Abstract
Menthol is commonly used in traditional medicine and is increasingly supported by pharmacological evidence. This review provides a comprehensive and updated overview of menthol's pharmacology, therapeutic applications, and safety profile, with particular focus on the underlying molecular mechanisms that may explain its actions. Evidence from both preclinical and clinical studies demonstrated benefits in inflammation, pain modulation, gastrointestinal function, microbial infections, and wound healing, while additional findings indicated potential roles in neuroprotection, anti-aging strategies, and cancer prevention. Clinical data further showed effectiveness in irritable bowel syndrome, headache disorders, and skin care. Menthol is generally considered safe, although high doses or unsuitable formulations may cause adverse effects, underscoring the importance of appropriate dosing and patient selection. This review suggests that menthol represents a promising natural therapeutic agent, yet emphasizes the urgent need for standardized clinical trials and long-term safety evaluations to clearly define its role within evidence-based medicine.
Introduction
For centuries, medicinal plants have played a significant role in healthcare due to the presence of bioactive constituents with therapeutic effects, including anti-inflammatory, analgesic, and antimicrobial properties. 1 These plant-based remedies are often perceived as gentler alternatives to synthetic pharmaceuticals, offering favorable tolerability and a lower incidence of side effects.2,3
Among these, Mentha × piperita L., commonly known as peppermint, stands out for its extensive use in traditional healing systems. 4 Peppermint is well-known for its distinctive fragrance and taste, primarily due to its rich content of volatile oils—particularly menthol. 5 In addition to peppermint, several other species of the Mentha genus are recognized for their high menthol content, including Mentha arvensis L., Mentha longifolia (L.) L., Mentha requienii Benth., Mentha pulegium L., Mentha haplocalyx Briq., and Mentha canadensis L. 6
Menthol exists as two optical isomers, d-menthol and l-menthol
Beyond its phytochemical profile, the clinical relevance of integrative medicine has gained increasing attention in recent decades, particularly in the management of chronic and lifestyle-related diseases. Natural products, including menthol, were frequently incorporated into complementary and integrative medicine strategies due to their favorable safety profile and therapeutic versatility. Several ethnopharmacological and clinical surveys highlighted the widespread use of medicinal plants in conditions such as diabetes, dyslipidemia, and viral respiratory diseases, underscoring their growing popularity as adjuncts or alternatives to conventional therapies.8–11
Preclinical investigations explored antioxidant, anti-inflammatory, antimicrobial, and immunomodulatory activities of menthol, alongside mechanisms that may underlie its therapeutic potential. 12 Clinical research further examined its utility in treating gastrointestinal disorders, neurological conditions, dental issues, and skin diseases. 13
Despite extensive research, key gaps persist in the literature. Evidence from preclinical and clinical studies is seldom distinguished, and safety aspects are inconsistently addressed. A concise synthesis that clarifies these issues and emphasizes therapeutic potential is therefore warranted. This review article aims to provide a comprehensive and up-to-date summary of current knowledge on the medicinal properties and clinical applications of menthol. It also addresses safety considerations and potential adverse effects associated with its use. By integrating evidence from both preclinical and clinical research, the review seeks to offer a well-rounded reference for researchers, clinicians, and healthcare stakeholders interested in leveraging menthol and other plant-derived compounds in therapeutic strategies.
Safety Profile
Toxicological evaluations indicated that the estimated oral lethal dose of menthol in humans is approximately 50–150 mg/kg body weight. 14 Preclinical studies in animals demonstrated that excessive administration of L-menthol can elicit central nervous system effects such as euphoria, convulsions, and coma, along with hepatic and renal alterations. These adverse outcomes are generally reversible upon cessation of exposure. 15
Documented cases of menthol-related toxicity in humans included allergic contact cheilitis and rhinitis linked to the use of mouth care items and various personal hygiene products containing menthol.16,17 Prolonged consumption of cough remedies formulated with menthol was linked to a variety of harmful outcomes, such as repeated occurrences of oral sores, occasional bouts of diarrhea, ataxia, persistent dizziness, heartburn, patchy skin discoloration, reduced urine output, darkly pigmented urine, and, in severe cases, coma resulting from shock. 18
The safety of menthol was examined in the context of tobacco products. Some in vitro investigations and limited cross-sectional studies suggested a potential increase in carcinogenic risk with mentholated cigarettes compared to non-mentholated varieties.19,20 However, results from larger, well-designed epidemiological studies did not support a causal link.21–23
Despite the lack of conclusive evidence for increased carcinogenicity, menthol cigarettes were marketed as smoother or less harmful alternatives. 24 Research indicated that menthol additives can facilitate smoking initiation among young individuals and reduce the likelihood of successful cessation.25–28 Consequently, in April 2022, the FDA proposed regulations to prohibit the marketing of cigarettes containing menthol flavoring and any cigars with added flavorings. 29
Peppermint has no well-documented contraindications. However, ingestion of peppermint oil rich in menthol may cause heartburn due to relaxation of the lower esophageal sphincter, thereby promoting gastroesophageal reflux, and can occasionally induce burning sensations in the anal or perianal region. 30 Current literature suggests that consumption of peppermint oil in culinary amounts is considered safe during pregnancy and lactation.31–34
Biological Activities of Menthol
Anti-Inflammatory Effects
Menthol was shown to exhibit notable anti-inflammatory effects across a range of experimental models. Evidence from in vitro assays and in vivo animal studies revealed multiple peripheral pathways through which menthol mediates its anti-inflammatory function. Immune cells contain members of the transient receptor potential family (TRPs)—such as transient receptor potential melastatin 8 (TRPM8), transient receptor potential vanilloid 1 (TRPV1), and transient receptor potential ankyrin 1 (TRPA1), which appear to mediate the inflammation-reducing activity of menthol through cationic signaling mechanisms, ultimately preventing the release of proinflammatory cytokines. 35 Specifically, in vivo murine models demonstrated that TRPM8 activation attenuated experimental colitis and mitigated post-myocardial infarction (MI) inflammation and cardiac remodeling.36,37 Moreover, menthol appears to modulate inflammation in gastroesophageal reflux by interacting with TRPV1 receptors within esophageal epithelial tissue. 38 These mechanisms were illustrated in Figure 1.
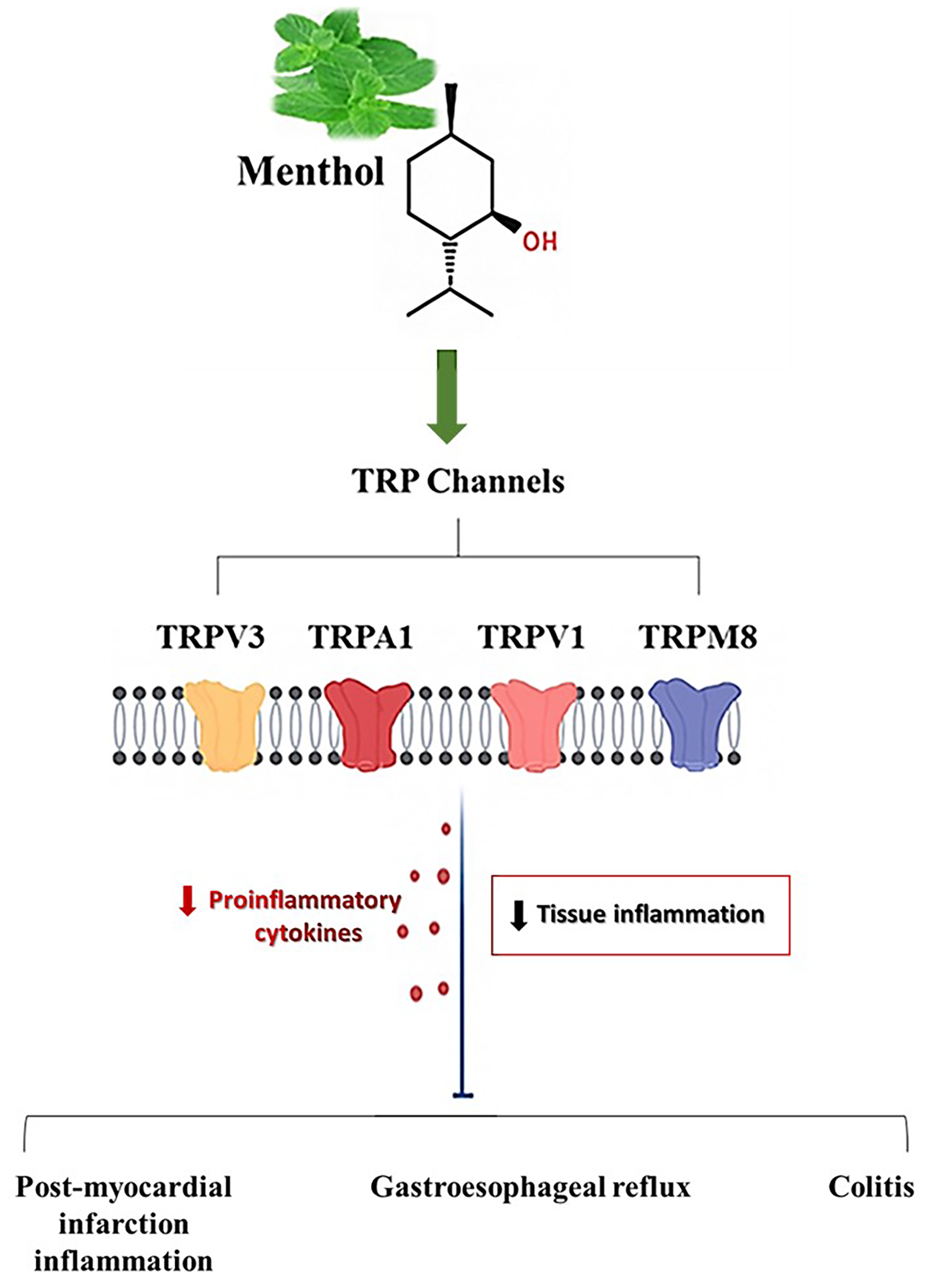
Proposed Mechanisms Underlying the Anti-Inflammatory Effects of Menthol Through TRP Channel Modulation.
The anti-inflammatory potential of menthol was further confirmed by classical in vivo models
Immunomodulatory Effects
Menthol exerts notable immunomodulatory activity, primarily through its anti-inflammatory and antioxidant mechanisms, though other biological pathways may also contribute. Most in vivo investigations into its immunomodulatory potential were conducted in aquatic and avian models. In fish, menthol oil administration was reported to boost innate immune defenses, including enhanced lysozyme secretion and phagocytic function, as well as a strengthened humoral response under immunostimulatory conditions. 42
In poultry studies, dietary supplementation with menthol led to an increase in lymphocyte populations, which was linked to the stabilization of lymphocyte cell membranes and protection against oxidative damage. 43 Consistent with these findings, menthol supplementation was also associated with elevated circulating levels of immunoglobulins (Ig), including total immunoglobulin G (IgG) and immunoglobulin M (IgM), reflecting a positive modulation of adaptive immune responses. 44
By modulating both innate and adaptive immune responses, menthol may offer therapeutic potential as a natural immunomodulatory agent in veterinary and possibly human medicine, particularly for conditions involving immune dysregulation.
Antioxidant and Cytoprotective Effects
Oxidative stress, characterized by a disruption in the balance between the production of reactive oxygen species (ROS) and the body's antioxidant defense capacity, is widely recognized as a key contributing factor in both the aging process and the development of long-term degenerative disorders. 45 Menthol demonstrated modest antioxidant activity, functioning as a free radical scavenger and enhancing endogenous antioxidant responses. Specifically, menthol was reported to enhance both the production and functional capacity of major antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx). By counteracting ROS, these enzymes help protect cells from lipid peroxidation and DNA injury. Through this mechanism, menthol provides cytoprotective benefits, particularly in tissues susceptible to oxidative injury, such as neuronal, hepatic, and dermal systems. The antioxidant properties of menthol also supported its use in anti-aging formulations and in mitigating oxidative stress–mediated inflammation in neurodegenerative and cardiovascular diseases. 45 Experimental evidence from animal studies supports menthol's antioxidant potential in vivo. In animal models, administration of menthol led to a marked decline in malondialdehyde (MDA) concentrations, an indicator of oxidative damage to lipids, and was accompanied by a recovery in the function of the body's intrinsic antioxidant enzymes.46,47
These antioxidant and cytoprotective properties supported the potential use of menthol in anti-aging formulations and in mitigating oxidative stress–related conditions, including cardiovascular and neurodegenerative diseases.
Antibacterial Effects
Most of the current evidence on menthol's antibacterial activity was derived from in vitro studies, which demonstrate its ability to interact with and disrupt microbial cell membranes. 48 Due to its lipophilic nature, menthol integrates into the lipid bilayer of microbial membranes, altering membrane fluidity and permeability. This disruption leads to leakage of essential intracellular components, such as ions, ATP, and nucleic acids, ultimately resulting in cell death. 49 The antimicrobial effect extended to a wide range of pathogens.
Among Gram-negative bacteria, inhibition was noted for Yersinia spp. (particularly Y. enterocolitica), Proteus spp., Pseudomonas aeruginosa, and various Enterobacter species. Several members of the genus Pseudomonas syringae—including the pathovars syringae, tomato, and phaseolicola—were also affected, along with plant-associated pathogens such as Xanthomonas campestris (pv. phaseoli and pv. campestris).
The Gram-positive spectrum included Listeria monocytogenes, Bacillus cereus, and methicillin-resistant Staphylococcus epidermidis.
In addition, the activity extended to Salmonella typhimurium and opportunistic pathogens like Klebsiella pneumoniae.50–52
Antimicrobial assessments showed that Mentha piperita possesses inhibitory effects on a range of multidrug-resistant pathogens. These include Enterococcus faecalis, Streptococcus pyogenes, carbapenem-resistant Escherichia coli, methicillin-resistant Staphylococcus aureus (MRSA), and methicillin-resistant Staphylococcus epidermidis (MRSE). 53
Such antibacterial activity highlighted menthol's therapeutic potential as a complementary strategy in infection control, particularly against multidrug-resistant bacterial pathogens.
Antifungal Effects
Menthol is known to exert fungistatic and fungicidal effects through several molecular pathways. One key target is the fungal plasma membrane H+-ATPase (PM-ATPase), a vital protein responsible for regulating intracellular pH, nutrient uptake, and cellular homeostasis. 54 Previously reported targets include Rhizopus solani, Alternaria alternata, Penicillium spp., and Fusarium spp.51,55 Additional susceptible strains include Pichia carsonii, as well as several species of Aspergillus and Candida. 56 Notably, essential oils rich in menthol also inhibited the growth of Candida albicans and Rhizopus nigricans. 50 Furthermore, menthol was demonstrated to have activity against drug-resistant fungal species and was reported to be effective against Cryptococcus neoformans, a major causative agent of fungal meningitis. 56
The demonstrated antifungal activity indicates menthol's promise as a natural antifungal agent, with potential applications in treating drug-resistant fungal infections and in food preservation.
Antiviral Effects
Menthol-rich essential oils were shown to disrupt the viral envelope, inhibit viral adsorption, block entry into host cells, and interfere with membrane fusion events. These actions are thought to be largely non-specific, driven by menthol's membrane-perturbing capabilities, but they nonetheless offer promising avenues for adjunctive treatment strategies, especially in topical and inhalation-based formulations.57–59
Mentha piperita and its essential oils, rich in menthol, were extensively studied for their antiviral potential across a variety of viral pathogens. One of the well-characterized mechanisms of menthol involves the inhibition of reverse transcriptase activity in the human immunodeficiency virus (HIV), as demonstrated by Geuenich et al 60 In the context of herpes simplex virus (HSV) infections, menthol was reported to disrupt the replication cycle of HSV-1 by targeting the late phases of viral development. 61 A key contributing factor to viral replication is the intracellular depletion of the antioxidant glutathione (GSH), which is commonly observed during HSV-1 infection. Menthol was shown to preserve intracellular GSH levels, implying a modulatory role on redox-sensitive pathways essential to viral maturation. Additionally, it demonstrated direct virucidal activity by binding to the viral envelope and glycoproteins, thereby preventing viral entry into host cells. 61
Recent pharmaceutical applications explored menthol for enhancing drug performance. For instance, menthol was utilized in the co-crystallization of Lopinavir, significantly improving its dissolution rate and intestinal absorption. 62 In another study, menthol-assisted microextraction methods for Favipiravir yielded a more accurate, eco-friendly, and safer analytical approach compared to conventional techniques. 63
These findings suggested that menthol and menthol-rich essential oils could be further developed as adjunctive antiviral agents, particularly in topical or inhalation-based formulations targeting respiratory and mucosal viral infections.
Analgesic and Antispasmodic Effects
Preclinical Evidence
The ability of menthol to modulate pain perception and smooth muscle tone is well established and has led to its widespread use in a variety of therapeutic applications, from tension-type headaches to gastrointestinal discomfort. The analgesic effects of menthol are primarily mediated by its activation of the transient receptor potential melastatin 8 (TRPM8) channels, which are expressed on cutaneous and visceral sensory neurons. Activation of TRPM8 induces a cooling sensation that counteracts nociceptive stimuli and desensitizes pain-transmitting nerve fibers. This sensory modulation reduces the perception of thermal and mechanical pain and is particularly effective in superficial applications such as topical pain relievers and cooling gels.64–66 In addition to TRPM8, menthol can also modulate the activity of other ion channels implicated in pain transduction, including voltage-gated sodium channels (NaV). By inhibiting these channels, menthol decreases neuronal excitability and impedes the propagation of action potentials, resulting in a local anesthetic-like effect. 67
Moreover, menthol exhibits central and peripheral antispasmodic effects by blocking voltage-dependent calcium channels (CaV) in smooth muscle tissues. 67 This action prevents calcium influx, leading to the relaxation of gastrointestinal, vascular, and bronchial smooth muscles.
Clinical Evidence
Muscle-relaxant properties of menthol were clinically validated in the management of irritable bowel syndrome (IBS), a functional gastrointestinal disorder characterized by visceral hypersensitivity and smooth muscle hypercontractility. Multiple randomized controlled trials and meta-analyses investigated that enteric-coated peppermint oil capsules, rich in menthol, significantly reduce abdominal pain, bloating, and cramping in IBS patients. These benefits are attributed to menthol's ability to suppress smooth muscle spasms without impairing normal peristalsis, making it a safe and effective treatment option.68–70
The analgesic action of menthol also extends to the management of primary headache disorders. The application of low-concentration menthol solutions to the temples and forehead was shown to alleviate tension-type headaches and reduce the intensity of migraine attacks. These effects are attributed to menthol's cooling sensation, vasodilatory action, and ability to modulate the release of neuropeptides such as calcitonin gene-related peptide (CGRP), which are implicated in headache pathophysiology.71,72
The therapeutic potential of menthol in the management of migraine was assessed in randomized controlled trials (RCTs).73,74 Among these, two trials reported a statistically significant reduction in migraine symptoms compared to placebo when higher concentrations were applied—specifically, 10% menthol, and undiluted peppermint essential oil.75,76
Furthermore, professional organizations, including the German Society for Neurology and the Italian Society for Headache Research, currently endorse the topical application of menthol products to provide short-term relief from tension-type headaches.77,78
Overall, these results underlined the therapeutic potential of menthol in the management of functional gastrointestinal disorders, such as irritable bowel syndrome, and in primary headache syndromes, including migraine and tension-type headaches.
Antiaging Effects
Multiple intracellular signaling pathways were implicated in the regulation of aging and the development of age-associated diseases. Among the proposed mechanisms, menthol may exert anti-aging effects via modulation of TRP channels—membrane proteins involved in thermosensation, nociception, and other sensory functions. Notably, TRP channels also play a regulatory role in cellular senescence, a process characterized by permanent cell cycle arrest and strongly linked to tissue aging and degenerative conditions. 79
A study investigating the potential anti-aging properties of menthol found that mesenchymal stem cells derived from rats exposed to peppermint extract, which is rich in menthol, showed increased expression of the pluripotency-associated Nanog gene. Additionally, there was a reduction in the expression of markers associated with cellular senescence, such as p53 and NF-κB. The same study also noted a lower percentage of cells present in the G2/M phase, suggesting a possible influence on cell cycle progression. 80
Despite these promising molecular insights, much of the current research on peppermint and menthol remains centered on dermatological and cosmetic applications. Studies have highlighted benefits such as enhanced skin hydration, improved elasticity, and better overall skin appearance.81,82 However, the broader implications of these compounds in systemic aging and age-related diseases remain underexplored, necessitating further experimental and clinical investigations to clarify their mechanisms and therapeutic relevance.
These preliminary findings suggested that menthol-containing formulations may contribute to anti-aging strategies in dermatology and potentially in age-related systemic disorders, though clinical validation is required.
Anticancer Effects
Extensive scientific investigations explored the therapeutic potential of menthol and neomenthol in the management of different cancer types, including prostate cancer, cervical cancer, leukemia, skin cancer/melanoma, neomenthol-associated skin cancer (Ehrlich ascites carcinoma, EAC), lung cancer, gastric cancer, and adenocarcinoma. Most of these studies utilized cellular assays and animal models to evaluate the effectiveness of these compounds and to clarify their mechanisms of action. Menthol was recognized as a strong inhibitor of cancer cell growth, acting mainly through cell cycle regulation, and it also exhibits suppressive effects on tumor cell invasion, metastasis, angiogenesis, and proliferation. 83
The cancer-preventive activity of peppermint is linked to its capacity to suppress cell proliferation by halting cells in the S phase of the cell cycle. Furthermore, it was reported to upregulate the expression of the p21 gene, which plays a role in triggering G1 phase cell cycle arrest. 84 The antiproliferative and anti-metastatic properties of menthol highlighted its potential as a complementary anticancer agent.
Wound Healing Effects
The potential of menthol-containing topical formulations in promoting wound healing was explored in various in vivo studies. In a study by Rozza et al, the wound healing activity of menthol cream was evaluated in rats using formulations containing 0.25%, 0.5%, and 1% menthol. Among the tested concentrations, the 0.5% formulation demonstrated the most favorable outcomes across all healing stages. It significantly downregulated proinflammatory mediators, while simultaneously enhancing antioxidant defenses via increased activity of peroxidase and dismutase enzymes. 85 Moreover, the treatment boosted the expression of Ki-67, a known marker of cellular proliferation. These biological responses collectively contributed to a reduction in wound size and accelerated healing throughout 3–14 days, corresponding to the different phases of repair. 85
In another in vivo investigation, Ansari et al assessed a hydrogel composed of menthol, a blend of chitosan and guar gum, in a rat model of burn injury. After 22 days of treatment, the infected wounds induced by Staphylococcus spp. showed approximately 90% contraction. The hydrogel formulation also inhibited the proliferation of Candida species, Staphylococcus spp., and Escherichia coli. 86 These results suggested that menthol-containing topical formulations may have therapeutic applications in wound management, particularly in accelerating healing and reducing microbial burden.
Psychological and Cognitive Effects
Preclinical Evidence
The therapeutic potential of menthol was explored in various neuropsychiatric disorders. In experimental models of depression, menthol demonstrated antidepressant-like activity in mice, mediated through modulation of serotonergic, dopaminergic, and γ-aminobutyric acid (GABA)ergic pathways. 87
Preclinical studies proposed several neuroprotective mechanisms, including attenuation of amyloid-β deposition in brain tissue, restoration of neuronal morphology, and reduction of oxidative stress-induced damage. In Alzheimer's disease models, cognitive enhancement was linked to modulation of arginine–proline, inositol phosphate, and cysteine–methionine metabolic pathways. 88 Additional mechanisms include improved cerebral blood flow and upregulation of brain-derived neurotrophic factor (BDNF) expression in the hippocampus.89–91
Clinical Evidence
A range of clinical studies investigated the anxiolytic effects of menthol in both healthy individuals, predominantly university students.92,93 These interventions were assessed in individuals undergoing invasive medical procedures, such as coronary angiography, and genetic amniocentesis, as well as in pregnant women and parturients during labor.94–97 Overall, the majority of these trials supported a beneficial role for menthol aromatherapy in alleviating anxiety symptoms. The proposed mechanism involves modulation of the hypothalamic–pituitary–adrenal (HPA) axis, leading to decreased cortisol secretion and subsequent reduction in anxiety manifestations.98,99
Accumulating clinical evidence indicates that menthol exerts positive effects on multiple domains of memory performance.100–103 The evidence indicates that menthol may hold therapeutic promise as a complementary approach for anxiety management and cognitive enhancement, including in neurodegenerative disorders such as Alzheimer's disease.
Collectively, the diverse pharmacological activities of menthol, supported by preclinical evidence, were summarized in Table 1.
Pharmacological Activities of Menthol, Including Mechanisms, Models, and key Preclinical Findings.

Abbreviations: TRPM8 – transient receptor potential melastatin 8; TRPV1 – transient receptor potential vanilloid 1; TRPA1 – transient receptor potential ankyrin 1; NaV – voltage-gated sodium channels; CaV – voltage-gated calcium channels; SOD – superoxide dismutase; CAT – catalase; GPx – glutathione peroxidase; MDA – malondialdehyde; BDNF – brain-derived neurotrophic factor; MI – myocardial infarction.
Besides preclinical evidence, clinical evidence on therapeutic applications of menthol in gastrointestinal, neurological, and psychological disorders was also summarized in Table 2.
Summary of Clinical Studies Investigating Menthol in Human Disorders.

Abbreviations: IBS – irritable bowel syndrome.
Study Limitations
Although menthol exhibits diverse pharmacological activities, several limitations must be considered. Much of the available evidence originates from studies in specific geographic regions, especially Asia, which restricts generalizability to other populations. Clinical trials are often small and heterogeneous in design, limiting the strength of their conclusions.
Methodological inconsistencies, including variable outcome measures, non-standardized protocols, and inadequate blinding, further increase the risk of bias, particularly for subjective endpoints. Safety data are mostly short-term, leaving gaps regarding long-term toxicity, drug–drug interactions, and delayed adverse effects. In addition, cost-effectiveness and subgroup analyses by demographic factors (eg, age, sex, comorbidities) are rarely addressed. Collectively, these issues highlighted the need for well-designed, large-scale randomized controlled trials with standardized formulations, validated endpoints, and extended follow-up to confirm menthol's clinical relevance.
Future Directions and Perspectives
Future research should prioritize large-scale, randomized controlled trials to validate the efficacy and safety of menthol across different therapeutic domains, including gastrointestinal, neurological, and dermatological conditions. Establishing standardized formulations and dosing regimens will be essential to improve reproducibility and clinical applicability. Mechanistic studies are warranted to further clarify the role of TRP channels, oxidative stress modulation, and neuroinflammatory pathways in mediating menthol's actions. In addition, exploration of innovative delivery systems such as nanocarriers, transdermal patches, and co-crystal formulations may enhance bioavailability and therapeutic outcomes. Long-term safety assessments and pharmacokinetic investigations are also necessary to fully realize menthol's potential as an integrative medicine option.
Conclusion
Menthol garnered increasing scientific interest due to its diverse pharmacological properties, favorable safety profile, and longstanding use in traditional and modern medicine. This comprehensive review underscores their multifaceted therapeutic potential, spanning anti-inflammatory, immunomodulatory, antioxidant, antimicrobial, antiviral, analgesic, antispasmodic, anti-aging, anticancer, and wound-healing activities.
Despite the encouraging data, clinical trials validating the systemic effects of menthol in humans—particularly in the domains of oncology, aging, and neurodegeneration are still limited. While menthol is generally well tolerated, attention to concentration, formulation, route of administration, and patient-specific factors is essential. Integrating menthol into evidence-based clinical medicine will depend on the implementation of standardized, high-quality clinical trials that determine appropriate formulations, dosing regimens, and validated outcome measures. Equally important are regulatory considerations, including the development of clear safety guidelines, long-term pharmacovigilance, and standardized quality controls, to ensure both efficacy and patient safety.
Footnotes
Author Contribution Statement
Kemal Husnu Can Baser, Emine Erdag, Ismail Celil Haskologlu: writing—original draft preparation. Kemal Husnu Can Baser: supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
