Abstract
Introduction
The Thai Herbal Wattana formula (WNF) is a multi-ingredient remedy used to promote overall health and mitigate age-related physiological degeneration, suggesting a potential adjuvant use in oncological treatments.
Aims
This ethnopharmacological study aimed to evaluate cytotoxic and antimigratory effects of the Ayurveda Siriraj WNF AVS073 variation (ASW) and its active/s botanical constituents in human liver cancer (HepG2) cells.
Methods
ASW and its ingredients were evaluated for cytotoxicity (Alamar blue), anti-migratory activity (2D gap closure). Mechanistic studies included cell death, apoptosis and cell cycle arrest (flow cytometry), and intracellular glutathione levels. Bioguided isolation was used to identify the active compound/s.
Results
ASW did not inhibit HepG2 cell proliferation at 200 µg/mL although it halved intracellular glutathione levels and reduced cell migration similarly to paclitaxel 0.01 nM. In contrast, the water extract from Biancaea sappan (syn. Caesalpinia sappan L.) (CSL) was the only ingredient showing cytotoxicity (IC50 = 44 µg/ml). It induces apoptosis and G2/M phase cell cycle arrest and significantly reduced 2D cell migration without modifying glutathione levels. Brazilein was dereplicated as the active cytotoxic component in CSL but it did not show any effect on glutathione levels or 2D cell migration.
Conclusion
This preclinical study demonstrates that the ASW formula lacks direct in vitro cytotoxicity towards HepG2 cells, but effectively reduced cell mobility and intracellular glutathione although at non-physiological concentrations. B. sappan L., exhibits potent cytotoxic and anti-migratory activities. Brazilein is its primary cytotoxic compound although is not endowed with the glutathione-depleting or anti-migratory effects observed in the ASW formula. These findings suggest that ASW's benefits in oncology may primarily link up with its established immunological and anti-inflammatory effects, while its lack of toxicity to HepG2 cells, a proxy for hepatocytes, plus its clinically proven lack of major adverse effects might indicate a positive safety profile.
Keywords
Introduction
The Thai Herbal Wattana formula (WNF) is a multi-ingredient remedy traditionally employed for its purported benefits in enhancing appetite, promoting overall health, and mitigating age-related physiological degeneration. Clinical evidence supports several potential medicinal applications for WNF. A Phase II clinical trial demonstrated that WNF possessed comparable efficacy to diclofenac tablets in alleviating pain and improving knee function in patients with knee osteoarthritis. 1 Furthermore, WNF has been shown to exert protective effects against UVA-induced melanogenesis, 2 as well as exhibit immunomodulatory, 3 anti-inflammatory, 3 and anti-amyloidogenic properties. 4
The precise composition of WNF exhibits variability, with formulations reportedly containing up to 18 distinct ingredients, often with qualitative differences documented over time.2–5 A set of core herbal ingredients are present in all the variations of this formula namely Aegle marmelos, Boesenbergia rotunda, Brachypterum scandens, Cinnamomun sp., Cladogynos orientalis, Criptolepsis dubia, Cyperus rotundus, Ferula assa-foetida, Piper nigrum, Putranjiva roxburghii, Saussurea lappa, Terminalia chebula and Tinospora crispa. Common additions to these are Caesalpinia sappan, Carthamus tinctorius, Citrus x aurantius, Ligusticum sinense and Mallotus repandus. The complete botanical details of the ingredients used in the recipe used for this work (hereby termed Ayurveda Siriraj WNF AVS073 variation or ASW) are presented in the materials and methods section (Table 1).
Summary of the Botanical Identity of the Ingredients of the ASW Formula.

While traditional indications suggest that WNF's potential lies in its use as an adjuvant in oncological treatments, its direct anticancer activity remains uninvestigated. Advanced cancer patients frequently experience a range of physical and psychological symptoms, including significant anorexia, which aligns with WNF's traditional use for appetite stimulation. A recent work demonstrated that the in vitro exposure of cytokine-induced killer cells and dendritic cells concluded that WNF upregulated Th1 and Th17, but downregulated Th2 and Treg phenotypes within CD3+CD56+ cells. 3 This specific immune profile, particularly the balance shift away from Th2 and Tregs towards Th1, is generally associated with a more favorable immune response against liver cancer, 6 the third leading cause of cancer death worldwide. 7
A survey of the literature unveiled that several of the ingredients such as Boesenbergia rotunda, 8 Biancaea sappan (syn. Caesalpinia sappan),9,10 Ligusticum sinense, 11 and Aucklacia lappa (syn. Saussurea lappa) 12 are reported as exerting direct cytotoxicity upon HepG2 cells, the most researched liver cancer cell line. 13 Despite its lack of consistent expression of phase 1 enzymes, 14 the HepG2 cell line has been also a popular model for hepatocytes metabolism 15 and toxicology. 16 Consequently, this study primarily aimed to evaluate cytotoxicity and antimigratory effects of botanical constituents of the Ayurveda Siriraj WNF AVS073 variation (ASW) and its active/s botanical constituents in human liver cancer (HepG2) cells.
Materials and Methods
Plant Extract Preparation
All plant materials (Table 1) were authenticated according to the Thai Herbal Pharmacopoeia, 17 cleansed by rinsing under purified water, dried in air-circulating ovens at 40 °C, grounded and sifted into fine powders with various pharmaceutical-grade milling equipment all following the in-house PIC/S GMP-certified Standard Operating Procedures of the Ayurved Siriraj Manufacturing Unit of Herbal Medicine and Products (Centre of Applied Thai Traditional Medicine, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand) 18 and phytochemical analyses (HPTLC CAMAG system, Switzerland; LC-MS-MS system Xevo TQ-XS, Waters, UK) and characterization of powders for particle size distribution, flowability, and tapped density using a powder flow analyser (TA.HD plus, Stable Micro Systems, Surrey, UK) were performed at the Siriraj Herbal—Drug Examination and Analysis Central Laboratory (SiHAC). 19 Batch certificates may be supplied upon reasonable request to the Office of Applied Thai Traditional Medicine, Piyamaharajkarun Building, seventh Floor (SiPH), Tel. 02-419-1705.
Water extracts were prepared according to Thai traditional methods for decoctions. 17 Briefly, each herbal powder was boiled with bi-distilled water (1:10) until the volume reached about 1/3 of the initial volume. The decoction was then filtered and freeze-dried. The dry extracts were stored in a cool, dry place and protected from light until use. Before cell-based assays, the extracts were prepared into stock solutions in water, filtered through a Millipore© 0.22 μm syringe filter (Merck, UK), and kept at −20 °C until use.
Cell Lines
HepG2 cells (ACC No 85011430, Lot 11C013) were cultured in Minimal Essential Medium (MEM) Alpha supplemented with 10% foetal bovine serum (FBS), 100 U/mL penicillin, 100 μg/mL streptomycin in a NuAire© DH Autoflow CO2 Air-Jacketed incubator (Avidity Science Ltd., UK) at 37 °C/5%CO2. All cell culture reagents were obtained from Gibco® (Thermo Fisher Scientific Inc., UK).
Cytotoxicity Assay
HepG2 cells (5000 cells/well) were incubated with the extracts, paclitaxel (≥95%, HPLC) from Taxus brevifolia (Sigma-Aldrich, UK), or medium as control for 48 h. The cell viability was measured using the AlamarBlue assay as recommended by Miret and co-workers 21 following the instructions of the manufacturer (Bio-Rad Abd Serotec Ltd., UK). The fluorescence intensity was measured at 560/590 nm using a Tecan Infinite© M200 microtiter plate reader (Tecan Trading AG, Switzerland). According to the American National Cancer Institute (NCI) criteria, crude plant extracts are considered cytotoxic if they exhibit an IC50 value less than 100 µg/mL. Extracts demonstrating an of 30 µg/mL or less are deemed particularly promising and warrant further pharmacological and phytochemical investigation The maximum non-toxic concentration (MNTC) was defined as the highest concentration of the compound that resulted in cell viability of >80% (IC20).22–24 As for pure compounds, the maximum concentration for being considered within pharmacological reasonable limits is 25 µM. 25 In our experiments Paclitaxel was used as a positive reference drug and exhibited an IC50 = 60 nM and an IC20 (MNTC) = 0.01 nM.
The extracts with IC50 below 50 μg/ml were further investigated for apoptosis induction and cell cycle arrest. Maximum non-toxic concentrations (MNTC) were determined from the maximum concentrations with <20% cytotoxic effect on the cells.
Cell Migration Assay
Bidimensional cell migration was measured using OrisTM Universal Cell Migration Assembly Kit (Platypus Technologies LLC, USA).
26
The assay utilizes proprietary stoppers made from a medical-grade silicone to restrict cell seeding to the outer annular regions of the wells. Removal of the stoppers reveals a 2-mm diameter unseeded region in the center of each well (the detection zone) into which the cells migrate offering more reproducibility than the classic “scratch assay”.
27
For the migration assay, the sterile stoppers were introduced in a flat-bottom 96-well NunclonTM plate (Thermo Fisher Scientific Inc., UK) before seeding HepG2 cells at a density of 105 cells per well. After 24 h of incubation the stoppers were removed, and the cells incubated with the maximum non-toxic concentrations (MNTC) of each treatment to avoid interference with any antiproliferative effect. We used complete cell media as control, and paclitaxel 0.1-0.01 nM (≥95%, HPLC) from Taxus brevifolia (Sigma-Aldrich, UK) as positive reference. At the end of the incubation period, the cells were fixed with 100 µl of cold 40% TCA. The plates were kept at 4 °C for 1 h and rinsed with water. The cells were stained by the addition of 50 µl of 0.4% trypan blue and incubated at room temperature for 1 h. Another washing step was performed, and the plate was then left to air-dry completely overnight. Measurement of zone closure was performed by digital images obtained by optical scanning (transmittance mode) by a Snapscan e50 (Agfa-Gevaert, Germany). Image J (National Health Institute, USA) was used to measure an average gap distance value and compare to the vehicle control to monitor the migratory capacity of the treated cells.
28
The migration rate was calculated as follows:
Determination of Glutathione Cellular Levels
HepG2 cells (50,000 cells/ well) were incubated for 48 h with camptothecin 5 μM, WNF 200 μg/ml, or CSL 50 μg/ml. The assay was performed with minor modifications from Allen et al (2000), as well as the calculation. 29 Briefly, HepG2 cells (4 × 104 cells/well) were incubated for 24 h with buthionine sulfoximine (10 μM) or plant extracts (100 μg/mL), then washed with PBS after which 60 μL of 0.1% Triton-X was added to each well of the plates to lyse the cells. Twenty-five microliters of 5% sulfosalicylic acid was added to the cell lysates and plates were shaken for 2 min. Twenty-five microliters of glutathione reaction buffer containing NADPH 2.39 mM, 5-5′-dithiobis(2-nitrobenzoic acid) 0.01 M and 500UI of glutathione reductase in sodium phosphate buffer 143 mM containing EDTA 6.3 mM was added to the cell lysates. Absorbance was read in a kinetic cycle every 30 s for 5 min at 405 nm (11 readings) using a Tecan Infinite© M200 microtiter plate reader (Tecan Trading AG, Switzerland). Absorbances were converted into absolute amounts by means of the i-slopes method using known concentrations of reduced L-glutathione. All chemicals were from Sigma-Aldrich (Merck Ltd, UK).
Cell Death Detection Experiments via Annexin V—Propidium Iodide Staining in HepG2 Cells
Detection of early apoptosis by quantitation of apoptosis and necrosis by annexin V binding, propidium iodide uptake, and flow cytometry was performed according the principles described in literature 30 with the eBioscience™ Annexin V Apoptosis Detection Kit BMS500FI Kit according to the manufacturer's instructions (Thermo Fisher, UK) using HepG2 cells (100,000 cells/ well) incubated with the extracts, camptothecin 5 μM (Sigma-Aldrich, UK), or control for 48 h after which a flow cytometric analysis was performed using a MACSQuant© Analyzer (Miltenyi Biotec Ltd., UK), and the data were processed using MACSQuantifyTM© Software (Miltenyi Biotec Ltd., UK).
Cell Cycle Analysis
HepG2 cells (100,000 cells/well) were incubated with the extracts, paclitaxel 1 μM or control for 48 h. PI staining was performed according to Leonce et al 31 The flow cytometric analysis was performed using MACSQuant Analyzers (Miltenyi Biotec Ltd., UK) and the data were processed using MACSQuantifyTM Software (Miltenyi Biotec Ltd., UK). using a MACSQuant© Analyzer (Miltenyi Biotec Ltd., UK), and the data were processed using MACSQuantifyTM© Software (Miltenyi Biotec Ltd., UK).
Fractionation of B. sappan L. Water Extract
Bioguided isolation pf the cytotoxic principles from B. sappan was conducted according to authoritative guidelines of the field.32,33 We fractionated 100 mg of CSL water extract using a glass column packed with Silica Gel 60 (Merck, UK). extract was eluted with 100 ml of Chloroform, Chloroform:Methanol (75:25; 50:50; 25:75), Methanol:Ethylacetate (75:25; 50:50; 25:75), and Ethylacetate, respectively. The fractions were analysed with a Waters™ HPLC consisting of a 2695-separation module and a 996-photodiode array detector (Waters Corporation, USA) using a Zorbax© RX-C18 column (5 μm; 4.6 × 250 mm, Agilent). The mobile phase consisted of 0.2% acetic acid in water (A) and methanol (B) were used in gradient mode from 0%(B) to 80%(B) in 60 min. Data were analysed using Empower© software (Waters Corporation, USA).
Statistical Analysis
The means ± standard deviation (SD) was calculated using Microsoft Excel 2010. Calculations of IC50, MNTC, and one-way Analysis of Variance (ANOVA) were performed using GraphPad Prism 8 software (GraphPad Software, Inc.) followed by Bonferroni post-ANOVA multiple t-test. The level of significance was set at p < 0.05. Experiments were run in technical duplicates in three or more independent biochemical or biological replicates (N ≥ 3). In all experiments a control (solvent as treatment) and positive drug treatment were added.
Results
In Vitro Cytotoxicity of ASW and its Ingredients to Human Hepatocellular Carcinoma HepG2 Cells
The screening of all individual ingredients (Figure 1) yielded three different types of effect: those with both IC50 and MNTC above 200 µg/mL (ASW, AM, BR, CT, CSC, CO, LS, PN and SL), extracts with IC50 ≈ 200 µg/mL and MNTC ≤ 50 µg/mL (CA, CR, DS, DR, FA, MR, TR, and TM), and finally the CSL extract was the only one exhibiting cytotoxicity in a linear dose-dependent manner with IC50 at 44 µg/ml and MNTC at 25 µg/ml. Therefore, the crude CSL extract was further assessed for its GSH depletion ability, anti-migratory activity, apoptotic effect, and cell cycle distribution as well as subjected to bioguided isolation.

Dose-Dependent Inhibitory Effects of the Ayurveda Siriraj Wattana Formula (ASW; AVS073 Variation) and its Constituent Ingredients (see Table 1 for Abbreviations) on Mitochondrial Activity in HepG2 Cells, Assessed after 48 h Using the Alamar Blue Assay.
In Vitro Effect of ASW and its Cytotoxic Ingredient to Glutathione Levels in HepG2 Cells
The treatment of HepG2 cells with WNF (200 μg/ml) significantly halved GSH control levels (21.17 + 1.0 down to 11.47 + 2.3 µM/mg cell protein; p < 0.05) similarly to camptothecin 5 µM. In turn, B. sappan water extract (50 μg/ml) showed no effect on total glutathione levels.
Effects of ASW and B. sappan Water Extract to Bidimensional Migration of Human Hepatocellular Carcinoma HepG2 Cells
We measured the effect of unrestrained proliferation and migration of liver cells in an in vitro bidimensional, membrane free model. Our findings are that treatments with WNF (at 200 µg/ml) or CSL (at 25 µg/ml) were as effective as the positive anticancer drug paclitaxel 0.01 nM in inhibiting this hallmark of cancer (Figure 2).

In Vitro Effect of the Water Extracts of the Ayurveda Siriraj Wattana Formula (ASW; AVS073 Variation) and its Cytotoxic Ingredient B. sappan (CSL) Compared with the Reference Drug Paclitaxel on the 2D Migration/Motility of HepG2 Cells. (A) Pie Chart Representing the Cell Migration Rate (%) for Each Treatment. (B) Bar Chart Showing Absolute Area of the Free Cell Gap before and after Treatments in Square Pixels (Average of Five Random Measurements Per Replicate, N = 3). (C) Representative Images of the Scratch/Wound before and after Treatment. (a) Difference is Significant (p < 0.05) Compared with Pre-migration Values; (b) Difference is Significant (p < 0.05) Compared with Control Migration Values (72 h).
Apoptotic Effects of B. sappan Water Extract in Human Hepatocellular Carcinoma HepG2 Cells
Cancer cells frequently develop resistance to apoptosis, making the early circumvention of this barrier a potential strategy to halt cancer progression. A hallmark event in early apoptosis is the translocation of phosphatidylserine (PS) from the inner to the outer leaflet of the plasma membrane, exposing it to the extracellular environment. In our study, dual Annexin V/Propidium Iodide (PI) staining of HepG2 cells after 48-h incubations with varying concentrations of B. sappan water extract revealed that this cytotoxic ingredient primarily induced cell death via necrosis, whereas the reference anticancer drug camptothecin significantly induced apoptosis (Figure 3).
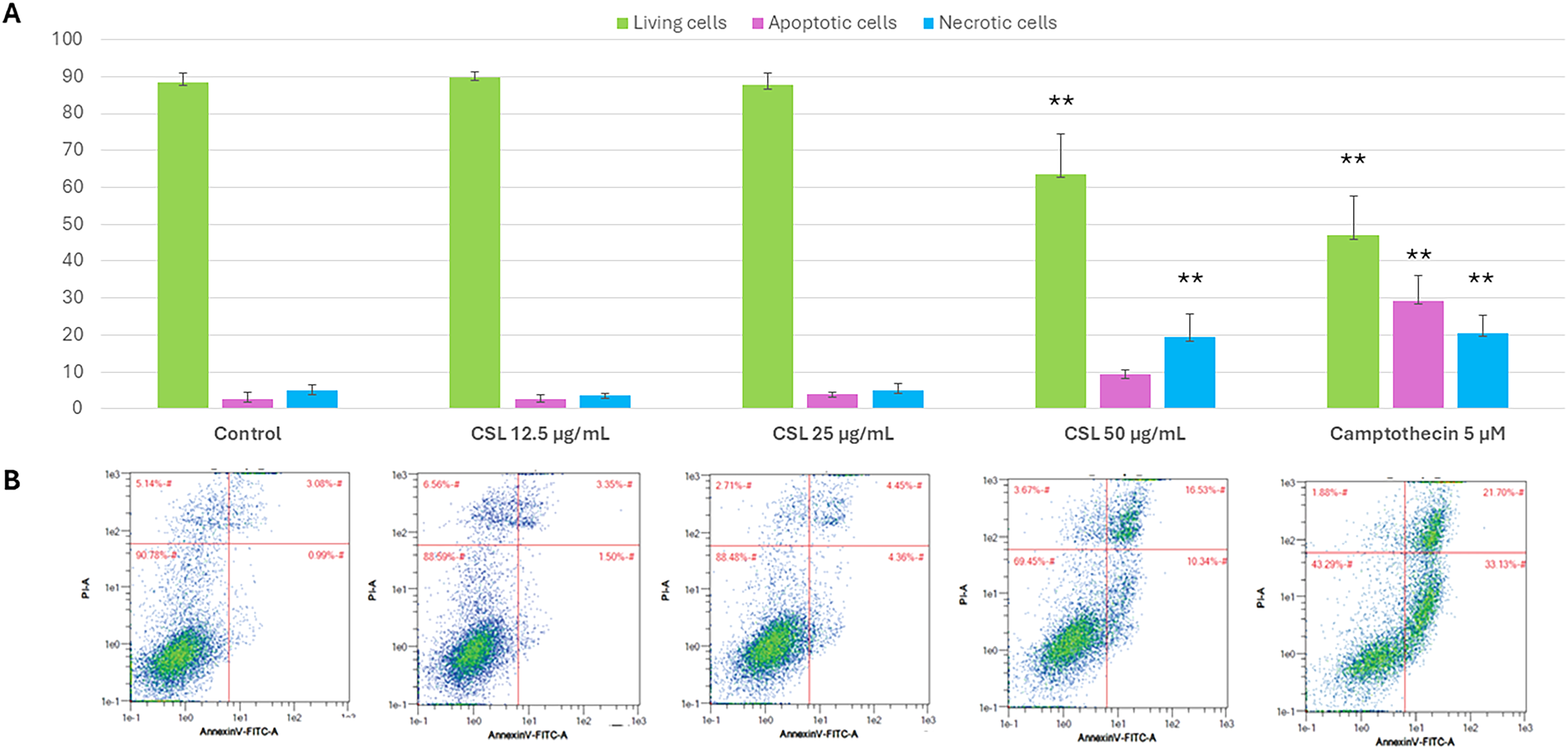
Apoptotic Effects of B. sappan (CSL) and the Reference Drug Paclitaxel on HepG2 Cells Assessed by Annexin V-FITC/PI Staining. (A) Quantitative Analysis of Data Presented as Mean + SD. (**) p < 0.01 Compared to Control (N > 3). (B) Representative Dot Plots of Cells for Annexin V-PI Counterstain Showing Annexin V-FITC and PI Channels (X and Y Axes, Respectively) Gated According to Untreated Cells for Analyses (Control, DMSO 0.08%). Each Plot Shows Cells Positive for Annexin V only (Lower-Right Quadrant, Early Apoptotic Cells), Positive for PI Only (Upper-Left Quadrant, Necrotic Cells), Positive for Both (Upper-Right Quadrant, Late Apoptotic Cells), and Negative for Both (Lower-Left Quadrant, Live Cells).
To understand how the cytotoxic ingredient affects cell cycle distribution, we synchronized HepG2 cells and analysed their DNA content using flow cytometry after 48 h of treatment (Figure 4). Both the positive drug of reference (paclitaxel) and the water extract of B. sappan significantly increased the fraction of cells in the G2/M phase, which was accompanied by a notable decrease in the S phase population. This data suggests that the B. sappan extract induces a cell cycle arrest or delay, primarily at the G2/M checkpoint.

Representative Flow Cytometric Analysis of Cell Cycle Distribution in HepG2 Cells Treated with B. sappan (CSL) and the Reference Drug Paclitaxel. (A) Quantification of Cells in G0/G1, S, and G2/M Phases Using the PI Channel. Data are Presented as Mean ± SD (N > 3). (*) p < 0.05; (**) p < 0.01 Compared with Control (DMSO 0.08%). (B) Representative PI Signal Plots (20,000 Single Events) Showing the Area Under the Fluorescence Signal Versus the Height of the PI Signal.
Bioguided Isolation of the Cytotoxic Component of B. sappan Water Extract
The extract was subjected to fractionation by vacuum liquid chromatography (VLC) prior to characterization/purification by high performance liquid chromatography (HPLC). Only two fractions, CSL1 (Chloroform/Ethylacetate 75:25; IC50 = 13 μg/ml; MNTC = 7 μg/ml) and CSL2 (Chloroform/Ethylacetate 50:50; IC50 = 41 μg/ml; MNTC = 24 μg/ml), retained the cytotoxic activity (Figure 5A). However, they did not significantly affect cell migration and GSH levels assays at MNTCs (Data not shown). Further HPLC analysis revealed that CSL1 (Figure 5B) consisted of a major peak with a characteristic UV (λmax = 445 nm). After prep-HPLC enrichment and purification, a compound matching the 1H NMR spectrum of brazilein (Figure 5C) was obtained.
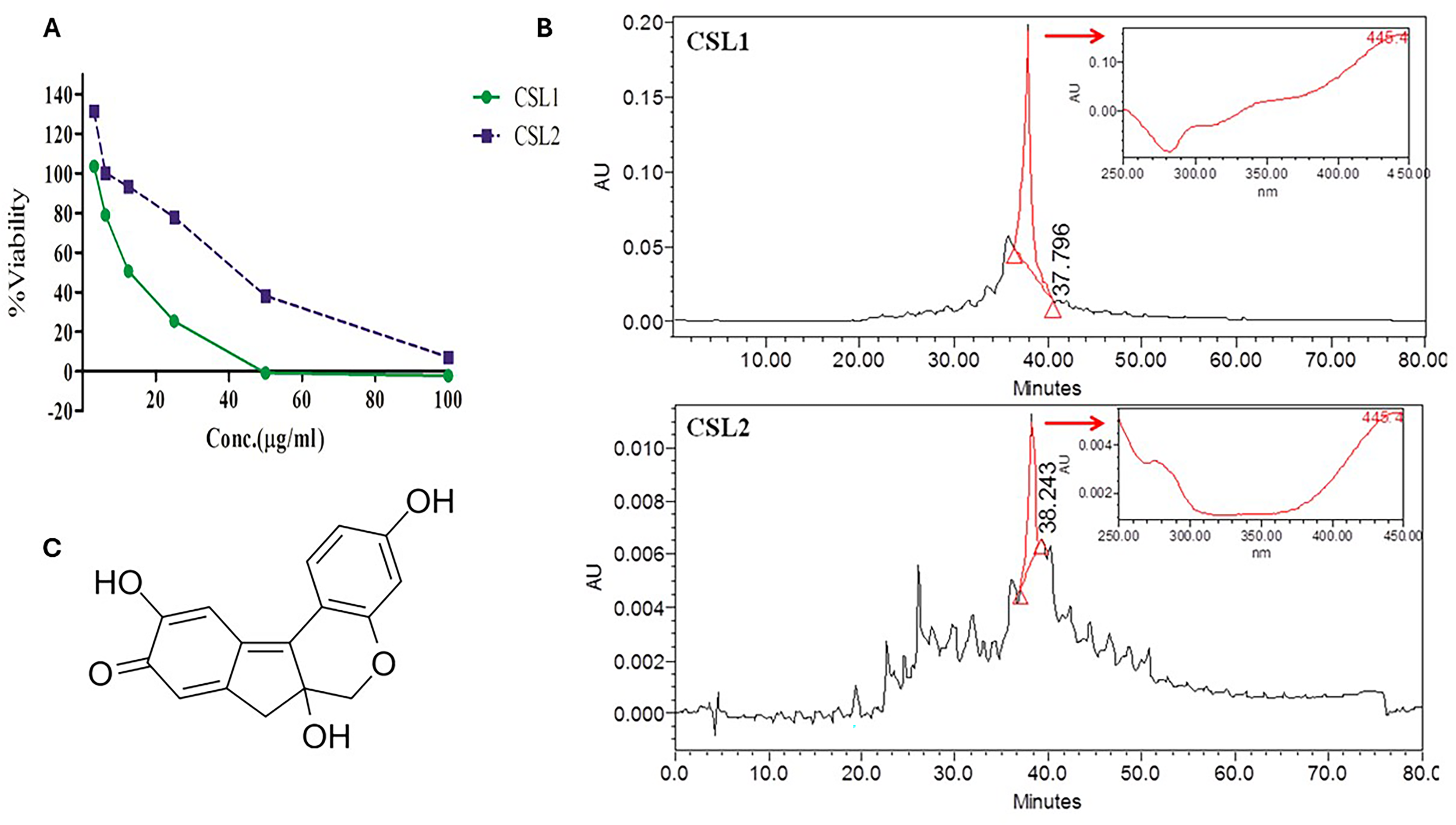
Bioguided Isolation of the B. sappan Water Extract (CSL). (A) Dose Dependent Cytotoxicity of CSL1 and CSL2 in the Alamar Blue Assay; (B) HPLC Chromatograms of CSL1 and CSL2 at 445 nm and UV spectra of the major Peaks; (C) 2D Chemical Structure of Brazilein.
Discussion
Cytotoxic Effects of the ASW Ingredients on HEPG2 Cells and Their Molecular Mechanisms
The ASW formula extract did not show any cytotoxicity towards HepG2 cells (IC50 > 200 µg/mL). The only cytotoxic herbal ingredient in the ASW formula was the water extract of Biancaea sappan. The double-digit IC50 of its water extract compares well with those of organic extracts reported in the literature: the methanolic extract is cytotoxic to HepG2 cells, with an IC50 of 20 µg/mL. 34 Meanwhile, petroleum ether root, leaf, and stem extracts were antiproliferative against HH-7 hepatoma cells (IC50 of 56 and 77 µg/mL, respectively) via intraperitoneal injections of petroleum ether extracts from the leaves and stems (20-65 mg/Kg) and oral administration (100-325 mg/kg, 12 days) in a H22 hepatoma-bearing mouse model, 35 thus confirming their antitumor activity.
Previous reports did not investigate the mechanisms behind these actions. Our study is therefore the first to provide data on the effects of sappanwood water extracts on the cell cycle and to characterize their pro-apoptotic effects. Flow cytometry data suggests that the B. sappan extract induces a cell cycle arrest or delay, primarily at the G2/M checkpoint. This means that after DNA replication, cells are prevented from entering or progressing through mitosis. The corresponding reduction in the S phase population is a direct consequence of this G2/M blockage, as fewer cells successfully complete mitosis to re-enter G1, thus decreasing the overall proportion of cells actively synthesizing DNA. This G2/M arrest could be triggered by the B. sappan water extract through mechanisms such as inducing DNA damage, interfering with DNA replication completion, or disrupting the machinery essential for proper mitotic entry. In this regard, the B. sappan water extract appears to mimic the action of the known cytotoxic natural drug paclitaxel. Unlike the apoptotic cell, which undergoes shrinkage and efferocytosis by neighboring living cells, necrotic cells swell and lyse, releasing their contents into the local tissue microenvironment. 36 This release subsequently recruits immune cells to survey damage and clear debris, triggering a pro-tumorigenic inflammatory process which may exacerbate pre-existing unresolved inflammation 39. However, some evidence suggests that the high inflammatory response characteristic of hepatocellular carcinomas might correlate with improved patient outcomes, highlighting the idiosyncratic nature of cancer biology. 37 Further studies are needed to fully characterize the pathways involved in these effects in HepG2. According to the literature, it may involve an increase in the expression of p53 and p21WAF1/CIP1 (as in head and neck cancer cells 38 ) and seems to be selective as it induced apoptosis in oral cancer cells but not in normal epithelial cell lines. 39
Our bioguided isolation efforts identified brazilein as the active principle of the B. sappan water extract, exhibiting an IC50 of 13 mg/mL. This finding aligns with a previous report on its cytotoxicity against human HepG2 cells and two additional hepatocellular carcinoma cell lines (SMMC7721 and SGC7901), with an IC50 of 10 μg/mL. Brazilein has also been shown to induce significant apoptotic cell death, as evidenced by DNA ladder assay, and growth inhibition in HepG2 cells, which the authors attributed to its effects via the inhibition of survivin protein and mRNA expression. 10 Other studies have reported cytotoxic activity on various cancer cells, including liver, MDA-MB-231, MCF-7, A549, and Ca9-22. 9 Brazilein is a natural, red-colored dye formed by the spontaneous oxidation of brazilin, a homoisoflavonoid found in the heartwood of trees such as brazilwood and sappanwood, which have been used for centuries as dyes for textiles, paints, and inks. 40 Our cell cycle analyses are consistent with the ability of the homoisoflavonoid brazilin to induce apoptosis and G2/M arrest through the inactivation of histone deacetylase in multiple myeloma U266 cells. 41 Furthermore, in other studies, the downregulation of survivin by brazilein was associated with strong and prominent activation of caspases-9 and −3, as well as PARP cleavage. Notably, similar homoisoflavonoid derivatives, such as sappanone A and protosappanin B, induce ferroptosis in hepatocellular carcinoma via the NRF2/xCT/GPX4 axis, 42 and pretreatment of H22 hepatoma cells (6.25 mg/mL) resulted in complete inhibition of tumor formation in KM mice, 43 respectively.
Other ingredients in our study's recipe, despite failing to show significant effects, have been reported to exhibit cytotoxic effects on HepG2 or other liver cells, typically as organic extracts or containing specific phytochemicals not readily soluble in water. For instance, Aegle marmelos fruit extracts inhibited chemically induced and promoted hepatocarcinogenesis in Wistar rats (25 mg/kg bw) 44 and Balb/c mice (100 mg/kg bw) 45 with positive effects on inflammatory (IL-1β, IL-6), anti-inflammatory (IL-4) cytokine expression, apoptosis (Bcl-2), and tumor-related (p53, c-jun) genes. The rhizomes of B. rotunda contain two chalcone derivatives with cytotoxic effects on HepG2 cells: Panduratin A induces G2/M phase arrest in cancer cells, significantly inhibits NF-κB 46 signaling pathways leading to decreased cancer cell viability, and shows anti-angiogenic effects, 47 while Boesenbergin A was cytotoxic to HepG2 (20 µg/mL) among other cell lines. 8 Hydroalcoholic extracts of Citrus × aurantium fruit peels are rich in bioflavonoids known to induce human HepG2 cell apoptosis via mitochondrial and death receptor pathways. Hesperidin activated and increased caspase-9, −8, and −3 activities in hesperidin-treated HepG2 cells, downregulated Bcl-xL protein, and upregulated Bax, Bak, and tBid protein levels in a dose-dependent manner, 48 and Wnt3a/β-catenin and Wnt5a pathways protected against chemically-induced early hepatocarcinoma models in rats. 49 Naringenin induced G0/G1 and G2/M phase arrests in HepG2 cells via the p53 pathway and induced apoptosis through the mitochondrial-mediated apoptosis pathway, shown by an increased Bax/Bcl-2 ratio, subsequent cytochrome C release, and sequential caspase-3 activation, 50 as well as via ROS-Mediated JAK-2/STAT-3 signaling pathways. 51 Nobiletin may reduce lipid accumulation by up-regulating the SIRT1-AMPK signaling pathway in HepG2 hepatocarcinoma cells, thereby preventing potential carcinogenic effects. 52 Naringin induces human hepatocellular carcinoma HepG2 cell apoptosis via mitochondria-mediated activation of caspase-9 and caspase-8-mediated proteolysis of Bid. 53 Sinensetin suppresses angiogenesis in liver cancer by targeting the VEGF/VEGFR2/AKT signaling pathway. 54
The root ethanolic extracts of Cladogynos orientalis exhibited cytotoxicity, apoptosis induction, and moderate alkylating activity in HepG2 cells. 55 Farnesiferol C from Ferula assa-foetida induces ROS-dependent apoptotic pathway in HepG2 cells. 56 58. The ethanol extracts of Aucklandia lappa (syn. Saussurea lappa) are rich in sesquiterpene lactones such as costunolide, dehydrocostus lactone, and lappadilactone, which inhibited HepG2 cell proliferation, 12 as well as showing antiangiogenic effects by inhibiting the VEGFR KDR/Flk-1 signaling pathway in HUVECs and reducing VEGF-induced neovascularization in vivo. 57 Mechanisms of action in other cancer cell lines have been described for cytotoxicity58–61 and inhibition of cell migration. 62
Hydroalcoholic extracts of the Terminalia chebula fruit are rich in chebulagic and ellagic acids, which possess cytotoxic activity against several cancer cell lines.63,64 Chebulagic acid increased the cytotoxicity of doxorubicin in HepG2 cells by 20-fold via a COX-2-dependent mechanism, accompanied by the downregulation of MDR1 expression. 65 Tinospora crispa aqueous crude extract (IC50 = 165 µg/mL) 66 had no anti-angiogenic properties in the rat aortic ring assay, no effect in the tube formation assay using human umbilical vein endothelial cells (HUVEC) on Matrigel, and no acute cytotoxicity in selected human cancer cell lines. 67 Non-mechanistic reports on HepG2 cytotoxicity were found for Cyperus rotundus (IC50 < 10 μg/mL) 68 and Conioselinum anthriscoides (syn Ligusticum sinense), whose ethanolic extracts reduced the viability of liver cancer cell lines CL-6, HepG2, and Hep-2 after treatment at 50 µg/ml by approximately 44%, 69%, and 27%, respectively. 11 Although no reports on direct cytotoxicity against HepG2 cells were found for Cinnamomum siamense, Cryptolepis dubia, Brachypterum scandens (syn. Derris scandens), Putranjiva roxburghii (syn. Drypetes roxburghii), or Mallotus repandus, it is plausible that some non-cytotoxic ingredients may act via other anticancer mechanisms such as immunological activities, increasing the bioavailability of other compounds in the recipe, or even inhibiting multidrug resistance. For example, Carthamus tinctorius dried flowers water extracts revert the avoidance of immune destruction of cancer cells by stimulating IFN-γ and IL-10 secretion of splenic T lymphocytes and enhancing the maturation of dendritic cells (DCs). Moreover, reduced tumor weight in tumor-bearing mice changing cytokine secretion toward the Th1 pathway and increasing the population of cytotoxic T lymphocytes ex vivo. 69 The seed oil extract also exhibited antitumor effects in a skin and breast cancer animal model and in a melanoma cell assay system. 69 Notably, Piper nigrum may help overcome low bioavailability of other plant metabolites and overcome P-glycoprotein-mediated multidrug resistance,70,71 although piperidine and piperine have been reported to have potent anticancer in vitro and in vivo effects in other cancer cells,72–76 as has β-caryophyllene oxide, an important component of black pepper's fragrance. 77
All the above biomolecular and pharmacological effects of the components of the ASW formula in the most important mechanisms associated to the hallmarks of cancer 78 -as proposed by Li and Mansmann 79 —have been summarized in Figure 6.

Summary of the Molecular Mechanisms of the Ayurveda Siriraj Wattana Formula (ASW; AVS073 Variation) and its Constituent Ingredients on a Map Linking Hallmarks of Cancer with Cell Signalling Pathways. The Abbreviation CSL in red Denotes new Information Provided by the Present Work on the Effects of B. sappan Water Extract. See Table 1 for plant species abbreviations.
Effects of the ASW Ingredients on HEPG2 Cells Migration and Their Molecular Mechanisms
This is the first study to report on B. sappan water extract's ability to reduce 2D cell migration in HepG2 cells. Its active compound, brazilin, may be directly responsible for this effect, as it has previously been reported to suppress the migration and invasion of MDA-MB-231 breast cancer cells. 9 The progression of hepatocellular carcinoma is driven by the unrestrained proliferation and migration of liver cells, a complex process in which multiple signal transduction pathways are critically involved.78,79 A significant portion of existing therapeutic approaches are designed to inhibit these pathways. Specifically, pathways such as Wnt/β-catenin, MAPK, PI3K/Akt/mTOR, PKB/Akt, HGF/c-MET, JAK, and VEGF contribute to hepatocellular carcinoma development to varying extents, often in concert with gaseous signaling molecules like nitric oxide (NO), carbon monoxide (CO), and hydrogen sulfide (H2S). 80 Based on the work of Hsieh et al, brazilin inhibits cancer cell metastasis primarily by disrupting some of these specific signaling pathways, namely by suppressing the phosphorylation of p38 MAPK, PI3K, and Akt, which are crucial for cell survival and proliferation. This inactivation of the p38 MAPK and PI3K/Akt pathways, in turn, inhibits the activation of NF-κB. The result of this cascade is the suppression of MMP-2 expression, a key protein involved in breaking down the cellular matrix and allowing cancer cells to spread. The authors also explicitly state that brazilin does not affect the phosphorylation of ERK1/2 and JNK. 9 While a comprehensive investigation, like that conducted by Hsieh et al, would be needed to formally establish whether these mechanisms apply to the effects of B. sappan and brazilin on the HepG2 cell line, the conservation of these signaling pathways across different cancer types suggests that a similar outcome is highly probable. A dedicated study would therefore likely serve to confirm, rather than discover, the underlying mechanisms as illustrated in Figure 7.

Summary of the Molecular Mechanisms of B. sappan Extract and its Active Principle Brazilein on Cell Pathways Related to Cell Migration.
Modulatory Effects of Glutathione Levels of the ASW Ingredients on HEPG2 Cells
Glutathione (GSH), the most abundant tripeptide antioxidant, is crucial for maintaining cellular redox homeostasis and plays diverse roles in detoxification, proliferation, differentiation, and programmed cell death. In malignant neoplasms, disruptions in GSH synthesis and the GSH/GSSG ratio are common, significantly impacting tumor cell viability, initiation, progression, and drug resistance. 81 Elevated GSH levels in various cancer types result from increased expression of key γ-glutamyl cycle enzymes and altered precursor amino acid transport. 82 Beyond its direct antioxidant function, GSH-related enzymes like glutathione peroxidases (GPxs) reduce hydroperoxides crucial for cell viability, while glutathione S-transferases (GSTs) catalyze GSH conjugation to electrophiles and regulate vital cellular signaling pathways, including metabolism and anti-cancer drug resistance via S-glutathionylation.82,83 Given that high GSH levels in cancer cells confer protection against oxidative stress and xenobiotics, GSH depletion strategies are emerging as promising therapeutic targets to enhance the efficacy of various cancer treatments, including chemotherapy, radiotherapy, and ROS-based therapies like ferroptosis.83–86
Although the treatment of HepG2 cells with ASW water extract (200 μg/ml) significantly halved GSH control levels similarly to camptothecin 5 µM this was considered as a modest result precluding further screening of the ingredients. It came as a surpise given the inhibition of UVA-induced glutathione depletion observed in G361 skin cells incubated with WTN. 2 In these skin cells, WTN appears to safeguard GST activity and the expression of γ-GCLC, γ-GCLM, and GST mRNA under radiation stress. Further research is warranted to determine if this differential bioactivity can be harnessed for future therapeutic applications.
On the other hand, B. sappan water extract (50 μg/ml) showed no effect on total glutathione levels. Other ingredients of the formula, such as A. marmelos hydro-alcoholic extract are reported to significantly reduced glutathione 87 and B. rotunda is known to contain metabolites that modulate glutathione levels, such as Panduratin A which reduced the (GSH) depletion caused by t-BHP in HepG2 cells, thus providing cell protection to oxidative stress in a similar dose-dependent (10-15 µM) effect than silybin 88 but their contribution to the effect of ASW seems to be dimisnish eiter by a dilution effect or by changes in the solubility of their bioactive metabolites.
Implications for the use of Herbal Formulae in Thai Traditional Medicine and Other Traditional Medicines in Clinical Setups
The Thai Herbal Wattana formula has been traditionally used for promoting overall health in Thailand, but clinical evidence supports potential medicinal applications and Phase II clinical trials demonstrated that WNF possessed a good safety profile in patients with knee osteoarthritis. 1 The precise composition of traditional recipes exhibits both complexity and variability, in this case containing up to 18 distinct ingredients, often with qualitative differences documented over time.2–5 Our study documents the in vitro effects of ASW in HepG2 cells in a similar way to the study carried out by Mahavorasirikul et al with 28 ingredients and 5 Thai traditional recipes who reported on the missing activity against human cholangiocarcinoma (CL-6) of the Pra-Sa-Prao-Yhai recipe. Moreover, these authors discuss how the human hepatocarcinoma HepG2 cell line proved to be the most resistant to most of the tested extracts. 89 This finding underscores the difficulty in unravelling the effects of complex traditional recipes, suggesting that the synergistic or combinatorial effects of multiple ingredients—a key principle in traditional Thai medicine—may be a crucial area for future investigation.
Traditional Chinese medicine also appears to utilize formulae to “soothe the liver” as an adjuvant therapy for liver cancer patients. An example is the Da-Chai-Hu-Tang formula, which dates back almost two millennia. The formula contains eight herbals: Bupleurum chinense, Scutellaria baicalensis, Paeonia lactiflora, Pinellia ternata, Rheum officinale, Citrus × aurantium, Zingiber officinale, and Ziziphus jujuba. Clinical studies have demonstrated the effectiveness of this formula in hepatocellular carcinoma patients both directly and boosting their immunity. In vitro studies corroborated that the formula could induce cell cycle arrest and apoptosis in HepG2 cells by regulating the expression of E-cadherin, N-cadherin, p53, Bax, Bcl-2, PI3K, p-AKT, AKT, and STAT3 through the PI3K/AKT/STAT3 signaling pathway. 90 Apparently, in China the concept of “soothing the liver” may transcend liver cancer and be used as a therapeutic approach for breast cancer. In a study to investigate this concept, Zhao et al developed a herbal formula using three liver-soothing herbs: Cyperus rotundus L., Citrus medica L. var. sarcodactylis Swingle, and Rosa rugosa Thunb, the first ingredients two being identically or similarly present in ASW. The results from both in vivo and in vitro experiments demonstrated significant anti-cancer effects. In vitro studies on MCF-7 and T47D breast cancer cells confirmed these findings. The formula exhibited a dose-dependent inhibitory effect on cell growth by downregulating serum estradiol, upregulated apoptosis rates, and significantly decreased the protein expression of SNCG, ER-α, p-AKT, and p-ERK. 91
In India and Persia, Triphala, a fundamental traditional Ayurveda formulation consisting of Terminalia chebula, Terminalia bellirica, and Phyllanthus emblica combined with honey, is also a remedy for malignant and chronic diseases. The cytotoxic activity of its hydroalcoholic extract significantly reduces HepG2 cell survival in a concentration-dependent manner, with the extract exhibiting a mild IC50 of 78 μg/mL. 92 This has inspired a recent network pharmacology approach unveiled the potential in silico mechanisms of Terminalia chebula fruit extracts in the treatment of hepatocellular carcinoma 93 illustrating the importance of traditional use to inform cutting-edge drug-discovery computational approaches.
It is known that progression from chronic liver disease to hepatocellular carcinoma involves a complex interplay of molecular mechanisms, often driven by the dysregulation of key signaling pathways. In this context, the inhibitory effects of brazilein 9 on the MAPK and PI3K/AKT/mTOR pathways, which are highly relevant to hepatocellular carcinoma pathogenesis. 94 The PI3K-AKT signaling pathway has been identified as a significant target for other compounds, such as aloe-emodin, in the treatment of HCC, 95 providing a strong molecular rationale for the historical inclusion of B. sappan in ASW, an important formula among the above mentioned traditional Asian “liver formulas”. For instance, research could focus on whether the recipe enhances the efficacy of existing treatments, such as gelation embolism agents used in TACE to combat pro-metastatic microenvironments. 96 Furthermore, its interaction with cellular pathways could be explored in conjunction with compounds like talazoparib, particularly in the context of specific genetic vulnerabilities like the loss of heterozygosity of CYP2D6. 97 Research could also explore whether the recipe's effects are mediated through a gut-liver axis, similar to how Lactobacillus acidophilus produces valeric acid to suppress HCC. 98
This paper, like other ethnopharmacological studies, seeks to elucidate the mechanisms of traditional remedies. The goal is to facilitate and guide future pre-clinical and clinical strategies that integrate natural compounds with established efficacy and long-standing safety with contemporary chemotherapy. An example of this approach is the synergistic cytotoxic and migration-inhibitory effect observed when brazilein is combined with cisplatin in 4T1 breast cancer cells 99 or how chebulagic acid is combined with doxorubicin in HepG2 cells. 65 However, systematic combination studies following strict mathematical protocols such as the one proposed by Chou-Talalay 100 are hampered by the number of ingredients of a typical herbal formula which offers hundreds of thousands of potential combinations.
Impact and Limitations of This Study
The present study advances the preclinical understanding of the Thai Herbal Wattana formula (ASW variation) providing a scientific basis for its traditional use in the context of hepatocarcinoma. Its uniqueness stems from its focus on ASW as a specific formulation with traditional applications, rather than its value in the field of drug discovery. A key impact is the identification and dereplication of brazilein as the primary cytotoxic principle within B. sappan, thereby linking traditional efficacy to specific secondary metabolites. The observed glutathione modulation occurs at concentrations considered non-physiological, therefore suggesting the safety of the formula in patients with compromised liver function and informing practical implications for integrative oncology. These findings suggest that the positive effects of ASW in treating cancer may be primarily linked to its established ability to modulate the immune system and reduce inflammation, an important facet in many tumor patients when the cancer-immunity cycles fail to run optimally, leading to tumor development and even endangering the host's life. 101
Despite these contributions, this study is subject to several limitations. Foremost, its exclusive reliance on an in vitro HepG2 cell model restricts the direct generalizability of findings to complex in vivo physiological systems, thereby emphasizing the critical need for subsequent animal and clinical studies. However, the genetic and phenotypic diversity of hepatocellular carcinoma (HCC) is here acknowledged. Different HCC cell lines, such as Huh-7 and Hep3B, carry distinct genetic mutations (eg, in TP53 and viral integrations) and display varying growth rates and signaling pathway activities, which can lead to differential responses to therapeutic agents. As comprehensively surveyed by Nwosu and co-workers, poorly differentiated HCC cells, which exhibit an aggressive phenotype, show altered metabolic pathways, including a higher reliance on glutamine for energy, making them uniquely susceptible to treatments that target the glutamine pathway. 102 Future work would certainly benefit from expanding the cell panel to include more HCC lines to validate and extend our observations. Such an approach would provide greater translational relevance by offering insight into how our findings might apply to different HCC subtypes. This is a critical next step, as the varied genetic backgrounds of cell lines can influence everything from chemosensitivity to the activation of key signaling cascades like the PI3K/Akt/mTOR pathway. 102 Ultimately, incorporating a diverse panel of cell lines will not only confirm our initial results but also allow us to identify specific genetic or phenotypic markers that predict a positive response to our agent, moving us closer to a personalized medicine approach for liver cancer treatment.
The limited direct cytotoxicity observed for the overall ASW formula, coupled with a modest reduction in glutathione levels that precluded broader screening, suggests a need for further optimization or exploration of its synergistic potential. Furthermore, the exclusive use of water extracts may have overlooked the bioactivity of lipophilic compounds present in the original herbs, but at the same time reduces the toxicity of the formula.
Future work should confirm the apoptotic pathway through molecular assays by investigating the effects of WNF and its constituents on an extended panel of liver cancer cells as well as normal hepatocytes or liver slices, 103 but we may avoid certain experimental work with in silico approaches such as exploring the anti-liver cancer mechanisms of natural entities using network pharmacology. 95
Finally, to advance preclinical testing, the Wattana recipe could be evaluated using sophisticated models like patient-derived precision-cut tissue slices, 104 which more accurately replicate the tumor microenvironment. If the recipe's effects are specific to certain tumor subtypes, a DNA methylation-based classifier could be used to differentiate tumor types and select patients who may benefit most. 105 In a clinical setting, NIR-II fluorescence-guided surgery could be used to track the recipe's effects or enhance tumor removal. 106
Conclusion
This preclinical study demonstrates that while the ASW formula lacks direct in vitro cytotoxicity towards HepG2 cells, it effectively inhibits their mobility and depletes intracellular glutathione although at non-physiological concentrations. Biancaea sappan (syn. Caesalpinia sappan), a core ingredient of ASW, exhibits potent cytotoxic and anti-migratory activities, with brazilein being its primary cytotoxic compound. However, brazilein is not endowed with the glutathione-depleting or anti-migratory effects observed in the full ASW formula. Further in vivo studies should be carried out to ascertain if long-term treatments with ASW may prevent invasion and metastasis of HepG2 cells without causing any significant cytotoxic effect. These findings suggest that ASW's benefits in oncology may primarily link up with its established clinical immunological and anti-inflammatory effects, while its lack of toxicity to HepG2 cells, a proxy for hepatocytes, plus its clinically proven lack of major adverse effects might indicate a positive safety profile.
Footnotes
Acknowledgements
We are grateful to Prof. Simon Gibbons (Liverpool John Moores University, UK) for kindly reading this paper and providing with invaluable feedback.
ORCID iDs
Ethical Considerations
Ethical Approval is not applicable for this article.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
