Abstract
Licorice is a renowned traditional Chinese medicine. Licoisoflavone A and licoisoflavone B are two compounds present in licorice. Previous studies have demonstrated their effectiveness in several pharmacological areas. This review aims to provide a comprehensive overview of licoisoflavone A and licoisoflavone B, including their biological sources, physicochemical properties, biotransformation pathways, and pharmacological activities. Licoisoflavone A and licoisoflavone B are primarily derived from different types of dried licorice roots, and pure forms of them can be readily obtained in the market for experimental research. While the exact biosynthesis, transformation, and metabolism pathway of licoisoflavone A and licoisoflavone B in vivo remain unclear, they likely share similar biotransformation pathways due to their classification as isoflavones. Licoisoflavone A and licoisoflavone B are commonly used as inhibitors of lipid peroxidation. They have been proved to exhibit antioxidant and antibacterial effects. Additionally, licoisoflavone A effectively inhibits myocardial hypertrophy, promotes non-specific immunity, and scavenges free radicals, thereby providing significant cardiovascular protection. At the same time, licoisoflavone B has been demonstrated certain anti-tumor effects. The wide range of pharmacological activities exhibited by licoisoflavone A and licoisoflavone B highlights their potential for various therapeutic applications.
Introduction
Licorice, a traditional Chinese medicinal herb and a natural flavoring agent, harbors a diverse array of chemical constituents. Among them, natural flavonoids in glycosidic form–widely present in vegetables and fruits—constitute essential phytochemicals in human diets. Licoisoflavone A and licoisoflavone B, two structurally analogous compounds derived from licorice (Figure 1), have attracted increasing scientific interest due to their broad spectrum of pharmacological properties. Both licoisoflavone A and licoisoflavone B exhibit significant anti-inflammatory, immunomodulatory, antitumor, and antioxidant activities, underscoring their potential application in preventing and treating various pathologies such as allergic disorders and cancers. 1 Nevertheless, their bioactivities, underlying mechanisms, and toxicological profiles remain to be fully elucidated.
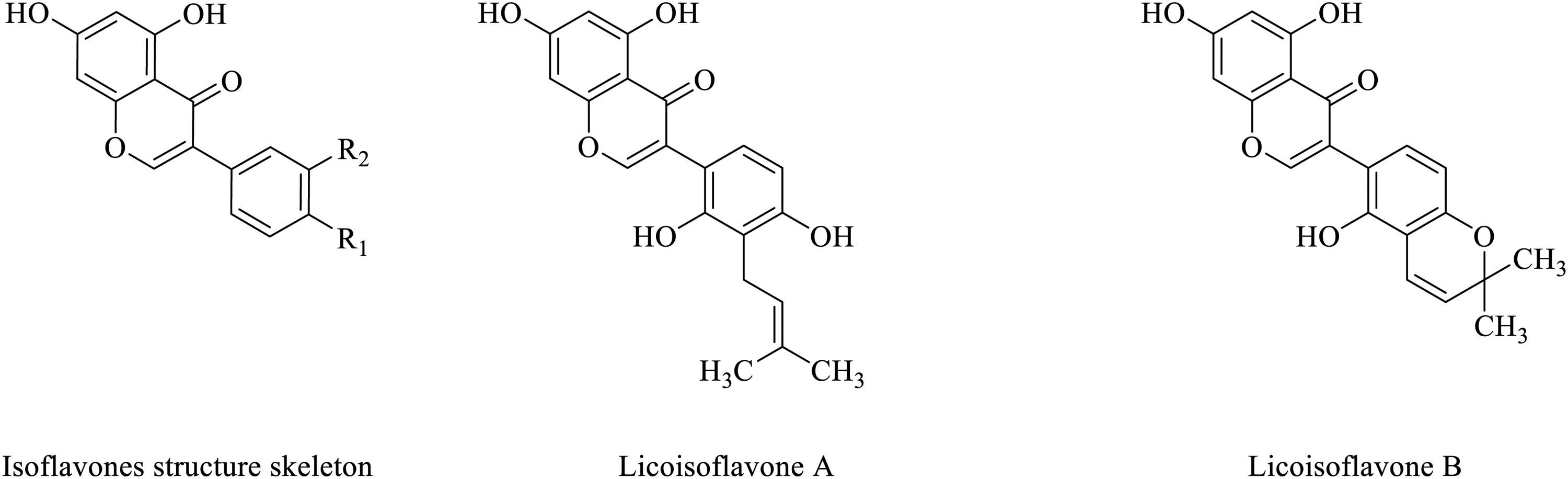
Chemical Structures of Various Compounds.
Current research on licoisoflavone A and licoisoflavone B remains largely embedded in broader investigations of licorice extracts, with studies focusing on the purified compounds still relatively limited. This chapter aims to synthesize and summarize contemporary findings regarding the in vitro and in vivo biological effects of these two distinct commercial applications and relevant patents pertaining to licoisoflavone A and licoisoflavone B, thereby highlighting their translational potential and guiding future research directions. All information collected is sourced from official databases, such as PubMed, Web of Science, China National Knowledge Infrastructure (CNKI), and X-MOL.
Resources
Licoisoflavone A and licoisoflavone B are natural compounds that can be obtained in high purity through conventional extraction methods. Licoisoflavone A and licoisoflavone B are primarily isolated from licorice, a traditional Chinese medicine derived from the dried roots and rhizomes of leguminous plants Glycyrrhiza uralensis Fisch., G. inflata Bat., and G. glabra L. Licorice is widely distributed at regions of China, including Northeast China, North China, Shanxi, Gansu, Qinghai, Xinjiang, and Shandong. It has been traditionally used to tonify spleen and qi, clear heat and toxins, resolve phlegm and relieve cough, alleviate acute pain, and moderated the effects of other medicinal compounds. It is commonly prescribed for conditions such as us spleen and stomach weakness, fatigue, palpitations, shortness of breath, cough with phlegm, epigastric and limb pain, as well as abscess and sores, and for counteracting drug toxicity.2,3
Apart from licorice, several other plant species are known to contain significant levels of licoisoflavone A and/or licoisoflavone B. These include Sophora moorcroftiana, 4 Lupinus albus (White Lupin),5,6 Echinosophora koreensis, 7 and Azorella madreporica. 8 These plants may serve as alternative sources for investigating the bioactivities and potential applications of the two compounds.
Biosynthesis Pathway and Biotransformation
Licoisoflavone A and licoisoflaovne B, both are classified as isoflavones, share common biosynthetic pathways in living organisms.9–11 Their biosynthesis originates from the phenylalanine pathway 12 (Figure 2). Glucose is enzymatically converted into phenylalanine and tyrosine, which serve as primary substrates. 13 These amino acids are then transformed into flavonoid intermediates through the catalytic actions of phenylalanine ammonia-lyase (PAL) and tyrosine ammonia-lyase (TAL). Subsequently, p-coumaroyl-coenzyme A ligase (4CL) and chalcone synthase (CHS) catalyze the formation of the central flavonoid precursor, trans-chalcone (1,3 diphenyl-2-propen-1-one). 14 Further enzymatic modifications lead to the synthesis of various flavonoid compounds, 15 including licoisoflavone A and licoisoflavone B.
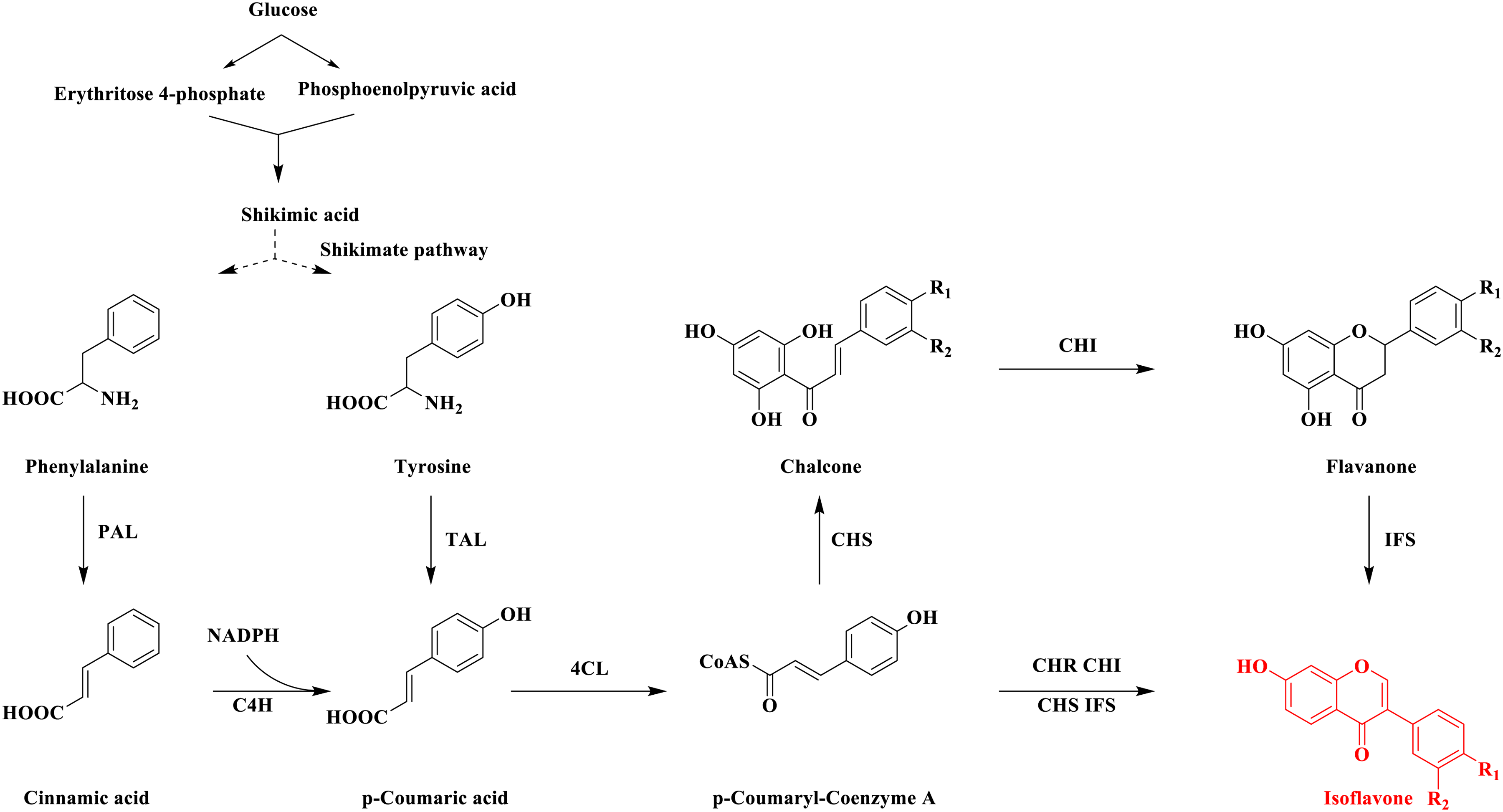
Detailed Pathway of Isoflavone Biosynthesis.
Beyond plant sources, several microorganisms—such as Escherichia coli 16 and Saccharomycetes,17–20 are also capable of producing isoflavones. However, microbial synthesis currently suffers from limited production efficiency and poor scalability compared to industrial chemical synthesis. Achieving high-yield production of specific isoflavone like licoisoflavone A and licoisoflavone B via microbial platforms remains challenging. Moreover, the biosynthetic mechanisms underlying the formation of these two compounds are not yet fully elucidated. Future studies are essential to identify the key enzymes and intermediate steps involved in their biosynthetic pathway, which may facilitate and intermediate steps involved in their pathway, which may facilitate more efficient bioproduction strategies. 21
Notably, systematic in vivo pharmacokinetic (PK) studies—evaluating absorption, distribution, metabolism, and excretion (ADME)—for licoisoflavone A and B are currently unavailable. A comprehensive understanding of their PK profiles is imperative to assess bioavailability, tissue distribution, metabolic stability, and potential accumulation. Therefore, conducting rigorous in vivo PK studies represents a critical and prioritized next step to advance these promising compounds toward clinical development.
Physicochemical Properties
Both licoisoflavone A and licoisoflavone B are isoflavones and possess similar physical and chemical properties which are shown in Tables 1 and 2.
Physical and Chemical Properties of Licoisoflavone A.

Physical and Chemical Properties of Licoisoflavone B.

Bioactivities of Licoisoflavone A and Licoisoflavone B
This section presents a comprehensive overview of the documented pharmacological activities of licoisoflavone A and licoisoflavone B. The accompanying Table 3 summarizes their key mechanisms of action—including anti-inflammatory, anticancer, antioxidant, antiviral, and cardioprotective effects—along with their associated molecular targets and signaling pathway.
Summary of the Pharmacological Activities of Licoisoflavone A and Licoisoflavone B.

Anti-Inflammatory Activity
Currently, there is no direct evidence demonstrating the anti-inflammatory activity of licoisoflavone A as a single compound. Existing research has primarily focused on chemical composition analysis and comprehensive pharmacological studies of complex formulations containing this compound. One such formulation, Huashi Baidu Decoction—a clinically approved treatment for coronavirus disease 2019 (COVID-19)—has demonstrated significant antiviral and anti-inflammatory effects. The anti-inflammatory properties are believed to arise from the synergistic action of multiple constituents, including licoisoflavone A. 22
The anti-inflammatory mechanism of Huashi Baidu Decoction may involve inhibition of phosphodiesterase type 4 (PDE4), a cyclin adenosine monophosphate (cAMP)-hydrolyzing enzyme that plays a crucial role in neutrophil activation and neutrophil-mediated inflammation during COVID-19. PDE4 is considered a promising target for anti-inflammatory drug developmet.23,24 Molecular interactions suggest that licoisoflavone A binds to the active site of PDE4 via hydrogen bonding—through its phenolic hydroxyl group and a free benzene ring hydroxyl—as well as through extensive hydrophobic interaction with PDE4 residues. These interactions collectively inhibit the catalytic activity of PDE4. 22
Although licoisoflavone A is not the most potent anti-inflammatory compound in Huashi Baidu Decoction, it exhibits favorable bioavailability and targeting properties, underscoring its potential therapeutic relevance.
Anti-Cancer Activity
Licoisoflavone A has been shown to possess potential anticancer properties, particularly in counteracting multidrug resistant (MDR)— a major obstacle in cancer treatment wherein tumor cells develop resistance to multiple chemotherapeutic agents following repeated or concurrent exposure. MDR is frequently associated with the overexpression or enhanced activity of multidrug resistance-associated protein (MRP),25,26 a transmembrane transporter responsible for efflux of anticancer drugs from cells, thereby reducing their intracellular concentration and therapeutic efficacy. By inhibiting MRP activity, licoisoflavone A effectively increased drug accumulation within tumor cells. Studies indicate that its inhibitory potency is comparable to that of indomethacin, a well-known MRP inhibitor. Through this mechanism, licoisoflavone A attenuated drug resistance and enhanced chemotherapeutic outcomes. 27
Licoisoflavone B also exhibits anticancer potential, primarily through anti-genotoxic activities. It counteracts the mutagenic effects induced by N-methyl-N-nitrosourea (MNU), a deoxyribonucleic acid (DNA) alkylating agent known to cause cancer in organs such as the forestomach, brain, and nervous system. By reducing DNA damage and mutation frequency, licoisoflavone B demonstrates protective effects against carcinogen-induced genotoxicity, highlighting its promise in chemoprevention. 28
Furthermore, a patented anti-skin cancer composition comprising licoisoflavone B, neoglycyrrhiza phenol, and glycyrrhiza chalcone A has been shown to synergistically inhibit skin cancer cell growth via multi-target mechanisms. Specially, licoisoflavone B contributes to this effect by suppressing the p38 mitogen-activated protein kinase (MAPK) pathway. This pathway, activated by stimuli such as ultraviolet (UV) radiation, inflammatory cytokines, and oxidative stress, played a complex role in skin carcinogenesis. Inhibition of p38 MAPK signaling helps modulate key cellular process—including proliferation and differentiation—thereby restraining the progression of skin cancer.
Antioxidant Activity
Licoisoflavone A and licoisoflavone B exhibit significant antioxidant properties, primarily through the inhibition of lipid peroxidation—a process critically involved in oxidative stress-related pathologies. Bothe compounds confer cellular protection against oxidative damage, with licoisoflavone B demonstrating superior efficacy in suppressing superoxide anion-induced lipid peroxidation compared to licoisoflavone A at equimolar concentration, suggesting an enhanced protective role in mitigating reactive oxygen species-mediated damage. 29
The antioxidant mechanism of licoisoflavone A involves modulation of the cytochrome 3A4 (CYP3A4) and nuclear factor erythroid 2-related factor 2 (Nrf2) pathways. 30 In murine models of paraquat-induced acute lung injury, licorice extract—containing licoisoflavone A—ameliorates pulmonary edema and fibrosis, reduces oxidative stress markers such as malondialdehyde (MDA) level, enhances superoxide dismutase (SOD) activity, and preserves lung tissue integrity. 31 These protective effects are mediated through upregulation of CYP3A4 and Nrf2 expression, which promotes detoxification, reduces paraquat accumulation, and improves hepatic and renal function, ultimately increasing survival. Among the five bioactive licorice compounds linked to this activity, licoisoflavone A was particularly implicated, as evidenced by diminished efficacy upon CYP3A4 inhibition or Nrf2 silencing.
Notably, the antioxidant effects of licoisoflavone A operate through a defined molecular pathway: Nrf2 activation, followed by reduced oxidative stress markers and enhanced SOD activity. The critical role of licoisoflavone A in paraquat detoxification is further evidenced by invention patent CN109953985A, entitled “Application of the combination of liquorice drugs for the treatment of paraquat toxicity”, which provides a comprehensive formulation of a licorice-based antidote containing this compound.
Antiviral Activity
Licoisoflavone A demonstrates notable antiviral activity and is a key bioactive component in traditional Chinese medicinal formulations used clinically to treat viral infection. As mentioned in section 4.1 above, it serves as one of the primary active ingredients in Hushi Baidu Decoction—a compound decoction that is clinically utilized for the treatment of COVID-19—known for its antiviral and anti-inflammatory properties. Molecular docking studies reveal that licoisoflavone A binds effectively to RNA-dependent RNA polymerase (RdRp), a critical enzyme for viral replication, via hydrogen bonding interaction with Tyr129 and Lys47 residues, thereby inhibiting coronavirus activity. 22
Licoisoflavone A also exhibits a wide therapeutic window and favorable drug properties, making it a relatively safe and promising candidate for anti-viral therapy. The anti-COVID-19 effect of licoisoflavone A is dose-dependent and characterized by a wide therapeutic window and favorable pharmacokinetic properties, underscoring its potential as a safe and promising antiviral agent.
Beyond its direct antiviral actions, licoisoflavone A exhibits immunomodulatory effects, thereby playing an antiviral role. For instance, Azorella compacta infusion, the herbal tea rich in licoisoflavone A, not only scavenges superoxide anion free radicals, but also activates the human natural killer (NK) cells and mildly stimulate T cells and granulocytes, suggesting a role in supporting innate immunity and potentially delaying immune aging. 32
Moreover, both licoisoflavone A and licoisoflavone B have demonstrated anti-human immunodeficiency virus (HIV) properties. An invention patent (Patent No.: CN105012289A) titled “Application of glycyrrhizin or its derivation in anti-acquired immune deficiency syndrome (AIDS) suggests that licoisoflavone A and licoisoflavone B may be used in combination with antiretroviral drugs to target and eliminate latent HIV-infected cells, showing potentially accelerating the eradication of the latent viral reservoir. This approach could provide a new avenue for achieving a complete cure for AIDS.
Regulating Drug Metabolism Effect
Licoisoflavone A modulates the activity of hepatic microsomal enzymes, particularly cytochrome P450 enzymes (CYP450) isoforms, which play a critical role in drug metabolism and detoxification. This compound enhances the activity of two major subtypes of CYP450, namely CYP3A4 and CYP1A2, through distinct mechanisms. For instance, CYP3A4 metabolizes a wide range of drugs such as simvastatin and clarithromycin, while CYP1A2 is involved in the metabolism of clozapine and theophylline. 33 The activity of these enzymes can be modulated by both endogenous and exogenous substances, as well as certain nuclear receptors such as pregnane X receptor (PXR) and aryl hydrocarbon receptor (AhR). When the PXR or AhR is activated, it can induce expressions of several genes, including those for drug-metabolizing enzymes and transporters. Licoisoflavone A has been reported to enhance the enzymatic activity on the two main subtypes of CYP450, namely CYP3A4 and CYP1A2, either directly or indirectly. 34 In the case of CYP3A4, licoisoflavone A not only acts both directly by binding to the enzyme and dramatically increases its activity,35–37 but also indirectly by activating the human pregnane X receptors (hPXR), as demonstrated by concentration-dependent increases in hPXR transcriptional activity in luciferase reporter assays. 38 Since CYP3A4 is a key hPXR-regulated enzyme, its enhanced expression accelerates substrate metabolism. 39 In the case of CYP1A2, licoisoflavone A does not directly enhance the activity of CYP1A2 upon contact, but increase transcription activity of AhR, which in turn promote CYP1A2 transcription and activity. The elevated activity of both CYP3A4 and CYP1A2 can result in accelerated drug clearance, potential reducing therapeutic efficacy, shortening duration of action, or leading to treatment failure.34,40
Similarly, licoisoflavone B also has the effect to cause pharmacokinetic drug interaction. primarily through inhibitory effect on specific CYP450 enzymes. It significantly and reversibly inhibits CYP2C8 and CYP2C9, while moderately inhibiting CYP2B6 via both irreversible and reversible mechanisms. 41 For the inhibition effect of licosoflavone on other enzymes in CYP450 family remain unclear and require further investigated.
Regulating the Body's Immune System
Licoisoflavone A demonstrates promising immunomodulatory properties by enhancing both innate and adaptive immune responses. Its ability to stimulate key immune effector cells suggests potential applications in supporting immune function. Azorella compacta, a natural source of licoisoflavone A, has shown to modulate nonspecific immunity in the human body by stimulating key immune cells, including NK cells, T cell, and granulocytes. 42 NK cells contribute to innate immune response through the recognition and elimination of virus-infected cells and tumor cells. T cells play a central role in adaptive immunity by orchestrating pathogen-specific immune responses, while granulocytes—such as neutrophils—mediate phagocytosis and clearance of microbial invaders. The immunostimulatory effects of Azorella compacta suggests that licoisoflaovne A may enhance immune function, though further research is necessary to definitively establish its immunomodulatory activity and mechanisms.
Licoisoflavone B has limited direct evidence for immunomodulation. Studies docus mainly on its anticancer and antioxidant properties. Potential immune effects may be inferred from structural similarity to other flavonoids, but specific mechanistic studies are lacking. No dedicated immune references are currently available.
Antibacterial Activity and Antifungal Activity
Licoisoflavone A and licoisoflavone B exhibit broad and potent antimicrobial activities against a spectrum of pathogenic bacteria and fungi, demonstrating significant potential in overcoming antibiotic resistance and supporting therapeutic development.
Licorice extract exhibits broad-spectrum inhibitory activity against various gram-positive and gram-negative bacteria, such as Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Candida albicans, and Bacillus subtilis. 43 Notably, licoisoflavone A demonstrates potent efficacy against E. coli with an IC50 of 0.03 μg/mL, 8 suggesting its potential role in preventing enteric infections and maintaining gastrointestinal health. Meanwhile, licoisoflavone B shows significant inhibitory activity against Helicobacter pylori—including strains resistant to clarithromycin and amoxicillin—highlighting its potential in managing peptic ulcers and reducing gastric cancer risk. 44 Furthermore, licoisoflavone B has been identified as the main antibacterial component in the traditional formula Jie-Geng-Tang (JGT), where it ameliorate S.aureus-induced acute lung injury in mice. 45 Likewise, an invention patent named “Antimicrobial Compounds of Plant Origin and their Application” punished in 2021 (Patent No.:CN114796188A) pertains to 31 compounds, including licoisoflavone A derived from plants that exhibit antimicrobial properties against Gram-negative bacteria carrying the mobilized colistin resistance (MCR) gene. The MCR gene produces an enzyme called phosphoethanolamine phosphotransferase, which modifies lipopolysaccharides on the bacterial cell walls. This modification results in resistance to polymyxin-based antibiotics such as Colistin. Along with other compounds, licoisoflavone A shows the ability to overcome this resistance mechanism. Importantly, these compounds do not exhibit hemolytic or cytotoxic effects and are considered safe for use. Overall, the invention patent highlights the antimicrobial potential of licoisoflavone A and other identified compounds, providing opportunities for their application in combating drug-resistant bacteria and addressing various bacterial infections and contamination.
Furthermore, both licoisoflavone A and licoisoflavone B have been reported to exhibit promising antifungal activity. Studies have demonstrated their effectiveness against two fungi species, Colletotrichum gloeosporioides and Cladosporium cladosporioides. 46
Protecting Cardiovascular Effect
Licoisoflavone A demonstrates significant potential in cardiovascular protection, primarily through promoting angiogenesis and inhibiting myocardial hypertrophy. Although licoisoflavone B has not been directly studied for cardiovascular benefits, its structural similarity suggests it may possess relevant biological activities worthy of further investigation.
Angiogenesis—the formation of new blood vessels from pre-existing ones—is crucial in physiological and pathological processes, such as tissue repair and the development of collateral circulation. The angiogenic potential of licoisoflavone A was investigated through an analysis of the active ingredients in Tongmai Yangxin Pills (TMYXPs), a traditional medicine used for regulation and maintenance of cardiovascular health. By screening compounds that stimulate human umbilical vein endothelial cells (HUVECs) proliferation under hypoxic conditions, licoisoflavone A, one of the active chemical components of TMYXP, was found to enhance HUVECs growth, implying its protective effect against damage caused by oxygen deprivation. These angiogenic properties were further corroborated by in vivo studies using transgenic zebrafish. 47
In addition, licoisoflavone A demonstrates anti-hypertrophic activity by activating sirtuin 3 (Sirt3), a histone deacetylase that acts as a key regulator of cardiac hypertrophy.48,49 Sirt3 activation results in improved mitochondrial function and reduced cardiomyocyte apoptosis, inducing cardioprotective effects. Myocardial hypertrophy, characterized by thickening of the heart muscle, is often associated with conditions such as hypertension and heart failure. Reduced Sirt3 activity or expression has been closely linked to the development and progression of cardiac hypertrophy. 24 In an initial study using TMYXPs, licoisoflavone A has been found to significantly attenuate angiotensin II-induced cardiac hypertrophy in a dose-dependent manner and emerged as the most potent anti-cardiac hypertrophy component among the chemical components tested. Further cellular experiment has confirmed that licoisoflavone A suppresses hypertrophic agonist phenylephrine-induced hypertrophy in cardiomyocytes by upregulating Sirt3 expression. These results indicate that licoisoflavone A has the potential to serve as a Sirt3 activator and exert therapeutic effects against myocardial hypertrophy.
A systematic summary of the major pharmacological activities, mechanisms of action, and evidence levels for licoisoflavone A and licoisoflavone B is provided in Table 4. It clearly compares the efficacy of both compounds across various domains. The table lists corresponding molecular targets and experimental models (from molecular docking and in vitro assays to in vivo studies). Serving as a valuable reference, it offers a quick overview of their therapeutic potential and research depth, highlighting promising avenues for future investigation.
Comprehensive Summary of Pharmacological Activities of Licoisoflavone A and Licoisoflavone B.

Patents
According to the search results from the official website of the China National Intellectual Property Administration (CNIPA), there are currently eight Chinese patents related to licoisoflaovone A and/or licoisoflavone B (Table 5). Among these patents, two are specifically pertain to licoisoflavone A, three pertain to licoisoflavone B, and the remaining three patents involve both licoisoflavone A and licoisoflavone B.
Patents Related to Licoisoflaovone A and/or Licoisoflavone B.

The patents related to licoisolavone A and/or licoisoflavone B are primarily invention patents. Many of them focus on medicinal or agriculture applications. Some patents specifically emphasize the therapeutic effects of licospflavone A and/or licoisoflavone B in treating certain diseases. One notable example is the patent named “Application of glycyrrhizin or its derivatives in anti-AIDS”, which suggests that glycyrrhizin and its derivatives (including licoisoflavone A) may possess therapeutic potential in the treatment of AIDS. These patents provide valuable information and insights into the potential applications of both licoisoflavone A and licoisoflavone B.
Patents related to licoisoflavone A and/or licoisoflavone B have been applied for not only in China but also internationally. For instance, the patent number WO2014042426A1 represents an invention filed through World Intellectual Property Organization (WIPO) under the Patent Cooperation Treaty (PCT). The “WO” prefix in the patent number indicates its international state. Such international patent applications with WO numbers are commonly used for conducting global patent searches and exploring intellectual property rights across multiple countries.
The international patent activity underscores the growing significance of licoisoflavone A and licoisoflavone B as natural compounds derived from Chinese medicinal materials within the global scientific and intellectual property landscape.
Limitations
Despite providing a comprehensive overview of the pharmacological activities, mechanisms, and potential applications of licoisoflavone A and licoisoflavone B, current research still presents several limitations and shortcomings:
Insufficient depth in mechanistic studies:
Current understanding of the pharmacological mechanisms of licoisoflavone A and B remains preliminary. Most evidence derives from studies on complex formulations or crude extracts, and these is a lack of systematic investigation into the molecular mechanisms of the individual compounds. Their specific signaling pathways, target proteins, and gene regulatory networks—particularly in anti-inflammatory and immunomodulatory activities —are not yet fully elucidated.
Limited in vivo data:
Although in vitro experiments demonstrate broad bioactivities, high-quality in vivo pharmacodynamic, pharmacokinetic, and toxicological studies are still relatively scarce. Most existing animal studies are based on extracts or multi-component preparations, which makes it difficult to accurately evaluate the efficacy, safety, and metabolic profiles of licoisoflavone A and B when administered alone.
Unclear structure-activity relationships:
Although structural modification has been proposed to optimize their physicochemical and pharmacological properties, research on the key pharmacophores of these compounds and their correlation with biological activities remains limited, thereby hindering rational drug design efforts.
Lack of clinical research support:
There are currently no clinical trial data on the individual application of licoisoflavone A or licoisoflavone B in humans. Their effectiveness, appropriate dosing, and potential adverse effects in actual disease treatment remain to be validated.
Challenges in bioavailability and formulation:
Both compounds exhibit poor water solubility and low bioavailability. Currently, there are no mature formulation strategies to address these issues, which impedes their translation into clinical applications.
Inadequate assessment of toxicity and drug-drug Interactions:
Particularly for licoisoflavone A, its modulatory effects on CYP450 enzymes suggest potential drug-drug interaction risks. However, research in this area remains limited and lack systematic evaluation.
In summary, further in-depth mechanistic studies, standardized preclinical and clinical evaluations, along with structural optimization and advanced delivery strategies, are essential to advance the development of these two compounds.
Perspectives
Licorice is a well-known herbal medicine in China with extensive clinical application. Licoisoflavone A and licoisoflavone B, which are among the main active ingredients of licorice and numerous traditional Chinese medicine preparations, have demonstrated significant potential as multifunctional compounds. They exhibit a broad spectrum of pharmacological activities, including anti-inflammatory, antioxidant, anticancer, antibacterial, antifungal, antiviral, and cardioprotective effects.
Further investigation into their specific molecular mechanisms—such as interactions with cell signaling pathways, target proteins, and gene expression regulation—would substantially advance the understanding of their pharmacological behavior and therapeutic potential. Moreover, systematic analysis of the active functional groups in their chemical structures could provide a foundation for structural optimization or modification. Retaining the core active scaffold while introducing targeted structural improvements may lead to compounds with enhanced physicochemical properties, increased pharmacological potency, and reduced toxic side effects.
This review highlights the promising prospects of licoisoflavone A and B in future medical development, particularly as candidate molecules for the treatment of inflammatory diseases, cancers, and cardiovascular disorders. It also provides a theoretical basis and structural insights for the development of novel derivatives, thereby supporting drug innovation based on natural products and promoting the integration of traditional Chinese medicine with modern pharmaceutical research.
Footnotes
Author Contributions
X.C. prepared the paper. W.S.C. supervised the project and revised the paper. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the University of Macau (grant number MYRG-GRG2023-00212-ICMS-UMDF and MYRG-GRG2024-00258-ICMS-UMDF), and the Young Talent Support Project of Guangzhou Association for Science and Technology (grant number QT2024-048).
Universidade de Macau, Young Talent Support Project of Guangzhou Association for Science and Technology, (grant number MYRG-GRG2023-00212-ICMS-UMDF, MYRG-GRG2024-00258-ICMS-UMDF, QT2024-048).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
