Abstract
Aldose reductase inhibitors are considered to be potential therapeutic agents for chronic diabetic complications. Diabetes mellitus can be accompanied by elevated blood levels of free fatty acids, which can cause lipotoxicity. Herbal extracts and their constituents are promising agents which have the potential for alleviating these complications. Our study was focused on the influence on these effects by flower infusions from Cornus mas L. and Cornus kousa F.Buerger ex Hance. Initially, phenolic compounds were quantified in the dried flowers. Next, we studied the ability of flower infusions from both plants to inhibit aldose reductase in vitro, the protective role in the cell model of lipotoxicity, and the cytotoxic action on fibroblast cell line NIH-3T3 by 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide assay. Both species are rich in phenolics; C. kousa flowers contain slightly higher amounts of phenolic acids (20.8%) and flavonoids (56.1%) than C. mas (20.2%) and (47.4%), respectively. Both extracts showed effective inhibition, expressed as half-maximal inhibitory concentration (IC50) (the concentration of inhibitor required to exhibit 50% inhibition of the enzyme reaction), of aldose reductase in non-toxic low concentrations (IC50 = 3.06 μg/mL for C. mas and IC50 = 2.49 μg/mL for C. kousa, respectively). In contrast, these concentrations of both extracts had almost no effects in the lipotoxicity cell model. To our knowledge, this study is the first report on C. mas and C. kousa flowers’ aldose reductase inhibitory activity and influence upon lipotoxicity.
The genus Cornus L., family Cornaceae, consists of 67 species 1 that are mostly shrubs or small trees, widely distributed in the northern hemisphere. Four of them, namely, Cornus mas L., C. officinalis Siebold & Zucc., C. controversa Hemsl., and C. kousa F.Buerger ex Hance. have edible fruits that are consumed in different parts of Europe and Asia. 2
The plant parts of C. mas, commonly known as cornelian cherry, have been used in ethnomedicine for the treatment of colds, flu, urinary inflammation, gastrointestinal disorders, stomach ulcers, colitis (fruits), and diabetes (leaves and seeds). 3 -5 This diverse range of ethnomedicinal utilization of C. mas plant parts was confirmed by several in vitro and in vivo preclinical studies in recent years. Fruits are the most studied. They showed antibacterial, cytotoxic, hypolipidemic, and antidiabetic activities. 6 -10 Leaves were studied for antiproliferative and immunomodulatory properties. 11,12
The biological activities of C. kousa (also called Kousa Dogwood or Japanese Dogwood) and Cornus sp. flowers have not been extensively studied until now.
Rat lens aldose reductase (RL-AKR) belongs to the wide aldo-keto reductase superfamily, which represents nicotinamide adenine dinucleotide phosphate (NAD(P)H) oxidoreductase enzymes that metabolize a broad spectrum of carbonyl-bearing endogenous compounds and xenobiotics. The human analog of RL-AKR is AKR1B1. 13,14 Aldose reductase is located in the lenses, in the retina, intestine, kidneys, and sexual organs and can reduce several carbonyls like steroid aldehydes, lipid peroxidation carbonyl products (4-hydroxynonenal), as well as glucose. Aldose reductase reduces glucose under hyperglycemic conditions to osmotically active sorbitol, which is accumulated intracellularly and can contribute to the development of chronic diabetic complications such as cataract, neuropathy, and nephropathy. 15,16 Several in vivo studies showed that aldose reductase inhibitors may reduce the development of these complications. 17 Water leaf extracts of C. mas and C. kousa showed effective inhibition of RL-AKR in vitro in low concentrations. 18
Decreased effectiveness of insulin leads to increased lipolysis. Higher serum levels of free fatty acids (FFA) commonly occurs in diabetes mellitus. 19 Under normal conditions, the plasma total FFA concentrations range from 200 to 600 µM. In pathological states such as diabetes, obesity, and cancer, the plasma total FFA levels range from 400 to 2000 µM. 20 -22 Cytoplasmic enrichment of FFA is associated with a deterioration of cellular function and an increased rate of apoptosis. 23 -25 One possible mechanism of apoptosis development could be oxidative stress induced by palmitic acid (PA) as well as other saturated FFA. 26,27 Thus, we shall also think about lipotoxicity.
In the current work, we have evaluated C. mas and C. kousa infusions—their constituents and biological activities of flowers with a focus on their influence on diabetes mellitus, including aldose reductase activity and lipotoxicity regulation.
The content of phenolic substances in infusions was determined using chromatographic methods as described further in the “Experimental” section. Summarization of the results is shown in Table 1.
Phenolics in the Dried Flowers of Cornus mas and Cornus kousa.

ND, not determined.
aMultipeak.
bQuercetin derivatives.
Based upon the data from liquid chromatography–mass spectrometry (MS) analysis, we can confirm that both samples are rich mainly in flavonoids and phenolic acids (major peaks representing total 77.1% in C. kousa, and 68.1% in C. mas flower infusions, respectively).
The inhibitors of aldose reductase, the first enzyme in the polyol pathway, are considered to be potential therapeutical agents in the development of chronic diabetic complications. The enzyme inhibition by the infusions was determined (Figure 1). IC50 values for both samples are similar and low. We previously observed comparable effective inhibition of aldose reductase by C. mas and C. kousa water leaf extracts and the well-known flavonoid quercetin (all in µg/mL range) as determined by the same method in the previous study. 18

Reducing activities of rat lens aldose reductase toward Cornus mas (a) and Cornus kousa (b) flower extracts. The data were shown in semilogarithmic plots. The values are expressed as the means ± standard deviation of 3 independent experiments.
According to the obtained results, we can confirm infusions of C. mas and C. kousa flowers as potent aldose reductase inhibitors with IC50 values of 3.06 ± 0.06 and 2.49 ± 0.07 µg/mL, respectively. Their activity nearly meets the IC50 values of the pure plant metabolites, for example, chlorogenic, gallic acids, and quercetin used as references for plant aldose reductase inhibitors. 28 -31 In our case, the above-mentioned acids and quercetin derivatives represent approximately 70% of the content.
Until today, only a limited number of cytotoxicity studies of Cornus flower extracts are available. We investigated the effects of infusions on cytotoxicity and proliferation on fibroblasts NIH-3T3 by 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay (Figure 2). The formation of formazan by reduction of the tetrazolium salt, MTT directly correlates with the number of viable cells containing active mitochondrial reductases. The IC50 values in the cell viability test are similar for both flower infusions. In our previous study, we determined cytotoxic activities of the water leaf extracts of C. mas and C. kousa. 18 The cytotoxicity on the NIH-3T3 fibroblasts of the leaf and flower extracts of C. mas is similar. In the case of C. kousa, the flower extract is more cytotoxic than that of the leaf extract (the IC50 is half as high).
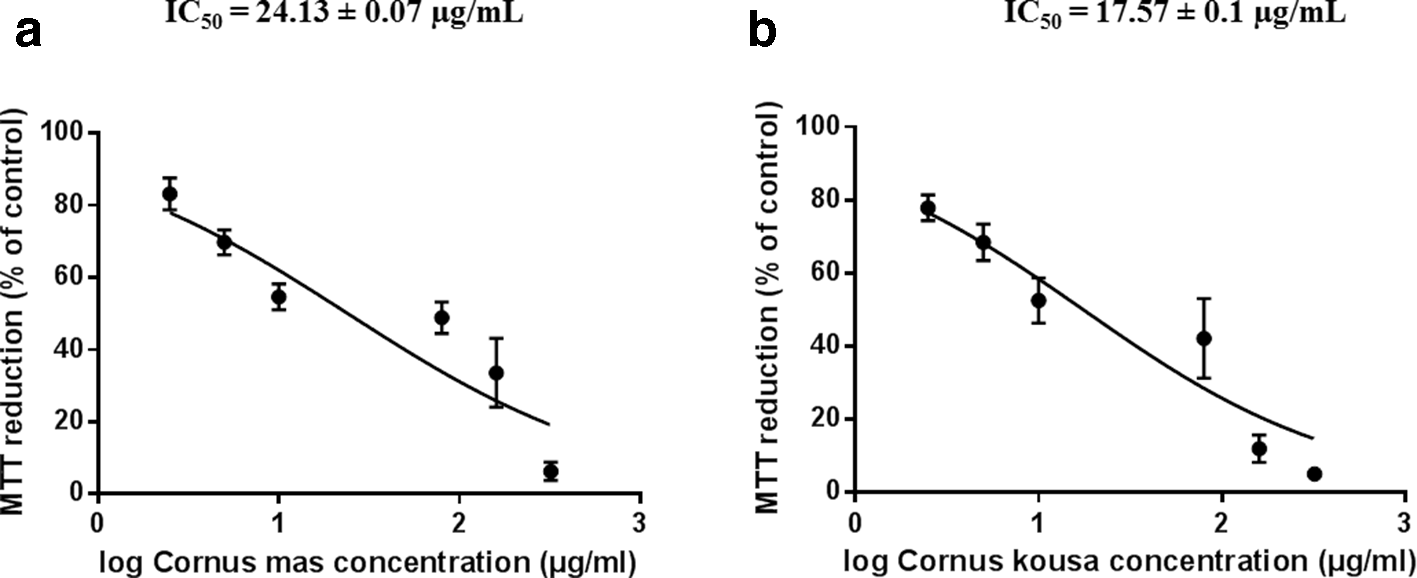
Comparison of viability (assessed by mitochondrial reduction of MTT) of 3T3 cells after 48 hours of incubation with Cornus mas (a) or Cornus kousa (b) flowers’ extracts. The data were shown in semilogarithmic plots. The values are expressed as the percentage of control (%) and are the means ± standard deviation of 3 independent experiments. MTT, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide.
Because diabetes mellitus is accompanied by an elevated blood level of FFAs causing tissue toxicity, we studied lipotoxicity. We examined whether phenolic-rich infusions of Cornus flowers and the well-known antioxidant quercetin played a potential protective role in the lipotoxicity cell model induced by a high level of PA coupled with the well-known carrier bovine serum albumin (BSA). Incubation with PA/BSA (0.15 mM/ 0.1%) for 24 hours caused a significant decrease in the fibroblasts’ viability (Figure 3). The assay was optimized in accordance with the published works.
32
-34
This combination of concentrations was selected because it caused almost a 50% decrease in the viability of the fibroblasts. Short pretreatment with either the extracts or quercetin did not cause significant changes in viability; thus, both samples probably did not play a protective role against lipotoxicity. For future experiments, other antioxidants or phenolic-rich samples could be studied for protection against lipotoxicity. In some previous studies, unsaturated fatty acids such as eicosapentaenoic, oleic, linoleic, and others,
32,33
as well as N-acetyl-

24 hours of incubation with PA/BSA (0.15 mM/0.1 %) caused significant decrease of the viability of 3T3 cells (assessed by mitochondrial reduction of MTT). Adding Cornus mas (“M”), Cornus kousa (“K”) flower extracts or quercetin (“Qc”) did not influence this effect significantly. The values are expressed as the percentage of control (%) and are the means ± standard deviation of 3 independent experiments. BSA, bovine serum albumin; MTT, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide; PA, palmitic acid.
The promising results show that C. mas and C. kousa flowers can act as effective aldose reductase inhibitors in the treatment of diabetes mellitus complications. Further detailed studies focused on other possible mechanism(s) of action and testing on an in vivo model will be desirable. In our previous work, we reported the aldose reductase inhibitory activity of leaf infusions of the above-mentioned species. As part of a continuing study aimed at diseases and complications connected with this important enzyme, we tested different plant parts. Based on available data, this is the first report on the influence of C. mas and C. kousa flower extracts on the tested enzyme activity and lipotoxicity.
Experimental
Chemicals
All the reagents—Dulbecco’s Modified Eagle’s Medium (DMEM), penicillin–streptomycin mixture, phosphate-buffered saline (PBS), fetal bovine serum (FBS), dimethyl sulfoxide (DMSO), PA, BSA, and MTT—were purchased from Merck (Merck Group, Germany).
Plant Material
Inflorescences of Cornus mas L. and C. kousa F.Buerger ex Hance (Cornaceae) were collected at the Tesárske Mlyňany Arboretum (Institute of Forest Ecology, Slovak Academy of Sciences, Slovakia) in March and May 2016. Botanical identification was performed by Ing. Peter Hoťka, PhD (Institute of Forest Ecology, Slovak Academy of Sciences, Slovakia). Voucher specimens have been deposited at the Department of Pharmacognosy and Botany (Faculty of Pharmacy, Comenius University in Bratislava, Slovakia) with accession numbers CM-2016, CK-2016.
Infusion Preparation
Harvested flowers were dried for 5 days at room temperature and milled in a laboratory mill. Particle size was adjusted using a No. 355 sieve (European Pharmacopoeia 9th Edition, “Particle-size distribution estimation by analytical sieving”). Water infusions were prepared according to the article “Decocta. Infusa.” in Pharmacopoeia Bohemoslovaca 4th Edition. In brief: flowers (10 g) were extracted in boiling water (100 mL) for 5 minutes and then the infusion was cooled at room temperature for 45 minutes, filtered through cotton wool and dried by freeze drying using a ScanVac CoolSafe 55-9 PRO system (LaboGene, Lynge, Denmark). The yields of individual extracts were 30% for C. mas (w/w) and 35% for C. kousa (w/w).
Extracts for HPLC Analysis Preparation
C. mas and C. kousa lyophilized flower infusions (4 mg) were redissolved in 1 mL of solvent prepared as follows: 0.2 mL of methanol was dissolved in 0.8 mL of water. Then 1 µL of this mixture was injected into the column and measured at 254 nm in electrospray ionization (ESI)-MS negative mode.
Phenolics Quantification and Identification by HPLC-diode array detector-MS
Compounds were quantified and identified using an Agilent 1260 high performance liquid chromatography (HPLC) system (Agilent Technologies, Inc., Santa Clara, CA, USA) coupled with an MS Instrument AB Sciex Triple Quad 3500 (AB Sciex Pte. Ltd., Woodlands, Singapore), with an ESI source. Separation was achieved with an Infinity Lab Poroshell 120EC-C18 column (4.6 × 100 mm, 2.7 µm particle size, Agilent). The column temperature was 30°C. The mobile phase composition: A (methanol with 0.1% formic acid and 1 mM ammonium formate) and B (water with 0.1% formic acid and 1 mM ammonium formate). Gradient conditions were as follows: 0 minutes 10% A, 36 minutes 100% A, 50 minutes 100% A, post-run time 16 minutes. The flow rate of the mobile phase was 0.3 mL/min.
The MS instrument was operated both in the positive and negative ion mode. ESI-MS conditions were: Scan type Q1 MS 50-1200 m/z, scan rate 2000 Da/s, CUR gas 25, temperature 450°C, gas 1 50, gas 2 40, ion spray voltage 4500 V.
Cell Culture
NIH-3T3 (mouse embryonic fibroblast cells) were obtained as a gift from Dr. Diana Vavrincová (Department of Pharmacology and Toxicology, Faculty of Pharmacy, Comenius University in Bratislava, Slovakia). Cells were grown at 37°C in a humidified atmosphere with 5% CO2 in DMEM supplemented with 10% FBS, 100 IU/mL penicillin and 100 µg/mL streptomycin in a BINDER BD 115 incubator (BINDER GmbH, Germany). Cells were subcultured twice a week.
Animals
The lenses, removed from the experimental rats at the Department of Pharmacology and Toxicology, Faculty of Pharmacy, Comenius University in Bratislava, Slovakia, were designated for other experiments. All experimental procedures involving the use of experimental animals were approved by the local Ethical Committee and the State Veterinary and Food Administration of the Slovak Republic. The investigation conforms to the Guide for the Care and Use of Laboratory Animals: 8th Edition (2010) published by the US Committee for the Update of the Guide for the Care and Use of Laboratory Animals; National Research Council; to the EU adopted Directive 2010/63/EU of the European Parliament and the Council on the protection of animals used for experimental and other scientific purposes.
Preparation of Purified RL-AKR
Aldose reductase from rat lenses (RL-AKR) was partially purified using a procedure adapted from Hayman and Kinoshita, 36 as described previously. 18,37
RL-AKR Enzyme Assay
The assay was adapted from previous work.
38
The assay mixture in the cuvette contained 4.5 mM
In Vitro Analysis of Cytotoxicity and Cell Proliferation
The effects of the infusions tested on the activity of mitochondrial dehydrogenases and proliferative functions of fibroblasts were assessed using the reduction of MTT. In short, cells were seeded (6500 cells/100 µL/well) in a 96-well plate (Sarstedt, Germany) in complete medium. After 24 hours, different concentrations of infusions, dissolved in complete medium, were added. The appropriate blanks were included in the experiment as well. Following 48 hours of incubation, an MTT solution was added to the wells (final concentration 0.4 mg/mL), except the blank, and after 4 hours of incubation, the medium was removed and 100% DMSO was added to lyse the cells. The absorbance was measured (λ = 570 nm) by an Infinite M200 plate reader (TECAN AG, Austria). Generated formazan amount (which corresponds to the number of viable and metabolically active cells) was calculated as a percentage of control cells, which was set to 100%.
Lipotoxicity Assessment
The effects of tested infusions on the viability and proliferation of the cells affected by the lipotoxicity induced by PA were assessed using the MTT test, as described above. The cells were seeded (8000 cells/100 µL/well) into a 96-well plate (Sarstedt, Germany) in complete medium. After 24 hours, the extracts were added to the cells. After 45 minutes, the mixture of PA and BSA (final concentrations 0.15 mM/0.1% w/V) was added. After 24 hours of incubation, the MTT test was performed. PA was dissolved in 0.1 mol/L NaOH at 70°C and then this solution was added to the BSA solution in the medium at 55°C. The assay was optimized as previously reported. 32 -34
Statistical Analysis
The data were visualized as means ± SD. Student’s t-test with equal variance was used for group comparisons. Statistical analyses were carried out using GraphPad Prism 7.0 software (San Diego, CA, USA).
Footnotes
Acknowledgments
Dr. Zuzana Kmecová and Dr. Eszter Bögy are acknowledged for their contributions and additional material. Mary Priestley (Curator of the Sewanee Herbarium) is acknowledged for reading of the article, corrections, and suggestions about grammatical English usage.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Science, Research and Sport of the Slovak Republic [grant numbers VEGA 1/0359/18, VEGA 1/0290/16, APVV-16-0207]; and by the Faculty of Pharmacy, Comenius University in Bratislava [grant number FaF UK/12/2019].
