Abstract
Background
Ginsenoside Rg1 (GRg1) is a natural compound derived from Panax ginseng, widely used in alternative medicine.
Methods
Olive oil, dimethyl sulfoxide, Tween-80, and distilled water were used to prepare blank SNEDDS. Then, 500 mg of GRg1 was added to formulate loaded SNEDDS, both of which were smoothly stirred for 72 h. Both preparations were characterized using dynamic light scattering (DLS), transmission electron microscopy (TEM), Fourier-transform infrared (FTIR), x-ray diffraction (XRD), and UV-Vis spectroscopy. Sixteen rats were divided into four equal groups: a control negative (CN) group that received distilled water by gavage; the others received oral GRg1-SNEDDS at a low dose (LD = 1.0 mg/kg), medium dose (MD = 5.0 mg/kg), or high dose (HD = 10 mg/kg). The growth and behaviour of the rats were checked, and 6.0 mL of blood was collected for biochemical and haematological analysis. Additionally, histopathological examinations were performed on liver and kidney sections.
Results
The characterization revealed that the GRg1 successfully loaded into its carrier, and the mean particle size (PS), polydispersity index (PDI), and zeta potential(ZP) of GRg1-SNEDDS were within accepted ranges (165.65 ± 45.58 nm, 0.27 and −20.8 ± 0.7 mV, respectively). Biochemical, haematological and histopathological studies revealed that the GRg1-SNEDDS was safe to use. ALT was significantly higher only in the HD group; AST was higher in both the LD and HD groups (all changes were within normal reference ranges). The granulocyte count was higher in the LD compared to CN. No signs of toxicity, such as weight loss, fever, infection, or bleeding, were detected.
Conclusions
Characterizations of the prepared GRg1-SNEDDS were within the accepted ranges. The toxicity profile of GRg1 SNEDDS was relatively safe in low and medium doses; the HD was associated with a marginal increase in ALT but more histopathological toxicity. No toxic effects were observed on the studied rats’ weight, physical wellbeing or behaviour.
Introduction
Ginseng was recognized over 5000 years ago as one of the most highly regarded herbs in ancient China, Korea, Japan, and the United States of America. 1 Ginsenoside Rg1 (GRg1) is one of the most widely studied bioactive compounds in ginseng, due to its broad range of therapeutic targets and minimal side effects. 2 Additionally, ginsenoside Rg3 (GRg3) has been shown to have anti-inflammatory and anti-tumor properties. 3
Recent studies have emphasized the ability of GRg1 to maintain glucose homeostasis and enhance insulin sensitivity. It has been shown to improve memory dysfunction and reduce synaptic loss in animal models. 2 GRg1 has been found to help alleviate congestive heart failure by inhibiting the phosphorylation of extracellular signal-regulated kinase 1/2 (ERK1/2) proteins. This, in turn, reduces apoptosis, promotes cell activity and proliferation, and decreases cardiac inflammation in rat models. 4 The combination of GRg1 has shown an anti-metastatic effect by inhibiting both the early and late stages of the neutrophil-initiated metastasis cascade in an experimental lung metastatic model. 5
Approximately one-third of newly identified drug compounds exhibit poor water solubility, resulting in low bioavailability when administered orally. Self-nanoemulsifying drug delivery systems (SNEDDS) have been investigated as an effective method to enhance the bioavailability and dissolution rate of drugs with limited water solubility.6,7 The application of SNEDDS in product development is expanding due to the flexibility of pharmaceutical lipid excipients and drug formulations, as well as their ability to work with liquid, semi-solid, and solid dosage forms. SNEDDS consist of a drug, oil phase, surfactant, and co-surfactant. 6
Additionally, these components can be easily adjusted to make SNEDDS suitable for hydrophilic and hydrophobic drugs. SNEDDS can be an efficient oral drug carrier for peptides and proteins by protecting them from gastrointestinal degradation and enhancing their permeability through the intestinal membrane.8,9
The water solubility of GRg1 is 0.304 mg/mL (poor), and the development of GRg1-loaded SNEDDS might improve the water solubility of GRg1 and thus improve its oral bioavailability. 10 Therefore, the primary goal of this study was to synthesize and characterize GRg1-loaded SNEDDS and evaluate the toxicity profile of low, medium, and high doses of the products in an in vivo study using healthy male Sprague-Dawley rats. Then, in the future, this formula can be used to treat various induced diseases in vivo, including diabetes, hypertension, and hypercholesterolemia.
Materials and Methods
Study Design and Setting
This experimental study was conducted at the College of Medicine's Animal House, University of Sulaimani, Sulaimaniyah, Iraq, from May to December 2024.
Self-Nanoemulsifying Drug Delivery System (SNEDDS)
Formulation of Blank SNEDDS
For blank SNEDDS preparation, 2.0 mL of olive oil was mixed with 2.0 mL of dimethyl sulfoxide [DMSO] as a co-surfactant and stirred for 2 h on a magnetic stirrer (at 200 rpm and 37° C). Then, 5.0 mL of Tween-80 [polyoxyethylene (POE) derivatives of sorbitan fatty acid ester] was added as a surfactant, homogenized by stirring for an additional hour. Using distilled water, the volume was completed to 100 mL. Then, the mixture was stirred continuously for 72 h at 37 °C, followed by ultrasonication for 2 h. Finally, the centrifuged/filtered nanoemulsions were stored at room temperature for characterization and toxicological analysis.11,12
Formulation GRg1-SNEDDS
When the blank-SNEDDS was successfully prepared, 500 mg of 98% purified GRg1 powder was added to it. The resulting GRg1-SNEDDS was gently stirred for 72 h at 37 °C, then ultrasonicated for two hours. The SNEDDS nanoemulsions were then centrifuged, filtered, and stored at room temperature for characterization and subsequent use (Figure 1).

The Formulation of Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
Characterization of GR1-SNEDDS
The particle size (PS), polydispersity index (PDI), and zeta potential (ZP) were measured using a particle analyzer (Litesizer 500, Anton Paar GmbH, Austria). At 25 °C, the nanoemulsions were diluted with Milli-Q water (1:10). Triplicate assessments were implemented using dynamic light scattering (DLS) measurement. Additionally, transmission electron microscopy (TEM) was used to examine the morphological characteristics of GRg1-SNEDDS by placing the nanoemulsions onto a copper grid (mesh size 300), covered with carbon, stained with phosphotungstic acid (1%, w/v), then inspected using a TEM device (Hitachi H-7100, Japan) after air drying. The chemical composition of SNEDDS and GRg1-SNEDDS was analyzed using Fourier-transform infrared (FTIR) spectrometry (Cary 640 Agilent, USA) in the 400-4000 cm−1 range. Their quantities were measured using a Shimadzu UV-160 UV-Vis spectrophotometer (USA) at wavelengths ranging from 200 to 600 nm. Additionally, the phase purity and crystallinity of the nanoemulsions were examined using x-ray diffraction (XRD) with a Panalytical X'Pert Pro diffractometer (Germany), operating at 40 kV, employing a nickel-filtered copper source (λ = 1.542 Å) in the range of 10–60°.
In Vivo Toxicity Study
Male Sprague Dawley rats (n = 16), aged 6-8 weeks and weighing 175-210 g, were acclimatized to regular tap water and standard rat chow at room temperature (25 ± 3° C) for 8 days. The rats were kept in an area with a 12-h day/night light accommodation cycle. Rats were divided into equal groups (n = four each), including Control Negative (CN) rats that were gavaged with tap water, low dose of GRg1-SNEDDS (1.0 mg/kg), GRg1-SNEDDS medium dose (5.0 mg/kg), and GRg1-SNEDDS high dose (10 mg/kg). According to Xie et al, the minimal studied doses of GRg1 in experimental animals were chosen, because of the tendency of SNEDDS to augment the efficacy and even the toxicity of medications. 13 For four weeks, the rats were treated via force-feeding, stainless steel, ball-tipped needles.
Body Weight Measurements and Clinical Observations
Body weights were recorded before treatment (day 0) and on days 7, 14, 21, and 28. Based on the rats’ body weight changes, the drug dose was adjusted weekly. Over the 28-day treatment period, the rats were checked daily for toxicological symptoms, clinical abnormalities, food intake, and general appearance.
Biochemical Parameters
After completion of the experiment (day 29), all rats were euthanized under deep anaesthesia using a high dose of a mixture of xylazine and ketamine. Then, 6 mL of blood was collected from each rat via cardiac puncture. Two over three of the blood (4.0 mL) was centrifuged at 3500 rpm for 10 min and used for the estimations of the liver function test (total protein, albumin, alanine aminotransferase [ALT], aspartate aminotransferase [AST], and alkaline phosphatase [ALP]), renal function test (urea and creatinine) and electrolytes (sodium, potassium, chloride, calcium, and inorganic phosphate) using Roche diagnostic kits (Cobas 311 system, Germany).
Hematological Parameters
Total white blood cells (WBC), red blood cells (RBC), haemoglobin (Hb), hematocrit (HCT), platelets (PLT), and differential WBC count (lymphocyte, monocyte, and granulocyte) were measured using the 2 mL blood that remained after being collected into EDTA vacuumed tubes after direct mixing. The blood was then analyzed using an automatic haematology analyzer (Cell Dyn 3700, Abbott, USA).
Histopathological Study
After animal euthanasia, the liver and kidneys of each rat were used for histopathological study. Briefly, the tissues were rinsed in normal saline, placed into plastic tissue cassettes and fixed with 10% neutral buffered formaldehyde solution for at least 72 h. Afterwards, the dehydration begins with passing the samples through a series of ascending ethanol alcohol (50%, 60%, 70%, 90%, and 100%), followed by three steps of xylene clearance. Later, the processed tissues were impregnated and embedded in melted paraffin blocks using an automated wax embedder machine at 60-70ᵒ °C. Then, after sectioning, we started using a semi-automated rotary microtome to cut the paraffinized tissue samples into 5.0 µm-thick sections, which were mounted on glass slides and dried using a hot plate tissue holder. Afterwards, glass slides with mounted tissue sections were deparaffinized and cleaned with a xylene solution for at least 30 min, then dried in a hot oven at 50 °C for 10 min. Finally, tissue sections were stained with Harris's hematoxylin and eosin solution, cleaned with xylene, and covered with a coverslip, then viewed, examined, and analyzed under a bright-field light microscope.
Semi-Quantitative Lesion Scoring Assay
Kidney sections were analyzed for vacuolar congestion within the tubular epithelial cells, glomerular atrophy, and vascular degeneration. In contrast, epithelial cellular degeneration and glomerular atrophy were counted in four randomly chosen fields under high-power magnification (100×), and the mean average was then calculated as a percentage. On the other hand, hepatocyte cellular degeneration and Kupffer cell hyperplasia were estimated and measured as a percentage of calculated cell numbers from randomly selected four fields in the tissue section. In contrast, vascular congestion was assessed in µm and evaluated as a mean percentage. The mean percentage of all calculated values was expressed using a lesion scoring and grading system. Score 0%–10% indicated as no lesions; score 10%–25% as mild; score 25%–50% as moderate; score 50%–75% as severe; and score 75%–100% as critical lesions. Lesion scoring was estimated semi-quantitatively via image analyzer software (AmScope, 3.7) using a microscope eye-piece camera (MD500, 2019), and tissue samples were analyzed under the light microscope (NOVEL XSZ-N107 T, China).
Statistical Analysis
The Statistical Package for the Social Sciences (SPSS, IBM, New York, USA, version 26.0) was used to analyse the data. Shapiro assessed the normally distributed data using the Wilk test and histogram. Independent samples T-test and ANOVA were employed to evaluate the relationship between normally distributed quantitative variables. For non-normally distributed quantitative variables, the Mann–Whitney U test was performed. A p-value of ≤0.05 was set as significant.
Results
Preparation of SNEDDS and GRg1-SNEDDS
The formulated blank-SNEDDS appeared as a slightly opaque homogeneous solution. Upon loading with GRg1, the emulsion appeared deeper, opaque, and hazy due to the nature of the white powder. No oil separation or sedimentation indicated that the oils were well-homogenized (Figure 2).

Formulated Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
Characterization of Blank and Loaded-SNEDDS
Dynamic Light Scattering Analysis
The PS, PDI and ZP of loaded and blank-SNEDDS were 165.65 ± 45.58 versus 32.03 ± 55.88 nm, 0.27 versus 0.25, and −20.8 ± 0.7 versus −10.7 ± 0.6 mV, respectively (Figure 3).

Showed Particle Size Distribution Curve, and Zeta Potential Curve of Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
Transmission Electron Microscopy (TEM) of GRg1-SNEDDS
The TEM revealed that GRg1-SNEDDS particles were nearly regular and seemingly spherical, with different diameters and no aggregation between particles (Figure 4).

Transmission Electron Microscopic Image (TEM) of Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
Fourier-Transform Infrared (FTIR) Spectroscopy Patterns of Blank and GRg1-SNEDDS
The FTIR spectrum showed peaks at 3438 that appeared broad with a rounded tip, indicating the presence of the hydroxyl (OH) group. In the region of 2800-3000, a peak of CH stretching (Alkyl) was seen, and CH bond vibrations were found at 1353, 633, and 648 (Figure 5).

Fourier-Transform Infrared Spectroscopy Patterns of Blank and Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
UV–Vis Spectroscopy Patterns of Blank and GRg1-SNEDDS
The UV–Vis spectroscopy showed a sharp peak at 260 nm, recorded between 200 and 600 nm. The absorbance intensity of loaded-SNEDDS was higher than that of the blank (Figure 6).

Ultraviolet Visible (UV-Vis) Spectroscopy Patterns of Blank and Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
X-RAD Pattern of Blank and GRg1-SNEDDS
There were identical X-RAD spectra of the broad peak powder between 15 - 30° and the broad diffraction peaks at 20° for blank SNEDDS and GRg1-SNEDDS. Loaded nanoemulsions had more diffuse x-ray scattering than blank-SNEDDS (Figure 7).

X-ray Powder Diffraction of Blank and Gensinoside-Self-Nano Emulsifying Drug Delivery System (GRg1-SNEDDS).
Toxicity Study
Daily oral administration of GRg1-SNEDDS at doses of 1.0, 5.0, and 10 mg/kg for four consecutive weeks did not affect the overall performance of the treated animals compared to the controls. The rats showed no signs of toxicity, such as segregation, lethargy, ruffled fur, cannibalism, loss of appetite, or abnormal water intake. No deaths occurred in either group, and significant (p ≤ 0.05) weight gain was observed in the CN group and all three doses of loaded nanoparticles throughout the study period compared to day zero (Table 1).
Estimation of Rat’s Body Weight Treated with Different Doses of GRg1-SNEDDS for Four Weeks.

Data have been analyzed using Paired T-test. significant difference indicates as letter “
The biochemical parameters did not show significant toxic alterations between the treated groups compared to the control negative (CN) group, apart from minor changes in some parameters. Compared to the CN group, AST was higher in low-dose (LD) (146.30 ± 5.31 U/l) and high-dose (HD) groups (146.50 ± 3.52 U/l); ALP was lower in all treated groups, and the albumin was slightly lower in the LD group. The most notable was the ALT, which was significantly (p ≤ 0.05) higher only in the HD rat group. None of the TP, blood urea or serum creatinine demonstrated significant differences between the groups. Neither the AST nor the ALT was elevated in the medium dose (MD) GRg1-loaded SNEDDS group. All the differences from the CN group fell within the normal range for rats according to different normative values (Table 2 and Appendix 1).
Mean of Biochemical Tests of Control and Treated Rats with Various Doses of GRg1-SNEDDS.

Independent samples T-test have been used to analyze all biochemical parameters except ALP that analyzed by Mann-Whitney U-test. Letter “
The serum electrolytes (sodium, potassium, chloride and calcium) did not alter significantly between the groups. Only the phosphate was elevated significantly (p ≤ 0.05) in the MD and HD groups compared to CN. The elevation did not follow the dose augmentation (Table 3). Regarding hematology, the granulocyte count was higher in the LD compared to the CN group. Minor variations within the normal range in several other parameters, including lower WBC, lymphocyte, and platelet counts, were observed in the HD group (Table 4 and Appendix 1).
Mean Blood Electrolytes of Control and Treated Rats with Various Doses of GRg1-SNEDDS.

Independent samples T-test have been used to analyze all blood electrolytes except Potassium that analyzed by Mann-Whitney U-test. Letter “
Mean Hematological Parameters of Control and Rats Treated with Various Doses of GRg1-SNEDDS.
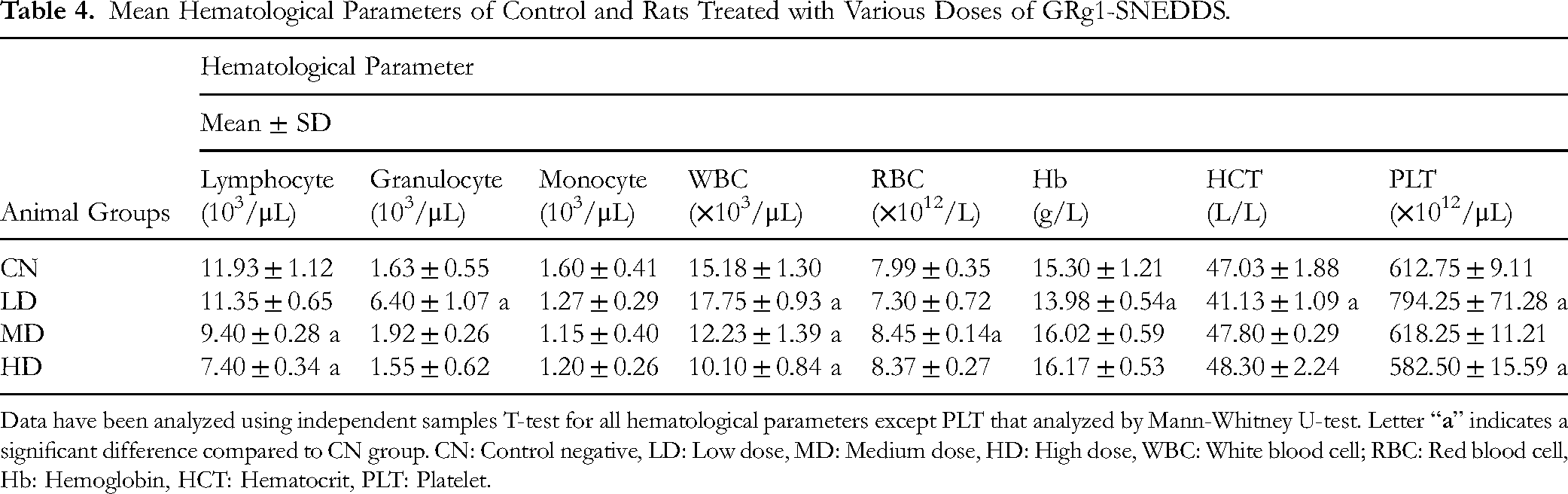
Data have been analyzed using independent samples T-test for all hematological parameters except PLT that analyzed by Mann-Whitney U-test. Letter “
Histopathological Analysis
Kidney
The semi-quantitative morphometric analysis and lesion scoring assay of kidney sections were illustrated in Table (5), which revealed a significant (p < 0.05) increment in the percentage of cellular swelling within the renal tubular epithelia with an overall lesion severity in terms of glomerular atrophy and vascular congestion from mild to moderate then to severe grade in a dose depended manner compared to CN group which received tap water. These findings indicate that animals receiving 10 mg/kg GRg1-SNEDDS exhibit severe and diffuse tubular epithelial vacuolar degeneration and glomerular damage. Likewise, animals in the MD group fed 5.0 mg/kg GRg1-SNEDDS show lower lesion severity scores, classified as a moderate lesion. On the other hand, animals in the LD group with only 1.0 mg/kg GRg1-SNEDDS showed a low grade of lesion severity expressed as a mild score compared to the other two regimen doses. In contrast, the CN group represents normal kidney tissue morphology without significant structural modifications. Thus, animals receiving 10 mg/kg GRg1-SNEDDS display a significant toxic influence on the renal micro-morphological architecture (Figure 8).

Photomicrograph of Kidney from Groups;
Semi-Quantitative Evaluation of Kidney Sections.
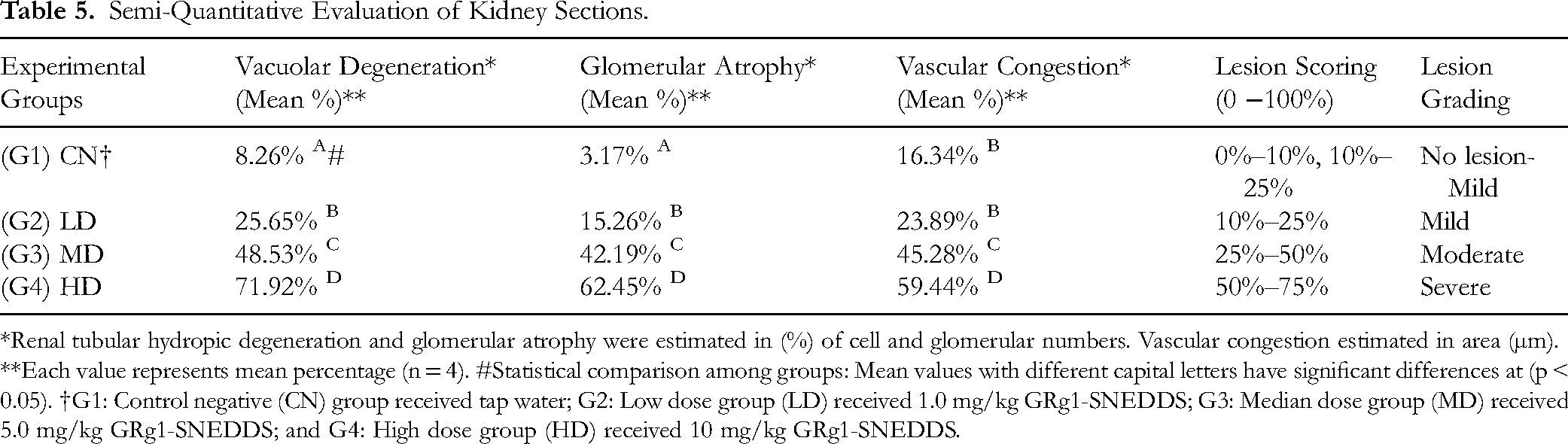
*Renal tubular hydropic degeneration and glomerular atrophy were estimated in (%) of cell and glomerular numbers. Vascular congestion estimated in area (µm). **Each value represents mean percentage (n = 4). #Statistical comparison among groups: Mean values with different capital letters have significant differences at (p < 0.05). †G1: Control negative (CN) group received tap water; G2: Low dose group (LD) received 1.0 mg/kg GRg1-SNEDDS; G3: Median dose group (MD) received 5.0 mg/kg GRg1-SNEDDS; and G4: High dose group (HD) received 10 mg/kg GRg1-SNEDDS.
Liver
Liver lesion severity score has been illustrated in Table (6), which reveals that animals in the HD group (10 mg/kg GRg1-SNEDDS) present significant augmentation in their lesion severity score, evident with severe cellular degeneration, vascular congestion and noticeable proliferation of the resident hepatic phagocytic cells compared to MD and LD prescriptions (5.0 and 1.0 mg/kg, respectively). Microscopical examination of the liver section offered a distinctive histological architecture of a typical ordinary liver in the CN group, which sustained a normal morphological organization of hepatocytes around the central vein. On the other hand, liver sections in LD and MD show mild to moderate lesion scoring compared to the HD group, as evidenced by their lower scores for hepatocellular degeneration, cellular proliferation, and vascular engorgement (Figure 9).

Photomicrograph of Liver from Groups;
Semi Quantitative Evaluation of Liver Sections.

*Hepatocytes hydropic degeneration and Kupffer cell hyperplasia were estimated in (%) of cell numbers per calculated fields. Vascular congestion estimated in area µm. **Each value represents mean percentage (n = 4). #Statistical comparison among groups. Mean values with different capital letters have significant differences at p < 0.05. †G1: Control negative (CN) group received tap water; G2: Low dose group (LD) received 1.0 mg/kg GRg1-SNEDDS; G3: Median dose group (MD) received 5.0 mg/kg GRg1-SNEDDS; G4: High dose group (HD) received 10 mg/kg GRg1-SNEDDS.
Discussion
Ginseng is an important herb described as a remedy for almost every illness 2 ; therefore, we conducted this study on GRg1, the most active ginsenoside, in nanoparticle form. SNEDDS consist of a mixture of oils, surfactants, and co-surfactants used as a drug delivery system to load GRg1 to increase bioavailability, improve efficacy, reduce total dose and minimize side effects. Khattab et al confirm that using a self-nanoemulsifying system as a carrier for GRg1 can bypass first-pass metabolism, enhance its presence in the blood stream, and result in a higher concentration reaching the brain compared to traditional extracts. 10 Regarding the characterization of GRg1-SNEDDS, various techniques, including PS, PDI, ZP, TEM, FTIR, and UV-Visible spectroscopy, as well as X-RAD, were used to measure the stability, purity, and structure of the nanoparticles before use. We used DLS analysis to study the size, uniformity and surface charge of the nanoparticles. The PS for GRg1-SNEDDS and blank-SNEDDS were 165.65 ± 45.58 and 32.03 ± 55.88 nm, respectively. Accordingly, both results were within the accepted nanoparticle range. 12 GRg1-SNEDDS had a larger size than blank, indicating a successful drug-loading process. Nanoparticles typically range in size from 40 to 400 nm, which is considered optimal for ensuring prolonged circulation times and enhanced accumulation at target sites with minimal renal clearance. Additionally, the shape and morphology of nanoparticles are considered crucial factors in passive targeting.
On the other hand, the PDI values were 0.27 and 0.25 for the loaded and blank SNEDDS, respectively, indicating that the samples consisted of monodisperse (homogeneous) particles. The droplet size stability further confirms the absence of minimum aggregation or agglomeration among the particles. PDI is a dimensionless value that indicates the variety of particle sizes in a sample, ranging from 0.1 to 0.7. 14 However, ZP of loaded and blank samples reported −20.8 ± 0.7 and −10.7 ± 0.6 mV, respectively, indicating a moderate degree of electrostatic repulsion between particles. This can contribute to the stability of the colloidal system because particles with similar charges tend to repel each other and remain dispersed in the medium. 15 The morphological study of nanoparticles was carried out by using TEM, 16 where GRg1-SNEDDS showed uniform nano-sized morphology, nearly regular, round to spherical, homogenous with no visible aggregations and PS less than what was detected by DLS.
FTIR is a valuable tool for characterizing organic molecules. As our samples were a mixture of different substances, a broad peak appeared around 3438 cm-1, indicating the presence of the OH stretching region due to interactions (such as hydrogen bonding) between the drug and the excipients. In the range of 2800–3000, a peak of CH stretching was observed due to an overlap between GRg1 and SNEDDS, while the alkyne chain appears at around 2100. Due to the effect of conjugation, the carbonyl group of carboxylic acid was seen at 1636 (instead of 1690-1700) in our sample. CH bond vibrations were observed at approximately 1353, 633, and 648 cm-1. The main objective of conducting comparative FTIR analysis between the blank and GRg1-loaded SNEDDS was to detect any possible chemical interactions between GRg1 and the formulation excipients. By analyzing the FTIR spectra, we can observe changes such as shifts, disappearance, or appearance of functional group peaks, which help assess the physical or chemical compatibility and verify the effective incorporation of GRg1 into the SNEDDS without causing degradation or unwanted interactions. 17 The UV-Vis spectrum indicates that the drug has been effectively incorporated into the SNEDDS formulation. This is evidenced by the higher absorbance observed at the drug's characteristic wavelength (approximately at 260 nm) in the drug-loaded sample compared to the blank formulation. UV–visible spectroscopy is the most commonly employed technique for analyzing the optical properties of nanoparticles. The optical characteristics of the GRg1 nanocomposite samples were examined using a UV–visible absorption spectrum at 200-600 nm. Within this range, electrons absorb light in the UV or visible regions, resulting in electronic transitions. An absorption edge peak was observed at 260 nm for GRg1-SNEDDS. The sharp peak observed in the nanoparticle combinations indicates monodispersion and minimal aggregation within the nanoparticle distribution.18,19
X-Ray Diffraction (XRD) analysis is employed to determine whether the components in Liquid Self-Nanoemulsifying Drug Delivery Systems (L-SNEDDS) exist in a crystalline or amorphous form. This assessment is crucial to determine if the drug has transformed from its original crystalline structure, which is less soluble, to an amorphous form with enhanced solubility after being incorporated into the SNEDDS formulation. 20 GRg1-SNEDDS had spectra with a broad peak between 15°-30°, and the broad diffraction peaks at 20° indicate amorphous or poor crystalline structures, which is common for nanoemulsions that contain oils, surfactants, and water. Narrow and sharp peaks were seen in highly crystalline structures. 21
This study critically assesses the physical, biochemical, haematological and histopathological toxicity of GRg1-loaded SNEDDS. Assessing the physical well-being and behaviour of rats is essential when evaluating drug toxicity. Key parameters include monitoring body weight, observing smooth and clean fur, assessing normal mobility, checking for clear eyes and nose, ensuring regular respiration, and confirming there are no signs of dehydration. 22
The toxicity profile showed that LD was very safe apart from minor biochemical or haematological adverse effects (MD and HD) that did not affect the physical well-being, behavioural aspects, or weight of the rats. The positive impact of GRg1 on the growth of broilers was also reported. 23 This study documented a normal increase in body weight in all animal groups. As an index of toxicity, changes in animal weight are used in most studies. When body weight is significantly reduced, it is considered a red flag for the toxic effects of the tested products on the studied animals. 24 There are scant studies on the safety assessment of rare ginsenoside mixtures, 25 and the literature on ginsenoside as separate subtypes is limited, with even less information available on GRg1 toxicity. 26 In our comprehensive search, we were unable to identify any article that specifically studied the toxicity of GRg1-loaded SNEDDS. In a prospective 4-week toxicity study, the Korean red ginseng extract (mainly containing GRg1 and Gb1) appeared safe when administered to male rats at 500, 1,000, and 2000 mg/kg/day. 26 They did not report significant haematological, biochemical, histopathological or gross physical toxicity. They regarded 2000 mg/kg/day of Korean red ginseng extract as having a no-observed-adverse-effect level (NOAEL).
Globally, the biochemical parameters alteration between the groups was within the normal range (Appendix 1)27,28 apart from ALT that was higher in the HD group (77.43 ± 7.25 U/l) with < one-fold rise (20% above the mean of CN group (64.35 ± 1.26 U/l)), which was of no clinical significance, just it needs further follow-up on chronic therapeutic use. In harmony with our safety belief in GRg1, Wu et al (2025) reported the hepatoprotective benefits of GRg1 in treating liver diseases. 1 On the contrary, Kim (2016) observed that GRg1 may exhibit severe hepatotoxicity when administered in high doses or combination with other substances. 29 Cao et al (2023) in their study on the toxicity profile of rare ginsenosides demonstrated that rats had significantly increased levels of ALT in HD ginsenoside (600 mg/kg), accompanied by inflammatory changes in the liver. 25 This finding is worth confirming in future research and considering when RGg1-loaded SNEDDS are studied for future therapeutic uses. Our impression was that the low- to medium-dose range was safe for use. Normal values of TP and albumin also indicated a safe liver profile of GRg1-SNEDDS, as it was not toxic at any dose.
Regarding the effects of GRg1-SNEDDS on renal function, normal urea and creatinine levels were announced with all three doses. The only electrolyte affected by the MD was phosphorus (11.09 ± 1.17 mmol/L), but the effect was less evident in the HD (10.04 ± 0.77 mmol/L) compared to the LD (8.90 ± 0.47 mmol/L) and CN (8.55 ± 0.44 mmol/L) groups. This may suggest other operating factors rather than the GRg1-SNEDDS dose effect.
Regarding the haematologic parameters, although changes in the estimated values (either increased or decreased) compared to the control group were detected, only a few parameters were outside the reference normal values (Appendix1)30,31 particularly granulocytosis in the LD group. No clinically correlated toxicity features were associated with any rat's haematological abnormality, such as fever, infections, or a bleeding tendency. The increased granulocytes in the LD group may be due to inflammation, immune response, stress, or other cofounding factor
Regarding the histopathological study, we were unable to compare our results with those of other studies due to the lack of histopathological data on GRg1-SNEDDS in the literature. 31 However, the effect of coupling minor ginsenoside Rg3 with magnetic components to obtain magnetic nanoparticles showed that the synthesized product could automatically target the mouse. In contrast, subsequent evaluation of organs in mice showed that nano-drugs were non-toxic and safe. 33 Also, ginsenoside compound K (CK) at 40 and 120 mg/kg in male and female rats, respectively, showed NOAEL in a 26-week toxicity study. Similarly, acute toxicity shows that CK administered orally to rats and mice did not cause mortality or toxicity at the maximum dosage of 8.0 and 10 g/kg, respectively. 34 At the same time, ginsenoside CK at a dose of 12 mg/kg indicates an NOAEL in dogs. 35 All these outcomes suggest that LD/MD GRg1 is safe for use in animals. Thus, ginsenoside nanoparticles, prepared using a simple and green method, are promising for delivering various insoluble drugs. 36 The study's limitations include a lack of advanced laboratories to conduct physical and chemical characterizations, which increases the time and cost burden on the researchers. There is a lack of recently updated sources for the standard reference values of the studied parameters in rats, as the comparison to the CN group may not reflect real toxicity when only the absolute values are considered as determinants for the levels of change in significance. Also, use of small convenient samples taking in consideration the animal rights.
Conclusions
Characterizations of the prepared GRg1-SNEDDS were within the accepted ranges, as determined by standard assessment techniques. The toxicity profile of GRg1-SNEDDS was relatively safe in LD/MD, while HD was associated with a marginal increase in ALT and severe histopathological alterations. The MD was associated with a minimal increase in phosphorus level, which was not augmented by HD. No potential effects were observed on the rats’ body weight, physical wellbeing or behavioural aspect. We recommend using GRg1-SNEDDS with lower doses in future therapeutic research for various medical indications to augment effects and minimize side effects of GRg1.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251374757 - Supplemental material for Ginsenoside Rg1 Loaded Self-Nanoemulsifying Drug Delivery System (SNEDDS): Preparation, Characterization and In-vivo Toxicity Study
Supplemental material, sj-docx-1-npx-10.1177_1934578X251374757 for Ginsenoside Rg1 Loaded Self-Nanoemulsifying Drug Delivery System (SNEDDS): Preparation, Characterization and In-vivo Toxicity Study by Nazaneen Akbar Omer and Saman Hussein Noori in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251374757 - Supplemental material for Ginsenoside Rg1 Loaded Self-Nanoemulsifying Drug Delivery System (SNEDDS): Preparation, Characterization and In-vivo Toxicity Study
Supplemental material, sj-docx-2-npx-10.1177_1934578X251374757 for Ginsenoside Rg1 Loaded Self-Nanoemulsifying Drug Delivery System (SNEDDS): Preparation, Characterization and In-vivo Toxicity Study by Nazaneen Akbar Omer and Saman Hussein Noori in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251374757 - Supplemental material for Ginsenoside Rg1 Loaded Self-Nanoemulsifying Drug Delivery System (SNEDDS): Preparation, Characterization and In-vivo Toxicity Study
Supplemental material, sj-docx-3-npx-10.1177_1934578X251374757 for Ginsenoside Rg1 Loaded Self-Nanoemulsifying Drug Delivery System (SNEDDS): Preparation, Characterization and In-vivo Toxicity Study by Nazaneen Akbar Omer and Saman Hussein Noori in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank the College of Medicine and the University of Sulaimani authorities for their support and assistance with this study.
Ethical Approval
The experimental protocol was approved by the scientific and ethical committees of the College of Medicine, University of Sulaimani, Iraq (No. 73 on 5/5/2024-CoM-UoS) following a thorough review (Appendix 2). The ARRIVE Guidelines for Animal Care and Use conducted the study, ensuring that procedures were implemented to minimize animal pain and discomfort (Appendix 3).37,38
Informed Consent
Not applicable.
Authors’ Contributions
NAO: Methodology, data collection, data analysis, prepared tables and figures, writing the original manuscript; SHN: Conceptualization, supervision, study registration, writing the original manuscript. Both authors agreed to submit the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets used and/or analyzed during the current study are all provided in the manuscript and supplementary file.
Human and Animal Rights
Measures were taken to minimize pain and discomfort, and experiments followed the ARRIVE Guidelines for animal ethics guidelines and protect animal rights.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
