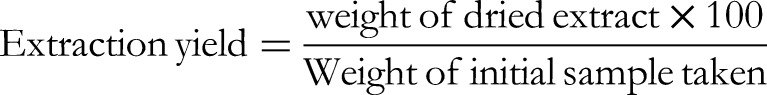Abstract
Objective
Ginger (Zingiber officinale) is well known for its therapeutic properties, particularly its anti-emetic effects and pain modulation potential. The research on nausea and pain modulation has primarily focused on ginger and shogaols. The potential role of other nonvolatile secondary metabolites in these activities remains largely unexplored. This study aims to comprehensively characterize secondary metabolites in fresh and oven-dried ginger using LC-MS and evaluate the pharmacological potential of identified compounds in nausea and pain modulation.
Method
Fresh and dried ginger rhizomes were extracted using multiple solvent systems (methanol, aqueous methanol, water, hexane, and acetone. The extracts were analyzed for phenolic content by the Folin & Ciocalteau method and antioxidant potential by β-carotene-linoleic assay. A comprehensive chemical fingerprint of all the extracts was generated using LC-MS. Compounds with favorable ADME profiles were subjected to molecular docking to evaluate their binding interactions with 5HT3 (5-hydroxy tryptamine Type 3 receptor) and TRPV1 (transient receptor potential vanilloid 1) receptors.
Results
LC-MS fingerprinting identified 61 bioactive compounds in a single run and highlighted distinct variations in compound distribution between fresh and oven-dried ginger extracts. Methanol was the most suitable solvent for the extraction of secondary metabolites, and the drying process showed a significant increase in shogaols content with relatively higher antioxidant activity. Diarylheptanoids, a relatively underexplored class of ginger secondary metabolites, exhibited strong binding affinity to 5HT3 and TRPV1 receptors, surpassing that of commonly studied gingerols and shogaols, suggesting their potential role in ginger's therapeutic effects.
Conclusion
The study presents a comprehensive LC-MS fingerprinting of fresh and oven-dried extracts of Zingiber officinale rhizome. Oven-dried ginger extracts showed significantly higher phenolic content, antioxidant activity, and levels of shogaols and diarylheptanoids. Several diaryl heptanoids demonstrated strong in silico binding to 5HT3 and TRPV1 receptors, suggesting that, these metabolites may contribute meaningfully to ginger's anti-emetic and analgesic potential.
Introduction
Zingiber officinale ROSCOE, or ginger, also known as “white ginger,” has been commonly consumed as a spice in many civilizations. Its medicinal use is well documented in traditional medicinal systems like Ayurveda for various ailments. Coming from the same family of Zingiberaceae as Curcuma longa, ginger also shares many of its biological activities like anti-inflammatory, anti-carcinogenic, and antioxidant activities. 1 Its traditional applications include use as a remedy for headache, pain, nausea, and vomiting. 2 In recent times, the use of Ginger has extended to managing joint pain, sciatic pain, chemotherapy-induced nausea, and vomiting (CINV). 3 Dry ginger is part of many Asian cuisines and has been known to absorb the water element of pitta dosha. 4
Many of Ginger's pharmacological and physiological activities have been studied in reference to its active compounds, gingerols and shogaols. Apart from these, the Zingiberaceae family is known to have several other compounds of interest, such as zingiberene, zingerone, and diarylheptanoids.
Ginger's widespread medicinal use suggests multifaceted mechanisms of action, influencing multiple physiological pathways in the body. A semantic relationship graph (Figure 1) constructed from a PubMed search for the word ‘ginger’, using SPACY 5 based Named Entity Recognition model, highlights primary disease conditions associated with research on ginger. Extracted entities in Figure 1 represent nausea and pain as the most commonly studied health conditions related to ginger. In addition to these conditions, CINV, migraine, fatigue, and vomiting were found to be closely related to ginger.
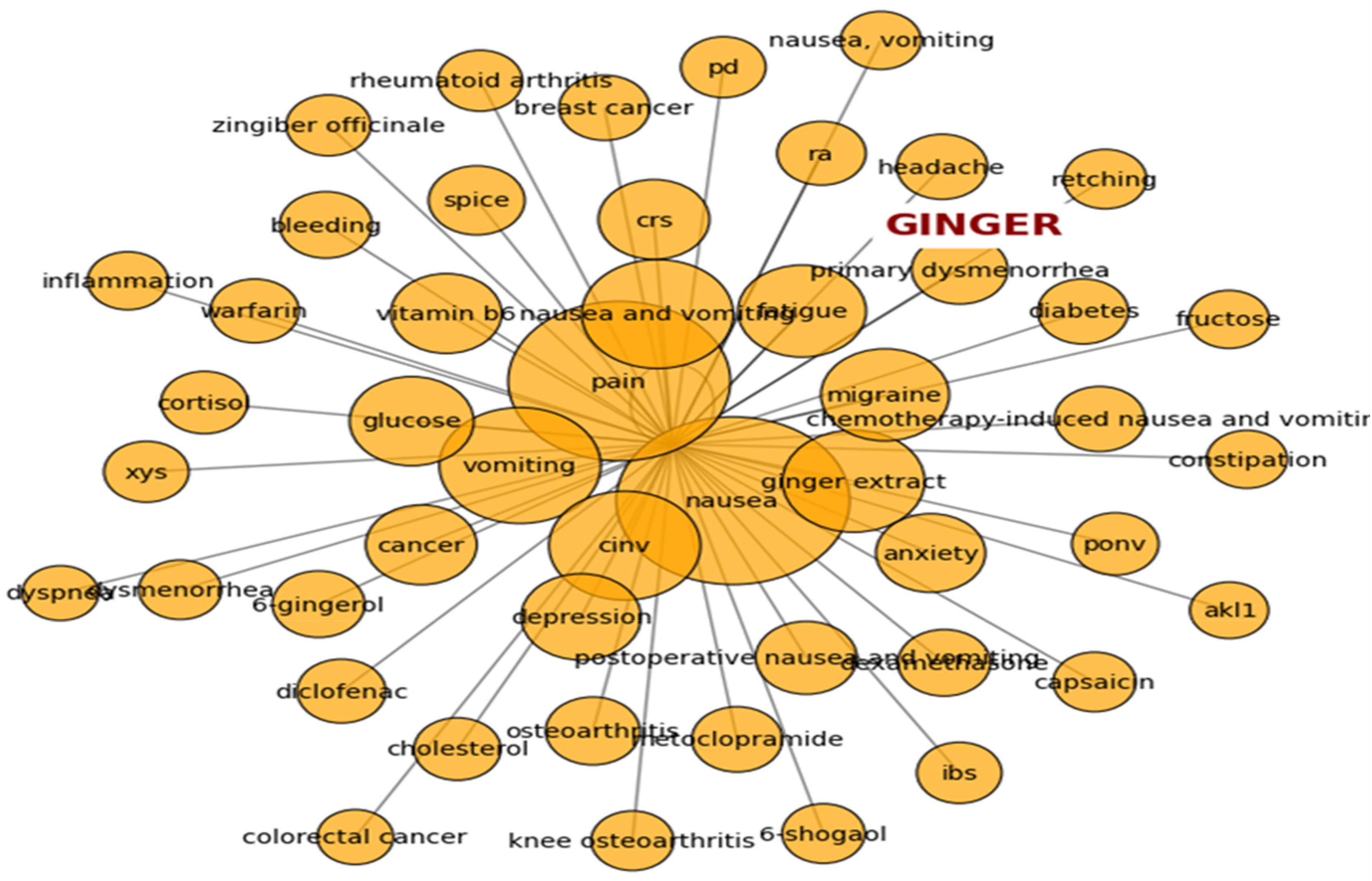
NLP Based Semantic Relationship Network for top 50 Terms Associated with Search Word “Ginger”.
Ginger's anti-emetic activity in CINV, nausea, and vomiting in pregnancy (NVP) has been studied in several clinical trials. 6 In a recently published review that included 23 randomized clinical trials (RCTs), most of the clinical studies exploring the anti-emetic activity of ginger were conducted using ginger powder, and 6-gingerol and 6-shogaol have been cited as the main bioactive contributing to its anti-emetic activity. 7 5HT3 receptor has been considered as a validated pharmacological target for potential anti-emetic compounds. Nausea is an outcome of 5-HT release from intestinal mucosal cells, which causes activation of 5HT3 receptors present in abdominal vagal afferents, stimulating emetic or vomiting reflex. Antagonistic action on this receptor is believed to result in anti-emetic action. 8
Ginger's pain-modulating activity has spiked interest in its application in sports nutrition, geriatric health supplements, and in painful diabetic neuropathy for managing pain and inflammation. Gingerols and shogaols have been reported as the main bioactive in ginger responsible for its pain modulating or analgesic activity. 9 To study the pain modulating activity of ginger compound, we chose TRPV1 or Transient receptor potential vanilloid 1 channel, as it is a key target for pain modulation, as a non-selective cation channel that can play vital role in pain perception and nociception. Role of TRPV1 channel has been supported by several pharmacological studies for pain modulation. 10
Given the well-established anti-emetic and pain-modulating properties of Zingiber officinale, most studies have focused on gingerols and shogaols as the primary active compounds. However, the potential role of other nonvolatile secondary metabolites in these therapeutic effects remains insufficiently explored. Furthermore, processing methods—particularly fresh versus oven-dried ginger—and solvent systems influence the extraction and composition of these compounds, which is not well understood.
This study aims to address these gaps by employing LC-MS fingerprinting to comprehensively characterize the secondary metabolite profiles of fresh and oven-dried ginger extracts. It also seeks to evaluate total polyphenolic content and antioxidant potential across different solvent systems. Furthermore, the study aims to explore the pharmacological relevance of identified compounds through in silico analysis targeting 5HT3 and TRPV1 receptors, better to understand their potential role in nausea and pain modulation.
Material & Method
Material
Zingiber officinale ROSCOE var Officinale was purchased from local market in new jersey USA and authenticated by in-house Botanist. Folin Ciocalteu's, Tween 40, Butylated hydroxy toluene (BHT), beta carotene, linoleic acid, gallic acid, sodium carbonate and all solvents were purchased from Millipore-sigma (Merck KGaA, Darmstadt, Germany). The column used in the study was Kinetex® (Phenomenex - Torrance, CA). HPLC-grade water was from the Millipore Direct-Q®3UV system.
Extract Preparation
Ginger extracts were prepared from fresh and oven-dried ginger rhizomes. The extraction process involved five different solvents: methanol, methanol: water (50:50), water, hexane, and acetone. A parallel extraction method was used. The extraction procedures for both fresh and dried ginger rhizomes was standardized and validated in our laboratory.
Fresh Ginger Rhizome Extracts (FGE)
Fresh ginger rhizomes were cut into small pieces, and 20 g of the material was separately extracted using five different solvents: methanol, aqueous methanol (50:50), hexane, water, and acetone. For each solvent, 20 g of ginger was soaked in 50 mL of the respective solvent, sonicated (Hash Branson Ultrasonic bath) for 1 h, and left for extraction at room temperature overnight. The filtrate was collected the next day, and the extraction process was repeated twice. Combined filtrates for each solvent were dried under vacuum (90 mm Hg, condenser temperature 5-10˚C, water bath temp 45˚C) at room temperature to minimize degradation of heat-sensitive compounds. The dried extracts were then weighed and stored for further analysis.
Oven Dried Ginger Rhizome Extracts (OGE)
Fresh ginger rhizomes were cut into small pieces, air-dried overnight, and subsequently oven-dried at 50 °C for 24 h. For each solvent—methanol, aqueous methanol (50:50), hexane, water, and acetone—2 g of the oven-dried ginger was separately soaked in 50 mL of the respective solvent, sonicated for 1 h, and left for extraction overnight at room temperature. The filtrate was collected the next day, and the process was repeated twice. Combined filtrates for each solvent were dried under vacuum at room temperature. The resulting dried extracts were weighed and stored for further analysis.
The extraction yields for both fresh and oven-dried extracts were calculated as below:
Antioxidant Activity (AOA)
The AOA was assayed by autoxidation of Beta carotene and linoleic acid as described by Emmons and Peterson. 11 100 mg of samples (fresh ginger rhizomes and oven dried ginger rhizomes) were extracted with 2.0 mL of following solvent system (a) methanolic (b) hydro-methanolic (c) Aqueous (d) hexane (e) acetone, separately for overnight. Reaction mixture was prepared as follows; 2 mg of Beta carotene was dissolved in 20 mL of chloroform and 3 mL of this solution was added to 40 mg of linoleic acid and 400 mg of Tween 40. After removing chloroform under reduced pressure, 100 µL of oxygenated water was added and mixed properly over a vortex mixture to obtain a stable emulsion. 3.0 mL aliquots of emulsion were then mixed with 40 µL of ginger extract prepared as above and incubated for 1 h at 50 °C. The absorption (λmax 470 nm) of test mixture was recorded at 0 and 60 min of incubation at 50 °C. The AOA was expressed as a percentage inhibition relative to the control. The activity of extracts was compared with a standard curve of BHT over a concentration range of 0- 1.4 µg in 40 µL of sample.
Standard Curve of Butylated Hydroxy Toluene (BHT)
Butylated hydroxy toluene (BHT), generally used as a commercial antioxidant, was taken as a standard to compare the antioxidant activity. Activities of extracts were compared with a standard curve of BHT activity over a concentration range of 0-1.4 µg in 40 µL of sample. Preparation of BHT standard curve was performed using protocol described by Emmons and Peterson. 11
Estimation of Polyphenolic Content
Fresh and oven dried ginger rhizomes (100 mg) were individually extracted with methanol, Aq methanol, water, hexane and acetone (2 × 10 mL), overnight at room temperature. The combined extractives from each solvent were centrifuged at 6000 g for 15 min, filtered, and diluted to 20 mL each. Total phenolic content (TPC) in different extracts was measured with slightly modified method of Ragazzi and Veronese. 12 In 1 mL of extract, 1 mL of Folin's Reagent (1N) and 2 mL of Na2CO3 (20%) were added subsequently. The test mixture was mixed properly on a cyclomixer, left at room temperature for 30 min and maintained to 25 mL with water. The absorbance of test mixture was measured at λmax 725 nm on Agilent Technology's Cary 60 Spectrophotometer. The reported TPC was expressed as gallic acid equivalent (GAE) mg/g on a dry weight basis. The quantities of phenols were calculated by comparison with standard curves of gallic acid, under identical experimental conditions.
Standard Curve of Gallic Acid
50 mg of gallic acid was dissolved in 50 mL of 50% methanol/ water to obtain the stock solution. It was diluted 1:5 to get an end concentration of 200 µg/mL, to prepare the standard curve 50 µL, 100 µL, 200 µL, 250 µL, 300 µL, 350 µL, 400 µL, 450 µL and 500 µL aliquots of diluted stock solutions were taken in triplicate maintained to 1 mL with 50% methanol/ water. Further process was followed as described above in the estimation of phenols by the method of Ragazzi and Veronese. 12
Analytical Conditions
LC-MS Parameters for Generating Chemical Fingerprints
LC-MS fingerprints of the various ginger extracts was planned to use an Agilent 1200 series HPLC system (Agilent Technologies, Waldbronn, Germany). Separation was performed on a KinetexTM core shell technology C-18 column (250 × 4.6 mm, 5 µ). Photodiode array (PDA) detector at wavelength 280 nm was used for detection. The mobile phase consisted of 0.1% formic acid (pump A) and acetonitrile: water (98:2, pump B). A gradient program was employed to achieve the optimal separation of a wide range of compounds. The gradient program commenced with a 20% acetonitrile composition and then progressively increased to 50% for 20 min. Subsequently, within the following 20 min, the composition rose to 65%, and in another 10 min, it further increased to 85%. After 55 min, the composition reached 95% and remained constant for 2 min. Within the subsequent 3 min, it reverted to the initial 20% composition, completing the entire run within 65 min. The flow rate was maintained at 0.8 mL/min throughout the run. FGE and OGE were dissolved in 5 mL of methanol and filtered through 0.45 µm polytetrafluoroethylene (PTFE) filter before injection into the system. The injection volume was 20 µL. LC-MS conditions used for fingerprinting of ginger extracts were standardized in our laboratory.
Mass spectrum was recorded by Agilent 6120 HPLC single Quad MS using ESI mode and ChemStation software version C.01.10[314]. The MS conditions were given as follows: capillary voltage 4000 V for both positive and negative, drying gas flow was 12 L/min, nebulizer pressure was 40 psig and drying gas temperature was 300 °C.
Estimation of Gingerols and Shogaols
To evaluate the efficiency of different solvents in extracting the gingerols and shogaols from both fresh and dried ginger samples, they were dissolved in 5 mL of methanol and filtered through 0.45 µm PTFE filter before injection into HPLC system. Quantitative estimation of gingerols and shogaols in extracts was done using USP monograph method NF 24.
ADME
For the determination of ADME, as a first step, the canonical SMILES information of the identified compounds in the LC-MS analysis was obtained through PubChem server (https://Pubchem.ncbi.nlm/nih.gov/). The SMILES for individual compounds were subsequently entered into the SwissADME online tool to evaluate their pharmacokinetic properties including absorption, distribution, metabolism and excretion. The ADME assessment was based on parameters such as molecular weight (MW), topological polar surface area (TPSA), Gastrointestinal (GI) absorption, Lipinski's rule of five and PAINS alert (Pan Assay Interference Compounds). In silico ADME profiling was carried out using SwissADME platform using the default settings for the prediction of GI absorption, Lipinski's rule of five and PAINS alert.
Molecular Interaction Analysis
Preparation of Proteins
To understand the individual anti-emetic activity and pain-modulating activity of various components of ginger extracts, we selected serotonin 5-HT3 receptor and TRPV1 receptor. Both the proteins, 5HT3 (PDB ID : 6HIN)) and TRPV1((PDB ID: 7L2S) with resolution of 4.10 A and 2.71A respectively, were downloaded from Protein Data Bank (rcsb.org). The Discovery Studio Visualizer software interface was used to prepare the proteins for molecular docking. The co-crystalized water molecule, heteroatoms (Hetatm) and non-essential chains were removed from both proteins, and polar hydrogens were added. Prepared proteins were loaded on PyRx and converted to PDBQT format.
Preparation of Ligands
3D Structures of reference compounds Ondansetron (PubChem CID: 4595), Capsaicin (CID 1548943), and selected compounds of ginger extracts based on their favorable ADME and Lipinski rule of five analysis, were downloaded from PubChem and saved in their SDF format. The energy minimization was carried on PyRx for all the ligands, using MMFF94 (Merck Molecular Force Field) with total 5000 steps (steepest descent method) and converted to their PDBQT format for docking.
Molecular Docking
Molecular docking simulations were performed using PyRx open-source software, which included Auto Dock Vina program. The prepared PDBQT files for both proteins and ligands were loaded in the Auto Dock Vina and Vina search space was performed on the proteins (6HIN, 7L2S) for ligand binding sites separately. The Vina Search space parameters were as below:
Molecular interaction was studied using Auto Dock Vina following the protocol by Trott and Olson 13
Statistical Analysis
Extraction efficiency, Total phenolic content, and anti-oxidant activity were determined in triplicates and results are expressed as mean ± standard deviation (SD). Statistical comparison between fresh and dried ginger extract across different solvents was reported using Student's t-test, with P < .05 considered significant. IBM SPSS statistics Inc software package was used for statistical analysis.
Results
Determination of Extraction Efficiency
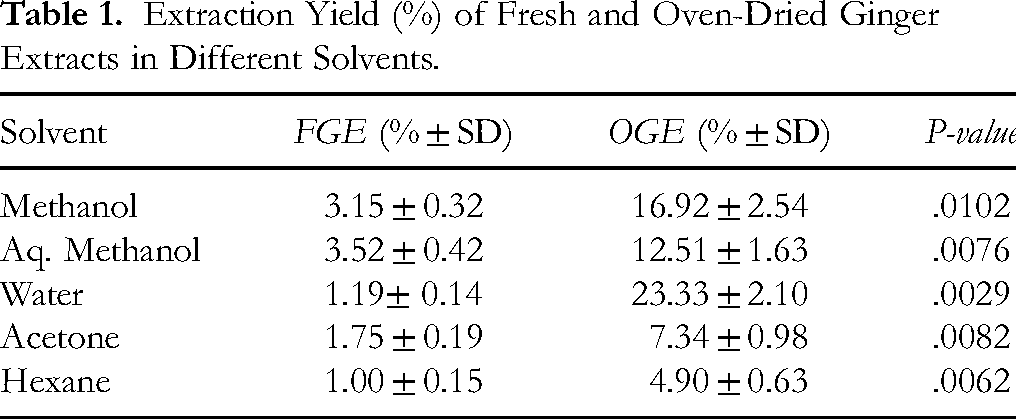
The extraction efficiency of various solvents for both fresh and dried ginger rhizomes was calculated. Results (Table 1) indicated that methanol was the most suitable solvent for FGE and OGE followed by Aq. Methanol. FGE generally showed lower extraction yield due to the presence of water in the rhizomes. All comparison shows statistically significant differences (P > .001) confirming that OGE consistently yields higher extract amount.
Extraction Yield (%) of Fresh and Oven-Dried Ginger Extracts in Different Solvents.
Antioxidant Activity
Antioxidant activity was calculated using protocol from Emmons and Peter, 11 which is based on the autoxidation of β carotene in the presence of Linoleic acid where it acts as an oxidative agent. This method evaluates how well an antioxidant (test substance) can prevent the degradation of β carotene caused by free radicals generated during oxidation of linoleic acid. Naturally occurring antioxidants in ginger are responsible for its biological activity against several chronic health conditions. The antioxidant activity (Table 2) of methanolic extract of OGE showed maximum antioxidant activity in relationship to BHT, followed by aq methanolic extract. FGE showed lower antioxidant activity compared to OGE in all solvents. Significant differences (P < .05) were observed for water, acetone and hexane.
Antioxidant Activity (% Inhibition, ±SD) of Fresh and Oven-Dried Ginger Extracts in Different Solvents.

Total Phenolic Content
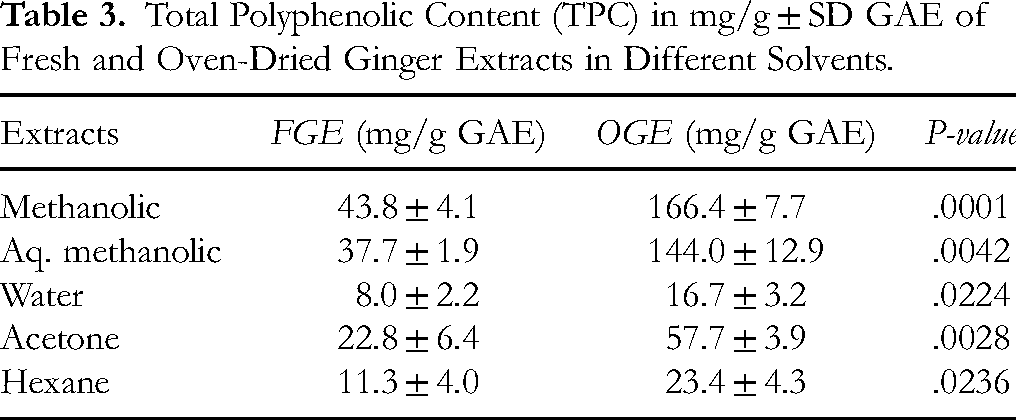
The phenolic content of the extracts was analyzed using Folin-Ciocalteu method. A calibration curve was constructed with gallic acid and individual extracts were compared to the graph for estimation. The reported total polyphenolic content (TPC) was expressed as gallic acid equivalent (GAE) mg/g on a dry weight basis. Ginger rhizomes contain several polyphenolic compounds such as 6, 8, and 10 -gingerols, shogaols, and paradols, which are responsible for the health benefits of ginger. Our results (Table 3) on polyphenolic content were directly correlated with the antioxidant activity of the extracts from both FGE and OGE, with the highest results obtained from the methanolic extract of OGE, followed by aq methanolic extract of OGE. Methanol was found to be the most effective solvent for the extraction of ginger polyphenols. Statistical analysis showed differences between OGE and FGE were significant (P < .01)
Total Polyphenolic Content (TPC) in mg/g ± SD GAE of Fresh and Oven-Dried Ginger Extracts in Different Solvents.
Quantitative Estimation of Gingerols and Shogaols by HPLC
The HPLC analysis of FGE & OGE (Figure2(a) and (b)) by the USP method showed a clear trend in the content of gingerols and shogaols in both FGE and OGE. OGE (Table 4) generally showed higher concentrations of gingerols & shogaols compared to fresh ginger extract, particularly methanolic extract (9.3 × 10−1mg/100 mg) and acetone extract (5.5 × 10−1mg/100 mg). This indicates that the drying process enhanced the extractability of gingerols & shogaols. The results highlighted that organic solvents such as methanol seem the most efficient in extracting these bioactives for dietary supplements.

Overlay HPLC chromatogram of FGE (a), OGE (b) . Blue color: methanolic extract, red color: aq methanolic extract, green color: hexane extract, pink color: acetone extract and olive green color: water extract. X axis: absorbance mAU and Y axis: RT (min).
Content of Total Gingerols (6, 8, 10) and Shogaols (mg/100 mg) in FGE and OGE in Different Solvents.

LC-MS Chemical Fingerprint
A comprehensive profiling was generated for methanolic OGE and a total of 61 nonvolatile secondary metabolite compounds were identified based on their mass spectra and fragmentation pattern. HPLC chromatogram and Total ion chromatogram are represented in Figures 3 and 4. Mass error of identified compounds was less than 1 ppm.

HPLC Chromatogram of Methanolic OGE. X Axis: Absorbance mAU and Y Axis: RT (min).

Total Ion Chromatogram (TIC) of Methanol Extract of Dried Ginger. X Axis Absorbance Y Axis RT.
The complete characterization of methanolic OGE using Mass spectroscopy is presented below (Table 5).
The Mass Spectral Characteristic of Methanolic OGE Detected by LC- ESI- MS.

ADME Screening Result
From the list of identified compounds in LC-MS analysis (Table 5), canonical SMILES of 33 compounds (Supplementary Table 1) were retrieved from PubChem database. These compounds were further subjected to In-Silico pharmacokinetic evaluation using SwissADME online tool. Based on the values of assessment parameters (in Table 6) such as molecular weight (MW), GI absorption, Topological polar surface area (TPSA), Lipinski Rule of Five violations and PAINS # alerts, 13 compounds were selected for further docking study with 5HTP-3, TRPV1 receptor proteins. (Table 6).
Pharmacokinetic Properties and Docking Selection of Bioactive Compounds from SwissADME Analysis.
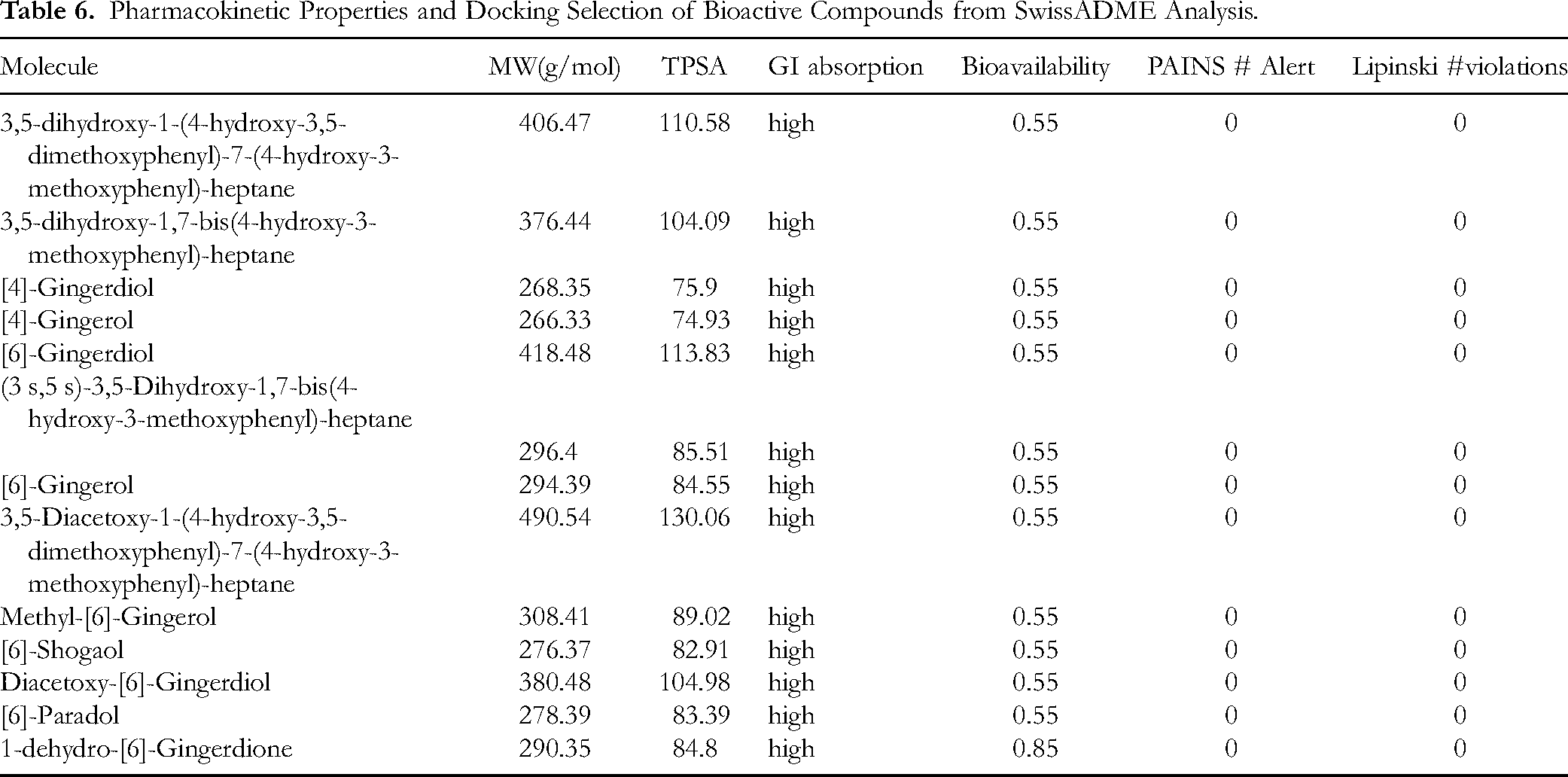
Molecular Docking Results
5HT3 Receptor Protein (PDB ID: 6 HIN)
The molecular docking results revealed that reference compound Ondansetron (CID: 4595) and selected bioactives from methanolic OGE (compounds
Molecular Docking Interaction with 5HT-3 Receptor Protein (6HIN) with Binding Energy and Interaction Types.

For the binding to 5HT3 receptor protein, Ondansetron (PubChem ID 4595) with IUPAC 9-methyl-3-((2-methyl-1H-imidazol-1-yl)-methyl)-2,3-dihydro-1H-carbazol-4(9H)-one was taken as a reference compound. Ondansetron is a selective 5HT3 antagonist, which is used in clinical practice as an anti-emetic compound for CINV. Based on the favorable ADME profile, 13 compounds from methanolic OGE were selected for molecular docking with the 5-HT3 receptor (PDB: 6HIN) (Table 8). Ondansetron, the reference ligand, exhibited the strongest binding affinity (−7.3 kcal/mol), interacting with key amino acid residues TYR64 and LYS127 through van der Waals forces and forming hydrogen bonds and π-π/π-alkyl interactions with residues such as TYR61, ARG65, and TRP63. Visual annotation of compounds docked with 5HT3 receptor proteins is represented in Supplementary Figure 1.
Molecular Docking Interaction with TRPV1 Receptor Protein (7L2S) with Binding Energy and Interaction Types.

Among the methanolic OGE compounds- 3,5-Diacetoxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3methoxyphenyl heptane [30), −7.0 kcal/mol] demonstrated the highest binding affinity to 5HT3 receptor protein (6HIN). It was closely followed by 3,5-dihydroxy-1-(4-hydroxy-3,5-dimethoxy phenyl)-7-(4-hydroxy-3-methoxy phenyl)-heptane [ (4), −6.9 kcal/mol] and 1-dehydro-
TRPV1 (PDB ID:7L2S)
For docking with TRPV1, we selected Capsaicin (CID 1548943) as a reference ligand. 13 compounds including diarylheptanoids, and gingerols from methanolic OGE were docked with TRPV1 protein receptor to evaluate the analgesic activity of methanolic OGE. The In-silico binding results are presented in Table 8.
The molecular docking of methanolic OGE compounds and capsaicin was performed with the TRPV1 receptor (PDB: 7L2S). The results indicated that several methanolic OGE compounds demonstrated stronger binding interactions than capsaicin (−6.5 kcal/mol). The diaryl heptanoids exhibited the highest binding affinity- 3,5-Diacetoxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl) heptane [(
The docking interactions included Van der Waals interactions and hydrogen bonding with residues such as TYR554, SER512, VAL567, ALA566, and ARG557 present in the binding pocket of the protein molecule.
These findings suggest in addition to gingerols, diarylheptanoid compounds, may play an important role in exhibiting the pain modulation effect of ginger.
The binding poses of the compounds are presented in Supplementary Figure 2.
Discussion
Ginger's phytochemistry includes a group of nonvolatile secondary metabolite compounds, such as gingerols, shogaols, and diarylheptanoids, which set it apart within the Zingiberaceae family. These active compounds undergo transformations during processing which further enrich their chemical complexity and biological potential. Major bioactives in Ginger can be categorized into five groups based on their structural relationships and transformations. Gingerols, homologs of 1-(3-methoxy-4-hydroxyphenyl)-3-keto-5-hydroxyhexane, serve as the foundational compounds in ginger's metabolic network. Through dehydration, gingerols transform into shogaols, which exhibit distinct chemical and biological properties. Paradols emerge as deoxygenated products of gingerols, forming through the loss of the beta-ketone hydroxyl group. Further oxidation of gingerols results in the formation of gingerdiones, with 1-dehydrogingerdiones representing a specific subset of these beta-ketone dehydrogenation products. Additionally, the reduction of the carbonyl group in gingerols produces gingerdiols, completing the transformation pathways. Along with the above groups we also identified many of di aryl heptanoids.
The present study offers a comprehensive evaluation of extracts obtained from fresh and oven-dried ginger rhizomes, incorporating multiple aspects such as extraction optimization, total phenolic content, antioxidant activity, quantitative HPLC analysis of gingerols, shogaols as well as complete characterization.
The extraction efficiency and phytochemical composition of ginger extracts were significantly influenced by the choice of solvent and drying process. In the case of oven-dried ginger extract (OGE), water exhibited the highest extraction yield, followed by methanol. In the case of OGE, our results showed much higher yields with all solvents except hexane which is in contrast with an earlier published study. 14 One of the reasons could be the difference in the raw material used for extraction, Nadeen WAA et al 14 took a crude powdered ginger extract from local herbalists for further extraction, whereas we used coarse oven-dried ginger rhizomes. However, the total phenolic content (TPC) and antioxidant activity were highest in the methanolic OGE, suggesting its superior ability to extract phenolic compounds. In contrast, despite the higher yield observed with water extraction, its lower TPC and antioxidant activity may be attributed to the extraction of non-polyphenolic constituents.
Quantitative HPLC chromatographic analysis provided additional insight into chemical transformation that occurred in the drying process with a noticeable increase in shogaol content in OGE compared to FGE. Overall methanol was the most effective solvent for extracting these compounds, reinforcing its use in optimizing the ginger extraction process for enhanced health benefits. Qualitative estimation of diarylheptanoids in OGE and FGE indicated that methanol was the most suitable solvent for extracting this class of bioactive compounds from ginger.
This study provides a comprehensive LC-MS fingerprinting analysis of FGE and OGE ginger extract. The key findings of this study are the distinct variation in compound distribution between different ginger extracts. Compound
Gingerols
Gingerols are the homologs of 1-(3-methoxy-4-hydroxyphenyl)-3-keto-5-hydroxyhexane. We identified - [4] (Compound
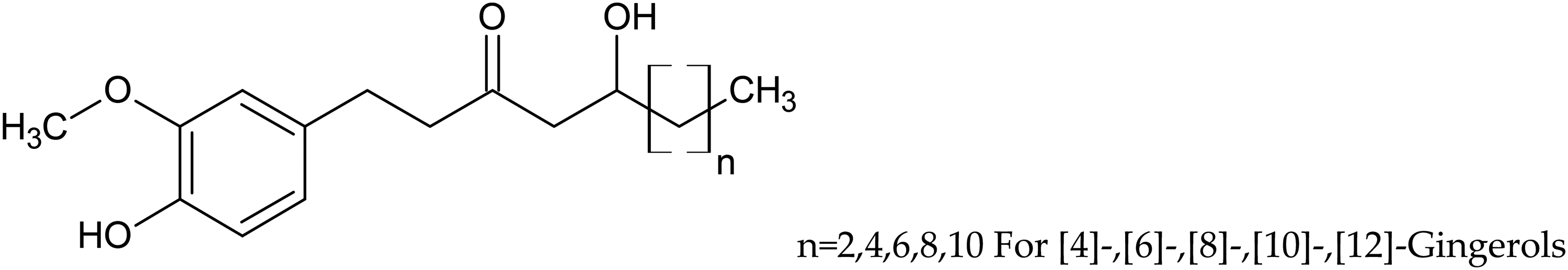
Structure of [4]-,[6]-,[8]-,[10]-,[12]-Gingerols.
Methyl- [6], - [8], and – [10] gingerol (compound

Structure of Methyl-{[6]/, -[8]/,-[10]}-Gingerols.
Despite the absence of ammonium ions in the mobile phase, the acetate derivatives of gingerols exhibited a distinct ammonium ion adduct may be due to its presence in water, in addition to the sodium ion adduct, in the positive ionization mode. The predominant product ion observed in the positive mode, (M + H-CH3COOH)+, strongly suggests the presence of an acetate group within the aliphatic chain. Acetoxy -[6], -[8], and –[10] gingerol (compound

Structure of Acetoxy-{[6]-,/[8]-,/[10]-}-Gingerols.
Shogaols
They are dehydrated products of gingerols and were found in higher concentrations in dried ginger extracts. We identified -[6],-[8], -[10]-shogaol (compound

Structure of [6]-,[8]-,[10]- Shogaols.
Paradols
Paradols are deoxygenated products of gingerols, and [6]-Paradol (Compound
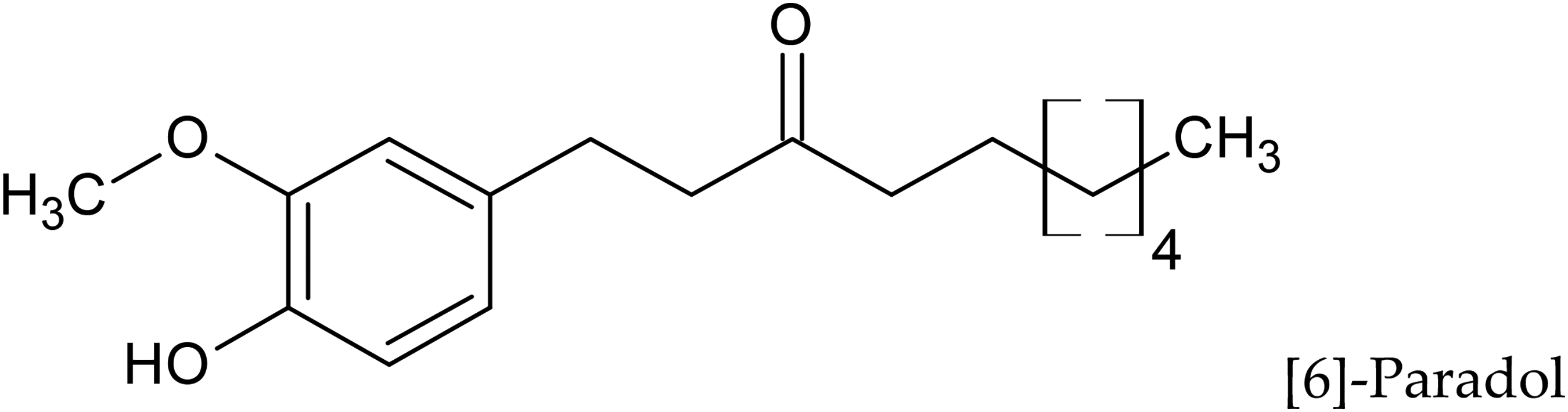
Structure of [6]-Paradol.
1-Dehydrogingerdiones
This class includes beta-ketone dehydrogenation products of gingerols. We identified 1-dehydro-[6]-gingerdione (Compound

Structure of 1-dehydro-{[6]-/[8]-/[10]-/[12]-}Gingerdiones.
Gingerdiols
Acetoxy gingerdiol (Compound 35) and methyl acetoxy gingerdiol (Compound 40) were identified based on their characteristic fragment ions, specifically (M + H-H2O-CH3COOH)+, which served as a distinctive marker as a base peak. Additionally, their RT were shorter as compared to corresponding gingerol (39, 44), supporting the presence of hydroxyl (-OH) groups in these compounds instead of keto group in 39, 44.
Diacetoxy gingerdiol exhibited a notable characteristic by generating ammonium and sodium adducts while lacking the (M + H)+ adduct. The fragmentation pattern further revealed the formation of (M + H-CH3COOH)+ and (M + H-2CH3COOH)+ ions. Diacetoxy -[4], -[6], -[8], and –[10] gingerdiols (Compound

Structure of Diacetoxy-{[4]-,/[6]-,/[8]-,[10]-}-Gingerdiols.

Structure of Methyl Diacetoxy-{[4]-,/[6]-,/[8]-}-Gingerdiols.
Diarylheptanoids
Compounds 1 and 2 show identical fragmentation pattern in both ESI + and ESI- modes with generation of m/z 391.2 (M + H)+ and 389.1 (M-H)-. Characteristic base peak m/z 373.2 was due to generation of (M + H-H2O)+, indicating -OH group on heptane base. Fragment m/z 153.1 corresponding to a fragment of [CH2C6H2(OH)2(OMe)]+ indicated dihydroxy methoxy phenyl group was present in both compounds.
Both compound
It's noteworthy to mention that 5-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)-3-heptanone possesses a chiral carbon atom at C-5 and is consequently present as an enantiomer (5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)-7-(3,4-dihydroxy-5-methoxyphenyl)-heptan-3-one). Previous data publications indicate their inseparability on HPLC, likely due to the requirement of a chiral column for the separation of these class of compounds. 21
The occurrence of two m/z 390 peaks was also observed by another research group led by Xiang Li. Notably, Xiang Li's group initially misidentified compound
Compound
Compound
Compound
Compound
Compounds
Compound
Compound
The fragmentation pattern unveiled the production of characteristic fragments with m/z 179 representing [CH2 = CH-CH+C6H2(OH)2(OCH3)]+, m/z 163 for [CH2 = CH-CH+C6H2(OH)(OCH3)]+ and m/z 149 for [CH2 = CH-CH+C6H2(OH)2]+. Based on characteristic fragments the subgroups were assigned on the alkyl chain and confirmed with previously reported compounds from Ginger. Figure 13 represents the structure of identified diaryl heptanoids based on compound numbering as in Table 5.

Diaryl Heptanoid Structures are Numbered as in Table 5.
1,7-bis-(4-Hydroxy-3-methoxyphenyl)-4-hepten-3-one {Gingerenone A} (compound
In contrast to previous study 15 that predominantly emphasized major gingerols and shogaols as principal bioactive constituents of Zingiber officinale, the present investigation broadens the analytical scope by identifying a structurally diverse set of acetylated, hydroxylated, and methoxylated derivatives of ginger phytochemicals. Xu Feng et al (2013) 26 employed LC-MS/MS to elucidate key bioactives in ginger, underscoring the chemical complexity of its phenolic profile. While their work provided an extensive metabolite overview, our study advances the field by systematically comparing fresh and dried ginger extracts across various solvents.
Some prior investigations have explored the phytochemical landscape of Z. officinale using chromatographic and spectroscopic methods. For example, Schwertner (2007) 27 applied HPLC for quantification of gingerols and shogaols in commercial products, reporting substantial variation attributed to processing conditions. Similarly, Semwal et al (2015) 28 emphasized the impact of thermal drying on the bioactive profile, particularly the conversion of gingerols to shogaols. However, these studies were either limited by narrow analyte focus or lacked comprehensive compound profiling. In contrast, our untargeted LC-MS-based chemical fingerprinting approach enables comprehensive chemical fingerprinting of both fresh and dried ginger. This detailed methodology not only facilitates in-depth chemical characterization but also establishes a robust analytical platform for authentication, quality control, and therapeutic differentiation of ginger preparations.
The comprehensive fingerprinting data generated in this study provide a robust analytical framework for standardizing dried ginger extracts used in dietary supplements. By identifying a diverse range of bioactive compounds beyond the commonly studied [6]-gingerol and [6]-shogaol, this research emphasizes the need for multi-compound standardization rather than single-marker quality control approaches.
To the best of our knowledge, this study is the first to report the comparative docking interactions of diaryl heptanoids, diacetoxy gingerdiols, paradols, and gingerdiones with serotonin 5-HT3 and TRPV1 receptors. The identification of 3,5-diacetoxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)-heptane (30) as the most effective component towards ginger's anti-emetic and pain modulation activity adds a new dimension to understanding ginger's pharmacological effects.
In a previous study 29 the mode of action of gingerols and shogaols on 5HT3 receptors was reported, suggesting their dominant role in ginger's anti-emetic activity. Our study adds several other compounds to the list of anti-emetic compounds from ginger.
In terms of pain modulation, prior research 30 has linked gingerols, zingerones, and shogaols to TRPV1 receptor activation. Our findings extend this understanding by demonstrating that diaryl heptanoids exhibit the highest binding affinity (−8.1 kcal/mol) to TRPV1, surpassing that of capsaicin (−6.5 kcal/mol). This suggests that methanolic OGE may be more effective than capsaicin in modulating TRPV1-mediated pain responses, making them valuable candidates for natural analgesic formulations. The variation in phytochemical properties observed across different extraction methods, aligns with previous studies demonstrating solvent polarity significantly influences gingerol and shogaols content. 31
While this study presents detailed LC-MS characterization and computational analysis of receptor interactions, one of the study's limitations is that it further needs validation through in vitro/ in vivo studies, to confirm the biological significance of these findings. We also restricted the molecular docking step to the compounds with favorable ADME, future studies can explore other minor compounds present in the Ginger extracts.
As a key finding of this study, the development of standardized ginger extracts, enriched in main bioactives such as diaryl heptanoids could pave the way for novel nutraceutical formulations targeting nausea, and pain. We also noticed that Dried ginger exhibited enriched content of shogaols and diarylheptanoids, which are formed or enhanced during the drying process. These compounds contribute significantly to its superior antioxidant activity and therapeutic potential. Investigating the synergistic effects of these compounds in clinical settings will be crucial in translating these findings into real-world therapeutic applications.
Conclusion
This study provides a comprehensive comparative analysis of fresh and oven-dried ginger (Zingiber officinale) extracts through LC-MS chemical fingerprinting, antioxidant assays, and in silico docking evaluations. The findings reveal that oven-drying significantly enhances the yield and concentration of key bioactives, particularly shogaols and diarylheptanoids, with methanol proving to be the most effective solvent for extraction. Antioxidant activity and total phenolic content were markedly higher in dried ginger extracts, indicating a favorable impact of drying on the bioactive profile.
A total of 61 bioactive compounds were identified from methanolic extracts, including structurally diverse classes such as gingerols, shogaols, paradols, gingerdiols, and notably, diarylheptanoids. We identified and corrected a few previously misidentified compounds.
Several diarylheptanoids exhibited strong binding affinities to 5HT3 and TRPV1 receptors, surpassing previously studied compounds like [6]-gingerol and [6]-shogaol. Our in silico findings suggest that ginger's anti-emetic and analgesic potential may be attributed not only to gingerols and shogaols but also to a broader array of secondary metabolites that have thus far been underexplored.
The study demonstrates how chemical fingerprinting can capture subtle variations in compound profiles influenced by processing method and solvent systems, enabling a more precise assessment of pharmacologically relevant constituents. This study reinforces the importance of comprehensive standardization strategies for dietary supplements, ensuring consistency in composition and therapeutic benefits. Further studies including clinical trials are warranted to explore the mechanism of anti-emetic and pain modulating activity of ginger bioactives.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251375515 - Supplemental material for Comparative LC-MS Chemical Fingerprinting, Antioxidant Potential, and in Silico Analysis of Anti-Emetic and Pain Modulation Activity of dry and Fresh White Ginger Extracts
Supplemental material, sj-docx-1-npx-10.1177_1934578X251375515 for Comparative LC-MS Chemical Fingerprinting, Antioxidant Potential, and in Silico Analysis of Anti-Emetic and Pain Modulation Activity of dry and Fresh White Ginger Extracts by Alpana Pande, Anju Majeed, Shaheen Majeed, Anurag Pande, Kalyanam Nagabhushanam, Muhammed Majeed, Muthusamy Sengodagounder and Senthil Kumar Thiruppathi in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251375515 - Supplemental material for Comparative LC-MS Chemical Fingerprinting, Antioxidant Potential, and in Silico Analysis of Anti-Emetic and Pain Modulation Activity of dry and Fresh White Ginger Extracts
Supplemental material, sj-docx-2-npx-10.1177_1934578X251375515 for Comparative LC-MS Chemical Fingerprinting, Antioxidant Potential, and in Silico Analysis of Anti-Emetic and Pain Modulation Activity of dry and Fresh White Ginger Extracts by Alpana Pande, Anju Majeed, Shaheen Majeed, Anurag Pande, Kalyanam Nagabhushanam, Muhammed Majeed, Muthusamy Sengodagounder and Senthil Kumar Thiruppathi in Natural Product Communications
Footnotes
Acknowledgements
The author gratefully acknowledges the guidance and support provided by Dr Muhammed Majeed and Dr Senthil Kumar Thiruppathi, whose insights and expertise greatly contributed to the development of this work.
Ethical approval statement
No human or animal subjects were involved in this study.
Informed Consent Statement
No human or animal subjects were involved in this study.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors Alpana Pande, Anurag Pande, Shaheen Majeed, and Anju Majeed are employed by Sabinsa Corporation. Kalyanam Nagabhushanam and Muhammed Majeed were employed at Sabinsa Corporation until their demise in 2024 (their contribution has already been mentioned in the submission for the preparation of this article). All the authors declare no conflict of interest, and research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Institutional Review Board Statement
No human or animal subjects were involved in this study.
Statement of Human and Animal Rights
No human or animal subjects were involved in this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.