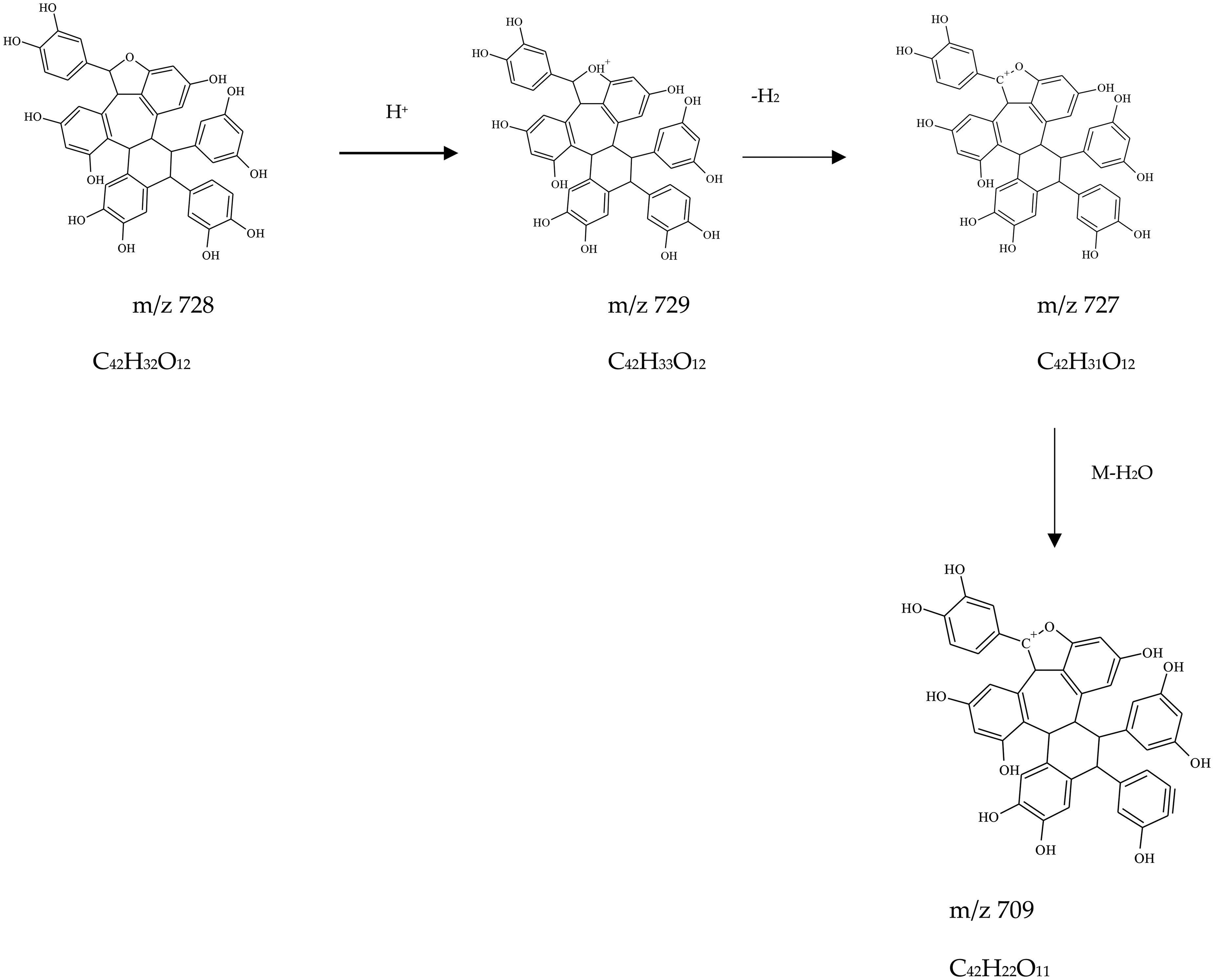
Compound 7 eluting at RT 17.49 min showed ions with m/z 727 in + ESI (Suppl Figure 8) and 725 in -ESI mode. The major fragment from compound

Fragmentation Scheme of Cyperus Phenol D (Compound

Structure of Fragment m/z 485 from Cyperus Phenol D (Compound
Compound

Fragmentation Scheme of Scirpusin A (Compound
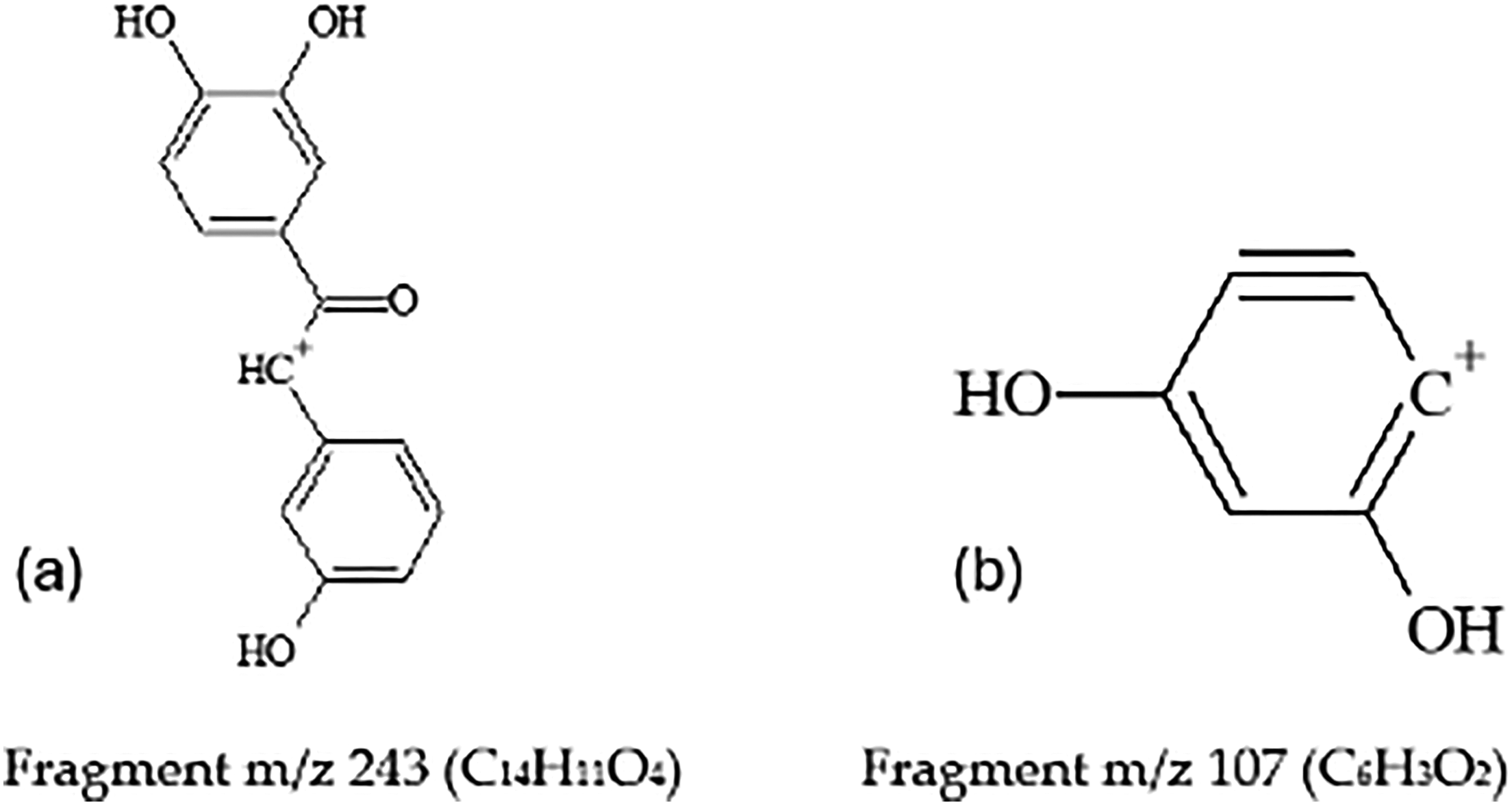
(a) and (b)- Major Fragments of Scirpusin A (Compound
Compound
Fragmentation Scheme of Cyperusphenol B (Compound

Fragment at m/z 619 from Cyperusphenol B (Compound
Compounds

Fragmentation Scheme for ion m/z 619.
The total phenolic content (TPC) of the CRRE was analyzed using Folin Ciocalteau's method. Reported TPC was expressed as gallic acid equivalent (GAE) mg/g on a dried basis after comparison with the calibration curve of gallic acid. The CRRE showed a TPC of 26.4 mg/g ± 0.29 GAE. This content includes the identified phenolic compounds identified by the LCMS analysis, as mentioned in the previous section. The total antioxidant activity of CRRE was calculated using protocol from Emmons and Peterson. 17 This method uses the autoxidation of beta carotene in the presence of linoleic acid, which acts as an oxidative agent. The method evaluates the capacity of test substance in preventing the oxidation of Beta carotene in the presence of linoleic acid. Stilbenoids, flavonoids present in the CRRE, such as resveratrol, piceatannol, Scirpusin a, b and cyperusphenols showed mild antioxidant activity, as evident from the 69.6% ± 3.3 inhibition of beta carotene auto-oxidation, which was lower than inhibition observed with BHT (89.0% ± 4.3 inhibition at the same concentration; p < .05).
Antiglycation Activity
Antiglycation activity was analyzed by monitoring the inhibition of the formation of Advanced glycation end products (AGEs) using the glucose-BSA system. Our results showed that CRRE showed excellent potential inhibiting the formation of AGEs (81.5% ± 4.7) in the system; AG, as a positive control, showed potent inhibition (94% ± 2.6; p < .05).
Molecular Docking
Molecular docking was carried out using PyRx. Table 2 summarizes the binding affinities as well as interaction with binding pocket residues for stilbenoid and phenolic compounds from CRRE, Epalrestat, and Beta glucogallin.
Binding Affinities and Interaction with major CRRE Compounds, β-Glucogallin and Epalrestat with Human Aldose Reductase 2 Enzyme (PDB ID: 1IEI).

Binding Affinities and Interaction with major CRRE Compounds, β-Glucogallin and Epalrestat with Human Aldose Reductase 2 Enzyme (PDB ID: 1IEI).

The strongest binding affinity was noticed with Scirpusin B −9.9 (
Molecular Docking poses and interaction with residues are given in Figure 16 (a-g).

Molecular Docking poses major CRRE compounds, β-glucogallin and Epalrestat
The ability of a drug molecule to penetrate membranes for transport throughout the body depends on certain physicochemical properties such as lipophilicity, solubility, size, and polarity of the molecule, as well as the flexibility of the molecule. For a drug-like property, lipophilicity represented as XLogP3 should be within −0.7 and +5.0, Molecular Weight (MW) range is 150 and 500 g/mol, Solubility in LogS <6, Saturation >0.25, Polarity in TPSA should be between 20 and 130A, and flexibility in a number of rotatable bonds <9 and Lipinski violation <0. The calculated physicochemical properties of the compounds are presented in Table 3.
Physicochemical Parameters of major CRRE Compounds, β-Glucogallin and Epalrestat.

Physicochemical Parameters of major CRRE Compounds, β-Glucogallin and Epalrestat

Based on the results obtained from SwissADME, most compounds exhibited optimal values of at least five parameters; however, both compounds

BOILED-Egg Plot of major Compounds in CRRE.
The antidiabetic activity was assessed using DPP-4, Lipase, and amylase enzyme inhibition. In another study,
In the present study, we characterized the stilbenoids in the CRRE through LC-MS. The study identified and characterized piceatannol, resveratrol, Scirpusin A, Scirpusin B, Cyperus phenol A,B,C,D and polyphenols such as p-coumaric acid, kaempferol & quercetin. In comparison to modern extraction techniques such as supercritical fluid extraction (SFE) and microwave-assisted extraction (MAE), the selective solvent partitioning method employed in this study offers a straightforward and cost-effective approach for enriching medium-polarity polyphenols like stilbenoids. While SFE and MAE provide enhanced extraction efficiency, reduced solvent consumption, and shorter processing times, they require specialized equipment and may not be accessible in all research settings. Similarly, advanced analytical methods such as UHPLC-MS/MS offer superior resolution and sensitivity, enabling more accurate identification and quantification of phytochemicals. Nonetheless, our combination of solvent partitioning with LC-MS and UV profiling provides a practical and reliable alternative for preliminary phytochemical characterization, particularly where resource constraints limit the use of more sophisticated technologies. This approach effectively balances selectivity, cost, and accessibility, facilitating the investigation of bioactive compounds in natural product research.
The selected stilbenoid compounds were studied for their ADME and potential for Aldose reductase inhibition using the in silico model. The study found the following order for strong ARI activity: Scirpusin B > Scirpusin A > Piceatannol > Resveratrol. The study compared these compounds against Epalrestat, an FDA-approved ARI, and β-glucogallin, a natural ARI from Indian gooseberry. Our results suggested the potential application of CRRE as an aldose reductase inhibitor. Further, we also studied the antioxidant and antiglycation activity; while the antioxidant activity was mild, the strong antiglycation activity exhibited the potential benefits of CRRE in inhibiting the formation of advanced glycation end products. Our study results also agreed with the antiglycation activity of
While the in silico docking simulations and ADME predictions offer valuable mechanistic insights, it is important to acknowledge the inherent limitations in directly extrapolating these results to in vivo pharmacological efficacy. Molecular docking provides a static snapshot of potential ligand-target interactions under idealized conditions, often neglecting dynamic biological variables such as metabolic biotransformation, protein binding, and efflux transporter activity. Furthermore, in vivo environments present complex systemic interactions, including enzymatic degradation, first-pass metabolism, and potential off-target effects that are not captured in computational models.
Thus, while our in-silico findings lay the groundwork for identifying potential aldose reductase inhibitors, they serve as hypothesis-generating tools rather than definitive evidence of in vivo efficacy. Future studies involving pharmacokinetic profiling, cellular assays, and animal models will be essential to validate the translational relevance of these computational predictions.
Although the current study is limited to in vitro assays and in silico docking, the observed biological activities—particularly strong aldose reductase inhibition and antiglycation effects—are consistent with pharmacodynamic mechanisms relevant to diabetic complications. Aldose reductase is a key enzyme in the polyol pathway, and its inhibition reduces intracellular sorbitol accumulation, thereby mitigating osmotic stress, oxidative damage, and formation of advanced glycation end products (AGEs). The high binding affinity of Scirpusin B, Scirpusin A, and piceatannol toward aldose reductase, as shown through docking simulations (ΔG: −9.9 to −8.6 kcal/mol), is comparable to the clinically approved inhibitor Epalrestat, suggesting a meaningful biological effect at the target site.
Moreover, the high antiglycation activity (81.5% inhibition in Glucose-BSA system) correlates with the mechanism by which polyphenolic stilbenoids may block Amadori product formation—an early pharmacodynamic event in glycation suppression. Since both aldose reductase activity and AGE formation contribute to vascular and neural complications of diabetes, the observed in vitro outcomes are indicative of a therapeutic PD effect. Although further pharmacokinetic and in-vivo evaluations are necessary, these findings provide a strong pharmacodynamic rationale for the potential use of CRRE in managing diabetic complications.
While our study explores potential ARI activity further animal and human studies are warranted. Another limitation of this study was that we only used selected stilbenoid compounds for docking purposes.
Conclusion
In this study, a total of 13 stilbenoid compounds were identified. A detailed LC-MS fingerprinting of the
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251409308 - Supplemental material for Phytochemical and Functional Insights into Cyperus rotundus Stilbenoids: LC-MS, Antioxidant, Antiglycation, and in Silico Assessment for Aldose Reductase
Inhibition Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X251409308 for Phytochemical and Functional Insights into
Footnotes
Acknowledgement
We acknowledge the receipt of raw material of
Informed Consent Statement
Not applicable.
Author Contributions
Conceptualization, Alpana Pande, Anurag Pande, T Senthil Kumar, S. Muthusamy.; methodology Alpana Pande; software, Anurag Pande.; validation Alpana Pande, formal analysis, Alpana Pande; investigation, Alpana Pande.; resources, Muhammed Majeed, Anju Majeed, Shaheen Majeed; data curation, T Senthil Kumar; writing—original draft preparation Alpana Pande, Anurag Pande.; writing-Review—Alpana Pande, Anurag Pande, Shaheen Majeed; visualization, Anurag Pande; supervision, Muhammed Majeed, Kalyanam Nagabhushanam, T Senthil Kumar; All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional Review Board Statement
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
