Abstract
Background
Ultraviolet (UV) radiation threatens ocular surface integrity, while the involvement of ferroptosis—an iron-dependent, lipid peroxidation-driven cell death—in UV-induced conjunctival damage remains unexplored. Chrysanthemum morifolium extract (CME), a traditional herb with vision-protective properties, was evaluated for its potential to mitigate this damage.
Methods
Human conjunctival epithelial cells (HCoECs) were exposed to UV to model solar injury. CME was characterized and its effects were assessed through cell viability (CCK-8), lipid peroxidation (MDA assay), ROS production, and mRNA sequencing. Bioinformatic tools (eg, Swiss Target Prediction, STRING) identified flavonoid targets and ferroptosis-related networks.
Results
UV exposure triggered ferroptosis in HCoECs, evidenced by iron overload (TFRC1 upregulation), elevated ROS/MDA, and suppressed cell viability. RNA-seq revealed activation of ferroptosis pathways (eg, TFRC1, ACSL4) and inflammation signaling (eg, IL-17/HIF-1). CME pretreatment (100 μg/mL) significantly attenuated these effects, lowering MDA levels and restoring cell viability. Mechanistically, CME inhibited TFRC1-mediated iron influx and disrupted ferroptosis effector networks.
Conclusion
Our findings demonstrate that CME could protect HCoECs from UV-induced ferroptosis by chelating iron and suppressing lipid peroxidation. This study provides mechanistic support for the traditional use of CME in ocular protection and underscores ferroptosis as a potential therapeutic target in ocular surface diseases.
Keywords
Introduction
Ultraviolet (UV) radiation, mainly comprising UVA (315-400 nm) and UVB (280-315 nm) at a ratio of approximately 20:1, represents the short-wavelength segment of solar energy reaching Earth's surface.1,2 With the growing popularity of outdoor activities such as camping, hiking and surfing, prolonged UV exposure has raised significant public health concerns.3,4 Although its damaging effects on the skin—including oxidative stress, accelerated aging, and carcinogenesis—is well-documented, UV radiation also poses substantial threats to ocular tissues, which warrants careful consideration.5–7
The ocular surface, particularly the conjunctiva, is highly susceptible to UV damage. The conjunctiva spans from the corneal limbus to the eyelid fornix and consists of conjunctival epithelial cells (CoECs), connective tissue, and stroma.8,9 CoECs play a vital role in maintaining ocular surface hydration through mucin secretion and contribute to innate immune responses, thereby protecting against infection and inflammation.10–12 As the first line of defense against external insults, these cells are vulnerable to UV-induced pathophysiological changes that can lead to various ocular disorders, including pterygium, dry eye disease, conjunctival squamous cell carcinoma, and keratoconus.13–16
A key mechanism implicated in light-induced cellular damage is ferroptosis—an iron-dependent programmed cell death driven by lipid peroxidation and reactive oxygen species (ROS) accumulation. 17 Under oxidative stress, CoECs exhibit dysregulated iron metabolism, characterized by upregulation of transferrin receptor 1 (TFRC1) and subsequent iron influx.18–21 This leads to iron overload, ROS generation, lipid peroxide (LPO) accumulation, and ultimately membrane rupture and cell death (Figure 1). Although ferroptosis has been identified in blue light-induced ocular injury involving retinal damage and age-related macular degeneration, its role in UV-mediated damage to conjunctival epithelium remains fully unexplored.22–25

Volcano Plot (A) and Heat map (B) Analysis Depicting the Results of Differential Gene Expression Analysis from RNA Sequencing of Ultraviolet-Induced Damage in Conjunctival Epithelial Cells. N = 3.
Therefore, this study aims to elucidate the involvement of ferroptosis in UV-induced conjunctival epithelial damage and to evaluate the protective efficacy of CME, along with their specific mechanisms of action. We further seek to decipher the underlying mechanisms, providing mechanistic support for the traditional use of chrysanthemum in eye care and offering novel insights into therapeutic strategies for UV-related ocular surface diseases.
Materials and Methods
Extraction, Purification and Identification of CME
CME was obtained from dried Chrysanthemum morifolium flowers through the following procedure. Firstly, the dried flowers were ground into a coarse powder. A measured quantity of this powder was subjected to triple extraction using 10 volumes (mL/g) of 75% (v/v) ethanol under reflux conditions at 80 °C, with each extraction lasting 2 h. The resulting extracts were combined and concentrated under reduced pressure at 45–50 °C until the ethanol odor was no longer detectable. After concentration, the solution was filtered to collect the supernatant. This supernatant was then applied to a macroporous adsorption resin column. Following a wash with distilled water, the column was eluted with 70% ethanol at a flow rate of 1 BV/h, using approximately 3–5 BV of eluent. The ethanol eluate was gathered, concentrated under reduced pressure at 45 °C , and dried via freeze-drying or vacuum-drying to produce CME powder. The identities of key active compounds, chlorogenic acid, luteolin, isochlorogenic acid A&B&C, and apigenin, were verified by Ultra Performance Liquid Chromatography (Figure. S1 and Figure. S2). The CME used in this study was internally prepared by Zhejiang CONBA Pharmaceutical Co., Ltd, with no commercial batch number assigned, and was provided under the patent application acceptance number 202510369176.2.
Cell Culture and Treatment
Human Conjunctival epithelial cells (HCoECs) were purchased from the Cell Bank of the National Institute for Materials Science, Japan (Cellosaurus No. CVCL_9700). The cells were cultured in DMEM /F12 nutrient medium with 10% fetal bovine serum at a temperature of 37 °C in a humified incubator with 5% CO2. The medium was altered every two days, in addition to one day before commencing experiments.
In the UV exposure group, we used handheld UV lamp (4w UVL-21 UVP Compact Ultraviolet Lamp, manufactured by Funaeko Co., Ltd, Japan) to emit 360 nm UV at an intensity of 1000 µW per square centimeter to HCoECs. The UVA irradiance was measured using an ultraviolet meter (UV-340C; Tokyo Corporation). The HCoECs were exposed to UV radiation (3 mW/cm2) for 30 min to achieve a UVA dose of 5.4 J/cm2 and the culture medium was replaced immediately after UV irradiation. 30 In the CME treatment group, HCoECs were pretreated with different concentrations of CME.
Cell Viability Assay
The HCoECs were placed in 96-well plates at a density of 1000 per well, with intervention as needed for the experiment. Then cells were incubated with Cell Counting Kit-8 assay (CCK8, Bioss, Beijing, China) at 37 °C for 2 h. The optical density (OD) value of each well was measured by spectrophotometry at 450 nm using a microplate reader (Model 680; Bio-Rad, Hercules, CA). The results are representative of three individual experiments in triplicate.
Measurement of Intracellular ROS Level
The HCoECs of different groups were seeded in 96-well plates to adhere and grow for 48 h and the supernatant was discarded and PBS was added for careful rinsing for three times. Then, the HCoECs were incubated with fluorescent probe DCFH-DA in the dark at 37 °C for 30 min, and the cell is flushed to remove the excess probe. Subsequently, the samples were placed on a flow cytometer to measure the average fluorescence intensity within the cells, which represents the level of ROS.
Measurement of Malondialdehyde (MDA) Level
After 48 h culture in 96-well plates to establish cellular adhesion and growth, the HCoECs from various experimental groups underwent three PBS washing cycles after complete supernatant removal. The MDA detection kit (S0131 M, Beyotime, China) was used for the assessment following the manufacture's instructions and the fluorescence intensity was assessed by SpectraMax i3× instrument (Molecular Devices, USA).
Total RNA Extraction and Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
Total RNA was extracted from the mice conjunctivas and HCoECs using TRIzol reagent (Invitrogen). After extraction, the RNA concentration and purity were determined by spectrophotometer, and the RNA was reverse-transcribed into complementary DNA using reverse transcriptase and oligonucleotide primers. For RT-qPCR, specific primers for target genes are designed and the cDNA template is amplified in real-time PCR using fluorescently labeled probes or SYBR Green dyes. The generated amplification curve is used to quantify the initial amount of target RNA in the sample, normalize it to the internal control gene, and analyze it to determine the relative expression level. The primers sequences are listed in Table 1.
Sequences of Primers.

RNA-Sequencing and Bioinformatic Analysis
Total RNA was extracted using Trizol reagent (Invitrogen) and assessed with an Agilent 2200, securing samples with an RNA integrity number > 7.0 for further analysis. RNA Sequencing libraries were constructed using the VAHTS universal v6 RNA-seq library prep kit (Vazyme, Inc.) following the manufacturer's protocol. Poly-A mRNA was isolated from 1 μg of total RNA and fragmented into 200–600 bp pieces. First- and second-strand cDNA was synthesized, with the second strand incorporating dUTP to facilitate its removal. The resulting cDNA fragments were end-repaired, A-tailed, ligated with indexed adapters, and treated with uracil DNA glycosylase. Enriched first-strand cDNA was PCR-amplified, quality-checked on an Agilent 2200, and sequenced on a DNBSEQ-T7 platform (150 bp paired-end). Raw reads underwent adaptor trimming and quality filtering before alignment to the Human genome (GRCh38, Ensembl104) using STAR. Gene counts were derived with HT-seq and expression levels calculated through RPKM. Differential expression was analyzed with the DESeq2 algorithm using criteria: Fold Change > 2 or < 0.5 and FDR < 0.05. Gene ontology (GO) and KEGG pathway analyses were conducted using Fisher's exact test with p < 0.05 for significance, leveraging data from NCBI, UniProt, and KEGG databases.
Data Analysis and Processing
We used GraphPad Prism software to analyze the data, and Adobe illustrator 20 to establish figures. The unpaired T-test, one-way or two away ANOVA analysis were used to determine statistic difference. P < 0.05 was regarded as significant difference.
Results
CME can Significantly Reduce UV-Induced HCoECs Viability Inhibition and Lipid Peroxidation
Firstly, we performed CCK-8 and MDA assays and observed that UV exposure significantly inhibited the cellular activity of HCoECs and induced a marked increase in MDA levels. Thereafter, CME was introduced following the concentration gradient. When the concentration reached 100 μg/ml, it significantly mitigated the reduction in HCoECs activity and the elevation of MDA levels induced by UV exposure (Figure 2A and 1B). Taken together, we realized that CME can effectively mitigate the damage to HCoECs and the associated elevation in lipid peroxidation levels induced by UV radiation.

CME Mitigates the Reduction in HCoE Viability (A) and Lipid Peroxidation (B) Induced by UV Exposure. N = 3. One way ANOVA Analysis and Tukeys’ Multiple Comparisons.
mRNA Sequencing Revealed That UV Significantly Damaged HCoECs in Vitro
Subsequently, transcriptome sequencing was performed on the HCoECs samples from both the UV-exposed group and the control group. Volcano plot analysis demonstrated that UV exposure markedly altered the mRNA expression profile of HCoECs (Figure 1A). Furthermore, hierarchical clustering based on heat map analysis highlighted distinct differences in the mRNA expression patterns between the two groups (Figure 1B). These results showed that UV can dramatically damage HCoECs in vitro.
The Ferroptosis-Related Pathways may be Involved in UV-Related Conjunctival Injury
After then, we further utilized GSEA analysis to find the potential pathways in UV-induced conjunctival damage. The results showed that ferroptosis, lipid and atherosclerosis, HIF-1 signaling pathway and IL-17 signaling pathway were significantly activated(Figure 3). Taken the above pathway into consideration, we speculate that the conjunctival damage induced by UV radiation may be closely associated with the lipid peroxidation and disruption of iron metabolism it triggers.
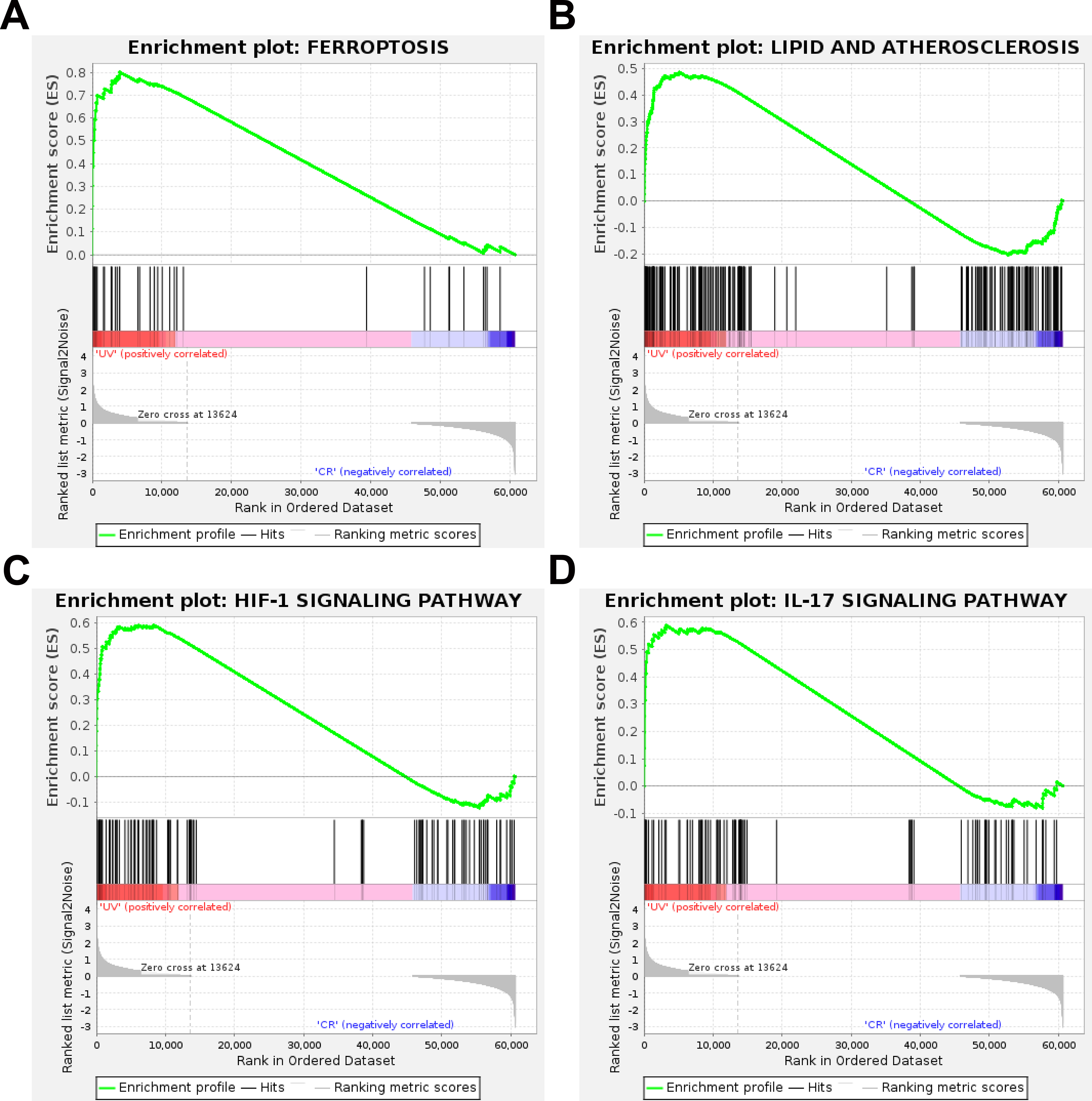
Gene Set Enrichment Analysis (GSEA) of Differentially Expressed Genes from RNA Sequencing of Ultraviolet-Induced Damage in Conjunctival Epithelial Cells. (A) Ferroptosis; (B) Lipid Metabolism and Atherosclerosis; (C) HIF-1 Signaling Pathway; (D) IL-17 Signaling Pathway. N = 3.
CME can Effectively Inhibit Ferroptosis of HCoECs Caused by UV in Vitro
According to the quantitative analysis results, through the compound screening website Swiss AMDE, it was found that flavonoids such as luteolin and acacetin in C. morifolium have good drug ability and bio-absorbency. The possible molecular targets of the above two components were examined using Swiss Target Prediction. By drawing Venn maps, the co-acting factors of the above component and the differentially expressed genes in transcriptome sequencing were screened out (Figure 4A). Summarizing the expression changes of the coding mRNAs of the above proteins in UV-exposed HCoECs, it was found that they were all significantly upregulated due to the influence of UV exposure (Figure 4B). Using STRING online analysis again, focusing on the interaction networks of the above four molecules, it was found that ACHE might have a potential interaction with TFRC1, a key molecule in the ferroptosis process (Figure 4C). The elevated expression of TFRC1 promoted the uptake of external iron ions by cells, leading to iron overload and ultimately causing ferroptosis (Figure 4D). Moreover, the fastp reports have been uploaded, please refer to Supplemental materials. Furthermore, we revealed that UV significantly increased mRNA expressions of TFRC1 and ACSL4, which is similar to the outcomes of RNA-Seq (Figure 4E). Therefore, outcomes of RNA-seq are highly effective.

Screening and Analysis of Drug Action Targets of C. morifolium Caused by HCoECs Ultraviolet Damage. (A) Venn Illustrates the Intersection of the Action Targets of two key Drug Components (Luteolin and Apigenin) and the Differentially Expressed Genes in Transcriptome Sequencing. (B) the Expression Differences of the mRNA Levels of the Above Genes Between the Control Group and the UV Exposure Group. N = 3. (C) Heat map of Differentially Expressed Genes Related to the Ferroptosis Pathway in Transcriptome Sequencing. N = 3. (D) Molecular Interaction Network Analysis of ACHE and Differentially Expressed Genes Related to the Ferroptosis Pathway in RNA Sequencing. N = 3. (E) the Expression Differences of the mRNA Levels of the Genes (TFRC1 and ACSL4) Between the Control Group and the UV Exposure Group by qRT-PCR. N = 3. One way ANOVA Analysis and Tukeys’ Multiple Comparisons.
Discussion
This study demonstrates that ferroptosis plays a pivotal role in mediating UV-induced damage in HCoECs and CME exerts protective effects by specifically suppressing this ferroptosis pathway. Our data revealed that UV exposure induced hallmark features of ferroptosis, including iron overload, lipid peroxidation, and subsequent cell death, all of which are significantly attenuated by pretreatment with CME.
UV is a well-documented environmental stressor with profound implications for human health, and its deleterious effects are primarily mediated through DNA lesions, which disrupt cell cycle processes and contribute to mutagenesis and disease development.31–34 UV exposure significantly impacts ocular health, which has been proven to induce various critical damages such as inflammatory responses and oxidative stress in lens epithelial cells and retinal cells.35–37 The conjunctival tissue, as the outermost layer of the eye, is highly susceptible to the effects of external stimuli, such as infectious agents, chemical exposures, mechanical trauma, and inflammatory processes.38,39 In our research, we demonstrated that UV exposure caused in vitro damage to HCoECs, with decreased cellular activity and increased lipid peroxidation, which might involve an imbalance in iron metabolism and oxidative stress. These findings provide insights into the molecular mechanisms of UV-induced damage and underscore the importance of protective measures against UV radiation. The UV dose (5.4 J/cm²) employed in this study effectively induces substantial cellular damage and ferroptosis phenotypes within a relatively short duration, thereby establishing a robust experimental model for investigating the molecular mechanisms underlying acute UV exposure. However, this approach does not directly replicate the conditions of chronic, low-dose environmental UV exposure encountered in daily life, which may involve distinct pathological pathways.
The central role of ferroptosis in light-induced ocular damage is an emerging research field. While previously linked to blue light injury in the retina, its involvement in UV-mediated conjunctival epithelial damage had not yet been established.20,24 Our study, through RNA-seq and GSEA analyses, effectively identified that ferroptosis as a major contributor in UV-related conjunctival epithelial injury, accompanied by the activation of related inflammatory signaling pathways (eg, IL-17, HIF-1). This finding extends the current understanding of UV-induced ocular surface phototoxicity, moving beyond the classical paradigm of DNA damage to encompass the molecular mechanisms underlying iron-dependent programmed cell death.
Chrysanthemum, a traditional Chinese medicine herb, has long been valued for its eye-protective properties, while the specific molecular mechanism has not yet been fully elucidated.40,41 Our research has demonstrated that CME could effectively counteract UV radiation-induced damage to HCoECs. Additionally, we also found that the protective effect of CME was mechanistically linked to the suppression of TFRC1-mediated iron influx, a key trigger of the ferroptosis cascade. This aligns with the known antioxidant and iron-chelating properties of flavonoid-rich plant extracts and provides a novel mechanistic basis for the traditional use of chrysanthemum in eye care. 42 Previous studies have demonstrated that CME possesses antioxidant and anti-inflammatory properties; however, this study directly associate its protective effects with a well-defined and tightly regulated form of programmed cell death—ferroptosis.
Despite the mechanistic insights gained, our study has several limitations that warrant consideration. First, the use of a monoculture system of HCoECs, while instrumental in delineating cell-autonomous responses, does not recapitulate the intricate architecture and physiological context of the intact ocular surface, which includes tear film dynamics, immune cell interactions, and neural regulation. Therefore, future studies employing in vivo models of UV-induced ocular surface damage are necessary to validate the role of ferroptosis and the efficacy of CME in a more integrated physiological setting. Second, the translational potential of CME is constrained by unresolved issues pertaining to its ocular pharmacokinetics, including corneal penetration, precorneal retention, and optimal delivery modalities. Further formulation studies are needed to enhance its bioavailability and therapeutic applicability. Third, although our bioinformatic analyses identified flavonoids such as luteolin and apigenin as putative active constituents, the precise composition and relative contributions of these compounds to the observed anti-ferroptotic effects remain unquantified. Subsequent phytochemical profiling and compound-specific functional assays are essential to identify the principal bioactive molecules. Lastly, while clinical translation represents an ultimate goal, such efforts are presently precluded by the preliminary nature of our findings and the requisite pre-clinical safety and efficacy evaluations.
Conclusion and Perspective
In summary, this study demonstrates that UV radiation induces ferroptosis in HCoECs, characterized by iron overload, lipid peroxidation accumulation and upregulation of key ferroptosis regulators like TFRC1. Crucially, CME effectively attenuated UV-induced damage by suppressing ferroptosis, reducing ROS generation and MDA levels to restore redox balance. These findings align with emerging evidence that ferroptosis is pivotal in UV-related ocular surface injury and highlight CME's potential as a protective agent (Figure 5).

The Model Diagram of This Study.
Supplemental Material
sj-html-1-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-html-1-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Supplemental Material
sj-html-2-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-html-2-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Supplemental Material
sj-html-3-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-html-3-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Supplemental Material
sj-html-4-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-html-4-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Supplemental Material
sj-html-5-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-html-5-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Supplemental Material
sj-html-6-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-html-6-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Supplemental Material
sj-docx-7-npx-10.1177_1934578X251401769 - Supplemental material for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro
Supplemental material, sj-docx-7-npx-10.1177_1934578X251401769 for The Extract of Chrysanthemum morifolium Ramat. Protects Conjunctival Epithelial Cells from Ultraviolet-Induced Damage in Vitro by Xiawei Wang, Jiafeng Li and Dongjie Song in Natural Product Communications
Footnotes
Ethical Approval
The research studies did not involve patients and did not require ethical approval.
Creditt Authorship Contribution Statement
Funding
This work was supported by the TCM Innovation Talent Support Program (2023ZR106); and the “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2025C01110): Key Technologies and Product Development for New Traditional Chinese Medicine (TCM) Drug Discovery Based on Artificial Intelligence; and the Zhejiang Provincial Natural Science Foundation of China under Grant NO.LQN25H060007.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
