Abstract
Background and objectives
Various parts of Macaranga capensis are widely used in traditional folk medicine due to their health-promoting effects. This study aims to investigate the phytochemical constituents in the stem bark of M. capensis.
Methodology
The stem bark of M. capensis was extracted using a methanol/dichloromethane (1:1) mixture, and the crude extract was subjected to chromatographic techniques to isolate the phytochemical constituents.
Results
Chemical investigation of the stem bark of M. capenesis led to isolation of eight previously reported metabolites: chrysoeriol (
Conclusion
The isolation of these compounds contributes to a deeper understanding of the chemical diversity within the Macaranga genus and underscores its potential therapeutic applications.
Introduction
Macaranga capensis (Baill.) Benth. ex Sim (Euphorbiaceae) is a deciduous tree characterized by pale gray bark, spirally arranged, broadly ovate leaves, and short thorns on its young stems. It bears densely packed, yellowish-green, dehiscent fruits and purplish brown to blackish seeds. M. capensis, an inhabitant of evergreen forests (ranging from 305-2133 m), is considered an indicator of forest invasion. It is found along lake and stream banks of East Africa (Kenya and Ethiopia) to South Africa and can grow up to 30 meters in height.1–3
Traditionally, M. capensis stem bark has been used in KwaZulu-Natal, South Africa, to treat various skin conditions, such as sunburn.1,4 The bark decoction is used to manage diarrhea in local communities in Kenya 5 and South Africa. 6 The Washambaa people of Tanzania use M. capensis leaves to manage allergies. 7 In South Africa, an aqueous decoction of the leaves is used to treat chest complications, coughs, and bloody sputum. 8 In Zimbabwe and Ethiopia, root-based formulations of M. capensis are used to manage male impotence.9–11 Additionally, traditional healers in Vietnam and Thailand utilize M. capensis to treat various diseases, including diabetes. 12
Phytochemical studies of various Macaranga species have identified the genus as a source of prenylated stilbenes and flavonoids, coumarin, ellagic acids, and terpenoids, which exhibit intriguing biological properties.13–18 Additionally, a previous study by Majeed 12 reported the presence of phytochemicals, such as 3α-hydroxyaleuritolic acid 2-p-hydroxybenzoate and 3-acetoxyoleanolic acid, in the crude methanolic extract of M. capensis stem bark. Our recent investigation examined the antibacterial activities of crude extracts from different parts of M. conglomerata, M. kilimandscharica, and M. capensis against 13 bacterial strains expressing multi-drug resistance (MDR) phenotypes. 19 Among these, the stem bark of M. capensis exhibited significant antibacterial activity, with minimum inhibitory concentration (MIC) values ranging from 4 to 32 μg/mL and bactericidal effects (with minimum bactericidal concentration (MBC)/MIC ratio ≤ 4) against Escherichia coli, Enterobacter aerogenes, Klebsiella pneumoniae, Providencia stuartii, Pseudomonas aeruginosa, and Staphylococcus aureus. 19 Despite the traditional uses of various parts of M. capensis and the promising pharmacological potential of their crude extracts, limited information is available on the phytochemical characterization of this species. Therefore, this study aims to fill this gap through the isolation and identification of chemical constituents from the stem bark of M. capensis. As a result, phytochemical investigation of the stem bark of M. capensis led to the isolation of different classes of secondary metabolites, and their structure elucidation is reported herein.
Methodology
Experimental Procedures
Silica gel (Kieselgel 60; mesh sizes 60-120, 70-230, and 230-400; Merck, Darmstadt, Germany) and Sephadex LH–20 (25-100 μm; Sigma Aldrich, USA) were used as stationary phases for column chromatography (CC) and gel filtration, respectively. Thin Layer Chromatography (TLC) was performed on pre-coated silica gel 60 F254 plates (0.25 mm; Merck, Darmstadt, Germany). Various solvent systems were tested, with optimal separation achieved using hexane-ethyl acetate or dichloromethane-methanol (v/v), depending on the polarity of the compound. TLC Plates were visualized under UV light at 254 nm and 365 nm, and further treated by spraying with a 5% (v/v) H2SO4–H2O solution followed by gentle heating. All solvents, including methanol, dichloromethane, and n-hexane, were of analytical grade and purchased from either Fischer Scientific or Sigma Aldrich (USA). NMR spectra were recorded on Bruker Advance Neo spectrometers operating at 400, 500, or 600 MHz for 1H NMR and at 100, 125, or 150 MHz for 13C NMR, respectively (Bruker BioSpin GmbH, Rheinstetten, Germany). Samples were dissolved in deuterated solvents, and all the spectra were recorded at room temperature using standard pulse sequences. Spectral data were processed using MestReNova 14.1 software and referenced to the residual solvent peaks.
Plant Collection
The stem bark of Macaranga capensis (Baill.) Benth. ex Sim was harvested in March 2019 from mature trees growing in Ngangao Forest, Taita-Taveta County, Kenya (3°25ʹ S, 38°20ʹ E). Collection was performed using a sterile spud to gently remove sections of bark without damaging the main trunk or compromising the health of the tree, ensuring a sustainable harvesting approach. Botanical identification of the plant was confirmed by Mr Patrick C. Mutiso, a taxonomist from the Faculty of Science and Technology (FST), University of Nairobi, where a voucher specimen (HIUON 2019/003) was deposited at the University of Nairobi Herbarium for future reference.
Extraction and Isolation
The stem bark of M. capensis was cut into pieces, air-dried under shade, ground into fine powder (600 g), and extracted three times at room temperature using a 1:1 mixture of methanol and dichloromethane for 24 h each. The combined extracts were filtered and concentrated under reduced pressure using a rotary evaporator, yielding a dark brown crude extract (65.9 g), corresponding to an extraction yield of 11.0%, calculated using the formula: yield (%) = (weight of dried extract/weight of dried plant material) × 100. Approximately 60.0 g of stem bark crude extract was subjected to column chromatography (CC) on silica gel (60-120 mesh), eluted with EtOAc/n-hexane (0:10 to 10:0), yielding 350 fractions of 100 mL each. These fractions were combined based on their TLC profiles into six fractions (FA−F). Fraction FC (580.5 mg) was then subjected to another CC on silica gel (230-400 mesh), eluted with an isocratic system of EtOAc/n-hexane (3:7) to give compound

Schematic Representation of the Isolation and Purification Workflow of the Compounds from the Stem Bark Extract of M. capensis.
Results
Identification of Compounds
Systematic fractionation of the stem bark extract, followed by chromatographic purification of subfractions, led to the isolation of eight secondary metabolites, including flavonoids (
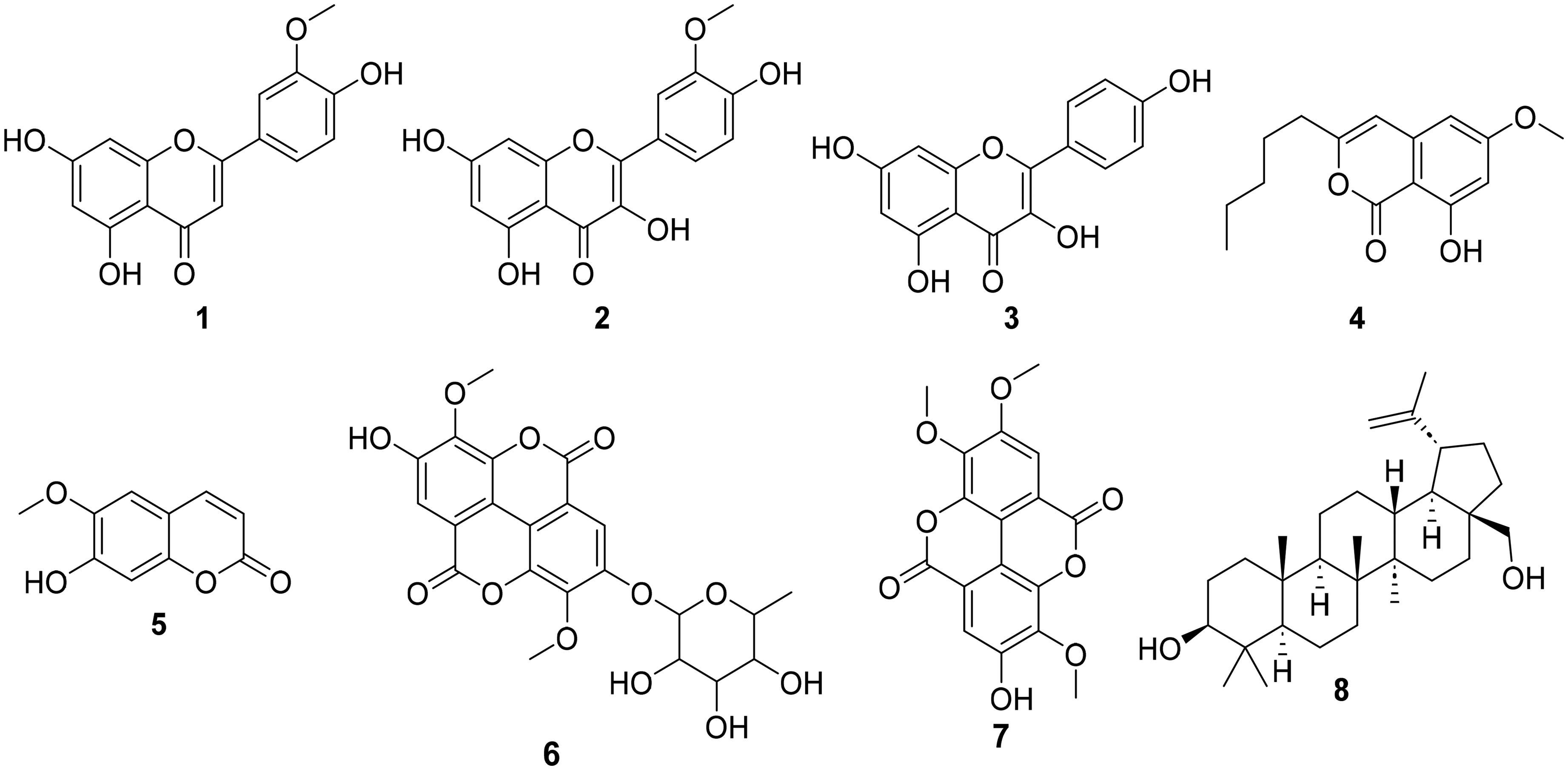
Chemical Structure of Compounds
Chrysoeriol (
Isorhamnetin (
Kaempferol (
8-Hydroxy-6-methoxy-3-pentyl-1H-isochromen-1-one (
Scopoletin (
3,3′-di-O-methylellagiciacid-4′-O-α-L-rhamnopyranoside (
3,3ʹ,4′-trimethoxyellagiciacid (
Betulin (
Discussion
This study presents the chemical investigation of M. capensis, a plant endemic to Kenya. All the compounds reported here were isolated from this plant for the first time. However, some of these compounds have been previously reported from other Macaranga species, supporting M. capensis's taxonomic position within the genus. Notably, this is evident for kaempferol (
The distinct chemical components isolated in this study may provide an explanation for some of the ethnomedicinal uses of M. capensis in regions where it is found. Additionally, these metabolites may contribute to the bactericidal activity observed in the stem bark extract, as demonstrated in our recent investigation against bacterial strains exhibiting MDR phenotypes.
19
It is important to note that several of these compounds have already been studied for various biological activities, including antibacterial. For instance, Nascimento et al
41
investigated the antibacterial activity of chrysoeriol (
Conclusion
Eight (08) secondary metabolites, including flavonoids, isocoumarin, and its chemical cousin, coumarin, ellagic acid, and terpenoids, are reported from the stem bark of Macaranga capensis. The occurrence of these compounds supports the plant's traditional medicinal use and underscores its rich phytochemical profile. Notably, the identification of certain compounds for the first time within the Macaranga genus may help chemotaxonomic studies. These findings contribute to a deeper understanding of the chemical diversity of secondary metabolites found in the genus Macaranga and their potential therapeutic applications. Future studies should focus on a comprehensive pharmacological evaluation of these compounds.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251372423 - Supplemental material for Chemical Constituents from the Stem Bark of Macaranga capensis (Baill.) Benth. ex Sim
Supplemental material, sj-docx-1-npx-10.1177_1934578X251372423 for Chemical Constituents from the Stem Bark of Macaranga capensis (Baill.) Benth. ex Sim by Hashim Ibrahim, Justus Mukavi, John Mmari Onyari, Leonidah Kerubo Omosa, Vaderament-A. Nchiozem-Ngnitedem and Inna Popova in Natural Product Communications
Footnotes
Acknowledgement
We thank Mr Patrick C. Mutiso of the Herbarium, Faculty of Science and Technology (FST), University of Nairobi, for plant identification.
ORCID iDs
Funding
This work was supported by the International Science Programme (ISP) through the KEN-02 project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
