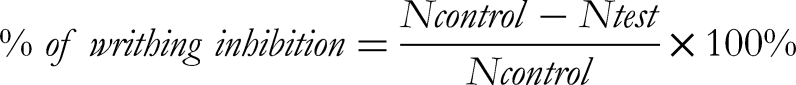Abstract
Objective
The investigation aimed to reveal in-vivo therapeutic efficacy of the D. trifoliata's leaves ethyl acetate extract (EACE) in mice model in addition to the isolation and detection of phyto-molecules.
Methods and materials
Phyto-molecules were isolated through chromatographic techniques and detected via nuclear magnetic resonance (NMR) approaches. Tail immersion, and acetic acid-induced writhing method were used for central and peripheral analgesic, respectively, whereas hypoglycemic and CNS depression efficacy were evaluated by oral glucose tolerance test, and thiopental-induced test, respectively. The 200, 400, and 600 mg/kg b.w. doses of EACE were used for all in vivo experiments.
Results
Four different compounds (4-hydroxy benzoic acid
Conclusion
EACE showed some specific in vivo efficacy in dose dependent manner such as analgesic and diarrhea. Further extensive investigation, is needed to identify the responsible lead compounds.
Introduction
Plants of the Fabaceae family are well known for their medicinal values, such as antibacterial, antifungal, antihypertensive, antiviral, insecticidal, anticancer, and so on. 1 Among the various genera of this family (having around 65 species), Derris is remarkable for its pharmacology values. For example, the stem of D. scandens has anti-inflammatory effect, as it is popular for the treatment of joint pain and osteoarthritis, along with anti-dysentery medicine.2,3 Furthermore, D. taiwaniana is popular for its therapeutic advantages against cancer and infertility, 4 while D. elliptica is well known in Asian countries for its insecticidal properties. 5 Additionally, the Derris genus is widely known as the blood glucose lowering agent. For example, D. elliptica has shown potential in maintaining β-cell performance and decreasing blood glucose levels in a study on streptozocin-induced diabetic rats. 6 Another species, D. reticulata, also lowered the blood glucose levels in alloxan-induced diabetic rats. 7 Similarly, recent studies demonstrated that some species of the Derris genus, including D. scandens, 8 D. indica, 9 and D. robusta 10 are popularfor their hypoglycemic aspect. On the other hand, a review of the Derris genus reported several phytochemical studies identifying numerous bioactive isoflavonoids and rotenoids, which may contribute to the cytotoxic, antimicrobial, anti-inflammatory, and antihyperglycemic activities. 11
Derris trifoliata Lour, a prominent Derris species, has significant therapeutic benefits. For instance, in Palau, it has been applied for dysentery, while in Papua New Guinea, it has been used for malaria. 12 In Bangladesh, it is traditionally used against rheumatoid arthritis and dysmenorrhea. 13 A variety of bioactive compounds, including flavonoids, rotenoids, 14 phenolics, and alkaloids 15 have been isolated from various parts of D. trifoliata.16,17Furthermore, a previous investigation showed the ethanolic extract of D. trifoliata increased the onset of sleeping and total sleeping time at around 500 mg/kg body weight, and at a 1000 mg/kg body weight dose, and a significant effect on ambulation and emotional defecation. 18 However, it was a very preliminary study, and further literature on the CNS impact of D. trifoliata leaves is not available.
Despite extensive phyto-pharmacological research on various parts of Derris trifoliata—including its fruits, bark, and seeds—systematic investigations into the in vivo efficacy of the plant's leaves remain scarce, particularly in the Bangladesh region. This gap is noteworthy, as leaves often contain distinct phytoconstituents that may contribute to unexplored therapeutic effects. Therefore, the present study is designed to evaluate the in vivo pharmacological potential of D. trifoliata leaves. According to the existing literature, D. trifoliata has traditionally been recognized for its rheumatoid and anti-diarrheal properties.12,13 In line with these traditional claims, its central and peripheral analgesic and antidiarrheal activities were examined through in vivo experimental mouse models. Although hypoglycemic activity of D. trifoliata has not been well-documented, structurally similar secondary metabolites, particularly flavonoids and rotenoids are present in this species, have been reported in other Derris plants to possess hypoglycemic potential. Thus, investigating this unreported property was scientifically reasonable, aiming to uncover an additional therapeutic facet of D. trifoliata. In addition, only one prior study has suggested potential CNS depressant activity of D. trifoliata, 18 to validate and extend this preliminary evidence, further CNS depressant experiments are performed in the current study. Finally, to support the pharmacological findings with the presence of bioactive molecules, phytochemical isolation and detection were conducted using multiple chromatographic techniques and nuclear magnetic resonance (NMR). The investigation may be beneficial for the possibility of discovering a new therapeutic potential against multiple widely available ailments.
Materials and Methods
Plant Materials
The fresh leaves of D. trifoliata were collected from the Sundarban in February 2022, the south-western region of Bangladesh. A taxonomist of the Bangladesh National Herbarium (BNH) in Dhaka, Bangladesh, identified and confirmed the plant sample. A voucher specimen has been preserved for future reference (accession number DACB 135402).
Reagents and Chemicals
Analytical grade reagents and chemicals were used in this investigation. Normal saline, diclofenac sodium, morphine, glibenclamide, diazepam, and loperamide were purchased from the Square Pharmaceuticals, while Incepta and Eskayef pharmaceuticals supplied thiopental sodium, and castor oil, respectively. Moreover, suspending agent Tween 80 was procured from Sigma-Aldrich.
Drying, Grinding, and Extraction
The freshly harvested leaves were promptly washed with clean water to eliminate any dirt or unwanted materials. Then, the leaves were allowed to air dry for several days under the shade until becoming brittle at ambient temperature (28 ± 2)°C to protect heat-sensitive phyto-constituents. The desiccated leaves were turned to coarse powder by utilizing a high-capacity grinding apparatus, (Grinder IKA®-WERKE, IKA MF10). Then, the powdered materials (300 gm) was macerated with 2.0 L of ethyl acetate in a sealed amber container for 15 days, with occasional shaking. Consequently, the extracted liquid was filtrated and subjected for the evaporation through a Rotavapor (BUCHI Rotavapor, Switzerland) under reduced pressure at around 45 °C to obtain a concentrated crude extract (EACE) (Figure S1). 19
Fractionation
The leaves extract (5 g, EACE) was sequentially partitioned into four fractions using a modified Kupchan method (Figure S1). 20 Firstly, the 5 gm extract EACE was dissolved in 90% methanol in aqueous solution for the partitioning. Then, the mixture was sequentially fractionated with n-hexane (DTHE), chloroform (DTCF), ethyl acetate (DTEA), and aqueous in order to their polarity (Figure S1). 20 Finally, all fractions were concentrated by evaporation and stored at low temperature until further use. The flowchart of the fractionation and their amounts were described at the figure S1 and table S1.
Column Chromatographic Separations
For the separations of secondary metabolites, the DTHE and DTCF were further fractionated using column chromatography (size exclusion chromatography), while lipophilic Sephadex LH-20 was the stationary phase, and solvent gradient system has been employed as mobile phase. Elution was performed initially with a solvent system of n-hexane:chloroform:methanol (2:5:1), followed by chloroform:methanol gradients. The different types of solvent systems that have been sequentially applied according to their polarity through the stationary phase for eluting and separation (Table S2). These column chromatography separation generated more than 130 resultant fractions from the both DTHE, and DTCF fractions that have been described in (Table S2).
Thin Layer Chromatographic (TLC) Separations and Purifications
The resulting fractions were analyzed and screened on TLC plates (aluminum plates coated with silica gel F254, 20 × 20 cm), where silica gel served as the stationary phase and various solvent systems at different concentrations were used as the mobile phase. A brief description of the solvent types and strengths applied for screening on the TLC plates is provided in the table S3. Initially, the TLC plates were observed under UV light at both short (λ = 254 nm) and long (λ = 366 nm) wavelengths to check for fluorescence quenching. Thereafter, the plates were heated for 2–3 min at 105 °C after spraying with 1% vanillin–sulfuric acid solution to detect the presence of colored compounds. Fractions showing identical patterns on the TLC plates were combined, which ultimately yielded sub-fractions. The total number of sub-fractions is presented in the table S3.
Finally, preparative TLC techniques were employed to purify and isolate phytomolecules from the sub-fractions. A total of four compounds were purified in this investigation. Compound A was isolated from the F-7 sub-fraction of DTCF (35% ethyl acetate in toluene) (Table S3). Similarly, compounds B and C were isolated from the F9 and F11 sub-fraction of DTCF using 5% methanol in chloroform (Table S3). On the other hand, compound D was obtained from the F2 sub-fraction of DTHE with 10% ethyl acetate in toluene (Table S3).
Molecular Detection Through Nuclear Magnetic Resonance (NMR)
For the structure elucidations, 600 MHz Bruker NMR machine has been engaged, where deuterated solvents CDCl3 and CD3OD were used to dissolve purified and isolated four phyto-molecules. After generating 1H-NMR, the spectrum data and chemical shift were calculated and compared with previous existing literatures.
Experimental Animals
Swiss albino mice (4 to 5 weeks, 25-30 gm) were purchased from the International Center for Diarrheal Diseases and Research in Bangladesh (ICDDRB). They were kept in cages (polypropylene) for two weeks at the as usual environment (25 ± 1 °C, 55 ± 0.5% RH, and 12-h light/dark cycle) before commencing the study. At these times, ICDDRB's standard food and water were supplied as their meal. All relevant precautions to minimize pain were ensured.
Dosing
As negative control, 1% tween 80 solution which was made by normal saline was employed to Group I (negative control). Testing materials (Group III-V) were suspended in 1% tween 80 solution by a vortex mixture and applied through oral route using a specialized feeding needle. Doses in the range of 200–600 mg/kg have been widely employed in previous similar in vivo studies on plant extracts.21,22 Furthermore, these doses were selected based on preliminary acute toxicity testing following OECD guidelines, which demonstrated no signs of mortality or observable adverse effects in mice more than 800 mg/kg dose. 23 Thus, three different doses including 200, 400 and 600 mg/kg of EACE were selected for the in vivo investigations.
Animal Groups
Based on the several past studies, the number of mice per group was five (n = 5) has been determined.24,25 In the in vivo investigation, a total of 25 mice were divided into five distinct groups (Group I–V). The five distinct groups are negative control (Group I), positive control/standard (Group II), 200 mg/kg b.w. dose of EACE (Group III), 400 mg/kg b.w. dose of EACE (Group IV), and 600 mg/kg b.w. dose of EACE (Group V).
Analgesic Activity
Central Analgesic Property
The central analgesic activity of EACE was assessed using the tail immersion experiment (a thermal technique) as per the previous studies.
26
Diclofenac sodium (5 mg/kg b.w.) was delivered subcutaneously as a positive control (Group II). The experimental animals’ tail were sub-merged in 55 °C hot water. Following this, the mice's latency period or pain reaction time (PRT) was recorded at 0, 30, 60, and 90 min after administration of the positive and negative control drug and test samples.
Peripheral Analgesic Property
An acetic acid-induced writhing approach was applied to determine the peripheral analgesic efficacy of EACE followed by the previous methods.
26
Group II received diclofenac sodium (50 mg/kg b.w.) as a standard drug. Each mouse was intra-peritoneal administered 1% glacial acetic acid (0.1 mL) in normal saline to generate visceral aching. The number of writhing in group I-V was recorded for 10 min following an intra-peritoneal administration of acetic acid. The percentage of writhing inhibition was computed as follows:
Where N = mean abdominal writhing for each group
Hypoglycemic Activity
The oral glucose tolerance test was used to evaluate the hypoglycemic effects of EACE as per the previous study.26,27 All of the animals were fasted for eighteen hours before the experiment. A one-touch glucometer (Bioland G-423S) was employed to determine the blood glucose level at zero hours. After that, each group was given their specified medication orally. Group II had the standard medication glibenclamide (10 mg/kg b.w.). All groups received an oral 10% glucose solution (2 mg/kg b.w.) after an hour. To measure blood glucose levels, blood samples were taken from each mouse's tail vein 30, 60, and 90 min after the glucose was administered. The hypoglycemic activity was quantified by calculating the percentage reduction in blood glucose levels through following equation.
Where A = Levels of blood glucose of the control group (Group I), and B = Blood glucose level of experimental or standard drug administering groups.
Antidiarrheal Efficacy
The anti-diarrheal investigation was assessed by employing the castor oil-induced diarrhea technique as per the prior study.
28
Prior to the trial, all animals were starved for 18 h. The standard medication loperamide (50 mg/kg b.w.) was suspended into the 1% Tween-80 solution and administered orally. After one hour of the administering, all of the mice received fresh castor oil (1 mL) to stimulate diarrhea. Each mouse was remained in individual crates coated with paper towels. The total count of diarrheal feces of every mouse was recorded after four hours of castor oil administration. The findings of the experiment were compared to both negative and positive control groups. The percentage of reduction of diarrheal feces was determined through the below formula:
CNS Depressant Activity Investigation
The precisely weighed extract was quantified and dissolved in 1 mL of distilled water to provide doses of 200 and 400 mg/kg b.w. Subsequently, a minimal quantity of Tween-80 (a suspending agent) was employed to triturate the samples uni-directionally. The negative control group received distilled water (0.1 mL/mouse, p.o) while the positive control group received diazepam (0.5 mg/kg, i.p). Following 30 min of treatment administration, each mouse was treated with thiopental sodium (40 mg/kg, i.p.) to induce sleep. They were monitored by placing each in different chambers for the latent period (time between thiopental sodium administrations and loss of righting reflex) as well as duration of sleeping time (time between the loss and recovery of the righting reflex) was recorded.
Statistical Analysis
The in vivo pharmacological test results are presented as mean ± SEM (n = 5) or as percentages. Statistical analyses were conducted using Microsoft Excel 2016 and GraphPad Prism (version 9.3.1). An independent t-test was used to determine statistical significance through the comparison of each treatment and standard group to the normal control group. The levels of significance were indicated by asterisks: * (p < 0.05), ** (p < 0.01), *** (p < 0.001), and **** (p < 0.0001).
Results
Detection of Isolated Compounds Through Nuclear Magnetic Resonance
The 1H-NMR spectrum (Table 1 and Figure S2) displayed two doublets in the aromatic region at δ 7.79 ppm (d, J = 8.0 Hz), and δ 6.84 ppm (d, J = 8.3 Hz) characteristic of a para-disubstituted benzene ring. Comparison of these spectral features with reported literature data supports the identification of compound
Isolated and Detected Phyto-Molecules from the Ethyl Acetate Leaves Extract of D. trifoliata Through Nuclear Magnetic Resonance Approache.

The 1H NMR spectrum (Table 1 and Figure S3) of the compound
The 1H NMR spectrum (Table 1 and Figure S4) of the isolated compound
The 1H NMR spectrum (Table 1 and Figure S5) of the isolated compound
Analgesic Activity
Central Analgesic Activity
Table 2 illustrates the central analgesic activity of the EACE as evaluated by the tail immersion method in mice. Reaction times were measured at 30, 60, and 90 min following administration of either the extract, standard drug, or negative control. The standard group treated with diclofenac sodium demonstrated significantly prolonged reaction times, increasing progressively from 11.74 ± 0.22 s at 30 min to 23.00 ± 0.44 s at 90 min. Among the experimental doses, Group III showed a moderate elevation (7.64 ± 0.13 s at 30 min, 8.62 ± 0.19 s at 60 min, and 8.49 ± 0.44 s at 90 min). The 400 mg/kg dose resulted in a peak effect at 90 min (11.29 ± 0.39 s), while the 600 mg/kg dose produced a statistically significant increase in response time (14.07 ± 0.44 s at 90 min) (Table 2). These findings suggest that the ethyl acetate extract of D. trifoliata leaves possesses significant central analgesic activity in a dose- and time-dependent manner.
Central Analgesic Activity of the D. trifoliata Leaves Ethyl Acetate Extract Through Tail Immersion Approach.

Peripheral Analgesic Activity
As shown in Table 3, positive control (Group II) produced the highest percentage inhibition of pain (83.96%, ***p < 0.001). Among the test groups, EACE demonstrated a dose-dependent effect. At 200 mg/kg, the extract produced 34.91% inhibition (**p < 0.01), which increased to 58.49% (**p < 0.01) at 400 mg/kg and reached 65.03% (***p < 0.001) at 600 mg/kg. These findings indicate that EACE possesses significant analgesic activity, though comparatively lower than the standard, and that efficacy increases with higher doses.
Peripheral Analgesic Potency of the D. trifoliata Leaves Ethyl Acetate Extract Through Acetic Acid-Induced Approach.

Anti-Diarrheal Efficacy
The antidiarrheal activity of the EACE was evaluated and presented at Table 4. Among the EACE-treated groups, the 600 mg/kg dose (Group V) exhibited the greatest inhibitory effect (82.20%), followed by the 400 mg/kg (78.81%) and 200 mg/kg (69.49%) doses, while the standard (loperamide) displayed 95.76% inhibition (Table 4). Thus, the outcome indicated a dose-dependent antidiarrheal activity.
Anti-Diarrheal Potency of the D. trifoliata Leaves Ethyl Acetate Extract Through Acetic Acid-Induced Approach.

Hypoglycemic Activity
Table 5 illustrates the anti-hyperglycemic activity of the EACE. The control group (Group I), exhibited relatively stable glucose levels throughout the study period and considered as baseline. The Group II (standard) showed a significant and time-dependent glucose reduction in plasma (4.75 ± 1.15 to 2.90 ± 0.41 mmol/L within 90 min) (Table 5). The 200 mg/kg dose (Group III) resulted in minimal glucose lowering efficacy (6.75 ± 0.78 to 6.3 ± 1.13 mmol/L). The highest dose, 600 mg/kg (Group V), produced the most notable effect among the extract groups (5.75 ± 3.17 at 30 min to 4.90 ± 0.42 mmol/L at 90 min) (Table 5). These findings suggest that the EACE leaves had a mild dose-dependent anti-hyperglycemic effect.
Hypoglycemic Potential of the Ethyl Acetate Extract of D. trifoliata Leaves Extract.

CNS Depressant Activity
Table 6 presents the sedative-hypnotic activity of the EACE in mice, as evaluated by the average onset of sleep and total sleeping time. Diazepam (Group II) significantly decreased the onset time (24.75 ± 5.43 min, p < 0.001) and markedly increased total sleeping time (225 ± 16.32 min, p < 0.001). At 200 mg/kg (Group III), the extract reduced sleep onset to 47 ± 4.92 min and increased sleep duration to 137.25 ± 10.93 min (p < 0.05). At 400 mg/kg (Group IV), the onset was further reduced to 39 ± 2.73 min (p < 0.05) and sleeping time extended to 165 ± 15.50 min (p < 0.01) (Table 6). The highest dose, 600 mg/kg (Group V), showed a significant reduction in onset time (33.75 ± 4.56 min, p < 0.01) and a sleep duration of 182.5 ± 10.50 min (p < 0.01) (Table 6).These results indicate that D. trifoliata leaves’ ethyl acetate extract exhibits moderate sedative-hypnotic activity in a dose-dependent manner.
CNS Depressant Activity of the D. trifoliata Leaves’ Ethyl Acetate Extract.

Discussion
This study investigated the phytochemical and pharmacological characteristics of the ethyl acetate extract from D. trifoliata leaves. The present work is significant, as it elucidated the phytopharmacological profile of D. trifoliata leaves from Bangladesh, which has been previously unclear, and compare it with other existing research. This study's aims was to examine the initial in vivo effectiveness in a mouse model. The current analysis revealed potential analgesic and antidiarrheal capabilities, as well as modest hypoglycemia and CNS depressive effectiveness.
The outcomes indicated four separated and detected compounds through chromatographic and 1H- NMR techniques, respectively; as per our best knowledge, the 4-hydroxy benzoic acid (compound
Herbal analgesic are considered as the alternate for synthetic medications due to its low adverse reaction.38,39 The results of current study indicate that the EACE of the plant caused a significant decrease in central and peripheral pain in mice model. One previous study of 2012 showed similar efficacy at same approach.
40
Some prior investigations demonstrated that this plant has several types of phyto-molecules such as steroid, flavonoid, reducing sugar, tannin, gum and saponin which can be found also in the sample materials.16,41 The analgesic properties of the D. trifoliata leaves are further solidified by the isolated phytomolecules; among the four isolated compounds
Diarrhea remains a major contributor to malnutrition among children below five years.
49
Comprehensively, it accounts for approximately 1.7 billion cases in children each year.
49
The use of medicinal plants and their formulations for managing diarrhea and its associated symptoms has been well-documented in traditional medicine.
50
Numerous validation studies have emphasized the therapeutic potential of these traditional remedies, including plant extracts and bioactive phytochemicals.
50
To evaluate the anti-diarrheal efficacy of D. trifoliata leaves, the widely accepted castor oil-induced diarrhea model was employed. In this model, the EACE demonstrated a dose-dependent inhibitory effect. These findings align with previous research, which reported that ethanol extracts significantly delayed the onset of diarrhea and reduced the frequency of diarrheal episodes in a similar experimental setup.
16
Moreover, the detected phyto-molecules
Throughout the years, natural therapies have been extensively employed worldwide in the treatment of CNS disorders. These botanical interventions are occasionally used as replacements for conventional drugs commonly prescribed for managing CNS conditions. Though one previous study revealed positive outcomes,
60
current investigation exhibited a moderate result. Probably, geographical location, collection time or variation of the phyto-molecules of the test materials are responsible for this outcomes. One study mentioned that saponins of this species may contribute to the spontaneous motor activity.
60
The ongoing phytochemical investigation also supports the in-vivo experimental outcomes. For instance, compound
Some previous studies postulated that Derris species have hypoglycemic potentiality.6–10,65 One investigation showed moderate hypoglycemic potency of D. trifoliata (23.45% and 29.62% glucose reduction within one hour). 66 Furthermore, another species of this genus, D. reticulata, has shown a potential antihyperglycemic effect on pancreatic cells through its antioxidant activity and inhibition of α-glucosidase. 67 Although several hypoglycemic molecules such as kaempferol, baicalein, and stigmasterol,68–70 have been isolated from the extract, the overall hypoglycemic effect of the crude extract was very mild. This discrepancy can be attributed to several factors. First, the concentrations of these active compounds in the extract may be low, insufficient to produce a strong glucose-lowering effect in vivo. Second, the presence of other non-active or antagonistic constituents in the extract may dilute or counteract the activity of the hypoglycemic compounds. Third, the bioavailability and pharmacokinetics of these molecules in the complex matrix of the extract may limit their systemic absorption and efficacy. Therefore, while the extract contains compounds with reported hypoglycemic properties, their cumulative effect in vivo appears modest due to limited concentration, interaction with other constituents, and reduced bioavailability. Further study is recommended even various in-vivo techniques to ensure the efficacy and accountable phyto-compounds from this species.
Limitations of the Investigation
The main limitation of this investigation lies in the isolation and detection of only a few bioactive molecules. A more comprehensive isolation and purification of this plant should be conducted in the future to identify the specific biomolecules which are truly responsible for the therapeutic outcomes. The biological activities reported here are based solely on preliminary plant extract studies. Therefore, further detailed biological investigations of the isolated phytochemicals are necessary to confirm their bioactive properties. Additionally, the preliminary assessment using only three extract dosages limits the establishment of a precise dose–response relationship. Another major shortcoming is the absence of in vivo safety and toxicity evaluations, as the extract doses were selected based on previous plant extract–based studies. Although this is a preliminary investigation, increasing the sample size (n ≥ 5) is strongly recommended to enhance the study's quality. A larger number of animals per group would help generate more reliable and reproducible results.
Conclusion
The current investigation mainly emphasizes the bioactive compound's isolation and the pharmacological properties through in vivo mice modelling. Four different compounds were isolated and detected, while 4-hydroxybenzoic acid (compound
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251387915 - Supplemental material for Pharmacological Activities and Phytochemical Profiling of Derris trifoliata Leaves: Isolation, Characterization, and in vivo Evaluation in Mice Models
Supplemental material, sj-docx-1-npx-10.1177_1934578X251387915 for Pharmacological Activities and Phytochemical Profiling of Derris trifoliata Leaves: Isolation, Characterization, and in vivo Evaluation in Mice Models by Mostari Alija, Md. Abdus Samadd, Najibah Nasrin, Md. Mobarak Hossain, Fahim Nakib, Masud Hassan Saimon and Md. Ashraful Islam in Natural Product Communications
Footnotes
Acknowledgments
The authors appreciate the scientific equipment support received from the Department of Pharmacy, International Islamic University Chittagong.
Ethical Clearance
The ethical committee of the State University of Bangladesh (SUB) also carefully passed the protocol and ethical procedures (Approval ID: 2024-12-09/SUB/I-ERC/006).
Authors’ Contributions
Funding
The authors received no financial support for the research.
Declaration of Conflicting Interests
The authors declare no conflicts of interest.
Statement of Human and Animal Rights
The study was done according to the rules of Federation of European Laboratory Animal Science Associations (FELASA) for ensure the ethical standard in investigations.
Statement of Informed Consent
Since there are no human subjects involved, any consent/permission is irrelevant.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.