Abstract
Background
Nourishing Blood Diuretic Formula (NBDF) is derived from the empirical formula of Prof. Fan Heng in the Department of Traditional Chinese Medicine (TCM), Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, which consists of 8 Chinese herbs, namely Abelmoschi corolla, Rhei Radix et Rhizoma, Astragali Radix, Angelicae Sinensis Radix, Poriae Cutis, Dioscoreae Rhizoma, Hirudo, Bombyx Batryticatus. This formula is designed based on the pathogenesis of chronic kidney disease (CKD) in TCM.
Objective
This study aims to investigate the efficacy and reveal the mechanism of action of NBDF for the treatment of chronic renal failure (CRF) by cell and animal experiments.
Methods
TGF-β1 protein was used to induce human renal tubular epithelial cells (HK-2) to produce fibrosis, and different concentrations of NBDF-containing serum were given to detect the levels of Fibronectin and E-cadherin in HK-2 cells of each group after drug administration. Adenine gavage was used to make a rat model of chronic renal failure, and after the groups were finished administering the drug, the blood and kidneys were collected and used to detect renal function indexes and protein expression.
Results
NBDF can significantly improve the fibrosis of HK-2 cells, and improve renal function and renal fibrosis and reduce renal injury in model rats, and its mechanism of action may be related to the regulation of TGF-β/Smad signaling pathway.
Conclusion
This study investigated the therapeutic effect and mechanism of action of chronic renal failure from cellular level and animal model, and provided experimental basis for the development of new drugs.
Keywords
Introduction
In recent years, the global prevalence of chronic kidney disease (CKD) has been estimated to be between 13% and 15%, and the number of patients with end-stage renal disease (ESRD) requiring renal replacement therapy has been estimated to be between 4.902 and 7.083 million. 1 Over time, CKD can lead to chronic renal failure (CRF) as well as cardiovascular disease,2,3 and renal fibrosis characterized by interstitial fibrosis and glomerulosclerosis is a common pathway for CKD to progress to ESRD and is the main cause of CRF.4,5 Modern medicine treats early and intermediate CKD with the aim of protecting renal function and delaying disease progression, however, once the disease progresses to the advanced stage, it is necessary to rely on dialysis or renal transplantation and other means to maintain life, and it is not possible to cure the disease thoroughly, which creates a heavy financial burden for patients, therefore, it is urgent to seek an effective treatment program. 6 Studies have pointed out that Chinese medicine has unique advantages in the treatment of CRF, which can effectively delay the progression of the disease, and plays a significant role in protecting renal function, relieving symptoms as well as improving the quality of life of patients, therefore, the development and research of new medicine from formulas with proven efficacy has a greater potential and a broader prospect.
Professor Fan Heng from the Department of Traditional Chinese Medicine at Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, developed the Nourishing Blood Diuretic Formula (NBDF) through years of clinical practice. This empirically derived formula was systematically refined through repeated clinical applications and theoretical refinement for treating CRF and chronic nephritis. Through continuous medication adjustments and rigorous practice-theory integration, the formula underwent progressive optimization before reaching its current definitive form. This formula is composed of eight herbs, including Abelmoschi corolla, Rhei Radix et Rhizoma, Astragali Radix, etc, and has been used for many years in clinical practice, which can significantly improve the levels of blood creatinine and urea, reduce urinary protein, protect renal function, improve the prognosis, and slow down the progression of chronic renal disease in patients with renal failure, but the specific mechanism of its action is not clearly yet. 7 Therefore, this study intends to reveal the mechanism of action of NBDF for the treatment of CRF, providing strong evidence and theoretical basis for this formula in the treatment of CRF.
Materials and Methods
Herbs
Abelmoschi corolla, Lot No. 210401, Rhei Radix et Rhizoma, Lot No. 210501, Hirudo, Lot No. 210101, Bombyx Batryticatus, Lot No. 200701, all purchased from Bozhou Jinshaotang Traditional Chinese Medicine Drinking Tablets Co., Ltd; Astragali Radix, Lot No. G03221006-07, Angelicae Sinensis Radix, Lot No. G01201117-21, purchased from Gansu Jiuzhu Tianrun Traditional Chinese Medicine Industry Co., Ltd; Poriae Cutis, Lot No. 211101, purchased from Hubei Jurui Biotechnology Co., Ltd; Dioscoreae Rhizoma, Lot No. D22090401, purchased from Huanggang Jingui Traditional Chinese Medicine Industry Development Co., Ltd These herbs were all characterization by Professor Duan Xue-yun from Hubei University of Traditional Chinese Medicine (Wuhan, China), which conforms to the provisions of the 2020 edition of Chinese Pharmacopoeia. All storing samples were stored in the herbarium of the Hubei Institute of Traditional Chinese Medicine (Wuhan, China).
Reagents
Fetal bovine serum and trypsin EDTA solution were purchased from Gibco, USA; DMEM/F12 culture medium, PBS solution, TGF-β1 protein, all purchased from Wuhan Pricella Biotechnology Co., Ltd; Cell Counting Kit-8, purchased from GLPBIO, USA; dimethyl sulfoxide, purchased from MP Biomedicals, USA; human Fibronectin ELISA kit and human E-cadherin ELISA kit were purchased from Wuhan Elabscience Biotechnology Co., Ltd; Adenine, purchased from BioFroxx, Germany; Uremic Clearance Granule (UCG), purchased from Guangzhou Consun Pharmaceutical Co., Ltd; Losartan Potassium Tablets (LPT), purchased from Zhejiang Huahai Pharmaceutical Co., Ltd; Creatinine (CREA) kit, Lot No. 30818027H, Urea (UREA) kit, Lot No. 30511024H, Uric acid (UA) kit, Lot No. 30703025Y, all purchased from Shandong Boco Biological Industry Co., Ltd; Urinary Protein (UP) kit, Lot No. 20230808, purchased from Nanjing Jianjian Bioengineering Institute; Cleaning solution for veterinary blood cell analysis, Lot No. VQ2206015, hemolytic agent for triple group veterinary blood cell analysis, Lot No. VB2303030, all purchased from Jiangxi Tekang Science and Technology Co., Ltd; Universal Tissue Fixative, Lot 182713, purchased from Wuhan Servicebio Technology Co., Ltd; HE Staining Solution Set, Lot No. S191003, Masson Staining Solution Set, Lot No. S191006, all purchased from Wuhan Pinofide Biotechnology Co., Ltd; E-CAD antibody, Lot No. 20874-1-AP, α-SMA antibody, Lot No. 14395-1-ap, fibronectin antibody, Lot No. 66042-1-Ig, all purchased from Wuhan Proteintech Group Co., Ltd; Vimentin antibody, Lot No. CY5134, purchased from Abways; COL-1 antibody, Lot No. ab270993, purchased from Abcam; TGF-β antibody, Lot No. BY0105, Smad2/3 antibody, Lot No. AY9594, p-Smad2/3, Lot No. AY8149, all purchased from Abways; Smad7 antibody, Lot No. BD-PK0168, purchased from Wuhan Elabscience Biotechnology Co.,Ltd; β-actin antibody, Lot No. GB11001, purchased from Wuhan Servicebio Technology Co.,Ltd; goat anti-rabbit secondary antibody, Lot No. PN0046, goat anti-mouse secondary antibody, Lot No. PN0080, all purchased from Wuhan Pinofei Biotechnology Co., Ld
Instruments
ME203E/02 electronic balance, purchased from Shanghai METTLER TOLEDO Co., Ltd; BK280 automatic biochemistry analyzer, purchased from Shandong BIOBASE Co., Ltd; Epoch type enzyme labeling detector, purchased from BioTeK, USA; TEK-VET3 veterinary automatic blood cell analyzer, purchased from Jiangxi Tekang Technology Co., Ltd; MX-F vortex mixer, purchased from Servicebio Co., Ltd; D3024R benchtop high-speed freezing centrifuge, purchased from Beijing DragonLab Co., Ltd; JT-12S dewatering machine, JB-L5 embedding machine, JB-L5 freezing table, purchased from Wuhan Junjie Electronics Co., Ltd; RM2016 pathology slicer, purchased from Shanghai Leica Biosystems Co., Ltd; KD-P water bath-slide drier, purchased from Jinhua Kedi Equipment Co., Ltd; 101A-3 electric thermostaticdrying oven, purchased from Shanghai Guangdi Equipment Co., Ltd; E100 biological microscope, ECLIPSE-Ci photomicroscope, purchased from Nikon, Japan; Pannoramic SCAN Ⅱ scanner, purchased from Jinan Tangier Co., Ltd; LDZX-40 autoclave sterilizer, purchased from Shanghai Shen'an Medical Instrument Factory; HCB-1300 V vertical laminar flow clean bench, purchased from Qingdao Haier Special Electric Appliances Co., Ltd; INCO 246 carbon dioxide incubator, purchased from Memmert, Germany; JXFSTPRP-48 high speed tissue grinder, purchased from Shanghai Jingxin Technology Co., Ltd; DS-S100 digital pendulum shaker, purchased from Wuhan Sevier Biotechnology Co., Ltd; Electrophoresis tank, electrophoresis power supply, purchased from Bio-Rad, USA.
Animals
Sixty-seven healthy male SPF-grade SD rats, weighing 180~200 g, were provided by Henan Skibbes Biotechnology Co. Ltd, License No.: SCXK (Yu) 2020-0005.They were housed in the Experimental Animal Center of Hubei Provincial Hospital of Traditional Chinese Medicine, Experimental Facility License: SYXK (E) 2017-0095, room temperature: 22°C ± 3°C, relative humidity: 55%~60%, no convective wind, artificial day and night (12 h day, 12 h night). The animal experiments involved in this study were approved by the Experimental Animal Committee of Hubei Provincial Hospital of Traditional Chinese Medicine, with the approval number: E Hospital of Traditional Chinese Medicine Animal (Fu) No. 202205. The reporting of this study conforms to ARRIVE 2.0 guidelines. 8
Preparation of Nourishing Blood Diuretic Formula
According to the prescription ratio of NBDF, weighed the eight herbs including Abelmoschi corolla, Rhei Radix et Rhizoma, Astragali Radix, etc, added 10 times the amount of water and soaked for 30 min, decocted for 1 h and then filtered, repeated thrice. The filtrate was combined and left overnight, and took the supernatant to be concentrated to 1.23 g·mL−1, which was stored in the refrigerator at 4°C for spare use.
Preparation of Drug-Containing serum
Seven male SPF-grade SD rats were adaptively fed for 7 days before starting gavage administration, NBDF was given by gavage at a dose of 12.3 g·kg−1 per day (the dose administered to the rat was approximately 6.25 times the dose administered to the adult by reference to the equivalent dose conversion method), gavage at a volume of 10 mL·kg−1 for 7 consecutive days, fasting for 12 h after the last administration, anesthetize the rats with 5% urethane 5 mL·kg−1, take 6~8 mL of blood from the abdominal aorta, leave it at 4°C overnight, centrifuge it at 3500 r·min−1 for 10 min, take the upper layer of serum, inactivate it at 65°C for 30 min, and then filter it through 0.22 μm micropore membrane, and keep it at 4°C for spare parts. The serum was stored at 4°C for reserve.
Preparation of Complete medium
Take a 50 mL sterile centrifuge tube, add 90% DMEM/F12 nutrient medium, 10% fetal bovine serum and 1% penicillin and streptomycin to it, mix well, seal it and keep it at 4°C for reserve.
Preparation of TGF-β1 Solution
10 μg of TGF-β1 protein was centrifuged at 12000 r·min−1 for 45 s at 4°C, and 100 μL of sterile water was added to formulate 100 μg·mL−1 TGF-β1 solution, which was divided into octuplex tubes of 20 μL each, with a total of 5 tubes, and stored in the refrigerator at −20°C, and then taken out and added into the medium to dilute to the required concentration at the time of using.
Preparation of Drug-Containing serum medium
2 mL blank serum medium: 200 μL blank serum + 1800 μL normal medium
2 mL 10% drug-containing serum medium: 20 μL drug-containing serum + 180 μL blank serum + 1800 μL normal medium
2 mL 20% drug-containing serum medium: 40 μL drug-containing serum + 160 μL blank serum + 1800 μL normal medium
2 mL 40% drug-containing serum medium: 80 μL drug-containing serum + 120 μL blank serum + 1800 μL normal medium
Effect of Drug-Containing serum on HK-2 Cell Viability Detected by CCK-8
Collect HK-2 cells in logarithmic growth phase, adjust the density of cell suspension to 5·103 cells/mL, add 100 μL of cell suspension into each well of 96-well plate, set up 6 replicate wells in each group, and wait for the cells to be attached to the wall in the incubator, after the cells were attached to the wall, they were divided into blank control group, 10%, 20%, and 40% of drug-containing serum group, each group was treated with the corresponding concentration of drug-containing serum. Each group was treated with the corresponding concentration of drug-containing serum for 24 h. After the drug effect was finished, 10 μl of CCK-8 solution was added to each well, and the incubation was finished after 2 h. The absorbance value was measured at 450 nm on an enzyme meter, and the survival rate of the cells was calculated.
Screening of Optimal Modeling Concentration and Modeling Time of TGF-β1 by CCK-8
HK-2 cells in logarithmic growth phase were collected, the concentration of cell suspension was adjusted to 5·103 cells/mL, 100 μL of cell suspension was added to each well of 96 plates, and 6 replicate wells were set up in each group, which were placed in the incubator to wait for the cells to be attached to the wall, and the cells were divided into the following groups after attachment to the wall: blank control group, 5 ng·mL−1, 10 ng·mL−1, and 20 ng·mL−1 TGF-β1 group. After adding the corresponding concentration of TGF-β1 solution to each group for 24 h, 48 h and 72 h, 10 μl of CCK-8 solution was added to each well. The culture was ended after 2 h of incubation, and the absorbance value was measured at 450 nm on the enzyme counter, after which the cell survival rate was calculated. 9
Grouping and Administration of Cells
After confirming the optimal modeling concentration and time and that each concentration of drug-containing serum had no effect on cell survival, cells in logarithmic growth phase were taken and inoculated into sterile 6-well plates. The experimental groups were as follows: blank control group, model control group, low-dose serum-containing group, medium-dose serum-containing group, and high-dose serum-containing group. In addition to the blank control group, the rest of the groups were stimulated with TGF-β1 for modeling, and the TGF-β1 solution was withdrawn and discarded after successful modeling. Blank serum medium was added to the blank control group and model control group, while 10% drug-containing serum medium was added to the low-dose drug-containing serum group, 20% drug-containing serum medium was added to the medium-containing serum group, and 40% drug-containing serum medium was added to the high-dose drug-containing serum group.10,11
Determination of E-Cadherin and Fibronectin in Cells by ELISA
After the cells in each group were administered, waited for the cells to attach to the wall, the original medium was removed, and the cells were gently washed with PBS, then digested with trypsin, centrifuged at 1200 r·min−1 for 3 min after which the cells were collected. The collected cells were washed with PBS twice, which were broken by repeated ultrasonication. The extracts were centrifuged at 1500 g for 10 min at 4°C, and the supernatant was taken and detected by ELISA kit to detect the levels of E -cadherin and Fibronectin.
Animal Model of Chronic Renal Failure
Sixty male SPF-grade SD rats were adaptively fed for 7 days, and then 10 rats were randomly selected as the normal group and modeled by gavage of CMC-Na solution at 10 mL·kg−1 per day, while the remaining 50 rats were modeled by gavage of adenine suspension at 250 mg·kg−1 per day for 21 consecutive days.12–14 At the end of the modeling, two rats in the normal group were randomly selected as well as two rats in the modeling group. After collecting urine for 24 h, the rats were anesthetized with 5% urethane at 5 mL·kg−1, and blood was collected from the abdominal aorta to detect creatinine and urea for evaluating whether the modeling was successful or not. Compared with the normal rats, the serum levels of creatinine and urea were significantly higher in the modeling group, and HE staining showed severe dilatation of renal tubules and Masson staining showed severe fibrosis of renal tissues, indicating that the modeling was successful. 14
Grouping and Dosing of Animals
Forty-eight model rats were randomly divided into the model group, the UCG, the LPT) group, the low-dose group, the high-dose group, and the drug combination group, with eight rats in each group. The normal and model groups were gavaged with CMC-Na solution at 10 mL·kg−1, (the dose administered to the rat was approximately 6.25 times the dose administered to the adult by reference to the equivalent dose conversion method), the UCG group was gavaged with UCG solution at a dose of 2.4 g·kg−1 once daily 15 ; the LPT group was gavaged with LPT solution at a dose of 10 mg·kg−1 once daily 16 ; the low-dose group was gavaged with NBDF solution at a dose of 12.3 g·kg−1 once daily; the high-dose group was gavaged with NBDF solution at a dose of 12.3 g·kg−1 twice daily; the combination group was given 10 mg·kg−1 dose of LPT solution in the morning, and 12.3 g·kg−1 dose of NBDF solution in the afternoon, once a day for 28 d, and weighed every 3 d. 17
Specimen Collection
After 28 days of continuous drug administration, rats in each group were fasted for 24 h and placed in metabolic cages to collect the urine for 24 h, rats were anesthetized with 5% urethane 5 mL·kg−1, and 6 mL of blood was collected from the abdominal aorta, of which 4 mL was loaded into the normal serum collection tube, and 2 ml was loaded into EDTA K2 anticoagulant blood collection tube. After centrifuging the blood tube at 3500 r·min−1 for 10 min, the upper layer of serum was taken and stored at 4°C; the EDTA K2 anticoagulation blood tube was stored at 4°C; the supernatant of urine was taken after centrifuging the blood tube at 3500 r·min−1 for 10 min, and the amount of urine was recorded, and then placed in the refrigerator at −80°C for storage. The rats in each group were dissected from both kidneys, stripped of the surface membrane of the kidneys, rinsed with physiological saline, placed in tissue fixative, to be paraffin-embedded, and the remaining kidney tissues were packed in sample bottles and stored in a −80°C refrigerator for freezing.
Indicator Detection
Set up the corresponding parameters on the automatic biochemistry analyzer, prepared the working reagent according to the ratio in the instruction manual of the kit, took the sample, and determined the content of creatinine (CREA), urea (UREA) and uric acid (UA) in rat serum. Set up the corresponding parameters on the automatic blood cell analyzer, took the sample, and determined the content of hemoglobin (HGB) in rat whole blood. Urine protein (UP) concentration in urine was detected using urinary protein and 24 hUP content was calculated. 18
Paraffin Embedding and Sectioning of Renal Tissue
Kidney tissues were fixed with 4% paraformaldehyde and put into 75% ethanol, 85% ethanol, 90% ethanol, 95% ethanol, and anhydrous ethanol solutions in order to remove the water; then they were put into alcoholic benzene, xylene, and paraffin wax in order to be embedded in paraffin wax. Cut the wax blocks into 4 μm slices by a slicer after solidifying into blocks at room temperature, and then put into a 37°C oven to be dried.
HE Staining
The sections were sequentially placed in dimethyl I for 20 min, xylene II for 20 min, anhydrous ethanol I for 5 min, anhydrous ethanol II for 5 min, 75% alcohol for 5 min and washed with tap water; the sections were then stained with hematoxylin stain for 3∼5 min and washed with tap water; differentiated with differentiation solution, washed with tap water, returned to blue, rinsed with running water, then the sections were dehydrated in 85% and 95% alcohol for 5 min respectively, and stained in eosin staining solution for 5 min. Finally, the sections were dehydrated in anhydrous ethanol I for 5 min, anhydrous ethanol II for 5 min, anhydrous ethanol III for 5 min, xylene I for 5 min, xylene II for 5 min and then sealed by neutral gum, the pathological and morphological changes of the renal tissues were observed under the light microscope.
Masson Staining
The sections were sequentially placed in xylene Ⅰ for 20 min, xylene Ⅱ for 20 min, anhydrous ethanol Ⅰ for 5 min, anhydrous ethanol Ⅱ for 5 min, 75% alcohol for 5 min and washed with tap water; potassium dichromate soaked overnight and washed with tap water; ferric hematoxylin staining was performed for 3 min and washed in tap water; differentiated with differentiation solution, washed with tap water, returned to blue, running water rinse; Li Chun red acidic magenta immersion staining 5∼10 min, tap water wash; phosphomolybdic acid aqueous solution immersion staining 1∼3 min; (do not need to wash) directly into the aniline blue staining solution immersion staining 3∼6 min; with 1% glacial acetic acid for differentiation.
Expression of Fibrosis-Related Proteins in rat Kidney Tissues Detected by Immunohistochemistry
The sections were sequentially placed in xylene Ⅰ for 15 min, xylene Ⅱ for 15 min, anhydrous ethanol Ⅰ for 5 min, anhydrous ethanol Ⅱ for 5 min, 85% alcohol for 5 min, 75% alcohol for 5 min, washed with distilled water; add appropriate amount of citric acid repair solution to heat the repair, and cooled to room temperature; incubated the sections with 3% H2O2 at room temperature for 20 min, and washed with PBS; added drops of serum to the tissue, and incubate for 30 min at 37°C incubation for 30 min for closure; added primary antibody (1:100 dilution) droply, incubated at 4°C overnight; dropwise addition of secondary antibody (1:100 dilution), incubated at 37°C for 1 h; dropwise addition of DAB working solution to develop the color, rinsed with tap water; stained with hematoxylin dye for 3~5 min, rinsed with water to remove the excess hematoxylin dye on the tissues, and the slides were put into 0.5% hydrochloric acid alcoholic differentiating solution when the nuclei turned blue. The slides were put into 0.5% hydrochloric acid alcohol differentiation solution for 1~2 s, and then put into water immediately foe rinsing. The slides were put into the anti-blue solution, immersed for 3~5 s, and then rinsed in water; dehydrated, transparent, sealed, and microscopically examined. The percentage of positive area of each protein in the tissue sections was determined and calculated by Image J software.
Western Blotting
The rat kidney tissue was rinsed 2∼3 times with PBS buffer to remove blood stains on the kidney surface, and the kidney tissue was cut into small pieces and placed in a homogenizer. Tissue protein extractor 10 times the volume of the tissue was added (protease inhibitor was added within minutes before use), and the homogenate was thoroughly homogenized in an ice bath. The homogenate was transferred to a centrifuge tube, shaken, and ice-bathed for 30 min, during which time the homogenate was repeatedly blown with a pipette to ensure complete lysis, then centrifuged at 12,000 r·min−1 for 15 min at 4°C, the supernatant was collected, which was the total protein solution. For each sample, 5 times loading buffer was added to the total protein solution, vortexed and mixed, heated at 98°C for 10 min for protein denaturation. According to the molecular weight of the protein, 10% separating gel and 5% concentrating gel were prepared, and a clean glass plate was fixed, then the pre-prepared separating gel was added, and water was added to liquid seal to remove air bubbles. After the gel was solidified, used filter paper to absorb the water in the glass plate, then added the concentrated gel and inserted the comb, and gently pulled out the comb after the gel was solidified again. Adjusted the amount of protein on the sample according to the results of protein quantification, added 1 μL of Protein Marker to the other end of the sample wells, processed samples of each group of proteins and start electrophoresis, the voltage of the concentrated gel was 80 V, and the voltage of the separated gel was 130 V. Terminated the electrophoresis when the bromophenol blue electrophoresis reached the bottom of the gel. The PVDF membrane was immersed in methanol and activated for 1 min, the black side of the clamp was placed in a glass dish containing transfer solution, and sponge pad, filter paper, separation gel, PVDF membrane, filter paper, sponge pad were put in from bottom to top, air bubbles were drained out, the clamp was tightly clamped, the pre-cooled transfer solution was added, and the current was set at 250 mA, and the membrane was transferred in an ice bath for 1 h. After the transfer was finished, the PVDF membrane was placed in 5% skimmed milk powder After the membrane transfer, the PVDF membrane was put into 5% skimmed milk powder solution and closed by shaking on a shaker for 1 h. The primary antibody dilution of the target protein was added proportionally, incubated at 4°C overnight, and the membrane was washed by adding TBST on a shaker for 5 times, each time for 5 min. The secondary antibody dilution was added and incubated on a shaker for 1 h. The membrane was was washed by adding a drop of the freshly prepared ECL mixture to the protein side of the membrane, and then luminescence was detected, and the exposure conditions were adjusted according to the different light intensities. The exposure conditions were adjusted according to different light intensities, developed and fixed, and the grayscale values of the target bands were analyzed using the Image J software processing system.
Data Processing
The results of the test were analyzed statistically using GraphPad Prism 8 software, a one-way ANOVA was used to compare whether there was a significant difference between multiple groups, P < 0.05 was considered statistically significant.
Results
Effect of Drug-Containing serum on HK-2 Cell Viability
As shown in Figure 1, there was no significant difference in the proliferation of cells in each group after 24 h of administration of blank serum, 10%, 20%, and 40% drug-containing serum, respectively, indicating that each concentration of drug-containing serum did not have a significant effect on cell growth.

Effect of Drug-Containing Serum on HK-2 Cell Viability After 24 h of Action.
Optimal Modeling Concentration and Duration of Modeling for TGF-β1
As shown in Figure 2, after 24 h of TGF-β1 action, there was no significant difference between the 5 ng·mL−1 and 10 ng·mL−1 TGF-β1 groups compared with the control group, and the cell proliferation of the 20 ng·mL−1 TGF-β1 group was significantly reduced (P < 0.0001); after 48 h and 72 h of TGF-β1 action, compared with the control group, cell proliferation of the 5 ng·mL−1, 10 ng·mL−1 and 20 ng·mL−1 TGF-β1 group cell proliferation rate was significantly reduced (P < 0.0001), but the survival rate was lower than 80%, so this experiment used 20 ng·mL−1 concentration of TGF-β1 action for 24 h modeling.

Effect of TGF-β1 on Cell Proliferation at Different Concentrations and Times (VS Control Group ####P < 0.0001).
Effect of Drug-Containing serum on E-Cadherin and Fibronectin Levels in a TGF-β1-Induced Fibrosis Model in HK-2 Cells
As shown in Figure 3, after 24 h of action with 20 ng·mL−1 concentration of TGF-β1, compared with the control group, the content of E-cadherin protein in the cells of the model group was significantly reduced, and the content of Fibronectin protein was significantly increased (P < 0.0001); compared with the model group, all concentrations of drug-containing serum groups could significantly increase the E-cadherin protein and decreased the content of Fibronectin protein (P < 0.0001) and improved cellular fibrosis compared with the model group at all concentrations of the serum-containing group.

Effects of Different Concentrations of Drug-Containing Serum on E-Cadherin and Fibronectin Levels in a TGF-β1-Induced Fibrosis Model of HK-2 Cells (VS Control Group ####P < 0.0001; VS Model Group ****P < 0.0001).
Effects of NBDF on Growth status and Body Weight of CRF Rats
Compared with the control group, rats in the model group showed depressed spirit, slow response, dull fur, and reduced diet, while rats in all dosing groups had a significantly better mental state than that of the model group, had a normal diet, and gained weight faster than that of the model group, as shown in Figure 4.

Plot of Body Weight Changes in Each Group of Rats.
Effects of NBDF on the Appearance of Kidneys and Organ Coefficients in CRF Rats
As shown in Figure 5 and Figure 6, compared with the control group, the kidneys of rats in the model group had a grayish-white appearance, hard texture, and significantly higher organ coefficients (P < 0.0001); compared with the model group, the kidneys of the rats in each administration group were significantly better, and the organ coefficients were significantly lower (P < 0.0001). The results indicated that NBDF could improve the renal condition of CRF rats.

Appearance of the Kidneys in Each Group of Rats.

Organ Coefficients of Rat Kidneys in Various Groups (VS Control, ####P < 0.0001; VS Model, ****P < 0.0001).
Effects of NBDF on Biochemical Indexes of Renal Function in Rats with CRF
As shown in Figure 7, compared with the control group, the CREA, UREA, UA, and 24 hUP contents of rats in the model group were significantly elevated, the HGB contents were significantly decreased, and the differences were statistically significant (P < 0.01, P < 0.0001); compared with the model group, the CREA, UREA, UA, and 24 hUP contents of rats in each dosing group were significantly decreased, and the HGB contents were significantly elevated, and the differences were statistically significant (P < 0.05, P < 0.01, P < 0.001, P < 0.0001). The results showed that NBDF had a certain restorative effect on renal function.
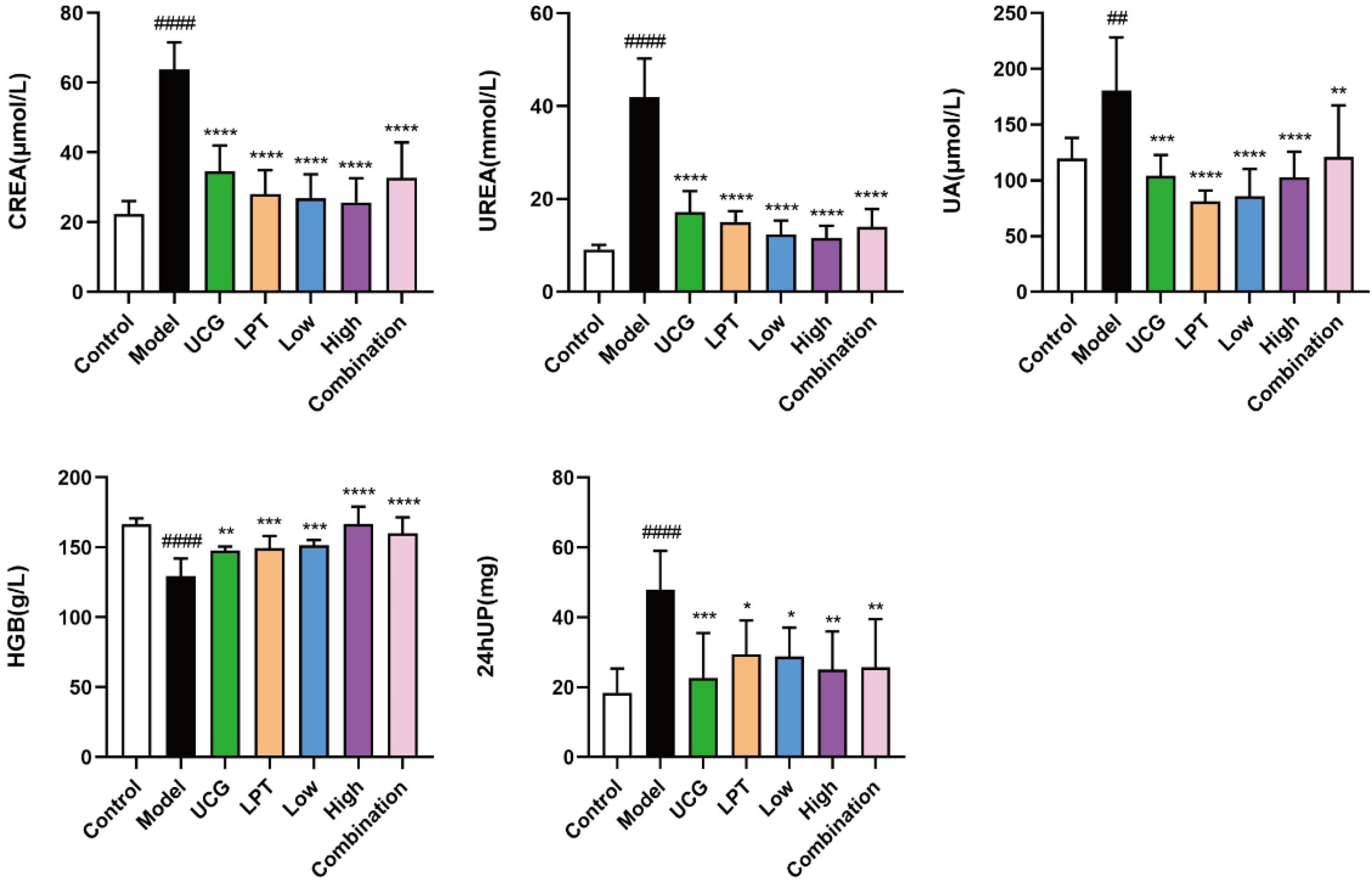
Levels of CREA, UREA, UA, HGB, and 24 hUP in Each Group of Rats (VS Control Group, ####P < 0.0001, ##P < 0.01; VS Model Group, ****P < 0.0001, ***P < 0.001, **P < 0.01, *P < 0.05).
Effect of NBDF on Histopathological Changes in the Kidney of Rats with CRF
The results of HE staining are shown in Figure 8, in the control group, the glomeruli and tubules were closely arranged with normal morphology and structure, and the proportion of renal interstitium was normal without inflammatory cell infiltration; in the model group, the glomeruli were reduced, the tubules were dilated or atrophied, there were vacuoles, and a large number of adenine crystals were deposited in the lumen of the tubule; compared with the model group, the tubular structure of the rats in the various groups of the drug administration was better, with fewer vacuoles and fewer adenine crystals, which showed that NBDF can improve the renal histopathological injury in CRF rats.

Effects of NBDF on Histopathological Changes in the Kidney of Rats with CRF (HE Staining, X200).
The results of Masson staining are shown in Figure 9, there were no obvious abnormalities in the glomeruli and tubules of the control rats, and only a trace amount of blue collagen fibers were deposited in the renal interstitium; a large number of blue collagen fibers were deposited in the renal interstitium of the model group rats; compared with the model group, the area of blue collagen fiber deposition in the renal interstitium of rats in each administration group was significantly reduced compared with the model group, indicating that NBDF can reduce the deposition of collagen fibers in the renal tissues of CRF rats.
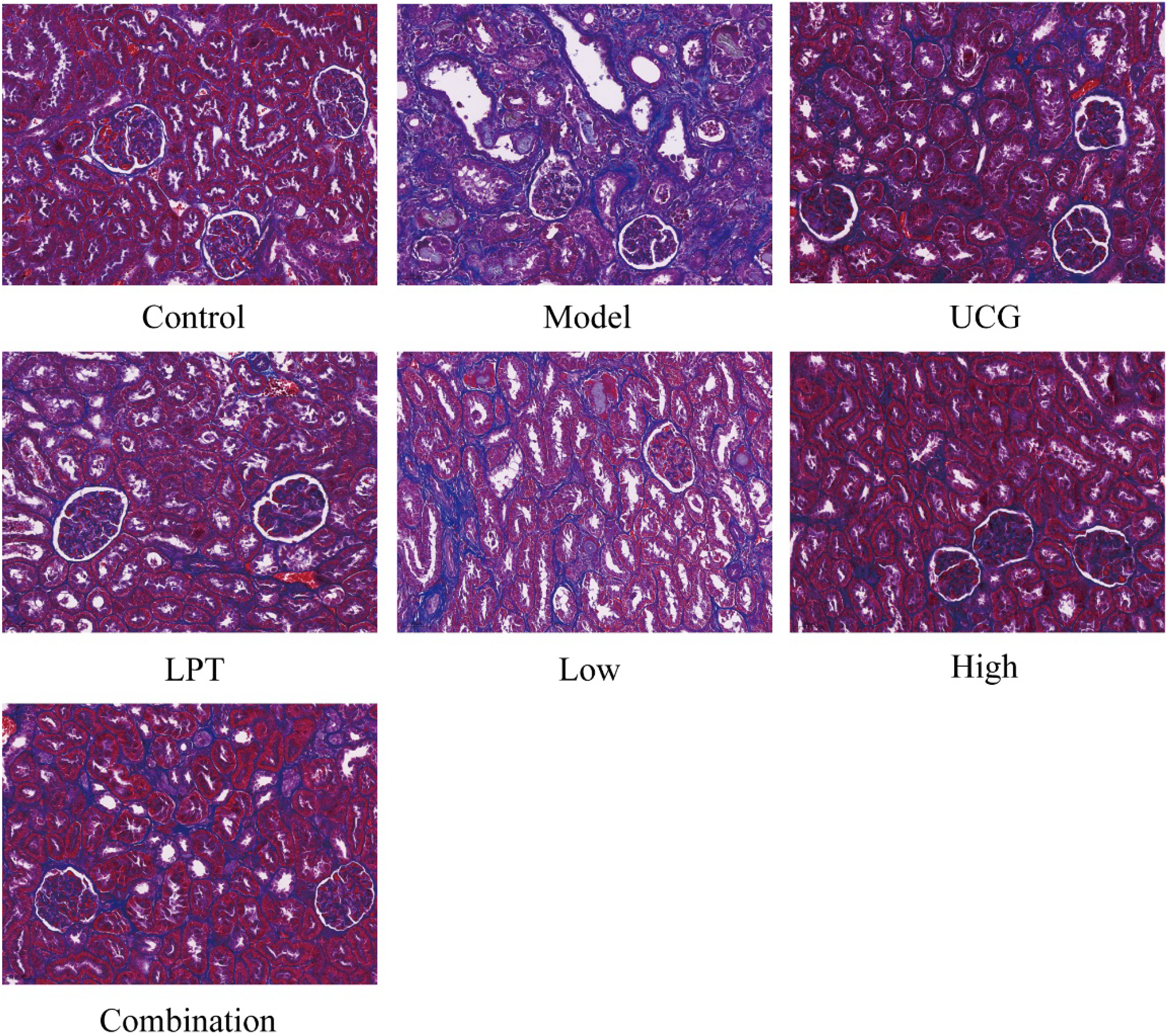
Effects of NBDF on Histopathological Changes in the Kidney of Rats with CRF (Masson Staining, X200).
Effect of NBDF on the Expression of Fibrosis-Related Proteins in Kidney Tissues of Rats with CRF
As shown in Figure 10, the immunohistochemical results showed that compared with the control group, the positive expression of E-cadherin in rats of the model group was significantly lower (P < 0.0001), and the positive expression of α-SMA, Vimentin, Fibronectin, and Collagen I was significantly higher (P < 0.0001); and compared with the model group, the renal tissues of rats of the various administration groups showed a E-cadherin positive expression was significantly higher (P < 0.001, P < 0.01, P < 0.05); α-SMA, Vimentin, Fibronectin, and Collagen I positive expression was significantly lower (P < 0.0001, P < 0.01, P < 0.05) in the kidney tissues of rats in each administration group.

A is the Expression of E-Cadherin, α-SMA, Vimentin, Fibronectin, Collagen I Proteins in the Kidney Tissues of Rats in Each Group (Immunohistochemistry, Ⅹ200), B is the Percentage of Positive Area of E-Cadherin, α-SMA, Vimentin, Fibronectin, and Collagen I Proteins in the Kidney Tissues of Rats in Each Group (VS Control Group, ####P < 0.0001; VS Model Group, ****P < 0.0001, ***P < 0.001, **P < 0.01, *P < 0.05).
Regulation of TGF-β/Smad Signaling Pathway-Related Proteins by NBDF
As shown in Figure 11, the expression of TGF-β, Smad2/3, p-Smad2/3 proteins were significantly higher (P < 0.05, P < 0.001) and the expression of Smad7 protein was significantly lower (P < 0.0001) in the kidney tissues of the rats in the model group, as compared with the control group. Compared with the model group, the expression of TGF-β, Smad2/3 and p-Smad2/3 proteins were decreased and the expression of Smad7 protein was increased in the administration group, the expression of Smad2/3 and p-Smad2/3 proteins were significantly decreased in the high-dose group and the positive-administration group (P < 0.05), the expression of Smad7 protein was significantly decreased in the UCG group, the low- and high-dose-administration groups, and the combination group of expression of Smad7 protein was significantly higher (P < 0.01, P < 0.05).

Results of Western Blotting on rat Kidney Tissue A is the Expression of TGF-β, Smad2/3, p-Smad2/3 and Smad7 Proteins B is the Calculation of Protein Gray Values (VS Control Group, ####P < 0.0001, ###P < 0.001, #P < 0.05; VS Model Group, **P < 0.01, *P < 0.05).
Discussion
Renal interstitial fibrosis (RIF) is the final pathological process and pathological basis of various chronic kidney diseases progressing to End Stage Renal Disease (ESRD), which is a dynamic and irreversible process. A distinctive feature of the pathological process of RIF is the deposition of a large amount of extracellular matrix (ECM) in the renal interstitium. The aggregation of myofibroblasts is considered to be one of the important reasons for the large accumulation of ECM in the renal interstitium and renal tubular Epithelial-Mesenchymal Transition (EMT) is an important source of myofibroblasts production, which is one of the important mechanisms of RIF.19–21
Transforming growth factor β (TGF-β) is a key regulator in the formation of renal fibrosis. On the one hand, which can promote ECM generation and reduce ECM degradation by promoting the synthesis of collagen fibers (collagen I, collagen III, etc), fibronectin (FN), laminin (LN), etc, while inhibiting matrix-degrading enzyme activities (MMP-2, etc); on the other hand, it can stimulate renal tubular EMT, in which renal tubular epithelial cells lose their epithelial cell marker proteins (eg, E-cadherin) and gain myofibroblast specific marker proteins (eg, α-SMA, Vimentin, etc), which further promotes the synthesis of the ECM, altered cell polarity and remodeling of tissue structure, which lead to renal fibrosis.22–24 E-cadherin is an adhesion molecule for intercellular junctions that plays an important role in tissue growth and development, and its loss of function leads to cellular dispersion, resulting in disruption of cellular junctions and infiltration and metastasis to the periphery. EMT is a process by which epithelial cells are transcriptionally re-edited and is induced by a specific set of transcription factors that regulate the expression of cadherins, in particular E-cadherin, so that its expression is reduced leading to reduced adhesion and enhanced cell migration. cadherin, resulting in decreased expression leading to reduced adhesion and enhanced cell migration.25–27 Fibronectin, a macromolecular glycoprotein, plays a central role in cell adhesion, regulates cell polarity, differentiation and growth, and can activate and repair senescent and damaged cells. It has been shown that EMT is associated with an increase in Fibronectin, and that blocking the expression of Fibronectin significantly inhibits various aspects of EMT, including adhesion junction breakdown and increased expression of mesenchymal markers.28,29
Numerous studies have shown that the TGF-β/Smad pathway plays a crucial role in renal fibrosis, and that overexpression of TGF-β can trigger renal fibrosis through activation of both the classical TGF-β/Smad signaling pathway and the non-classical signaling pathway, which leads to activation of myofibroblasts, excess of ECM components, and inhibition of ECM degradation. 30 It has been shown that Smad2/3 is an important downstream gene of classical TGF-β signaling that is activated in fibrotic kidneys,31,32 and it is now well-documented that Smad2/3 is activated by TGF-β to regulate renal inflammation and fibrosis. 33 Smad7 is a negative regulator that acts negatively in renal fibrosis and inflammation, and an imbalance between Smad2/3 and Smad7 is the a key mechanism of renal fibrosis, which leads to activation and accumulation of myofibroblasts, overproduction of ECM, and decreased ECM degradation. 34
Creatinine, urea, uric acid and urinary protein are important indicators for evaluating renal function. Changes in the concentration of serum creatinine are mainly determined by the glomerular filtration rate, and a decrease in the filtration capacity results in an increase in the concentration of creatinine35,36 ; urea refers to blood urea nitrogen, which is excreted through glomerular filtration, and the concentration is elevated during periods of renal failure, so it is used as an indicator of glomerular filtration function clinically; uric acid is the end product of purine metabolism, the increase of serum uric acid suggests that the accumulation of uric acid in the body increases, which can be interpreted as the metabolism of too much intake, too little discharge, suggesting that may be suffering from gout, nephritis and other diseases37,38; urinary protein, the protein contained in the urine, is a kind of important urinalysis indicator, normal human urine can contain a very small amount of protein, urinary protein abnormalities are commonly found in a variety of renal diseases39,40; anemia is a common complication of CKD, and the insufficient production of erythropoietin is considered to be the cause of renal disease, 41 so patients with CRF may have reduced levels of hemoglobin in their blood. 42
In our preliminary research, our team utilized LC-MS technology to analyze the chemical constituents and blood-entry components of the NBDF. 7 Our analysis identified 125 distinct compounds, including 48 blood-entry components, among which six key prototypical compounds—emodin, epicatechin, formononetin, chlorogenic acid, senna glycoside A, and astragaloside III—demonstrated notable efficacy in the treatment of CRF. Through the TGF-β/Smad signaling pathway, the natural components in rhubarb (such as rhein, emodin, chrysophanol, aloe-emodin, gallic acid and catechin) can ultimately reduce the levels of FN and ɑ-SMA, reduce the ECM deposition, and inhibit the expression of apoptosis-related factors.43,44 Studies demonstrate that Astragalus membranaceus exerts its anti-fibrotic effects mainly via TGF-β1 suppression, with astragaloside IV serving as its key bioactive constituent. Astragaloside IV, Calycosin, and Astragalus polysaccharides can inhibit TGF-β1 to exert anti-fibrotic effects.45,46
In the process of exploring the expression of proteins related to the TGF-β/Smad signaling pathway in animal kidneys, we only used the western blotting method for detection. Although western blotting can effectively detect the expression of target proteins, it is regrettable that we did not detect the protein expression of E-cadherin and Fibronectin in cells. E-cadherin and Fibronectin are important proteins related to cell adhesion and matrix, and they play a key role in the pathological process of renal fibrosis. Their expression levels can provide important supplementary information for studying the molecular mechanisms of renal fibrosis. Therefore, the failure to detect these two proteins is undoubtedly a major shortcoming of this study, which to some extent limits our comprehensive understanding of the molecular mechanisms of renal fibrosis. In future research, we plan to use multiple detection methods, such as immunohistochemistry and western blotting, to conduct in-depth studies on the protein expression of E-cadherin and Fibronectin, in order to more comprehensively reveal the molecular mechanisms of renal fibrosis.
Conclusion
In summary, on the basis of the clinical efficacy of NBDF, this study investigated the therapeutic effect and mechanism of CRF from cellular level and animal model, and provided experimental basis for the development of new drugs.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251386976 - Supplemental material for Study on the Mechanism of Action of Nourishing Blood Diuretic Formula in the Treatment of Chronic Renal Failure and Anti-Renal Fibrosis Through TGF-β/Smad Signaling Pathway
Supplemental material, sj-doc-1-npx-10.1177_1934578X251386976 for Study on the Mechanism of Action of Nourishing Blood Diuretic Formula in the Treatment of Chronic Renal Failure and Anti-Renal Fibrosis Through TGF-β/Smad Signaling Pathway by Sicheng Yang, Xia Lei, Jingbo Wang, Bin Yan, Ju Huang, Fengqin Yue, Sidi Chen, Wei Peng, Wanjin Sun, Heng Fan and Xueyun Duan in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
All authors thank all the people who helped and provided comments to complete this research.
Ethics Approval and Consent to Participate
The animal experiments involved in this study were approved by the Experimental Animal Committee of Hubei Provincial Hospital of Traditional Chinese Medicine, with the approval number: E Hospital of Traditional Chinese Medicine Animal (Fu) No. 202205.
Authors’ Contributions
Wanjin Sun, Heng Fan provided fund support. Xueyun Duan designed this experiment. Sicheng Yang, Xia Lei carried out this experiment and wrote the article. Jingbo Wang, Bing Yan, Kai Deng, Ju Huang, Fengqin Yue, Sidi Chen, Wei Peng provided assistance in conducting experiments and performing statistical analysis. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hubei Provincial Natural Science Foundation—Joint Fund for Innovation and Development (2025AFD520), the Seventh Batch of Traditional Chinese Medicine Experts in China (No. [2022] 76), the National Training Project for Traditional Chinese Medicine Technology Inheritance Talent (No. [2023] 96), the Hubei Province Major Difficult Diseases Collaboration Project and Provincial Key Laboratory Research Project (2021-06) and the Research on Integrated Traditional Chinese and Western Medicine for Crohn's Disease (2025YFC3508502).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data and Materials Availability
Data are available upon request from the frst author, Sicheng Yang; ysc570721074@163.com
Statement
All the methodologies have been standardized in our laboratory.
Supplemental Material
All supplemental material mentioned in the text is available in the online version of the journal.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
