Abstract
Objectives
This study aimed to optimize the extraction and purification processes of polyphenols from daylily (Hemerocallis citrina) to enhance the utilization of this plant.
Methods
Using response surface methodology (RSM), the effects of ethanol volume fraction, solid-to-solvent ratio, and ultrasonic power on polyphenol concentration were investigated. The impact of temperature, pH, and feed concentration on the adsorption rate was examined and optimized using an orthogonal experimental design. The phenolic compounds in the extract were qualitatively profiled using liquid chromatography–mass spectrometry (LC–MS). Finally, the antioxidant activity was evaluated by determining the DPPH radical scavenging rate.
Results
The optimal extraction conditions were as follows: an ethanol volume fraction of 70% (v/v), a solid-to-solvent ratio of 1:13 g/mL, and an ultrasonic power of 150 W. Under these conditions, the polyphenol concentration reached 0.769 milligram gallic acid equivalents per milliliter. The optimal adsorption conditions for the macroporous resin were 40 °C, pH 4.0, and a feed concentration of 1.54 mg/mL, which provided an adsorption rate of 12.06%. A total of 43 phenolic compounds were detected in the extract using LC–MS, among which rutin exhibited a relatively high abundance. At a concentration of 20 mg/mL, the DPPH radical scavenging rate of the extract was 42.47%.
Conclusion
These findings provide optimized process parameters for the utilization of daylily and could guide its further development.
Introduction
Daylily (Hemerocallis citrina), commonly known as the golden needle flower, is a perennial herbaceous plant belonging to the genus Hemerocallis, in the family Hemerocallidaceae. In China, the edible flower buds of this plant have long been consumed as a vegetable.1,2 Historical records indicate that daylily has been cultivated for both dietary and medicinal purposes for over a millennium.3,4 Although daylily contains a diverse range of bioactive constituents, it is particularly rich in sugars, polyphenols, and alkaloids. The contents of protein, crude fiber, flavonoids, and carotenoids in daylily have been reported to reach 12.95 g/100 g, 5.65 g/100 g, 0.35 g/100 g, and 0.16 g/100 g, respectively, 5 demonstrating that this plant is a valuable natural source of functional phytochemicals.
Among the major production regions in China, the Datong area of Shanxi Province is particularly renowned for its high-quality daylily, production owing to its volcanic soil and unique climatic conditions. Daylily from this region is characterized by thick petals and long stems, which provide distinct quality advantages. 6 However, despite the widespread cultivation and traditional significance of daylily, the industrial utilization of its bioactive compounds remains fairly limited. Daylily harvesting requires intensive manual labor, which increases production costs and hinders industrial competitiveness. Additionally, the extraction and high-value application of the active constituents of daylily are still in its infancy.
Polyphenols are important plant secondary metabolites and contain one or more aromatic rings bearing hydroxyl groups. These compounds can be categorized into major groups such as phenolic acids, flavonoids, and lignans. 7 Numerous studies have demonstrated that plant polyphenols possess significant antioxidant, anti-inflammatory, and anti-tumor properties.8–11 Thus, these compounds not only play protective roles in plant defense but also contribute substantially to human health upon consumption.
Recent studies have deepened our understanding of the chemical composition and functional potential of H. citrina. Through high-resolution ultra-high-performance liquid chromatography (UHPLC)-Orbitrap tandem mass spectrometry (MS/MS) profiling more than 70 phenolic constituents of daylily, predominantly flavonoids and phenolic acids, have been identified. These findings have highlighted the value of this species as a rich reservoir of radical-scavenging compounds. 12 Meanwhile, widely targeted UPLC-MS/MS metabolomics analysis of daylily produced in different Datong production areas have revealed region-specific metabolite patterns and identified biomarkers useful for quality assessment. 13 Furthermore, integrated metabolomic–transcriptomic studies have revealed developmentally regulated flavonoid biosynthesis mechanisms, showing that peak accumulation occurs during the initial flowering stage and supporting optimized harvest timing and post-harvest processing. 14 In parallel, evidence has shown that several newly isolated Hemerocallis phenolics demonstrate measurable free radical-scavenging activities, highlighting the continued discovery of potent antioxidant scaffolds within this genus. 15 Collectively, these advances indicate that the bioactive potential of daylily polyphenols remains to be fully leveraged.
A systematic investigation of the extraction and purification of polyphenols from daylily is crucial for developing natural daylily-derived antioxidants and enhancing the value of this traditional plant. Therefore, in this study, response surface methodology (RSM) was employed to optimize extraction conditions, enabling the simultaneous evaluation of factor interactions and efficient determination of optimal parameters with fewer experimental runs when compared to one-factor-at-a-time approaches. Through this strategy, extraction yield and reproducibility could be improved while reducing solvent use and time consumption. Macroporous adsorption resins were subsequently used for purification, leveraging their tunable polarity and high surface area to selectively enrich phenolics from complex plant matrices. This approach was scalable, regenerable, and compatible with food-grade solvents, facilitating industrial applications. Subsequently, the phenolic composition was characterized using liquid chromatography–mass spectrometry (LC-MS). Finally, the antioxidant activity of the extract was evaluated using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay. Overall, this study establishes an efficient and scalable workflow for the high-value recovery of polyphenols from daylily. The findings not only improve the scientific understanding of the functional properties of daylily but also provide a theoretical and technical foundation for its high-value utilization and industrial development.
Materials and Methods
Materials
Daylily samples were collected from a cultivation base in Datong, Shanxi Province; Gallic acid standard, sodium carbonate, and Folin-Ciocalteu reagent were purchased from Shanghai Yuanye Biotechnology Co., Ltd. Meanwhile, DPPH was obtained from Sigma Aldrich Co., Ltd, and AB-8, HPD-600, HPD-100, D101, NKA-9, and S-8 macroporous resins were purchased from Solarbio Science Technology Co., Ltd. Methanol and acetonitrile were of chromatography grade, while all other chemicals used in this study were of analytical grade.
Methods
Analysis of the Total Polyphenol Content
The content of total polyphenols was determined using the Folin-Ciocalteu reagent method. Gallic acid was used as the polyphenol standard to prepare a calibration curve. Each working solution (1.0 mL) was added to 5.0 mL of Folin–Ciocalteu reagent (10%). After mixing and 5 min of incubation, sodium carbonate solution (7.5%, 4.0 mL) was added. Following 60 min of incubation, the absorbance was measured at 765 nm using a TU-1810 UV/Visible Spectrophotometer. The results were expressed as gallic acid equivalents (GAE) per milliliter.
Optimization of Extraction Conditions Using Response Surface Method Experiments
The effects of extraction time (10, 20, 30, 40 min), the solid-to-solvent ratio (1:5, 1:10, 1:15, 1:20 g/mL), ethanol volume fraction (60%, 70%, 80%, 90%), extraction temperature (50, 60, 70, 80 °C), and ultrasonic power (100, 125, 150, 175 W) on polyphenol yield were evaluated. Ethanol extracts of dried daylily samples (0.5 g) were obtained at the specified solid-to-solvent ratios. Ultrasonic treatment was applied under the defined temperature and power conditions, and the extracts were subsequently filtered through a 0.45-µm membrane. The polyphenol concentration was determined as described above.
Based on the single-factor experiments, an RSM experiment was designed to identify the optimal extraction conditions. Three key variables, ie, the ethanol volume fraction, solid-to-solvent ratio, and ultrasonic power, were selected for further optimization. The experimental factors and levels are listed in Table 1.
Factors and Levels Used in the Response Surface Design for the Extraction Test.

Optimization of Static Adsorption on Macroporous Resins
Macroporous resins (AB-8, HPD-600, HPD-100, D101, NKA-9, and S-8) were each weighed (2.0 g) into 50 mL test tubes and mixed with 15 mL of the daylily polyphenol extract. Samples were incubated at 40 °C for 3 h under static adsorption conditions. The supernatant was collected, and its absorbance was measured to calculate the adsorption rate (%) according to Eq. (1). After adsorption, the resins were rinsed with distilled water and subsequently desorbed with 70% ethanol. The desorption rate (%) was calculated according to Eq. (2).

In Eqs. (1)–(2). C0, C1, and C2 denote the polyphenol concentrations in the supernatant before adsorption, after adsorption, and after desorption (mg/mL), respectively. Meanwhile, V1 and V2 denote the volumes of the adsorption and desorption solutions (mL), respectively.
Kinetic model fitting was performed according to Eqs. (3) and (4):

Here, t is the adsorption time (h); k1 is the first-order kinetic constant; k2 is the second-order kinetic constant; qe is the adsorption capacity at equilibrium time; and qt is the adsorption capacity at time t.
Optimization of Adsorption Conditions via Orthogonal Experiments
To determine the adsorption equilibrium time, 15 mL of the daylily polyphenol extract (adjusted to pH 4.0) was mixed with 2.0 g of the selected resin and incubated at 40 °C. Samples were collected every 30 min for absorbance measurement.
The polyphenol extract obtained under optimal extraction conditions was concentrated to final concentrations of 2.33, 1.54, and 1.02 mg/mL. The effect of AB-8 resin (2.0 g) was then evaluated under varying feed concentrations (2.33, 1.54, and 1.02 mg/mL), temperatures (30, 40, and 50 °C), and pH values (3.0, 4.0, and 5.0) to assess adsorption efficiency. Three factors namely, feed concentration, temperature, and pH were examined using an orthogonal experimental design. The experimental factors and corresponding levels are summarized in Table 2.
Factors and Levels Used in Orthogonal Experiments for Assessing Macroporous Resin Adsorption.

Detection of Polyphenols in Daylily Extract
Daylily extracts were mixed with two different volumes of a methanol/acetonitrile solution (1:1, v/v). The mixtures were subjected to ultrasonic extraction at low temperature for 30 min, followed by centrifugation (4 °C, 12000r, 10 min). The supernatants were collected and incubated for 1 h to precipitate proteins. After a second round of centrifugation (4 °C, 12000r, 10 min), the resulting supernatants were dried under vacuum and re-dissolved in 100 µL of 30% acetonitrile solution. The solutions were vortexed and centrifuged, and the final supernatant was subjected to analysis.
Chromatographic separation was performed using a UPLC system (Vanquish, Thermo Fisher Scientific, USA) coupled with a high-resolution mass spectrometer (Q Exactive HFX, Thermo Fisher Scientific, USA). Spectral acquisition was carried out on the Q Exactive HFX system equipped with an electrospray ionization (ESI) source. Instrument parameters were set as follows: sheath gas flow rate, 40 arb; auxiliary gas flow rate, 10 arb; ionization voltage, + 3000 V (positive mode) and −2800 V (negative mode); source temperature, 350°C; ion transfer tube temperature, 320°C; scan mode, Full MS-ddMS2; polarity mode, positive/negative; scan range, 70–1050 m/z; resolution: Full MS, 70,000; MS2, 17,500.
Raw data were processed using Progenesis QI software (Waters Corporation, Milford, USA). Data were analyzed by R package, where it was subjected to multivariate data analysis, including principal component analysis (PCA) and orthogonal partial least-squares discriminant analysis (OPLS-DA). The variable importance in the projection (VIP) value of each variable in the OPLS-DA model was calculated to indicate its contribution to the classification. Metabolites with the VIP value >1 were further applied to Student's t-test at univariate level to measure the significance of each metabolite, the p values less than 0.05 were considered as statistically significant.
Antioxidant Activity Determination
A 0.1 mmol/L DPPH radical solution was prepared. The daylily polyphenol extract was mixed with the DPPH solution and incubated at 37 °C for 1 h in the dark. Three experimental groups were established to determine the DPPH radical scavenging rate: a blank group (DPPH solution only), a test group (DPPH solution + sample solution), and a control group (sample solution only). Following incubation, the absorbance was measured at 517 nm, and the DPPH radical scavenging rate was calculated. The IC50 value (the concentration required to achieve 50% DPPH scavenging) was calculated using SPSS software (version 27.0, IBM Corp., Armonk, NY, USA). Regression and probit analyses were applied to fit the data and estimate the IC50 value with corresponding confidence intervals.
Statistical Analysis
All statistical analyses were performed using SPSS Statistics 27.0 (IBM Corp., Armonk, NY, USA). One-way analysis of variance (ANOVA) followed by Tukey's post hoc test (for parametric data) or Dunn's post hoc test (for non-parametric data) was used to compare different extraction parameters (eg, solvent type, extraction time, and temperature). RSM was applied to optimize extraction conditions, and the interaction between variables was assessed using regression analysis. Optimal conditions were predicted based on the model's coefficient of determination (R²). ANOVA was also conducted to optimize adsorption conditions for the macroporous resin. Factors such as resin type, pH, and adsorption time were analyzed using one-way ANOVA to identify the conditions that significantly affect adsorption efficiency. Post hoc analyses were conducted using Tukey's HSD test for pairwise comparisons. A P value < 0.05 was considered statistically significant for all tests.
Results
Gallic Acid Calibration Curve
A calibration curve was established by plotting the concentration of gallic acid versus the absorbance at 765 nm (Fig. 1). The regression equation was y = 0.0087x + 0.0211, with the coefficient of determination (R2) being 0.9996, indicating excellent linearity within the tested range.
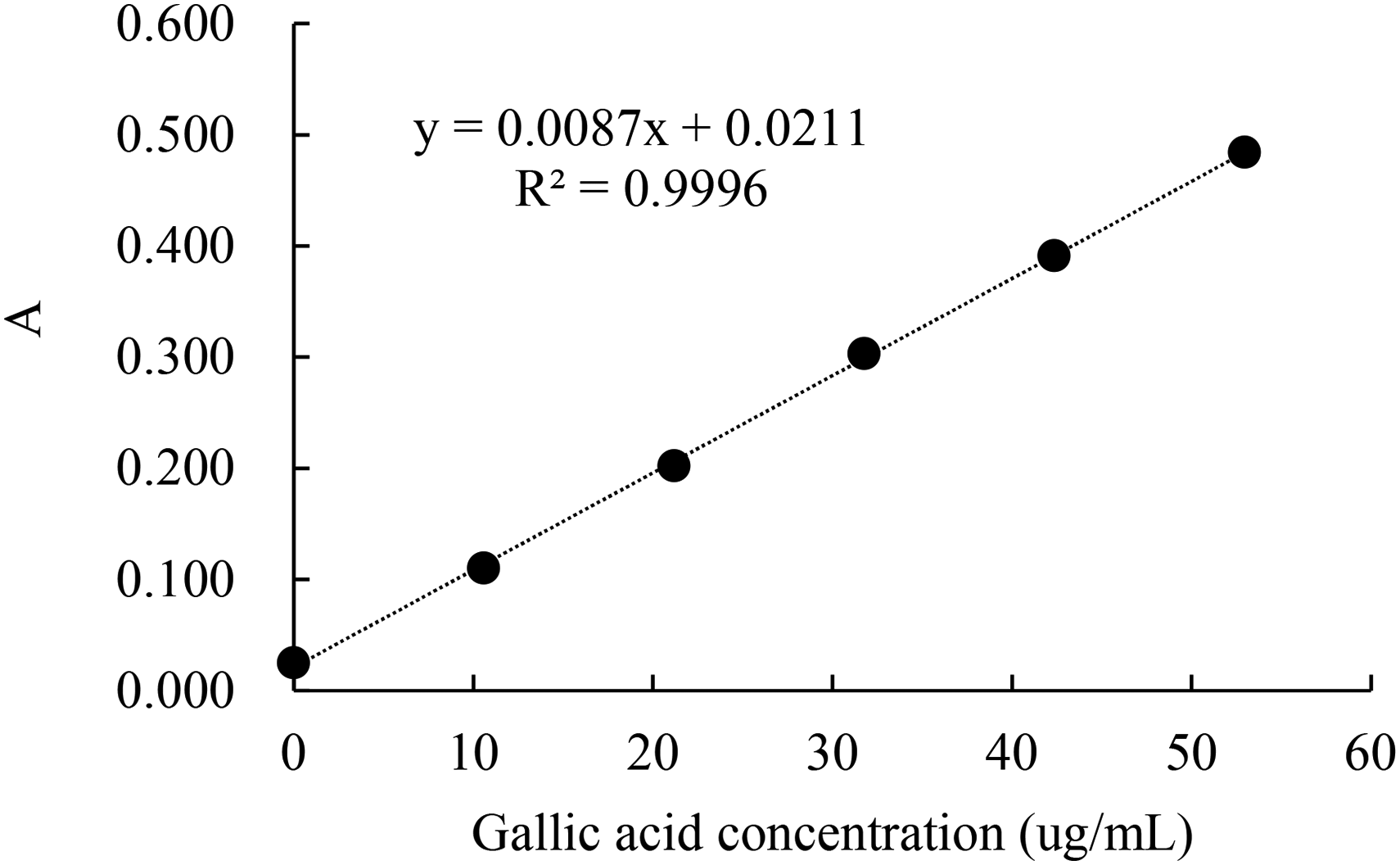
Calibration curve of the gallic acid standards.
RSM Results for Polyphenol Extraction from Daylily
The extraction efficiency of polyphenols is affected by solvent concentration, temperature, extraction time, and the solid-to-solvent ratio. Previous studies have reported that the yield of polyphenols increases with the ethanol volume fraction in the range of 50%–70%. However, the yield decreases beyond this range because the enhanced polarity reduces polyphenol solubility. Moreover, temperature influences extraction efficiency by improving molecular diffusion and solubility, although excessive heating may lead to polyphenol degradation. Similarly, extraction efficiency does not significantly improve beyond 30 min of ultrasonic treatment. 16 In the extraction experiments conducted in this study, the ethanol volume fraction and the solid-to-solvent ratio were found to significantly influence polyphenol recovery. Notably, the ethanol volume fraction directly affects solubility and diffusion. Indeed, evidence shows that an appropriate ethanol content optimizes solvent polarity and permeability, facilitating cell wall disruption and the release of target compounds. 17 Meanwhile, the solid-to-solvent ratio governs mass transfer and concentration gradients with a suitable ratio increasing the solvent–substrate contact area and improving extraction efficiency. 18 Thus, based on single-factor experiments, the ethanol volume fraction, solid-to-solvent ratio, and ultrasonic power were selected for the subsequent response surface design experiments. The specific experimental results are presented in Table 3.
Results of the Response Surface Test for Polyphenol Extraction.

In this study, RSM was applied to optimize the extraction conditions for daylily polyphenols. ANOVA and regression analysis confirmed the significance and reliability of the models. The resulting regression equation was as follows: Y = −11.62712 + 0.173710A + 0.012910B + 0.082409C − 0.000127AB − 0.000065AC − 0.000066BC − 0.001176A2 + 0.000761B2 − 0.000256C2, with R2 = 0.9852.
As shown in Table 4, the model was highly significant (P < 0.0001) with an F value of 51.62. The lack of fit was not significant (P > 0.05), indicating a satisfactory fit. In this model, the factors A and B exerted significant main effects on the polyphenol content, and the quadratic terms A2 and C2 were also significant. In contrast, the interaction terms AB, AC, and BC were not significant. Based on F values, the relative influence of the different factors on polyphenol content was found to be as follows: B (solid-to-solvent ratio) > A (ethanol volume fraction) > C (ultrasonic power).
Variance Analysis of Results Derived from Response Surface Experiments.

Note: “*” indicates significant, P < 0.05; “**” indicates highly significant, P < 0.01.
The response surface plots and contour maps illustrating the interactions among the experimental factors are presented in Fig. 2. All response surfaces exhibited convex downward trends, indicating the existence of a maximum polyphenols yield. The data shown in Fig. 2, combined with the ANOVA results, confirmed the consistency between graphical and statistical interpretations, with the interaction between ethanol volume fraction and ultrasonic power appearing more pronounced.

Response surface and contour plots illustrating the interaction effects of extraction parameters.
Overall, response surface analysis indicated that the optimal process parameters for extracting polyphenols from daylily were as follows: an ethanol volume fraction of 67.875%, a solid-to-solvent ratio of 1:12.78 g/mL, and an ultrasonic power of 149.26 W. Under these conditions, the predicted polyphenol content was 0.764 mg GAE/mL. Considering practical constraints, the experimental conditions were set as follows: ethanol volume fraction, 70%; solid-to-solvent ratio, 1:13 g/mL; and ultrasonic power, 150 W. Three parallel experiments were conducted using these parameters (Table 5). The average polyphenol content of the daylily extract obtained under these conditions was 0.769 mg GAE/mL, largely consistent with the predicted value.
Polyphenol Concentration Under the Optimized Extraction Conditions.

Static Adsorption on Macroporous Resins
Adsorption–desorption experiments were also carried out using six different macroporous resins (Table 6). Phenolic compounds generally show intermediate molecular weights (200-1000 Da), and factors such as specific surface area and pore size thus need to be considered for resin selection. In addition, resin polarity also plays a key role such that resins with polarities closer to those of polyphenols show stronger adsorption capacities.19,20 As demonstrated in Table 6, in this study, AB-8 and HPD-600 displayed higher adsorption rates for daylily polyphenols, while AB-8 and D101 exhibited higher desorption rates. Considering both adsorption and desorption performance, AB-8 was selected for subsequent experiments.
Adsorption and Desorption Rates of Polyphenols on Different Types of Macroporous Resin.

Determination of Static Adsorption Time
As shown in Fig. 3, the slope of the adsorption curve was relatively steep within the first hour, indicating a large concentration gradient between the resin and the extract as well as a rapid adsorption rate. Between 1 and 2 h, the slope decreased, and the adsorption rate increased more slowly. After 2 h, the curve approached equilibrium as a balance was reached between adsorption and desorption. Based on the adsorption profile, the static adsorption time was determined to be 3 h.

Time course of the static adsorption of daylily polyphenols.
The adsorption kinetics data were fitted using a first-order kinetic model and a second-order kinetic model, respectively (Table 7). The pseudo-second-order (PSO) model provided a superior description of the static adsorption behavior of the macroporous resin, yielding a higher correlation coefficient (R2 = 0.9923). Moreover, the equilibrium adsorption capacity predicted by the PSO model was closer to the experimental value, while the pseudo-first-order (PFO) model tended to underestimate the equilibrium capacity. These findings suggested that the adsorption rate was predominantly influenced by processes associated with the occupation of active sites or electron exchange interactions on the resin's surface.
Parameters of Adsorption Kinetics.

Orthogonal Test Results for the Adsorption of Daylily Polyphenols on Macroporous Resin
Table 8 shows the orthogonal test results for the adsorption of daylily polyphenols on macroporous resin. Notably, concentration was found to have the most significant effect on the adsorption rate. Higher concentrations accelerated the transfer of polyphenols from the solution to the resin's surface. However, at excessively high concentrations, the unadsorbed polyphenols caused resin blockage, thereby reducing adsorption efficiency. 21 The pH value was the second most influential factor, affecting both the affinity of the resin and the ionization state of the solute, thus indirectly impacting adsorption. As acidic solutes, polyphenols largely remain in non-ionized forms under low pH conditions, leading to enhanced hydrophobic interactions and better adsorption onto AB-8 resin. 22 Finally, temperature exerted the least influence. This was expected, since resin adsorption primarily relies on van der Waals and hydrophobic forces, which weaken at higher temperatures, thus facilitating desorption. 23
Orthogonal Test Data for Polyphenol Adsorption on Macroporous Resin.

In conclusion, the optimal adsorption conditions for daylily polyphenols were determined to be as follows: temperature, 40 °C; pH value, 4; and concentration, 1.54 mg/mL. Validation experiments conducted under these conditions yielded an adsorption rate of 12.06%.
Detection of Phenolic Compounds
Phenolic compounds are bioactive substances that are widely distributed in plants. These compounds are mainly classified as flavonoids, tannins, and phenolic acids. To further characterize the phenolic compounds present in the daylily extract, LC–MS analysis was performed in this study. The LC–MS diagram (Fig. 4) revealed the presence of several prominent peaks corresponding to typical phenolic constituents in the daylily extract. Moreover, a total of 43 phenolic compounds were identified in the crude extract of daylily polyphenols (Table 9).

LC–MS diagram.
Analysis of Variance of Orthogonal Test Data.

Note: “*” indicates significant, P < 0.05; “**” indicates highly significant, P < 0.01.
Antioxidant Activity Analysis
To further validate the antioxidant capacity of the samples, antioxidant assays were conducted (Fig. 5). In these assays, vitamin C (VC, 10 mg/mL), a well-known antioxidant, served as the positive control. Overall, the DPPH radical scavenging assay revealed that the daylily polyphenol extracts possessed notable dose-dependent antioxidant capacity. At a concentration of 20 mg/mL, the scavenging rate reached 42.47%. The IC50 value for the DPPH radical scavenging capacity of daylily polyphenols was calculated to be 25.62 mg/mL, indicating a moderate antioxidant capacity (Table 10).

DPPH free radical clearance rate of daylily polyphenols.
Phenolic Compounds Detected in the Daylily Extract.

Discussion
In this study, an efficient workflow was established for recovering polyphenols from daylily. The optimization of the extraction process resulted in satisfactory polyphenol recovery, with LC–MS profiling identifying 43 phenolic compounds in the crude extract. Notably, rutin emerged as one of the most abundant constituents of the daylily polyphenol extract. Furthermore, the extract exhibited notable DPPH free radical-scavenging activity, which reflected its antioxidant potential and functional significance.
However, the study has some limitations, which should be acknowledged. Notably, the adsorption rate observed in our adsorption experiments was relatively low. One plausible explanation is that the high amount of ethanol (a polar solvent) in the mixture favored the retention of polyphenols in solution and hindered their adsorption. 24 Moreover, dried daylily is known to contain up to ∼63% total sugars. It is possible that the carbohydrates and proteins in the crude extract competed with polyphenols for active sites on the resin, thereby decreasing adsorption efficiency. Additionally, the LC–MS analysis conducted in this study was primarily qualitative, and comprehensive quantification was not performed. Future studies should thus focus on desugaring pretreatments and quantitative profiling to establish the link between polyphenol composition and functional potential.
Conclusion
The optimal extraction conditions for daylily polyphenols identified in this study were as follows: ethanol volume fraction of 70%, solid-to-solvent ratio of 1:13 g/mL, and ultrasonic power of 150 W. These conditions yielded a validated polyphenol concentration of 0.769 mg GAE/mL. Subsequently, adsorption on macroporous resin was optimized. AB-8 was selected as the adsorbent, and the effects of temperature, concentration, and pH were evaluated under static conditions, followed by orthogonal design experiments. The optimal adsorption conditions included a temperature of 40 °C, pH of 4.0, and concentration of 1.54 mg/mL, providing a validated adsorption rate of 12.06%. LC–MS analysis of the crude extract enabled the identification of 43 phenolic compounds, among which rutin displayed a relatively high abundance. Collectively, the findings of this study highlight the value of daylily as a source of bioactive polyphenols and provide a process-based foundation that can be refined for the industrial translation of daylily polyphenols. These advances could accelerate the value-added utilization of daylily and inform quality control strategies for functional ingredient development.
Footnotes
Acknowledgements
The authors would like to thank Xiaojie Nan, Min Zhu, and Weiwei Guo for providing the necessary equipment and research facilities. The authors are also grateful to Cheng Zhang for his assistance with manuscript proofreading.
Ethical Approval
No ethics approval was required for this study.
Consent to Participate
This study did not involve any human subjects, and informed consent requirements were thus not applicable.
Consent for Publication
Not applicable. This study does not contain any individual person's data or identifiable images.
Author Contributions
G.A. conducted the experimental work and prepared the initial draft of the manuscript. Y.Z. performed data analysis and contributed to manuscript revision and improvement. S.G. supervised the study and provided the necessary experimental resources and guidance.
Equal Contribution: Y.Z. and G.A. contributed equally to this work.
Corresponding author: S.G.
Funding
This work was supported by the School Science and Technology Innovation Project (grant number CXGC202410) and the project “Research and application demonstration of key technologies in the daylily vegetable industry” (grant number 2021YFD1600301).
Declaration of Conflicting Interests
The authors declare that there are no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
This study did not involve any experiments on human or animal subjects.
