Abstract
Objectives
This study aimed to evaluate the safety of daily use and skin viscoelasticity in vivo and under long-term conditions (14 and 28 days) of a cosmetic cream containing an extract of dragon's blood (Croton lechleri Muell) and vegetable oils from aguaje (Mauritia flexuosa Lf.), goldenberry (Physalis peruviana L.), super sacha inchi (Plukenetia huayllabambana), and sacha inchi (Plukenetia volubilis), all of which are of Peruvian origin.
Methods
Dermatological safety and viscoelasticity tests were conducted using the International Contact Dermatitis Research Group evaluation scale for measuring reactions and the Cutometer® MPA580, respectively.
Results
The results reported that the cosmetic cream in the safety evaluation did not induce any significant skin sensitisation or irritant phenomena; and in the long-term viscoelastic evaluation significant increases were achieved with respect to the baseline control (p = 0.000): the indices of firmness, viscoelasticity during the suction phase and total recovery, obtained average improvements of 29.5%; 39.0%; 37.7%, respectively.
Conclusion
The cosmetic cream is dermatologically safe and demonstrates a sustainable improvement effect on the viscoelasticity parameter and increases firmness gradually after 14 and 28 days of daily application.
Introduction
Intrinsic aging leads to a reduction and degeneration of collagen and elastin proteins, progressively decreasing elasticity (Ue). Viscoelasticity (Uv) increases because of fragmentation and increased intermolecular bonds, whereas the viscosity of the interstitial fluid decreases because of reduced glycosaminoglycan and soluble collagen content. 1 The viscoelastic properties of the skin protect against injury. However, this protective function decreases because of intrinsic aging and photoaging. 2
Skin elasticity and firmness can be measured using Cutometer® MPA580, a high-tech noninvasive skin bioengineering device from Courage + Khazaka Electronics, Cologne, Germany. The values obtained from in vivo measurements indicate the physiological state of the skin. 3 This device creates negative pressure in the area under study for a specific duration (suction). Subsequently, the pressure is removed, and the skin can return to its initial position (relaxation).4,5
Bioactive compounds, such as fatty acids and polyphenols, are found in the seeds, fruits, and extracts of various plant species, including those found in Peru. In terms of chemical structure, the hydrophobicity of the acyl chain allows fatty acids to spontaneously and easily penetrate the stratum corneum up to the epidermal–dermal junction, thus contributing to skin permeability. 6 The antioxidant activity of vegetable oils may increase in direct proportion to its bioactive compound levels. 7 In catechins, because of the presence of 3- and 5-OH groups with 4-oxo function in A and C rings, they have good free radical scavenging and inhibitory effects on proteinases that degrade extracellular matrix.8,9
Interest in creating cosmetic products with natural active ingredients has grown considerably, leading to increased scientific studies on promising vegetable oils with different levels of monounsaturated and polyunsaturated fatty acids. 10 In addition, there is an increasing demand for plant extracts for cosmetic application worldwide. 11 Cosmetic formulations containing unsaturated fatty acids, such as oleic and linoleic acids, are noted for their emollient and occlusive properties, preventing the loss of transepidermal water and increasing skin hydration. 12 A deficiency of these fatty acids may induce the expression of keratins K6, K16, and K17, which lead to hyperproliferation and innate immune activation of keratinocytes, altering the epidermis.13,14
Peru is a source of unique botanical ingredients with dermocosmetic potential. Andean Skin Care (ASC) Cream is a topical cosmetic product in the form of an oil-in-water emulsion. Its oily phase comprises natural oils of Mauritia flexuosa Lf., Physalis peruviana L., Plukenetia huayllabambana, and Plukenetia volubilis. Its aqueous phase contains a resin extract of Croton lechleri Muell, which significantly increases hydration and maintains skin pH, as reported in a previous study consisting of in vivo instrumental efficacy tests. 15
Other phytochemical characterization studies have reported unsaturated fatty acids in these vegetable oils. For example, P. volubilis seed oil contains 44% linolenic acid, 33.5% linoleic acid, and 10.7% oleic acid, 16 and P. huayllabambana contains 9.33%, 28.09%, and 54% oleic, linoleic, and linolenic fatty acids, respectively. 17 Mauritia flexuosa Lf. pulp oil contains approximately 74.73% oleic acid and smaller quantities of linoleic and linolenic acids, with 1.92% and 1.19%, respectively.18,19 Furthermore, Physalis peruviana L. seed oil contains 11.9% oleic acid, 76.1% linoleic acid, and 0.02% linolenic acid.20,21 Croton lechleri Muell latex is rich in flavanols and diverse catechins, known for their strong free radical inhibition.22,23 Proanthocyanidins, catechins, epicatechins, gallocatechins, and epigallocatechins have been isolated from this latex and identified as its main constituents. 24
Regarding the dermatological safety of cosmetics, products, such as creams, serums, masks, lipsticks, scrubs, and conditioners, that include different concentrations of vegetable oils have been reported to be safe in skin irritation and sensitization tests conducted in humans, highlighting the importance of clinical studies. 25 Although oils appear safe when used topically, they can cause redness, burning, or allergic reactions. 12 However, some cosmetics containing oils or extracts have potential benefits in the treatment of itching. 13 Therefore, the safety of cosmetic formulations should be evaluated under dermatological supervision.
For instance, one study evaluated the irritation potential of P. volubilis oil applied every 12 h to the lower legs over 14 days. The assessments conducted after 7 and 14 days revealed no adverse effects, such as signs of erythema, desquamation, or edema, at the application site. Therefore, it was considered safe and beneficial for dry skin. The study also suggested that further studies should be conducted with larger sample sizes over longer periods for further confirmation of its safety and beneficial effects. 26 Considering that ASC Cream incorporates bioactive compounds from one extract and four vegetable oils, we performed a safety assessment for its use under the supervision of a dermatologist. Furthermore, dermatological data on the tolerability of these oils in topical formulations remain limited.
Studies have recommended the daily application of cosmetic moisturizers and hydrating agents to help prevent or provide timely treatment for skin conditions, such as diabetic foot syndrome, ichthyosis, xerosis, and keratosis. These products can relieve the symptoms of itching, erythema, and cracking.27,28
Considering the aforementioned points, no firmness or viscoelasticity studies of a cosmetic formula containing a mixture of an extract and four vegetable oils have been conducted to date. Therefore, this study aimed to determine the dermatological safety of ASC Cream and its positive effect on skin mechanical properties of elasticity and firmness, parameters after continuous use.
Materials and Methods
Formulation and Development of the Cosmetic Product
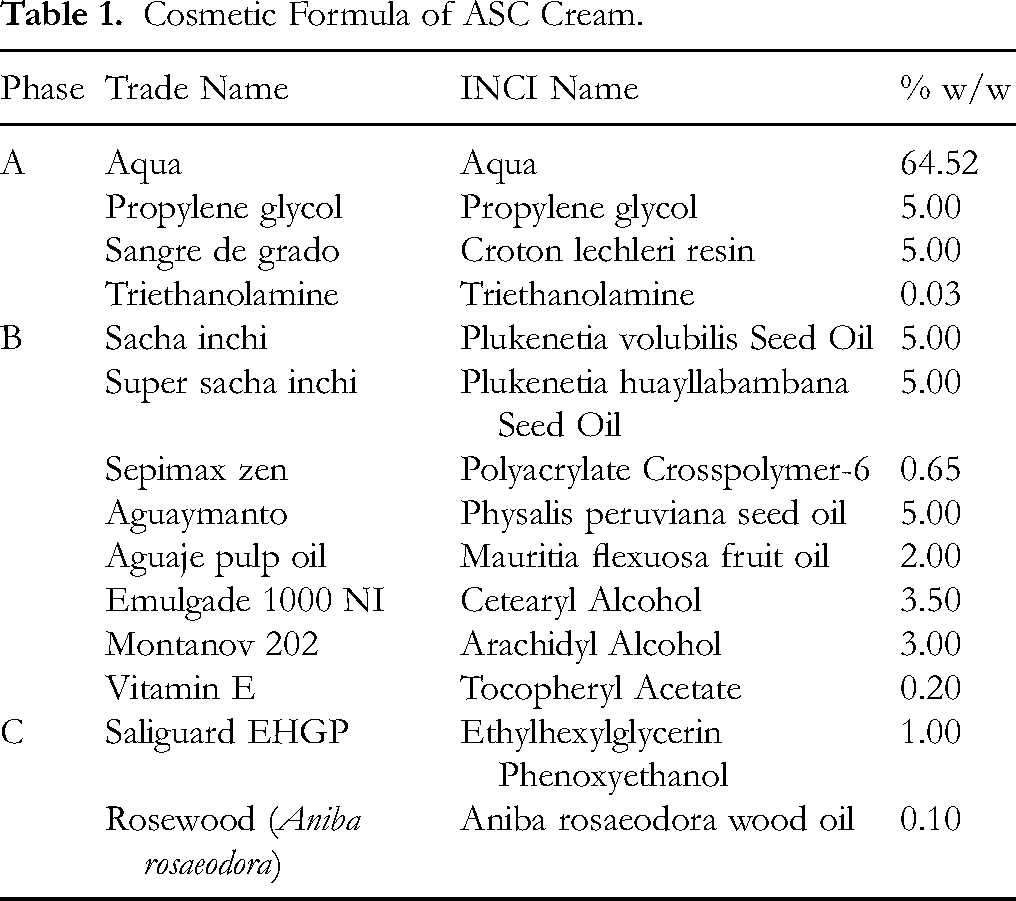
Table 1 presents the ASC Cream cosmetic formula, which was used in a previous study. 15 The cream is an emulsion in which the aqueous phase (Phase A) is mixed with the oily phase (Phase B) at a temperature between 65 °C and 75 °C under constant agitation. When the system reaches a temperature below 50 °C, Phase C is added.
Cosmetic Formula of ASC Cream.
Safety Evaluation Protocol
This evaluation was conducted in healthy volunteers (from April to May 2024) under the supervision of a dermatologist with CMP 65253 - RNE 35800 for a period of 3 weeks, including visits at baseline and at the end of the study.
At the first visit, 30 healthy volunteers were recruited from the CITIC database by the pharmaceutical chemist. The dermatologist, considering the inclusion and exclusion criteria, selected only 27 volunteers, examined both arms and chose the arm that did not show any signs of erythema or alteration for the evaluation of the cream.
Inclusion Criteria
This evaluation included male and female volunteers in the age range of 40–65 years with healthy skin (cohort of the present study), who signed a consent form agreeing to comply with the study procedures and requirements and to come to the laboratory at the scheduled test time.
Exclusion Criteria
Individuals with tattoos or scars on the posterior surfaces of the legs were excluded. Pregnant women and women with skin conditions, such as vitiligo, psoriasis, lupus, and atopic dermatitis, and other diseases that could directly interfere with the study or put the participant's health at risk, such as diabetes mellitus, opportunistic infections, hypothyroidism, history of hypoglycemia, and immune insufficiency, were excluded. Volunteers using topical or systemic drugs, such as corticosteroids, immunosuppressants and antihistamines, as well as women with a history of allergic and irritant reactions to plant and cosmetic raw materials were also excluded.
One of the main indications for the study is that the volunteers should not use any other treatment product or cosmetic on the anterior surface of the forearm (left or right), to ensure that the results are only due to the ASC Cream cosmetic.
The amount of cream to be used is a dose of 0.50 grams, which is equivalent to two pumps from the cream container, the amount necessary to ensure coverage of the forearm area, the product placed on the skin is gently massaged until completely absorbed.
In the second and final assessment visit, according to a clinical score, the parameters of erythema, oedema, papules and vesicles were assessed in each of the volunteers. 29
Table 2 shows the evaluation scale used for reading reactions recommended by the International Contact Dermatitis Research Group (ICDRG).30,31
Assessment Parameters Used for Reaction Readings.

The skin irritation index was calculated based on the scores assigned to adverse reactions (Table 2).
The evaluation scores obtained at different evaluation times were calculated to obtain the Individual Skin Irritation Index (Isii). The value represents the sum of all individual scores obtained for each reaction observed at all reading times.
The mean skin irritation index (Msii) was calculated using the following equation:
With the irritation index obtained, the product was classified according to the following scale shown in Table 3: 30
Classification of Potential Skin Irritation.

Protocol of the in Vivo Skin Mechanical Properties Study
Study Design
This was an experimental, analytical, longitudinal, and prospective study conducted at the CITIC.
Study Population
The study included healthy female volunteers, who signed an informed consent form before participating in the study. The study followed the Declaration of Helsinki and the provisions of the National Institute of Health (INS) of Peru. The clinical trial regulations were approved by Supreme Decree No. 021-2017-SA24.
32
Personal data collected were used exclusively for the purposes set forth here and were protected in accordance with the provisions of the Directorate for Personal Data Protection pursuant to Article 34 of Law No. 29733 (Personal Data Protection Law) and Articles 76, 77, 78, 79, and 81 of the Regulations of the Personal Data Protection Law approved by Supreme Decree No. 003-2013-JUS.
33
Study Sample
The inclusion and exclusion criteria were the same as those established for the safety test (sections 2.2.1 and 2.2.2), with the only difference being that in this assessment of the mechanical properties of the skin, only female volunteers were included; male volunteers were not included. The sample in this test consisted of 24 healthy female volunteers aged between 40 and 65 (study cohort), who gave their informed consent before participating. These 24 volunteers constituted a separate group from the participants included in the dermatological safety assessment.
This age range was chosen because after the age of 40 there is an alteration in collagen synthesis and a decrease in the production of ceramides responsible for skin hydration. Skin viscoelasticity and firmness progressively decrease with age, particularly after the fourth decade of life, due to reduced collagen synthesis and alterations in dermal extracellular matrix organization.34,35
Skin Bioengineering Instruments
The mechanical properties of the skin were measured using the dual Cutometer MPA 580, Courage and Khazaka, Cologne, Germany. This is a non-invasive device that measures skin elasticity in vivo by suction and has a 2 mm measuring probe. It uses the time/deformation mode with a 3-s application of constant negative pressure of 450 mbar, followed by a 3-s relaxation period. 36 The following study parameters were evaluated: R0 = Uf (firmness or amplitude at the end of the suction phase), R2 =Ua/Uf (gross elasticity or viscoelasticity during the relaxation phase), R5 = Ur/Ue (net elasticity or elastic part of the suction phase vs immediate recovery during the relaxation phase), R6 = Uv/Ue (viscoelasticity during the suction phase or ratio of viscoelastic to elastic region), R7 = Ur/Uf (immediate skin recovery vs maximum deformation), and R8 = Ua (total skin recovery). Ue = immediate skin relaxation within the first 3 s; Uv = delayed relaxation.37,38 These parameters were chosen because they are results of a correct synthesis, degradation and conformational structure of collagen and elastin.4,5
Procedures and Techniques
The study lasted 28 days (from October to November 2023), with three assessment points: on day 1, baseline measurements (before applying the cream); on days 14 and 28 (during daily application of the cream). In order to analyze the changes in the skin, the values obtained in the baseline measurements were compared with those on days 14 and 28.
The application of ASC Cream was a dose of 0.50 grams on a surface of 7 cm × 5 cm on the anterior part of the leg, an amount equivalent to two pulses of the cream container and necessary to ensure its coverage on the skin, where it is gently massaged until total absorption. The leg (left or right) was selected randomly, and this part of the body was chosen because the skin on the legs is more prone to dryness than the forearms. 39
Working Procedure
At the baseline visit (day 1), participants signed informed consent and received instructions on the correct mode and frequency of product application (twice daily, morning and evening). They were advised to refrain from applying the product on the day prior to the follow-up assessments (days 14 and 28).
For each evaluation (baseline, day 14, and day 28), volunteers were acclimatised for 30 min in a controlled room (20 °C ± 2 °C; 50% ± 5% relative humidity) before measurements were performed. Skin mechanical parameters were assessed at the same site following the procedure described in sections 2.3.4 and 2.3.5. Comparative analyses were then conducted between baseline and follow-up values to determine changes in firmness and viscoelasticity. The choice of days 14 and 28 for follow-up evaluations is supported by both clinical evidence and skin physiology. Previous cosmetic studies demonstrated significant improvements in hydration and elasticity already at day 14, with more pronounced effects by day 28. Considering that the epidermal renewal cycle in adults takes about 20–28 days, these time points allow the assessment of both early and mid-term cutaneous responses. 40
Statistical Analysis
Measurements of each parameter were performed in triplicate for each participant. The results were analysed statistically with IBM SPSS Statistics 29.0. A 95% confidence interval was considered, and values are expressed as mean, standard deviation, standard error, percentage improvement and percentage of long-term positive cases.
Means were compared at each time point, baseline versus 14 day and 28 day means. In addition, 14-day means were compared with 28 day means. Shapiro-Wilk normality tests, Student's t-test for parametric tests, and Wilcoxon tests for non-parametric tests were used.
Results
The results obtained correspond exclusively to the main objectives proposed in the research.
Safety
The 27 participants selected by the dermatologist completed the study. Table 4 shows that ASC Cream did not cause any irritation or significant sensitization during the study period, resulting in a skin irritation index of 0.00.
Results of the Daily use Safety Tests.

None of the participants had significant skin reaction (erythema, edema, papules, or vesicles) in the areas of application of the investigational product during the 3 weeks of application.
Skin Mechanical Properties
The application of ASC Cream produced increases in skin firmness and viscoelasticity values. These results can be seen in Tables 5 and 6.
Results of R0, R2, and R5 Parameters of Mechanical Properties Tests.

Results of R6, R7, and R8 Parameters of Skin Mechanical Properties Tests.
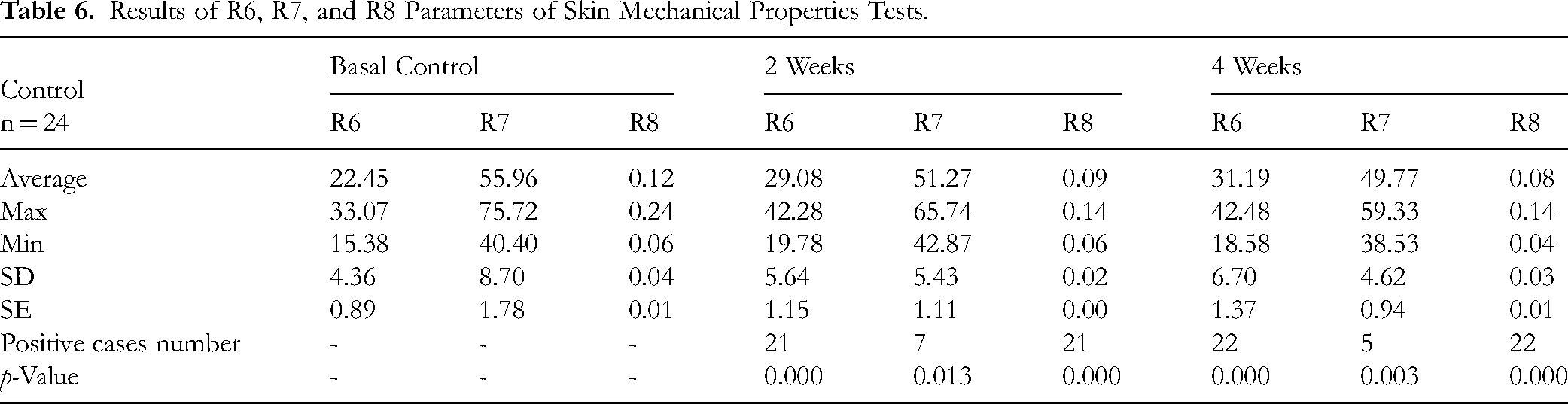
Significant changes were observed in skin firmness (R0), which showed a progressive reduction from baseline to week 2 (p = 0.001) and week 4 (p < 0.001). Viscoelasticity (R6) also increased significantly at both week 2 (p < 0.001) and week 4 (p < 0.001). Total recovery (R8) decreased significantly at week 2 (p < 0.001) and week 4 (p < 0.001).
In contrast, the parameters R2 (gross elasticity) and R5 (net elasticity) did not show significant long-term improvement compared with baseline (p > 0.05). Immediate recovery (R7) decreased significantly at both week 2 (p = 0.013) and week 4 (p = 0.003).
Figures 1–6 illustrate the percentage changes for each parameter, highlighting significant differences between baseline, week 2, and week 4.

Comparison of the percentages of improvement in the parameter R0: second and fourth week with respect to the baseline control and the gradual increase in firmness between the second and fourth week.

Comparison of the percentage changes in the R2 parameter, second and fourth week with respect to the baseline control.

Comparison of percentage changes in parameter R5, second and fourth week with respect to baseline control.

Comparison of the percentages of improvement in parameter R6: second and fourth week with respect to the baseline control and the sustainable behaviour of viscoelasticity between the second and fourth week.
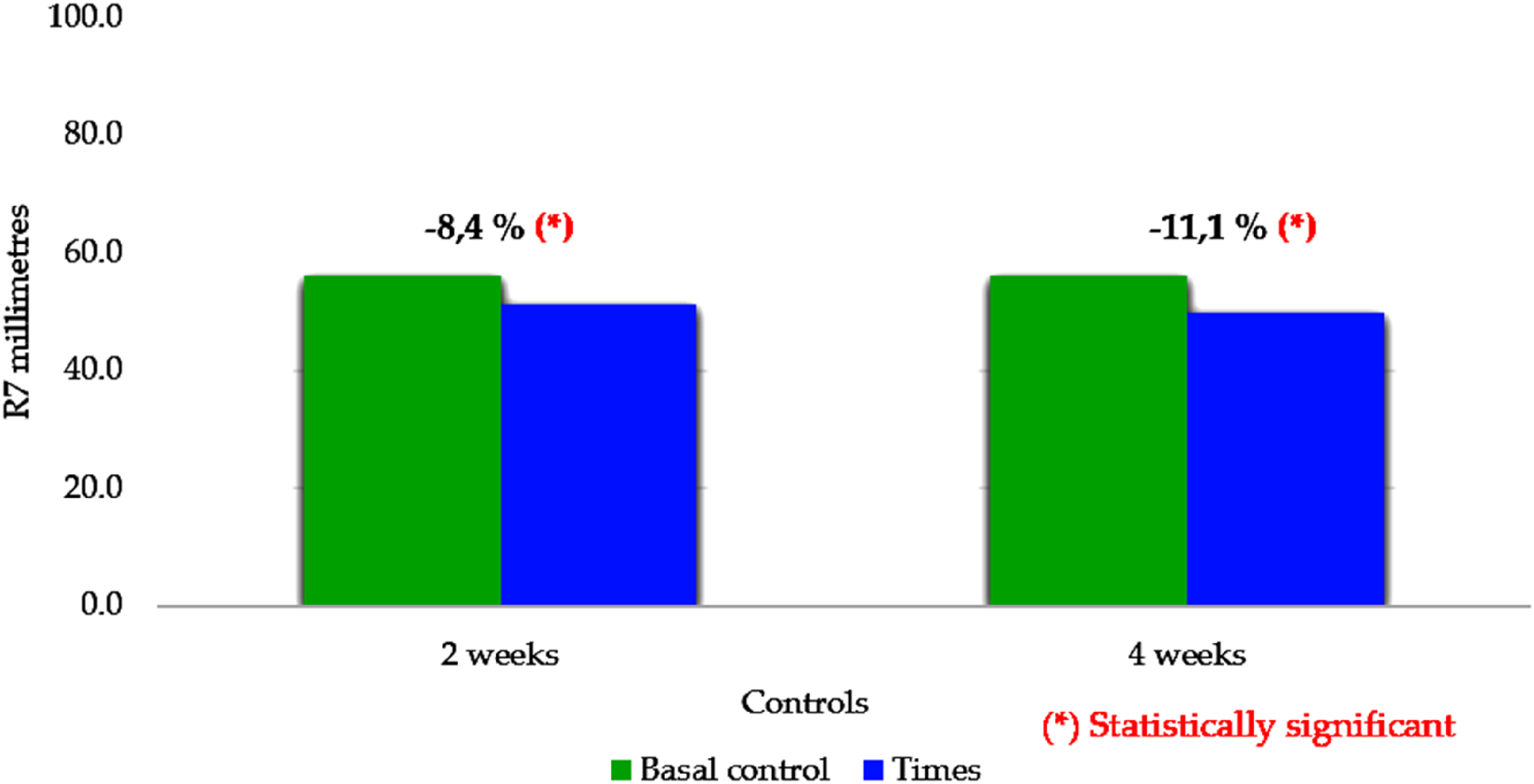
Comparison of percentage changes in parameter R7, second and fourth week with respect to baseline control.

Comparison of the percentages of improvement in parameter R8, second and fourth week with respect to the baseline control and the incremental behaviour of the total skin recovery between the second and fourth week.
The values obtained for the parameter R0 show a significant incremental decrease in the second and third week, 21.0% and 29.5%, respectively, which is interpreted as a gradual increase in firmness with 10.6%. 37
When assessing the R5 parameter, we did not observe significant changes between baseline average values and average values after 4-week treatment during the suction (Ue) and relaxation phases (Ur).
When evaluating parameter R7, we observed a significant decrease in elasticity, which may be explained by the significant increase in skin firmness at the second and fourth week of use, which simultaneously includes a decrease in retraction (Ur) and maximum distensibility (Uf) values.
The parameter R8 is the total skin recovery in millimetres 41 the results showed a significant decrease in the values, this is caused by significant firmness.
Discussion
ASC Cream is a cosmetic product with a natural extract and natural oils. In a previous study, it was shown to significantly increase skin hydration while maintaining pH levels. 15 Our study demonstrated that ASC Cream is a safe cosmetic formulation for daily use, showing no signs of irritation or sensitization in volunteers. The cream produced significant improvements in key biomechanical parameters, particularly firmness (R0), viscoelasticity (R6), and progressive recovery capacity (R8) sustained throughout the 28-day period. Consistent with previous findings, we found a direct relationship between hydration improvements and improvements in skin mechanical properties.3,42,43
The fatty acid profile of ASC Cream can be inferred from the published phytochemical characterizations of its components.16–21 As described in the Introduction, these vegetable oils are particularly rich in linoleic, oleic, and linolenic acids, with linoleic acid being the predominant constituent in most of them. This composition is relevant because linoleic acid contributes to epidermal barrier function and recovery. Acylglucosylceramide, along with lamellar bodies, conform to the lipid envelope that allows cohesion between the stratum corneum and stratum granulosum. 44 Considerable quantities of linoleic acid and oleic acid provide good hydration. 12 Oleic acid increases the fluidity of intercellular lipids, making the “brick” structure of the epidermis more continuous and improving the distribution and speed of transport of water-soluble molecules through the stratum corneum. 45 This enables ASC Cream to support significant hydration, as the fatty acid helps retain water within the epidermis. Moreover, linolenic acid modulates the immune response of T-lymphocytes in the epidermis, which are potent PGE2 inhibitors. Linolenic acid also acts as a ligand for peroxisome proliferator-activated receptors, which act as receptors in inflammation, immune regulation, and skin barrier homeostasis processes,46,47 all of which contribute to skin health. These combined properties justify ASC Cream's significant moisturizing effects and dermatological safety for topical application.
About security, Plukenetia volubilis oil was found to be safe for daily use in a previous study. 26 However, ASC Cream, which is a cosmetic formula containing other vegetable oils in addition to this species, contains higher levels of bioactive compounds. Therefore, conducting a long-term dermatological safety study with a representative sample was essential. ASC Cream had a skin irritation index of 0.00, failing to induce any irritant phenomena or significant skin sensitization during the study period. Consequently, the cream is considered safe for daily skin application.
The EEMCO guidelines recommend the Cutometer® MPA580 for the evaluation of biomechanical properties of skin in vivo, this methodology allows the cosmetic industry to substantiate significant changes and improvements in skin, such as firmness, overall elasticity, viscoelasticity, extensibility and recovery after long-term use of their commercial cosmetic products. 48 Cosmetic efficacy studies focus on strict comparison under different conditions that make it possible to identify the true impact of the product and its components. For example, evaluation using skin bioengineering equipment, comparing an area with and without product application 49 ; product with actives and base product 50 ; it is also compared with a commercial reference product. 51
Hydration, which is essential for skin health, produced by the combination of natural oils and an extract, is far superior to that provided by other formulas. 15 A direct correlation between the parameters R6 (Uv/Ue) and R8 (Ua) was reported when using occlusive hydrating products 36 ; however, our study showed that this relationship is not necessarily direct. In our study, viscoelasticity (R6) increased because of the increase in the absolute parameters of viscoelasticity (Uv) and the reduction in elasticity (Ue). This was also evidenced in the results obtained in R2 and R5, which showed reduced elasticity. We also observed a decrease in R8, which is justified by the significant increase in firmness (R0). Further clinical studies should be conducted in patients with dehydration problems and loss of skin firmness, such as those with diabetes.
Collagen is a fibrillar support protein that provides elasticity and mechanical resistance to the vessel walls of the extracellular matrix, intervenes in the process of cell regeneration, and ensures adequate skin hydration because of its water retention capacity. When collagen deficiency occurs, either due to intrinsic or extrinsic factors, the primary symptom is the loss of skin firmness. 52 In our hydration study, ASC Cream produced a significant increase in hydration. In parallel, in the cutometric study, firmness values also increased during 28 days of daily use, which may be interpreted as supporting optimal collagen function.
Reduced viscoelasticity, diminished firmness, and sagging of the skin are caused by alterations in the extracellular matrix (including elastin, collagen, and hyaluronic acid). This results in wrinkles and sagging skin. Fibroblasts are also responsible for synthesizing these structures in addition to fibronectin, glycosaminoglycans, and metalloproteinases. 53 In the present study, daily application of ASC Cream was associated with a significant increase in the firmness parameter (R0) (Figures 1 and 4). This finding may suggest that the cream helps stimulate fibroblast activity, thereby promoting the production of collagen and elastin. Previous research has reported that a decrease in R0 may indicate improved elastic properties, while increases in R5 and R7 reflect an emollient effect. 41
Regarding elasticity, the R2 parameter which reflects gross elasticity and is mainly influenced by elastic fibres, did not show any increase. 54 This finding can be interpreted as greater skin stiffness over the 28 days of application. Similarly, R5, representing net elasticity, decreased at weeks 2 and 4, which may be attributed to the marked increase in firmness observed in R0. However, because R6 parameter (viscoelasticity) increased consistently, the reductions in R2 and R5 may be transient and could stabilise or reverse in longer treatments (Figure 2 and 3).
R6, which represents viscoelasticity, is closely related to hydration levels: When water content increases, friction between fibres decreases and interstitial mobility is enhanced. 38 This parameter increased significantly in our study, rising by 7.3% from week 2 to week 4, confirming that ASC cream improves both hydration and viscoelasticity. Viscoelastic behaviour involves skin stretching and recovery, meaning that the skin responds elastically to an applied force and returns smoothly to its original state. 55 This mechanism was clearly observed in our results, supporting the moisturising effects previously reported for ASC cream. 15
The R7 parameter (Figure 5), which indicates the skin's immediate recovery following maximum deformation, 37 also supported this interpretation: although recovery was gradual, ASC cream promoted a slow and sustained return to the initial skin state, consistent with the improvements detected in R6.
A cutometric study of Korean women aged 30–50 reported that R6 increases with ageing as viscosity becomes more dominant, while R5 and R7 decrease due to the degeneration of elastic fibres. In contrast, ASC cream produced a significant increase in R6, likely due to enhanced hydration leading to greater amounts of interstitial fluid, proteoglycans and hyaluronic acid, all of which contribute to viscoelasticity. 37 Meanwhile, changes in R5 may also reflect modifications in collagen fibres, 54 as observed in our study where Ue did not increase despite significant gains in firmness.
Firmness (R0) is largely governed by the tension of collagen and elastin fibres. 54 A decrease in R0, together with an increase in R2 and R5, would typically indicate improved skin ageing. However, in our study, R2 and R5 decreased alongside a significant increase in firmness, suggesting that ASC cream does not improve gross or net elasticity within the 28-day period. Concomitant decreases in Uf and Ua (R8) values indicated reduced extensibility and greater resistance to suction, further supporting the interpretation of increased firmness. In turn, Uv (R6) increased, consistent with enhanced hydration, while Ue decreased, again confirming higher resistance to deformation.
Age-related reference values have shown that R2, R5 and R7 are highest in young women under 35 years of age. 56 Elastin degeneration during intrinsic and extrinsic ageing contributes to flaccidity and wrinkles, 57 whereas collagen particularly type I collagen, which accounts for 80%–85% of dermal protein is the primary determinant of tensile strength. 58 In our study, the early decrease in R2 from day 14 may be explained by the rapid increase in firmness (collagen-related) observed at the same time point.
Evidence from other vegetable oils further supports our findings. Creams containing Mauritia flexuosa, P. volubilis, and Oenocarpus bataua oils improved both elasticity and firmness (decreased R0 and increased R2 and R5) after 28 days in women. 59 ASC Cream contains 5% M. flexuosa and P. volubilis oils in a single formulation, which may explain the significant increase in firmness observed at days 14 and 28. However, the presence of two additional oils and a plant extract in ASC Cream's complex formulation may account for the reductions in R2 and R5. This suggests a synergistic action whereby firmness and viscoelasticity are enhanced through collagen and elastin cross-linking without necessarily improving gross or net elasticity. A longer clinical study would be needed to clarify the effects of ASC Cream on skin elasticity.
It is important to note that the main limitation of the study was not considering a control measurement, either by applying the base cream (without plant extracts and oils), or a commercial reference product, which could have strengthened the in vivo instrumental efficacy of the cosmetic cream. Nevertheless, the results of the second and fourth week were compared to the baseline measurements. In addition, the results were compared between the second and fourth week, where a highly significant incremental efficacy in the parameters of skin firmness and total skin recovery and a sustainable effect on viscoelasticity were demonstrate.
Non-invasive bioengineering evaluations are of utmost importance, and further research should be conducted on this ASC Cream product in older people or vulnerable groups with skin conditions characterised mainly by loss of skin hydration and firmness.
Another limitation was the absence of a statistical power calculation to determine the sample size. The number of participants was estimated based on feasibility and representativeness, which should be refined in future similar studies.
Conclusions
ASC Cream proved safe for topical application and demonstrated significant improvements in skin firmness and viscoelasticity after 14 days of daily use. These findings support the efficacy of incorporating natural plant-derived oils and extracts into dermocosmetic formulations to enhance skin biomechanical properties. In order to observe and confirm more robustly the beneficial properties obtained on the skin, it is suggested to continue the study, involving more weeks of topical application with a larger sample size over a wider age range.
Footnotes
Acknowledgments
Our own CITIC Research Centre carried out the entire development of the study, therefore, we thought we would omit this section.
Ethical Declaration
The study does not require an ethics committee; this is mentioned in national regulations, point 2.4.1. According to Supreme Decree No. 021-2017-SA24: “An investigational product is a pharmaceutical product or medical device that is investigated or used as a comparator in a clinical trial, including products with health registration when used or combined, in the formulation or packaging, in a manner different from that authorised, or when used to treat an unauthorised indication, or to obtain further information on its authorised use
Consent to Participate
The study included volunteers in the age range of 40-65 years with healthy skin, who signed a consent form agreeing to comply with the study procedures and requirements and to come to the laboratory at the scheduled test time. The study followed the Declaration of Helsinki and the provisions of the National Institute of Health (INS) of Peru. The clinical trial regulations were approved by Supreme Decree No. 021-2017-SA24. 32
Consent for Publication
Personal data collected were used exclusively for the purposes set forth here and were protected in accordance with the provisions of the Directorate for Personal Data Protection pursuant to Article 34 of Law No. 29733 (Personal Data Protection Law) and Articles 76, 77, 78, 79, and 81 of the Regulations of the Personal Data Protection Law approved by Supreme Decree No. 003-2013-JUS. 33
Author Contributions
Conceptualization, L.V.-T. and P.L.
Methodology, J.H. and J.R.
Formal analysis, L.V.-T.
Investigation, L.V.-T. and P.L.
Writing—original draft preparation, L.V.-T. and P.L.
Writing—review and editing, AMM.
Visualization, L.V.-T., J.H., J.R. and P.L.
Supervision, P.L.
All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
