Abstract
Antagonism in traditional medicine has long been documented to warn practitioners against the concurrent use of certain herbal pairs that may lead to adverse effects or toxicity. With scientific advancements, understanding the interaction mechanisms of herbal pairs in Thập Bát Phản (Eighteen Incompatibilities), particularly licorice with Euphorbia kansui, Euphorbia pekinensis, Sargassum, and Daphne genkwa, has become crucial. A review of 87 scientific publications retrieved from three databases – PubChem, Google Scholar, and CNKI – suggests that these interactions are variable, depending on the combination ratio and duration of use. Additionally, some antagonism combinations of Licorice exhibit beneficial pharmacological effects, such as reducing ascites and treating goiter, highlighting their potential for clinical applications.
Introduction
Drug interactions are defined as occurrences in which the effect of a drug is altered due to its simultaneous use with another pharmaceutical drug, medicinal herb, food, or environmental factor. In modern clinical medicine, drug interactions are commonly classified into two types: pharmacokinetic interactions and pharmacodynamic interactions, each with specific subcategories. Pharmacokinetic interactions occur during absorption, distribution, metabolism, and excretion, whereas pharmacodynamic interactions involve synergistic or antagonistic effects. 1
Traditional medicine also classifies drug interactions into six types, including mutual reinforcement, mutual assistance, mutual restraint, mutual incompatibility, mutual inhibition, and mutual antagonism. In traditional medicine, mutual incompatibility refers to the enhancement of toxicity when two herbs are used together. This type of interaction has been documented in ancient medical texts, particularly in the Shennong Ben Cao Jing (Shennong's Classic of Materia Medica), where 18 pairs of herbs are known as the “Eighteen Incompatibilities”. 2
Licorice, the dried roots and rhizomes of Glycyrrhiza uralensis Fisch., Glycyrrhiza inflata Bat., or Glycyrrhiza glabra L., is a widely used herb in traditional medicine, often serving as an Adjuvant or Guiding agent in prescriptions and formulations. The main bioactive chemical groups in licorice include flavonoids, saponins, polyphenols, carbohydrates, and alkaloids. Nearly 80 saponin compounds have been identified and isolated from licorice, with glycyrrhizic acid being a key marker saponin, characterized by its triterpenoid structure. 3 Regarding its pharmacological effects, licorice exhibits various biological activities, including anti-inflammatory, antibacterial, antiviral, antiparasitic, antioxidant, and hepatoprotective effects. 4 In traditional medicine, it is classified as a Qi-tonifying herb with a sweet taste and neutral nature, entering the Liver and Spleen meridians while connecting to all twelve meridians. Licorice functions to tonify Qi, nourish Blood, moisten the Lung to relieve cough, clear Heat, detoxify, and alleviate pain and spasms. It is commonly used to treat Qi and Blood deficiency, sore throat, pharyngitis, abdominal pain, gastric pain, and inflammatory skin conditions such as swollen boils. 5
Licorice is generally regarded as a non-toxic herb in traditional medicine and can beneficially interact with certain other medicinal herbs such as Paeonia lactiflora, Rheum palmatum, Cinnamomum cassia, etc to exert useful biological effects. 3 However, historical texts have reported cases of toxicity when licorice is used in formulations containing Euphorbia kansui (Cam toại or Kansui), Euphorbia pekinensis (Đại kích), Sargassum sp. (Hải tảo), or Daphne genkwa (Nguyên hoa). 2 Numerous experimental studies have examined the incompatibility between licorice and these herbs, with findings published in various scientific journals. This review aims to compile scientific evidence on the antagonistic interactions of licorice with other traditional herbs, providing a comprehensive overview and exploring the underlying mechanisms of these interactions.
Antagonism Interactions Between Traditional Medicine Herbs and Licorice
Studies on the chemical composition and pharmacological effects of the traditional herbal pairs Licorice – Euphorbia kansui (LK), Licorice – Euphorbia pekinensis (LP), Licorice – Sargassum sp. (LS), and Licorice – Daphne genkwa (LD) were searched in three databases: PubMed, Google Scholar, and CNKI up to June 20, 2025 (Table 1). Because we found that the pairs licorice – Euphorbia kansui and licorice – Sargassum are present in the Gansui Banxia Decoction and Haizao Yuhu Decoction respectively, we included these two keywords to the search process.
Search Terms on the Databases.

The search yielded 94 research studies on the mutual incompatibility between licorice and other traditional medicinal herbs, including 28 studies on the LK, 8 studies on the LP, 38 studies on the LS, 14 studies on the LD, and 6 studies investigating two or more incompatible herb pairs with licorice simultaneously (as illustrated in Figure 1).

Number of Publications on the Interaction Between Licorice and Four Antagonistic Herbs.
The Interaction Between Licorice and kansui
Kansui (Euphorbia kansui or Cam toại) is a purgative herb with a bitter taste, cold nature, and toxic properties, entering the Spleen, Lung, and Kidney meridians. It has the function of draining water and purging downward, commonly used to treat abdominal edema and urinary retention. 5 Regarding its chemical composition, approximately 100 phytochemicals have been extracted, isolated, and structurally identified from Euphorbia kansui, belonging to various groups such as ingenane-diterpenes, jatrophane-diterpenes, triterpenes, steroids, and organic acids. Several pharmacological effects of Euphorbia kansui have been reported, including diuretic activity, detoxification effects, anti-tumor properties, treatment of pancreatitis, and inhibition of fibroblast proliferation. 6
Ancient medical texts document the simultaneous use of Euphorbia kansui and Licorice in the formula Gansui Banxia Decoction, proposed by the renowned physician Zhang Zhongjing in his work Jin Gui Yao Lue (Essential Prescriptions from the Golden Cabinet). This formula consists of 1.5 g of Euphorbia kansui, 9 g of Pinellia ternata, 15 g of Paeonia lactiflora, 10 g of honey-processed Licorice, and 15 g of honey. Gansui Banxia Decoction is traditionally used for draining excess fluids, resolving phlegm, dispersing accumulations, eliminating chronic conditions, unblocking meridians, and relieving pain, particularly in cases of diarrhea caused by fluid retention. In this formula, E. kansui acts as Monarch herb, promoting the elimination of retained fluids. P. ternata transforms dampness and disperses phlegm-retention. P. lactiflora serves as Minister herb, assisting in dissolving phlegm, dispersing accumulations, soothing the Liver, and stabilizing Yin. Honey-processed Licorice functions as Guide herb, helping to moderate the harshness of the formula and detoxify. According to Jin Gui Yao Lue Tang Zheng Lun Zhi, the mutually incompatible pair LK is intentionally combined in this formula to enhance the water-draining effect of Euphorbia kansui, demonstrating a strategic use of incompatibility in traditional medicine. 7
Toxicity of Licorice – kansui Combination
The effects of the combination of licorice and Euphorbia kansui on various organ functions have been studied and reported. Research by Huang et al showed that a 1:1 ratio of licorice – kansui exhibited toxicity to the heart and liver in mice (as evidenced by increased biochemical markers ALT, CPK, LDH, and HBDH and mild congestion in the tissues) but had little impact on kidney function. 8 Sun et al provided additional evidence in 2014 and 2015, indicating that a 1:1 combination of licorice – kansui caused minimal kidney damage by promoting cellular apoptosis (with increased Bcl-2 gene expression, decreased Bax, reduced BUN and CRE levels, and little significant microscopic renal injury) compared to kansui alone.9,10 Long-term use of kansui may accelerate apoptosis and affect kidney function in mice. However, combining it with licorice can mitigate its nephrotoxicity, with effects dependent on dosage. The licorice – kansui combination (1:1 ratio) was also reported to increase the absorptive permeability of Rhodamine 123 in the jejunum of mice while reducing secretory permeability. 11 The decrease in R123 permeability and CF (fluorescent sodium) transport further suggests that the combination inhibits P-glycoprotein function, reducing the excretion of toxic compounds from Euphorbia kansui, thereby increasing its toxicity. Moreover, licorice – kansui combinations at ratios of 6.25:1 and 6.5:1 did not exacerbate gastrointestinal damage but disrupted the gut microbiota more severely than kansui alone, particularly affecting Mucispirillum, Roseburia, and Desulfovibrio strains. 12 Thus, it appears that a ratio of licorice greater than or equal to that of Euphorbia kansui leads to toxicity when these two are used in combination.
The toxicity of the LK combination is not only dependent on the dosage of E. kansui but also positively correlated with the proportion of licorice in the mixture, as reported by Shen et al in 2015. Specifically, increasing the ratio of licorice in the combination may exacerbate its toxicity. Histopathological analysis of mice administered a 10:1 LK decoction revealed moderate mucosal alterations and necrosis in the intestines, with a significant infiltration of inflammatory cells. 13 The study also found that LK ratios of 1:2 and 1:4 were the most effective in treating malignant pleural effusion in mice, as they reduced pleural fluid accumulation, increased urine volume, and improved several biochemical markers. However, as the proportion of licorice increased, adverse effects on the liver and heart became more pronounced. The optimal balance between efficacy and toxicity in the LK combination was suggested to fall within the range of 1:1 to 1:2. 14 This ratio has been confirmed in a recent study by Li et al. The authors proposed that when the proportion of licorice is high, the solubilizing effect on kansuinin A (a bioactive compound of Euphorbia kansui) decreases due to precipitation or colloidal aggregation. Moreover, the H2S – AQP2 metabolic pathway is believed to be a key link in the efficacy–toxicity mechanism of the licorice – Euphorbia kansui combination. Finally, the authors suggested that when the human dose of Euphorbia kansui is 1.5 g, co-administration with 0.75 g of licorice produces a beneficial synergistic effect, whereas at higher doses of 7.5 g and 15 g, licorice begins to exert toxic effects. 15
In addition to studies on the toxicity of the LK combination, in vivo safety assessments of the Gansui Banxia Decoction formula have also been conducted. Research by Liu et al in 2013 demonstrated that combining licorice – kansui with other ingredients in Gansui Banxia Decoction increased the LD50 value compared to using the LK combination alone (LD50 values of 51.01 g/kg and 48.26 g/kg, respectively). 16 Interestingly, the ratio of licorice in the Gansui Banxia Decoction is higher than that of Euphorbia kansui (6.67:1). This suggests that other ingredients in the formula, such as P. ternata (Banxia) and P. lactiflora (Paeoni), may help reduce the toxicity of the licorice – kansui combination. Additionally, the licorice – kansui within Gansui Banxia Decoction did not exhibit adverse effects on kidney function but reduced the activity of CPK, an enzyme associated with heart function.17,18 However, there were conflicting conclusions regarding hepatotoxicity between two research groups. Liu et al reported no significant liver toxicity from Gansui Banxia Decoction, while Wang et al suggested that if the amount of E. kansui in the formula exceeded 1.5 g, it could cause a certain degree of liver damage. Based on data analysis, Wang's team recommended avoiding LK combinations with a ratio lower than 7:1 or higher than 20:1.17-19 Notably, the Gansui Banxia Decoction formula used in Liu et al's study had a LK ratio of 15:1 (1.67 g of honey-processed licorice and 0.11 g of kansui), which aligns with Wang's findings. Interestingly, the ratio recommended by the renowned physician Zhang Zhongjing was 6.67:1 (10 g of honey-processed licorice and 1.5 g of kansui), highlighting differences in the potential toxicity of this combination between animal models and human applications.
Efficacy of Licorice – kansui Combination
Mouse models inducing fluid accumulation were used to evaluate the efficacy of the LK combination. The LK ratio of 0.39:1 significantly enhanced diuretic effects, reduced body weight, ascites severity, ascitic fluid volume, and abdominal circumference in mice with ascites induced by H22 hepatocellular carcinoma (HCC) cells. In contrast, ratios of LK ≥ 1.1:1 showed no statistically significant difference compared to the disease model group, as reported by Lin et al. 20 Moreover, the mRNA and protein expression levels of AQP2 (aquaporin-2) and AVPR2 (arginine vasopressin receptor 2) were upregulated in the group treated with an LK ratio of 0.39:1, while no significant changes were observed in the group treated with an LK ratio of 1.1:1. 20 By 2017, Shen et al reported that E. kansui and the LK combination decoctions were most effective in reducing pleural effusion volume in mice with malignant pleural effusion induced by Walker 256 cells. E. kansui helped regulate disrupted metabolic processes in diseased mice, with this effect being enhanced at a 1:4 ratio but diminished when the licorice proportion increased to 4:1. Additionally, seven endogenous metabolites associated with disease conditions were identified, including niacinamide, nicotinic acid, selenocystathionine, Se-adenosylselenomethionine, 4-hydroxycyclophosphamide, and L-alanine. The E. kansui and LK (1:4) treatment groups had metabolic indices closer to the control group, whereas the licorice – kansui (4:1) and licorice groups had indices similar to the disease group. 21 A year later, Chen et al, using the ultra performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry (UPLC-QTOF/MS) combined with heatmap analysis, provided further evidence that the LK (1:4) combination improved metabolic abnormalities in mice with malignant pleural effusion, restoring them to normal levels. Conversely, the LK (4:1) combination exacerbated disease symptoms. 22 The LK (1:4) ratio was suggested to modulate abnormal nicotinate and nicotinamide metabolism (resulting from tumor invasion-induced stress), selenoamino acid metabolism (due to oxidative damage or tumor-induced inflammation), primary bile acid biosynthesis, and sphingolipid metabolism, thereby exerting beneficial effects in malignant pleural effusion mice. 22
Although classified as antagonistic (increasing toxicity when used together), licorice and kansui are both present in the traditional formulation Kansui Banxia Decoction, where they function to expel retained fluid and disperse accumulations (Figure 2). A study by Wang et al (2013) demonstrated that the LK ratio of 15:1 in Gansui Banxia Decoction could increase urine output and reduce vascular endothelial growth factor (VEGF) concentrations in abdominal fluid. This suggests that this ratio has a diuretic effect and counteracts the increase of cytokine-related ascitic factors, acting as an effective cytokine antagonist. 23 Continuing research on the malignant ascites model, Liu et al (2014) and Huo et al (2022) investigated the effects of Gansui Banxia Decoction with or without the LK combination. Results showed that the full formulation exhibited superior diuretic effects compared to modified versions where one or both of the antagonistic herbs were omitted.24,25 Not only did the 15:1 LK combination in the Gansui Banxia Decoction reduce ascitic fluid caused by Walker 256 tumor cells, but it also affected the immune system, according to a study by Huo et al (2024). Specifically, the formula containing the 15:1 LK combination promoted the infiltration of CD4⁻CD8⁺ T cells into the tumor, thereby enhancing antitumor immunity. It inhibited excessive T-cell proliferation in the bone marrow while promoting T-cell proliferation in the thymus, increased tumor cell killing capacity (by raising the proportion of natural killer cells and cytotoxic lymphocytes), and suppressed immunosuppression (by reducing the proportion of regulatory CD8⁺CD25⁺Foxp3⁺ T cells). 26 One year later, the research team reported additional findings on the Gansui Banxia Decoction with a 15:1 LK ratio. These included reduced concentrations and increased mRNA expression of ANP (Atrial Natriuretic Peptide) and BNP (B-type Natriuretic Peptide) in the kidneys, upregulation of mRNA expression of NPR-A and PKG II genes, and elevated levels of cGMP. Furthermore, the authors confirmed that removing one or both herbs of the incompatible pair diminished the therapeutic effects of the formula. 27

Mechanism of Malignant Ascites Formation and the Therapeutic Mechanism of Gansui Banxia Decoction (Created with BioRender.com).
Walker 256 cells induce malignant ascites in rats by increasing the volume of ascitic fluid and reducing peripheral blood volume, thereby activating the renin –angiotensin – aldosterone system (RAAS). When the RAAS is activated, the adrenal glands secrete more aldosterone, which promotes sodium and water retention.24,25 In response, natriuretic peptides are secreted in greater amounts to counteract RAAS overactivation. However, due to RAAS activation, the kidneys increase renin secretion, which converts angiotensinogen into angiotensin I, and subsequently into angiotensin II through the action of ACE. Elevated levels of angiotensin II reduce the interaction between atrial natriuretic peptide (ANP) and its receptor NPR-A and promote the degradation of cGMP, thereby worsening ascites.
27
Furthermore, the reduction in peripheral blood volume stimulates the pituitary gland to release more antidiuretic hormone (ADH, also known as vasopressin), leading to increased water reabsorption in the renal collecting ducts via upregulation of AQP2. This is mediated through the activation of the Gs-AC-cAMP-PKA signaling pathway, which also exacerbates ascites.
25
Therefore, the proposed mechanisms by which the Gansui Banxia Decoction alleviates ascites include:
Interestingly, the LK ratio that is considered toxic (greater than 1:1) has been applied for anticancer purposes when present within a multi-herb formula. Specifically, a study by Feng et al (2021) showed that the Gansui Banxia Decoction with an LK ratio of 1:1 exhibited cytotoxicity against H22 hepatocellular carcinoma cells (IC50 = 463.22 ± 34.77 mg/L). In in vivo experiments, the formula reduced tumor volume and weight, increased the proportions of T cells, B cells, and NK cells, and decreased the frequency of CD11b⁺F4/80⁺ macrophages and CD11b⁺Gr-1⁺ myeloid-derived suppressor cells (MDSCs). It also reduced the levels of cytokines IL-1β, IFN-γ, and IL-10, and markedly downregulated the phosphorylation of signaling proteins including p-AKT, p-mTOR, p-ERK, p-STAT3, p-ERK1/2, and p-STAT3.Based on these findings, the authors proposed that the mechanism by which the formula treats hepatocellular carcinoma involves the inhibition of the accumulation and proliferation of MDSC precursors, as well as the suppression of the AKT/STAT3/ERK signaling pathways. 28
Most recently, a study by Huang et al (2025) found that the 2:1 LK ratio in the same formula also exhibited cytotoxicity against H22 hepatocellular carcinoma cells. Through network pharmacology, in silico analysis, and experimental validation, the authors identified schaftoside, lactiflorin, and violanthin (Figure 3) as potential active compounds in the formula responsible for its anticancer effects. These compounds exert their effects by upregulating p53 expression and downregulating Bcl-2, contributing to apoptosis and inhibition of liver cancer cell proliferation. 29

Chemical Structure of 3 Potential Anticancer Phytocompounds.
Changes in Chemical Composition of Licorice – kansui Combination
Additionally, substantial evidence has demonstrated and reported that chemical composition changes contribute to the increased toxicity of the LK combination. Shen et al suggested that licorice and kansui regulate each other's active compound levels, with licorice increasing the content of toxic diterpenoid compounds in kansui. Specifically, the concentrations of 12 ingenane-diterpenes and 7 jatrophane-diterpenes were significantly elevated in proportion to the LK ratio, leading to enhanced adverse effects such as tumor promotion, inflammation, and cytotoxicity. 30 Euphol, a steroid compound known for its hepatotoxic effects, 31 was found to be nearly three times higher in the LK combination (1.3060 mg/g) compared to E. kansui alone (0.5399 mg/g). 32 Furthermore, this combination influenced pharmacokinetics by inhibiting CYP2C19 activity, thereby reducing the metabolism and elimination of kansuinin A and B (toxic diterpenoids in E. kansui), ultimately increasing toxicity when licorice and kansui are used together. 33
Conversely, the safety of LK in the Gansui Banxia Decoction is attributed to the role of P. lactiflora. A study by Cui et al (2018) found that adding P. lactiflora to the LK combination increased the bioavailability of isoliquiritigenin, liquiritin, glycyrrhetinic acid, and glycyrrhizic acid compared to the LK group alone. This suggests that P. lactiflora enhanced the absorption of these compounds and counteracted the inhibitory effects of E. kansui on licorice. 34 Figure 4 illustrates the chemical structures of certain compounds in kansui that are altered when combined with licorice.

Chemical Structures of Several Phytocompounds Affected by the Combination of Licorice and Kansui.
Additionally, several other studies on the chemical composition of the LK combination have been documented. A study by He et al (2018) showed that differences in solvent polarity affected the type and quantity of chemical components in the extracts. Furthermore, an increase in licorice caused the peak maximum heat loss rate to occur earlier. For petroleum ether, chloroform, and ethyl acetate extracts, the peak maximum heat loss rate appeared earlier; however, the heat loss rate increased for chloroform, while it decreased for petroleum ether and ethyl acetate. 35 A study by Zhang et al (2021) proposed an optimal processing method for E. kansui fried with licorice decoction to maximize euphorbiadienol content and alcohol-soluble components. The optimal parameters included a licorice ratio of 27%, a roasting temperature of 180 °C, and a roasting time of 11 min. The average euphorbiadienol content in three batches of optimally processed E. kansui was 0.225%, and the average alcohol-soluble content was 16.621%. 36 However, these two studies have not demonstrated a correlation between changes in toxicity and the efficacy of the licorice – kansui combination.
The Interaction Between Licorice and Euphorbia pekinensis
Similar to E. kansui, Euphorbia pekinensis (Đại kích) is a purgative herb in traditional medicine. It has a bitter taste, cold nature, and is toxic, entering the Liver, Kidney, and Spleen meridians. Its functions include expelling water retention, purging downward, and detoxifying. It is used to treat constipation, abdominal distension, and edema. 5 The primary chemical components of E. pekinensis are diterpenoids with various skeletal structures, including cembrane, isopimarane, and abietane. Regarding pharmacological effects, phytochemicals in E. pekinensis have been demonstrated to possess antibacterial and cytotoxic activities. 37
Toxicity of Licorice – Euphorbia pekinensis Combination
Both E. pekinensis and E. kansui belong to the Euphorbia genus and are classified as “water-expelling” herbs in traditional medicine. Consequently, the antagonistic interaction between licorice and Euphorbia pekinensis has also been observed in clinical practice. Similar to E. kansui, the combination of licorice and E. pekinensis at a 1:1 ratio enhances the hepatotoxicity of E. pekinensis, reduces CYP3A2 enzyme activity, and affects cardiac function while having minimal impact on kidney function.38,39 From a chemical perspective, co-administration of licorice and E. pekinensis may reduce hepatoprotective effects (by decreasing liquiritin content) and increase the herb's water and sodium retention toxicity (by elevating glycyrrhizic acid levels). 40 However, studies on the toxicity of this combination remain limited, and further research is needed.
Efficacy of Licorice – Euphorbia pekinensis Combination
The hepatocellular carcinoma (HepG2) cytotoxic effects of the licorice and E. pekinensis combination at a 3:1 ratio were reported by Wu et al in 2014. Specifically, the LP 3:1 combination induced cell contraction, reduced cell count, and inhibited the proliferation of HepG2 cells. 41 However, the diuretic effect of E. pekinensis may be suppressed by licorice at equivalent doses based on the 2010 Chinese Pharmacopoeia, as observed in the study by Xu et al (2014). 42 In 2020, Ding et al conducted experimental studies that further explained the reduction in diuretic efficacy when licorice is combined with E. pekinensis. Specifically, licorice attenuated the water-expelling effects of E. pekinensis by decreasing intestinal motility, prolonging the retention time of substances in the intestines, and delaying the excretion of ascitic and pleural fluids. This led to renal interstitial edema, increased renal capillary permeability, and ultimately, elevated protein levels in the urine. 43
Additionally, in 2017, Yan et al investigated the efficacy of the licorice and E. pekinensis combination in treating ascites in mice with hepatocellular carcinoma (H22 HCC ascitic tumor model). Their study showed that, compared to the disease group, mice treated with E. pekinensis decoction and a LP decoction at a 2:1 ratio exhibited significant reductions in ascitic fluid volume, abdominal circumference, body weight, and the expression of AQP2 and vasopressin receptor 2 (V2R) proteins. Among these, the 2:1 combination group demonstrated greater therapeutic effects than the E. pekinensis monotherapy group. Conversely, the 4:1 combination significantly reduced the therapeutic efficacy of E. pekinensis in treating ascites. 44 The authors suggested that the enhanced therapeutic effect of the LP combination on ascites might be associated with the regulation of the AQP2 and V2R signaling pathways in the kidney. Using the same model, Zhang et al further supported the effectiveness of the LP combination and proposed that the mechanism underlying ascitic fluid reduction at a 2:1 ratio might be linked to the downregulation of the Frk-Arhgdib-Inpp5d-Avpr2-Aqp4 axis. 45 It appears that the 2:1 LP ratio represents a synergistic interaction between licorice and Euphorbia pekinensis, while the 4:1 ratio reflects an antagonistic effect.
The Interaction Between Licorice and Sargassum
The medicinal herb Sargassum (Hải tảo) has a salty taste and cold nature, entering the Liver, Spleen, and Kidney meridians. It functions to dissolve phlegm and promote diuresis and is commonly used in conditions such as testicular swelling and pain, edema, and swollen legs. The primary bioactive compounds in Sargassum include polysaccharides such as alginic acid derivatives, fucoidan, and laminaran. 5 According to modern medicine, Sargassum exhibits various pharmacological effects, including antioxidant, anti-tumor, immune-boosting, anti-aging, hypoglycemic, anti-platelet aggregation, antibacterial, and antiviral properties. In traditional medicine, Sargassum is commonly derived from Sargassum pallidum and Sargassum fusiforme. 46
The concurrent presence of licorice and Sargassum has been documented in the Haizao Yuhu Decoction Formula, recorded in Orthodox Manual of External Medicine (Wai Ke Zheng Zong, Ming). This formula consists of 11 herbal ingredients: 299.52 g of Thallus Sargassi Pallidi, 116.14 g of Thallus Laminariae Japonicae, 116.14 g of Rhizoma Pinelliae Preparata, 116.14 g of Bulbus Fritillariae Thunbergii, 116.14 g of Pericarpium Citri Reticulatae, 116.14 g of Pericarpium Citrus reticulata, 116.14 g of Radix Angelicae Sinensis, 116.14 g of Rhizoma Chanxiong, 116.14 g of Radix Angelicae Pubescentis, 116.14 g of Fructus Forsythiae Suspensae and 696.96 g of Radix Glycyrrhiza. In clinical practice, Haizao Yuhu Decoction is used for its functions in soothing the Liver, resolving phlegm, softening hardness, and dissipating nodules. It is commonly prescribed for conditions related to Liver Qi stagnation, phlegm accumulation, and goiter. 47
Toxicity of Licorice – Sargassum Combination
Sargassum is a heat-clearing and dampness-drying medicinal herb in traditional medicine. It is non-toxic and widely consumed as a food ingredient in household meals. However, when combined with licorice, the mixture has been documented in classical medical texts to possess potential toxicity. Similar to the LK and LP combinations, the toxicity of the LS combination depends on the ratio of the two herbs in the mixture. A study by Ding et al (2002) demonstrated that LS combinations at ratios of 1:1 and 3:1 induced hepatic CYP450 enzyme activity and caused hepatotoxicity, as evidenced by focal necrosis and increased lymphocyte infiltration in liver tissue. 48 Particularly, the toxicity of LS was further supported by findings from Yan et al (2007) and Zhao et al (2014). Their studies showed that a 3:1 ratio affected blood composition, liver, and kidney function, while a 1:1 ratio induced liver damage associated with decreased expression of the sodium taurocholate cotransporting polypeptide (Ntcp) protein, leading to bile acid transport imbalance.49,50 Moreover, acute toxicity studies reported by Ji et al (2012) revealed that LS combinations at 1:1 and 3:1 ratios resulted in 100% mortality in experimental mice. 51 Additionally, other adverse effects of the LS combination have been documented. In 2014, Ding et al observed that the combination inhibited intestinal smooth muscle stimulation in mice. 52 By 2018, Meng et al proposed that the primary mechanism of toxicity in the LS 1:1 combination was related to increased accumulation of glycyrrhetinic acid and inhibition of 11β-hydroxysteroid dehydrogenase type 2 (HSD11B2) expression, leading to dysregulation of the aldosterone-cortisol system. 53 It seems that the toxicity of the licorice – Sargassum combination is manifested when the ratio of licorice is greater than or equal to that of Sargassum.
An interesting aspect is that the toxicity of the LS combination also depends on the species of Sargassum used, the processing method of licorice, and the incorporation of the LS combination in Haizao Yuhu Decoction. In 2016, Xiu et al compared the acute toxicity between the two Sargassum species, S. pallidum and S. fusiforme, in combination with licorice. Their results indicated that the licorice – S. pallidum combination exhibited low toxicity in mice (LD50 = 40.83 g/kg), while S. fusiforme showed no apparent toxicity. 54 However, the researchers also observed hepatocyte and renal cell contractions in mice administered decoctions containing S. fusiforme, whereas this toxicity was absent in those treated with formulations containing S. pallidum. 55 Regarding the processing method of licorice, Li et al reported that the acute toxicity of the combination decreased in the following order: Sargassum > Sargassum – honey-processed licorice > Sargassum – raw licorice > honey-processed licorice > Haizao Yuhu Decoction (with honey-processed licorice) > Haizao Yuhu Decoction (with raw licorice) > raw licorice. 56 By 2013, Liu et al published results on the acute toxicity of Haizao Yuhu Decoction formulations with or without licorice and Sargassum, revealing that the LD50 values decreased in the following order: the formula without Sargassum and licorice > the formula with raw licorice but without Sargassum > the formula with raw licorice > the formula with honey-processed licorice > the formula without licorice. Additionally, in mice treated with decoctions containing honey-processed licorice but without Sargassum, the LD50 could not be determined (indicating no observed toxicity). 57 A 2018 study by Liu et al further demonstrated that the Haizao Yuhu Decoction formula containing S. pallidum increased liver biochemical markers (globulin (GLB), aspartate aminotransferase (AST), AST/ALT ratio) and induced hepatic CYP2E1 and CYP3A1 enzyme activity to a greater extent than S. fusiforme, though it did not significantly affect liver structure. 58
The toxicity study of Haizao Yuhu Decoction was not only evaluated under normal physiological conditions but also assessed in pathological models in mice. Interestingly, there is consistency in the reports by Li et al (2013, 2014) and Liu et al (2014), which found that in the pathological model of goiter induced by propylthiouracil (PTU), Haizao Yuhu Decoction containing contrasting herbal pairs at certain combination ratios did not cause significant adverse effects on the liver, kidneys, or heart. Furthermore, different combination ratios were able to improve abnormally elevated creatine kinase levels induced by PTU-induced hypothyroidism, bringing them back to normal levels. This therapeutic effect tended to decrease as the dose of Sargassum increased.59-61
Efficacy of Licorice – Sargassum Combination
The notable effect of the LS combination is its efficacy in treating goiter. In in vivo experiments, PTU was the selected agent for inducing a goiter model in mice. The modeling results by Ding et al (2003) showed that PTU administration for 10 days reduced serum triiodothyronine (T3) and thyroxine (T4) levels in mice, but the difference was not statistically significant compared to the normal group. 62 Therefore, in many subsequent studies, the PTU administration period was adjusted from 10 days to 14 days to induce a significant reduction in thyroid hormone levels. However, the key finding of Ding et al's study was that decoctions of LS (at ratios ranging from 1:3 to 3:1) reversed the PTU-induced increase in thyroid microsomal antibody (TM) and thyroglobulin (TG). 63 Using the same model in 2010, Xie et al discovered that the licorice – Sargassum combination reversed PTU-induced downregulation of Fas expression and upregulation of Bcl-2 expression, with the effect being positively correlated with the licorice ratio. 64 In 2011, Zhu et al reported an improvement in PTU-induced thyroid damage after 10 days of PTU administration when treated with a LS combination at a 2:1 ratio. Specifically, thyroid follicles were observed to have a uniform size and morphology, follicular epithelial cells appeared cuboidal with no apparent proliferation, follicular cavities were filled with eosin-stained thyroglobulin, and very few surrounding capillaries showed dilation and congestion. 65 It was not until recently that a study by Wu et al (2025) reported on the effects of the licorice – Sargassum pair. Specifically, at a ratio of LS 1:1.2, the combination improved PTU-induced goiter by modulating the autophagy process regulated by Beclin1. 66
The Haizao Yuhu Decoction, which contains the contrasting pair of LS (1:1.2), is used clinically for treating thyroid disorders and goiter. Interestingly, in vivo studies on the effects of this formula consistently indicate that treatment with the complete formula is more effective in regulating thyroid hormone levels and improving histopathological changes in goiter-induced mice compared to treatment with modified formulas lacking one or both of the contrasting herbs. This suggests that the LS is a crucial component of the formula.67,68 The proposed mechanisms of action of the formula, primarily driven by the LS combination, include:
Beyond its effect on goiter treatment, the LS combination has also been demonstrated to exert hepatoprotective effects, as evidenced by the study conducted by Ye and Zhao in 2006. Specifically, in a mouse model of liver injury induced by carbon tetrachloride (CCl4), treatment with a decoction of the LS mixture at various ratios, particularly 1:1 and 3:1, significantly improved AST and ALT enzyme levels compared to the disease model group. 76 These findings align with the results of Ding et al (2002), which indicated that while the LS combination at 1:1 and 3:1 ratios induced hepatotoxicity under normal conditions, it provided hepatoprotective effects in cases of liver injury. The protective effect of LS on liver cells became even more pronounced when incorporated into the Haizao Yuhu Decoction. Through in vivo experiments, Liu et al (2019) proposed that the hepatoprotective effect of the Haizao Yuhu Decoction is associated with the activation of the nuclear factor erythroid 2-related factor 2/heme oxygenas-1 (Nrf2/HO-1) pathway, enhancing the antioxidant capacity of liver cells. 77 More recently, in 2024, Dong et al further supported this hepatoprotective mechanism, linking it to the activation of the Nrf2/HO-1 signaling pathway to reduce oxidative stress in the liver. Additionally, they proposed an alternative protective mechanism: inhibition of the p53/Caspase-3 signaling pathway to reduce hepatocyte apoptosis. 78
Most recently, in 2025, Qian et al published a study on the therapeutic effects of Haizao Yuhu Decoction (with an LH ratio of 1:3) on silicosis. Key findings included alleviation of alveolar structural damage, reduction in inflammatory cell infiltration, a significant decrease in collagen volume fraction, and lower levels of cytokines such as IL-1β, IL-6, TNF-α, and TGF-β1. Based on results from network pharmacology, molecular docking, and experimental validation, the Haizao Yuhu Decoction along with three active compounds (glycitein, diosmetin, and limonin; Figure 5) was shown to suppress cell invasion, reduce the expression of TGF-β1, p-Smad2, p-Smad3, and vimentin, while enhancing E-cadherin expression. From these findings, the authors proposed that the silicosis treatment mechanism of Haizao Yuhu Decoction is through inhibition of the TGF-β1/Smad signaling pathway and suppression of EMT (epithelial – mesenchymal transition). 79 Figure 6 summarizes the therapeutic mechanisms of Haizao Yuhu Decoction.

Chemical Structure of 3 Potential Anti- Silicosis Phytocompounds.

The Therapeutic Mechanisms of Haizao Yuhu Decoction.
In addition to studies evaluating the efficacy of the LS combination and the Haizao Yuhu Decoction, comparative studies on the effects of different Sargassum species and/or Glycyrrhiza species have also been reported. The findings of Xiu et al (2017) indicated that in PTU-induced goiter mice, the Haizao Yuhu Decoction containing S. pallidum and S. fusiforme both reversed the stimulation of toxins and carcinogens in the liver, promoted drug metabolism, and led to similar PTU-induced liver damage. 80 The decoction formulated with both Sargassum species exhibited comparable regulatory effects on thyroid hormone synthesis, as reported by Xiu et al (2017) and Liu et al (2019).55,81 According to Liu et al, the Haizao Yuhu Decoction containing S. fusiforme reduced PTU-induced thyroid-stimulating hormone (TSH) levels in serum and activated TG mRNA expression. 82 At a high therapeutic dose (with LS at 2.0 and 2.4 g/kg, respectively), Wang et al observed that serum T3 levels in mice treated with the decoction containing S. fusiforme were higher than those in the group treated with S. pallidum, while free-T3 (FT3) levels showed the opposite trend. 83 Furthermore, the S. fusiforme group exhibited increased Bcl-2 mRNA expression and a higher Bax/Bcl-2 ratio compared to the S. pallidum group. 84 Table 2 compares the toxicity and efficacy between the two Sargassum species in the formula.
Comparison of Toxicity and Efficacy Between the two Sargassum species in Haizao Yuhu Decoction.

The efficacy of the LS combination varied not only between Sargassum species but also among different Glycyrrhiza species. A study by Chen et al (2021) showed that the decoction containing G. uralensis increased thyroid hormones (T3, T4, FT3, free-T4 (FT4)) and reduced TSH levels compared to the disease model group. G. glabra also restored PTU-induced reductions in FT3 and increases in TSH, though its effect was less pronounced than G. uralensis. Meanwhile, G. inflata showed a general trend of restoring thyroid hormone and TSH levels in serum. Additionally, decoctions containing G. uralensis and G. inflata significantly reduced TRH levels compared to the disease model group. Notably, the decoction with G. inflata was more effective in reducing thyroid follicular cell diameter, while the G. uralensis decoction demonstrated the most significant effects on TPO and TG levels, apoptosis rate, and ERK1 expression. 72
Changes in Chemical Composition of Licorice – Sargassum Combination
The chemical interactions of the LS combination were studied by Liu et al in 2011. Their results showed that when the LS ratio was 1:3, 1:2, and 1:1, the glycyrrhizic acid content significantly decreased, reaching its lowest level at a 1:1 ratio. When the proportion of licorice was increased from 2:1 to 3:1, the glycyrrhizic acid content gradually increased but remained lower than that of licorice alone. 82 In 2019, the research team continued to publish quantitative analyses of active compounds in decoctions containing S. fusiforme, showing statistically significant increases in hesperidin, osthol, and forsythin levels, while peimine and peiminine levels significantly decreased compared to decoctions containing S. pallidum. 80 Meanwhile, a study by Chen et al in 2021 found that decoctions containing G. uralensis had the highest liquiritin and hesperidin content, decoctions with G. inflata had the highest glycyrrhizic acid, forsythin, and ferulic acid content, while decoctions with G. glabra had the highest osthol content. 72 In 2020, Lyu et al conducted a simultaneous quantification of five compounds (liquiritin, glycyrrhizic acid, phyllyrin, hesperidin, and ferulic acid) in different decoction samples, including licorice decoction, licorice – Sargassum decoction (1:1.2 ratio), Haizao Yuhu Decoction, the decoction without Sargassum, the decoction without licorice, and the decoction without both Sargassum and licorice. The results showed that liquiritin and glycyrrhizic acid levels gradually decreased in the order: decoction without Sargassum > Haizao Yuhu Decoction > licorice decoction > LS decoction. Meanwhile, the remaining three compounds decreased in the order: decoction without Sargassum > decoction without both Sargassum and licorice > decoction without licorice > LS decoction. 83 Finally, in 2023, research by Chen et al found that glycycoumarin was detected only in decoctions containing G. uralensis, while licochalcone A was only found in decoctions containing G. inflata. The chemical composition of the decoctions using different Glycyrrhiza species did not show differences in compound types but exhibited variations in the content of glycyrrhizin, glycyrrhizic acid, hesperidin, and forsythin. 84 Studies on the chemical interactions between licorice and Sargassum reveal significant changes in active compound content when adjusting their ratio or using different species. While the overall chemical composition remains unchanged, variations in the levels of key compounds such as glycyrrhizic acid, hesperidin, forsythin, and liquiritin may influence the pharmacological effects and toxicity of the decoction.
The Interaction Between Licorice and Daphne genkwa
Many compound groups have been identified in Daphne genkwa, including diterpenoids, sesquiterpenoids, triterpenoids, flavonoids, coumarins, lignans, and others. Regarding pharmacological effects, various biological activities of Daphne genkwa have been studied and published, such as cytotoxic effects, neuroinflammation inhibition, anti-inflammatory activity, antioxidant properties, growth inhibition, and anti-allergic effects. 85
Toxicity of Licorice – Daphne genkwa Combination
The toxicity of the LD combination (1:1 ratio) on the function and morphology of the heart, liver, and kidneys in rats has been studied and reported. The combination led to an increase in biological markers (ALT, CPK, LDH, HBDH), congestion of interstitial blood vessels in the heart, hepatic vascular congestion, hepatocyte edema, glomerular interstitial congestion, and mild renal tubular epithelial edema. 86 Furthermore, the LD combination at ratios of 1:1 and 3:1 caused diffuse hepatocyte edema, granular and ballooning degeneration, inflammatory cell infiltration, focal hepatocyte necrosis, and scattered eosinophilic hepatocytes, with the 1:1 ratio exhibiting more severe toxicity than the 3:1 ratio. 87 Through metabolomics analysis, the authors suggested that the 1:1 combination of licorice and Daphne genkwa exerts toxicity by disrupting the biosynthesis pathways of phenylalanine, tyrosine, and tryptophan, as well as the metabolism of tyrosine, glycerophospholipids, and glycerolipids. 88 At an LD ratio of 3.3:1, the toxicity of the combination was manifested through increased production of H2S in the colon, disruption of the gut microbiota, and the promotion of Desulfovibrio species. Additionally, there was upregulation of genes involved in both assimilatory and dissimilatory sulfate reduction pathways. 89 At a ratio of 6.6:1, the combination induced toxicity by disrupting epithelial integrity, exacerbating edema, and causing more severe inflammation compared to the Daphne genkwa decoction alone. 90 Thus, a licorice-to-Daphne genkwa ratio that is greater than or equal to 1 may potentially manifest toxicity.
Similar to E. kansui and E. pekinensis, Traditional medicine classifies D. genwka as a purgative herb. Several proposed mechanisms underlying the toxicity of the LD combination include:
Efficacy of Licorice – Daphne genkwa Combination
Beyond toxicity studies, the therapeutic efficacy of the LD combination has also been investigated. Specifically, a ratio of 3.3:1, within a certain range, has been shown to inhibit spontaneous activity in mice. 94 However, when combined at ratios of 5:3, 10:3, and 20:3, the diuretic effect of D. genkwa was diminished, as evidenced by a significant reduction in urine output within two hours after administration. 95 Another studied effect of the LD combination is its impact on ascites caused by liver cancer. Research by Li et al demonstrated that different ratios of the combination could either enhance (1:1 ratio) or reduce (4:1 ratio) the anti-ascitic effects in hepatocellular carcinoma-induced mice, which was associated with the regulation of AQP2 and V2R protein expression in renal tissue. 96 At the molecular level, Yu et al suggested that glycyrrhetinic acid, a component of licorice, is responsible for upregulating AQP2 channels and counteracting the downregulation of AQP2 induced by yuanhuacin and genkwanin from D. genkwa. A pharmacological network analysis revealed that glycyrrhetinic acid, yuanhuacin, and genkwanin interact with MEK1/FGFR1 proteins, influencing the ERK-MAPK pathway. Based on these findings, the authors proposed that in the LD combination at a ratio of 5.36:1, the active compounds modulate AQP2 expression through the ERK1/2 – CREB – AQP2 signaling axis. 97
Changes in Chemical Composition of Licorice – Daphne genkwa Combination
From a chemical transformation perspective, research by Shi et al suggested that the decoction process involving licorice and D. genkwa induces a series of chemical reactions, leading to the precipitation, binding, or modification of certain active components, thereby reducing efficacy. 98 In 2012, Chen et al conducted experimental studies and proposed that interactions between licorice and D. genkwa, particularly vinegar-processed D. genkwa, resulted in an increased concentration of toxic diterpenoid compounds. As the ratio of licorice increased (from 4:3 to 20:3), the levels of diterpenoid and flavonoid compounds in both raw and vinegar-processed Daphne genkwa increased, especially genkwanin, while the levels of luteolin-7-methoxy-5-O-β-D-glucoside remained relatively unchanged, and genkwanin-5-O-β-D-glucoside decreased. 99 Furthermore, with an increasing proportion of licorice, the concentrations of several active compounds in the combination were reduced (as Figure 7), including liquiritin apioside, apigenin-5-O-β-D-glucoside, isoliquiritin apioside, hydroxy-genkwanin, and glycyrrhizic acid, leading to a decrease in pharmacological efficacy. 100
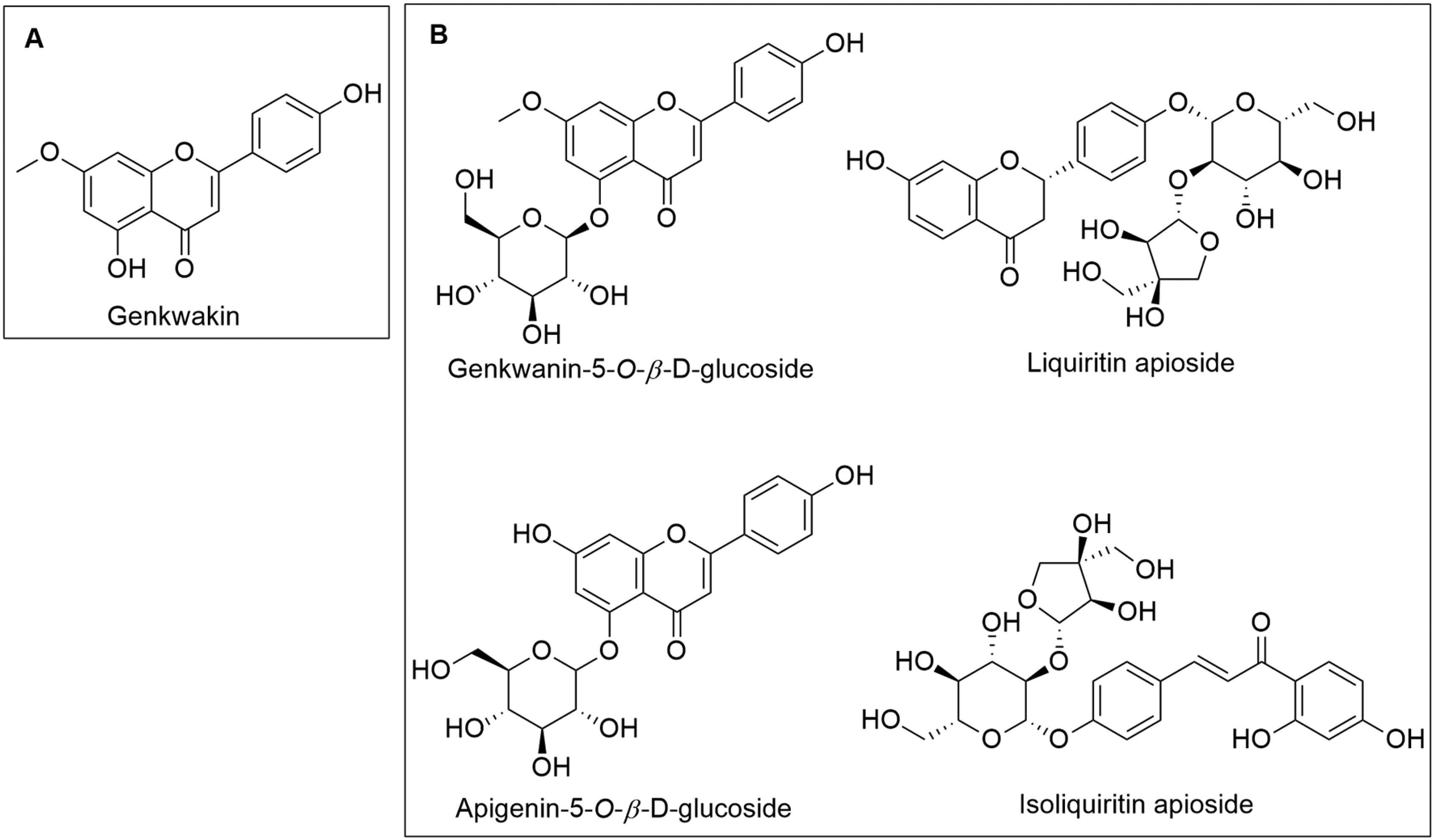
Chemical Structures of Several Phytocompounds Affected by the Combination of Licorice and D. genkwa. A. Increase in Content; B. Decrease in Content.
Comparative Studies on the Toxicity and Efficacy of Licorice in Combination with Antagonistic Herbs
In addition to individual studies on the toxicity and efficacy of combining licorice with antagonistic medicinal herbs, several comparative evaluation reports have also been documented. Studies from 2012 to 2016 have contributed to comparing and clarifying the effects of licorice when combined with antagonistic herbs such as E. kansui, E. pekinensis, Sargassum sp., and D. genkwa on liver enzyme activity, intestinal motility, intestinal tissue permeability, and the active compound content in licorice. The results showed that licorice could stimulate the activity of the liver enzyme CYP1A2 when combined with kansui but had little effect when combined with E. pekinensis and D. genkwa. 101 Regarding its effects on the intestines, licorice inhibits the contractions of the mouse small intestine, while contrasting medicinal substances promote contractions. However, when combined, the contractile effect is reduced. 102 Regarding intestinal effects, licorice inhibited the contraction of the mouse small intestine, whereas the contrasting herbs promoted contraction. However, when combined, the contraction effect was reduced. The influence of licorice on intestinal motility depended on the ratio and dosage of the combination: LP reduced motility when the licorice dose increased, whereas LD only increased motility when the total dose exceeded 5 g/kg. 103 Intestinal tissue permeability also changed upon combination, with licorice and D. genkwa increasing FD4 (FITC-dextran 4 kDa) permeability. However, when combined with E. pekinensis, Sargassum, or kansui, FD4 permeability decreased. For FD40 (FITC-dextran 40 kDa), LS and LK increased permeability, whereas LP and LD decreased permeability. 104 Licorice also enhanced R123 secretion, but this effect was diminished when combined with Sargassum, E. pekinensis, and D. genkwa. 105 Chemically, the combination with antagonistic herbs affected the active compound content in licorice. Specifically, E. pekinensis increased ammonium glycyrrhizinate but reduced isoliquiritigenin and 18β-glycyrrhetinic acid, while Sargassum significantly decreased 18β-glycyrrhetinic acid. D. genkwa increased isoliquiritigenin but sharply reduced 18β-glycyrrhetinic acid, whereas kansui reduced all major compounds in licorice. 106 These findings indicate that combining licorice with contrasting herbs can alter the pharmacokinetics and biological effects of the formulation, emphasizing the importance of determining appropriate ratios and dosages to optimize therapeutic efficacy and minimize adverse effects.
Conclusion
Antagonism is a type of interaction in traditional medicine that has been documented in ancient medical texts based on thousands of years of clinical experience. The antagonistic herbal pairs in traditional medicine, known as the Eighteen Incompatibilities (Thập Bát Phản), were observed and compiled to warn traditional medicine practitioners against their simultaneous use when formulating prescriptions or modifying classical formulas. This precaution aims to prevent severe adverse effects or toxicity in patients. With advancements in scientific research and the increasing emphasis on evidence-based medicine, studying, proving, and elucidating the mechanisms underlying the Eighteen Incompatibilities has become crucial. Our review compiles evidence on the toxicity and efficacy of traditional medicinal herbs that exhibit antagonistic effect with licorice. Overall, the antagonism between licorice and E. pekinensis, E. kansui, Sargassum, and D. genkwa is relative, depending on the ratio of herbal combinations and the duration of use. Conversely, these contrasting combinations of Licorice also demonstrate potent pharmacological effects, such as reducing ascites and treating goiter, which suggests their potential application in clinical practice. However, this work has several limitations. Despite the summary of the available pharmacological studies, clinical evidence directly supporting incompatibility interactions involving licorice remains limited and mainly based on empirical reports. In addition, due to the complexity of multi-herb prescriptions and the diversity of processing methods, this review was unable to comprehensively evaluate their potential influences on the outcomes of herb – herb interactions. Therefore, it is essential to conduct more pharmacological experiments and clinical investigations in the future to further clarify the mechanisms underlying these antagonistic effects, particularly in relation to the generation of toxic compounds, changes in drug metabolism, and the modulation of specific molecular targets.
Footnotes
Acknowledgements
The authors declare that no specific funding was received for this research. All contributions to the study were made by the listed authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
