Abstract
The genus Micromelum belongs to the Rutaceae family. As its rich bioactive constituents its stems, flowers, leaves, and roots have been used in traditional medicine, for the treatment of various diseases from ancient time. Phytochemically, many bioactive compounds, including coumarins, polyoxygenated flavonoids, phenylpropanoic acid derivatives, quinolone alkaloids, and also carbazole alkaloids, have been reported as secondary metabolites of the Micromelum spp. including many new compounds. Therefore, Micromelum spp. are considered potential for drug leads. In this article, we present an overview of secondary metabolites isolated from genus Micromelum and their bioactivities that have been reported between 1982 and 2019.
Introduction to Chemical Constituents of Genus Mocromelum and Their Bioactivity
Micromelum spp. (Rutaceae) consist of 9 species found mostly in Samoa, Fiji, and Tonga islands, Australia, Southeast Asia, southern China, Ceylon, northeastern India, and West Pakistan. 1,2 Micromelum spp. are known for being rich in coumarins such as the 6- and 8-prenylated coumarins. In addition, the carbazole alkaloids, dihydrocinnamic acid derivatives, and flavonoids have been isolated from it. 3,4 The leaves and stems of Micromelum species have been found to contain coumarins, phenylpropanoic acid derivatives, polyoxygenated flavonoids, and also alkaloids. 5,6 The following Micromelum species have been well investigated due to their pharmaceutical properties.
M. integerrimum is a tree that grows up to 8 m high. Its young parts have rust-colored pubescent. It usually grows in moist mountain forests, maritime thickets in sandy soil; near sea level to 2000 m and is widely distributed in China, Bhutan, Cambodia, India, Laos, Myanmar, Nepal, Thailand, and Vietnam. 7 M. integerrimum is highly diversified for its secondary metabolites, and apart from acridone and carbazole alkaloids, a variety of coumarin derivatives, especially 6,7-di and 7,8-disubstituted coumarin core structures, having at least a prenyl unit have been isolated. Naturally occurring coumarin derivatives from Micromelum spp. exhibit a variety of biological activities including anti-corpulence, cytotoxicity, anti-platelet, and antimutagenicity. 3
M. minutum is a small spineless tree that can reach up to 3 m in height. It is widely distributed in Southeast Asia and in Pacific islands. The stems, flowers, leaves, and roots are used pharmaceutically for a variety of indications. 8,9 The plant is known to contain coumarins, some of which are active compounds showing strong cytotoxic activities. The leaves are traditionally used to treat fever and giddiness. The poultice of the boiled roots is used for ague. 10 In Indochina, the roasted and crushed leaves are rubbed to skin irritated by scabies and are considered to be emmenagogues. 11 Previous phytochemical investigations on the different parts of M. minutum have showed the presence of coumarins, triterpenes, alkaloids, and phenylpropanoids. 12
M. falcatum is a tree of 1-3 m high and its leaflet blades are alternate, ovate to lanceolate, in equilateral. Flowers are ellipsoid or broad in bud. Petals are white, oblong, outside glabrous, or pubescent. Fruits are ellipsoid to ovoid, 1- or 2-seeded. Its flowering period is from January to April. It is usually found in the mountain area at heights below 1200 m in China, Cambodia, Laos, Myanmar, Thailand, and Vietnam. 13 Its leaves and roots are used for treating infected wounds, odynolysis, rheumatism, muscular atrophy, and insect bites. 4,14 M. falcatum has been found to contain several coumarins, such as 7-oxygenated coumarins, dihydrocinnamic acid derivatives, the dimeric indole alkaloid (yuehchukene), the carbazole alkaloid, 5,6-pyranoglycozoline, the 6-prenylated coumarin, micromelin, and the 8-prenylated coumarins, phebalosin and murpanidin. 6,15,16 The dimeric indole alkaloid yuehchukene was found to have potent anti-implantation activity. 17
M. compressum is a tree described as unarmed and glabrous. Its leaves are odd-pinnate, the leaflets are alternate, and the fruits are ovoid. Phytochemical efforts on the CH2Cl2-methanol extract of M. compressum have resulted in the isolation and identification of 3 polymethoxylated flavones. 1
M. hirsutum is a tree of up to 10 m height with greenish-yellow flowers and aromatic leaves. It is widely distributed in peninsular Malaysia (Melaka and Pahang northward), Andaman Islands, Myanmar, Laos, Thailand, and Vietnam. 18 In 2010, Rodphukdeekul research group investigated antitumor activity of the dichloromethane extract from branches of M. hirsutum on human B lymphoma cells, Ramos. The result demonstrated that the extract has antitumor potential against B lymphoma. 19 In Myanmar its leaves are widely used as spices in curries. Phytochemial analysis of this species has reported the presence of many carbazole alkaloids, as well as the γ-lactone compounds. 18
M. glanduliferum is a tropical or subtropical small tree found in Thailand. Coumarins and some carbazole alkaloids compounds, such as osthol, phebalosin, minumicrolin, murrangatin, murralongin, micormarin A, microminutinin, micromelin, and 2,7-dihydroxy-3-formyl-1-(30-methyl-20-butenyl)carbazole and 7-methoxy heptaphylline, were reported from the roots of M. glanduliferum. 20
The chemical analysis of leaves and stems of M. zeylanicum has reported an oxazole alkaloid, O-methylhalfordinol, a coumarin, micromelin, and a flavone, 5-hydroxy-3,3ʹ,4ʹ,7-pentamethoxyflavone. In addition, the stems contained 6-formyl-7-methoxycoumarin, and the leaves contained the carbazole alkaloid, koenigine, and p-sitosterol. 21
In this review the articles were collected from 1982 to 2019. The main purpose of this article was to evaluate the diversity of secondary metabolites isolated from Micromelum spp. in order to provide a complete reference of the diverse secondary metabolites from Micromelum spp. To the best of our knowledge this is the first review about secondary metabolites isolated from genus Micromelum.
Phytochemical Aspects of Micromelum Spp. and Their Tested Bioactivities
Coumarins
Coumarins are highly active biological substances and major secondary metabolites isolated from Micromelum spp. They are widely used in medicine, perfumes, cosmetics, and laser dyes.
6
Coumarins have antioxidant activity and they preserve the levels of other antioxidants in human plasma. Coumarins also inhibited lipid peroxidation and increased the activity of antioxidants, superoxide dismutase, and catalase.
22
Many bioactive coumarins have been reported from genus Micromelum, and the basic core coumarin compound, umbelliferone (
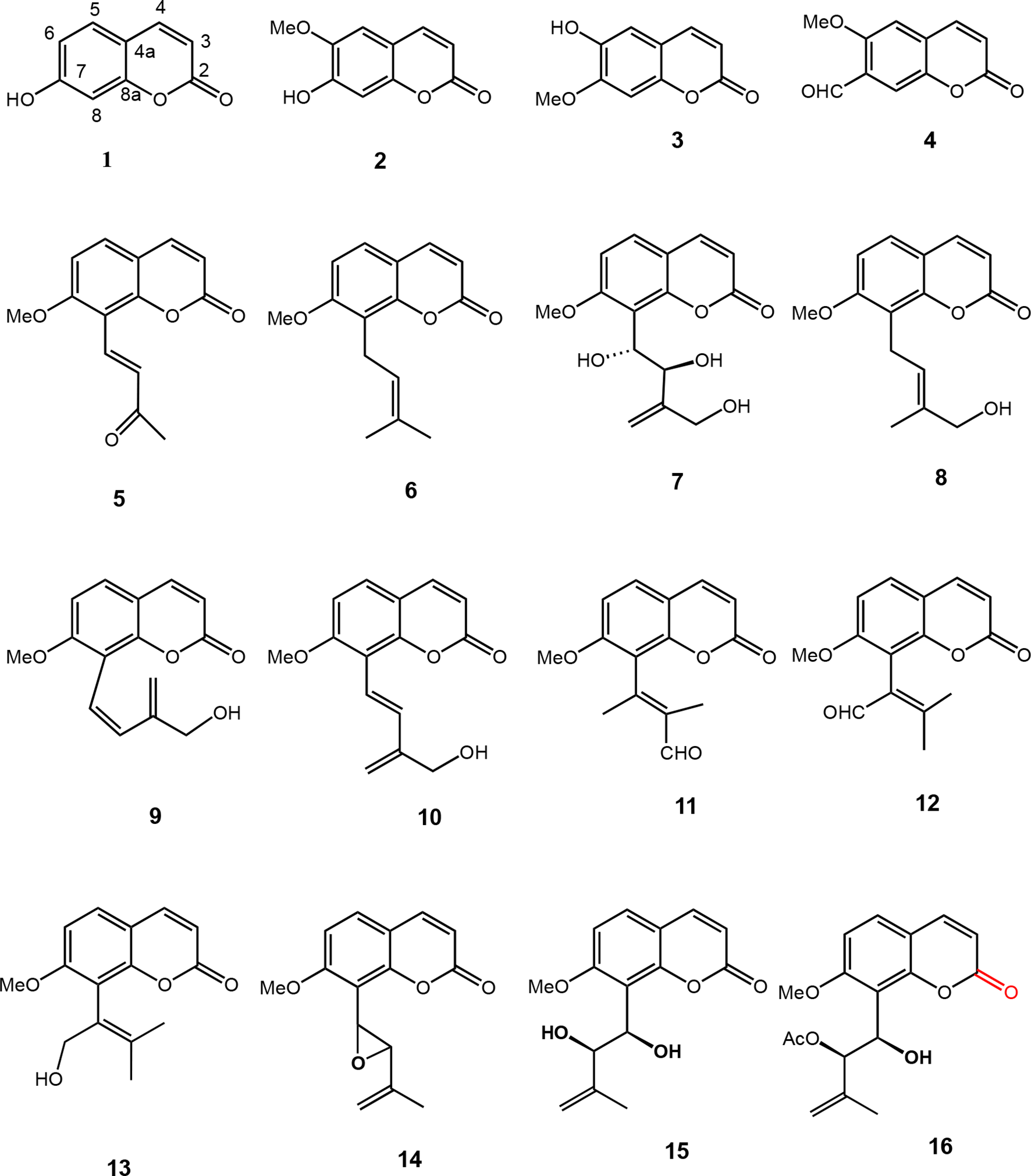
Coumarins from Micromelum minutum, M. integerrimum, M. falcatum, and M. zeylanicum.
One-prenylated coumarins named micromarin-F (

Coumarins isolated from Micromelum falcatum.
Micromelin or 7-methoxy-6-((1R,2R,5R)−5-methyl-4-oxo-3,6-dioxabicyclo[3.1.0]hexan-2-yl)−2H-chromen-2-one (

Some coumarins isolated from Micromelum integerrimum and M. minutum.
Ester coumarinoids, (3S,4S)−3,4-dihydroxy-4-(7-methoxy-2-oxo-2H-chromen-8-yl)−2-ethylenebutyl 3-methylbutanoate (

Coumarins from Micromelum minutum and M. falcatum.
Murralonginol isovalerate-1 (
Five other geranylcoumarins, such as 8-hydroxyisocapnolactone-2ʹ,3ʹ-diol (

Coumarins from the Micromelum minutum.
Two monoterpene coumarins, minutin A (
The 7-methoxylated coumarin, micromelosidester (
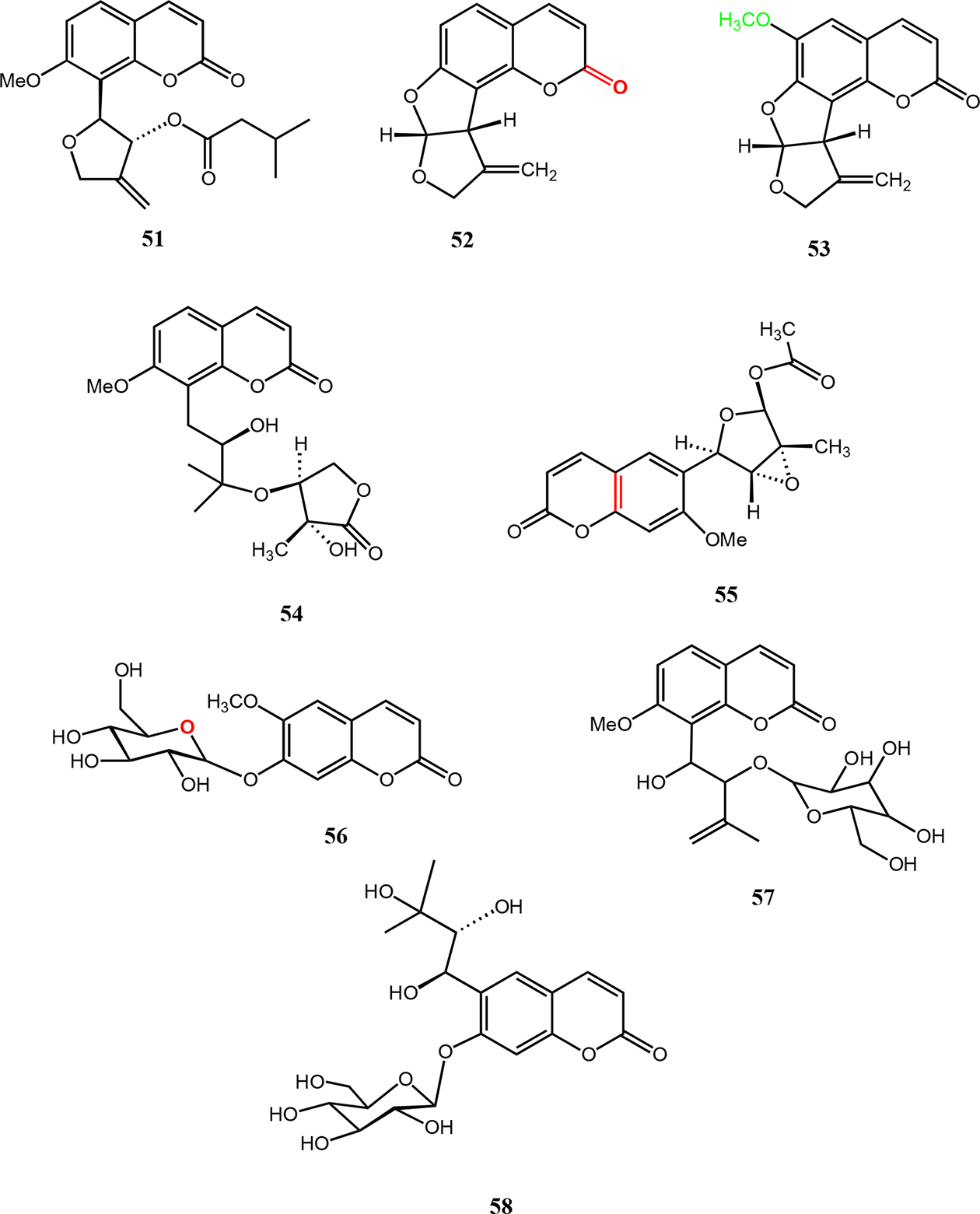
Coumarins from the Micromelum minutum, M. integerrimum, and M. falcatum.
In 2018, Kassim et al reported a lignan sesamin (

Miscellaneous compounds from the Micromelum integerrimum, M. hirsutum, M. minutum, and M. falcatum.
Coumarins from the Micromelum minutum.
Alkaloids
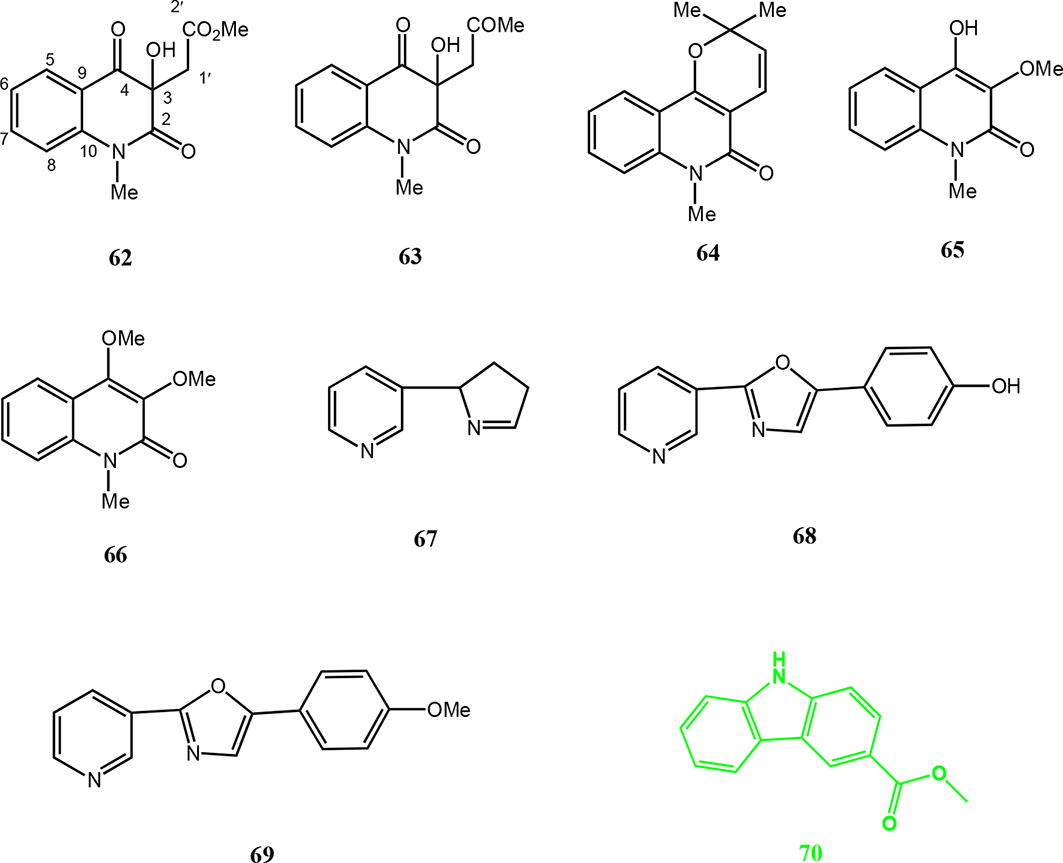
Alkaloids have inspired the development of several antibacterial drugs through the synthesis of quinine serendipitously yielding the quinolones, structural alteration of azomycin yielding metronidazole, and working with the quinoline scaffold yielding bedaquiline. In other drugs, alkaloids are present as scaffold substructures, eg linezolid and trimethoprim. Alkaloids remain the focus of much research. Their development as antibacterial drugs pursued within academia, industry, and jointventures. 35 A large number of carbazole alkaloids have been isolated from higher plants of the genera Murraya, Glycosmis, Micromelum, and Clausena, all belonging to the family rutaceae. 36,37
The quinoldione alkaloids 2-(3-hydroxy-1-methyl-2,4-dioxo-1,2,3,4-tetrahydroquinolin-3-yl)acetate (

Alkaloids from Micromelum falcatum, M. zeylanicum, and M. integerrimum.
The alkaloids myosmine (
Increased death rates due to lung cancer have necessitated the search for potential novel anticancer compounds such as carbazole derivatives. Carbazoles are aromatic heterocyclic organic compounds, with a tricyclic structure composed of 2 benzene rings each fused onto a 5-membered nitrogen-containing ring with anticancer, antibacterial, and anti-inflammatory activity.
Anti-TB bioassay-directed fractionation led to the isolation of 6 carbazole alkaloids from the CH2Cl2 extract of the stem bark of M. hirsutum. The carbazoles including 3-methylcarbazole (

Alkaloids from Micromelum minutum, M. hirsutum, M. zeylanicum, M. glanduliferum, M. falcatum, and M. integerrimum.

Alkaloids from Micromelum falcatum.
Two carbazole alkaloids, 2,7-dihydroxy-3-formyl-1-(30-methyl-20-butenyl)carbazole (
In addition, a carbazole alkaloid, mahanine (
Phenethyl cinnamides, micrometam A (
Flavones
A number of phytochemical studies dealing with polymethoxylated flavones from Micromelum spp. have been reported and a large number of flavonol-type metabolites were identified with the occurrence of partially methylated derivatives. 1
Polymethoxylated flavones, 5-hydroxy-3,4ʹ,7,8-tetramethoxyflavone (

Flavones from Micromelum minutum, M. zeylanicum, and M. compressum.
Three compounds, 5,7-dihydroxy-4’,6,8-trimethoxyflavone (
Coumarin and Flavone Condensed Binary Compounds
The compounds 8-((1S,2R)−1-hydroxy-2-(5-hydroxy-6, 8-dimethoxy-2-(4-methoxyphenyl)−4-oxo-4H-chromen-7-yloxy)−3-methylbut-3-enyl)−7-methoxy-2H-chromen-2-one (

Coumarin and flavone condensed compounds from Micromelum minutum.
Miscellaneous Compounds
The stigmasterol (

Miscellaneous compounds from Micromelum minutum and M. hirsutum.
In 2014, Zhi-yao et al have reported 2 phenylpropanoids, microintegerrin A (
Conclusion
Micromelum spp. possess very rich sources of bioactive secondary metabolites. As a member of rutaceae family it contains mainly coumarins (from simple to prenly, and geranyl), alkaloids (quinolidine and carbazole), polymethoxyflavones, and a few glycosides and phenylpropanoic acid derivatives (Table 1). The coumarins are the most abundant compounds from Micromelum spp., and some compounds, such as 8-hydroxyisocapnolactone-2ʹ,3ʹ-diol, have positive effects on the anticancer activity. Currently, at least 118 compounds have been reported from different plant parts of M. integerrimum, M. minutum, M. falcatum, M. compressum, M. glanduliferum, M. hirsutum, and M. zeylanicum (Table 1). Among them, M. integerrimum, M. minutum, and M. falcatum were well studied chemically and pharmacologically and others, M. compressum, M. glanduliferum, M. hirsutum, and M. zeylanicum, have been studied less. So, the review suggests that there is a great potential for researchers to search drug lead secondary metabolites from the genus Micromelum.
Isolated Compounds Reported From Micromelum Spp.

Footnotes
Acknowledgments
Appreciation is conveyed to Prof M. Iqbal Choudhary from H.E.J. Research Institute of Chemistry, International Center of Chemical and Biological Sciences, University of Karachi, Karachi-75270, Pakistan, for his help in improving this review article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
