Abstract
Background
Panax ginseng root extract (PGRE), rich in bioactive ginsenosides, has been widely recognized for its potential health benefits. However, its skincare efficacy, particularly in anti-inflammatory and antioxidant activities, remains to be fully elucidated.
Objectives
This study aimed to evaluate the skincare efficacy of PGRE using primary keratinocytes and a 3D human epidermal model (EpiKutis®), focusing on its anti-inflammatory, antioxidant, and barrier repair mechanisms.
Methods
The cytotoxicity of PGRE was assessed at a concentration of 50 μg/mL, and its anti-inflammatory effects were evaluated by measuring the suppression of IL-1α, PGE2, and NF-κB activation. The antioxidant activity of PGRE was tested under 4-HNE-induced oxidative stress by quantifying reactive oxygen species (ROS) reduction. Additionally, the modulation of the NF-κB signaling pathway was investigated to understand its role in mitigating inflammatory feedback loops.
Results
PGRE at 50 μg/mL showed no cytotoxicity (>80% cell viability) and demonstrated potent anti-inflammatory effects by suppressing IL-1α (34.17%), PGE2 (20.70%), and NF-κB activation (43.65%). Under oxidative stress conditions, PGRE significantly reduced ROS levels by 51.6%, highlighting its strong anti-carbonylation activity. Furthermore, PGRE effectively modulated the NF-κB signaling pathway, mitigating inflammatory feedback loops and underscoring its multitarget mechanism in anti-inflammatory and antioxidant activities.
Conclusion
PGRE exhibits significant anti-inflammatory and antioxidant properties, making it a promising natural ingredient for anti-aging and anti-inflammatory skincare formulations. These findings suggest that PGRE could play a vital role in barrier repair and inflammation modulation. Further clinical studies are warranted to validate these in vitro results and to explore the role of NF-κB in PGRE's anti-inflammatory and barrier repair mechanisms.
Introduction
The use of plant extracts in cosmetics has a long history, dating back to ancient civilizations such as Egypt, Greece, Rome, and China, where natural components from plants were employed for beauty and skincare purposes.1,2 Over time, the understanding of plant extracts deepened, with systematic research and documentation during the Middle Ages and the Renaissance further advancing their applications. 3 By the twentieth century, scientific and technological progress had significantly enhanced extraction and purification techniques for plant-based compounds. 4 Modern advancements in chemistry and biology have allowed researchers to precisely isolate and identify active components in plants, including antioxidants, vitamins, minerals, and plant hormones.5–7 These components are now widely utilized in various cosmetics to deliver diverse skincare benefits, such as hydration, anti-aging effects, anti-inflammatory properties, skin brightening, and repair.8–10 In recent years, consumer demand for natural and organic products has grown significantly. 11 As a result, many brands have introduced cosmetics enriched with natural plant extracts, highlighting their mildness, safety, and effectiveness. 12 The application of plant extracts in cosmetics has thus evolved from traditional practices to highly refined modern techniques. Despite some safety concerns—such as a heightened risk of allergies and increased photosensitivity—plant extracts have significantly enriched the variety and functionality of cosmetics. This evolution offers consumers a broader range of options to address their increasingly sophisticated skincare needs.
Ginseng has a long history of use in East Asia, particularly in traditional Chinese medicine, as a health food, and as a substance recognized as GRAS (Generally Recognized As Safe) by the FDA (Food and Drug Administration). Furthermore, evaluations by the Cosmetic Ingredient Review indicate that ginseng-derived materials, including ginseng extract, are relatively safe for use. The main components of panax ginseng root extract (PGRE) include a variety of bioactive compounds such as ginsenosides, polysaccharides, volatile oils, flavonoids, amino acids, trace elements, vitamins, and organic acids.13–15 Among these, ginsenosides are the primary active components, offering antioxidant, anti-inflammatory, anti-fatigue, immune-enhancing, and cell-regenerating properties.16,17 Polysaccharides provide immune-modulating and anti-tumor effects, 18 while volatile oils contribute aromatic properties as well as antibacterial and anti-inflammatory benefits. 19 Flavonoids exhibit strong antioxidant and anti-inflammatory activities, 20 amino acids play a key role in skin hydration and repair, 21 and trace elements are essential for maintaining skin health. 22 Additionally, vitamins support skin hydration, antioxidant defense, and repair, 23 while organic acids help regulate skin pH and promote metabolic processes. 24 These components work synergistically, enabling ginseng root extract to deliver a wide range of skincare benefits, including anti-aging, hydration, anti-inflammatory effects, skin brightening, and the repair of damaged skin. 25 As a result, ginseng root extract is widely incorporated into various cosmetic products, such as creams, serums, masks, eye creams, and sunscreens, to provide comprehensive skincare effects.26,27 With ongoing research, the applications of ginseng root extract in cosmetics are expected to become even more widespread and diverse, further enhancing its role as a key ingredient in skincare.
Cellular-level experiments are generally less influenced by environmental factors, making them well-suited for studying various molecular mechanisms. However, due to the absence of transdermal absorption and interactions between different cell types, these experiments cannot fully represent a substance's efficacy in skincare. 28 3D skin models, as an advanced in vitro testing platform, can closely replicate the structure and function of human skin, offering a reliable foundation for evaluating the efficacy and safety of cosmetic products. Tissue-level experiments, which build upon cellular studies, are less affected by external factors and feature stratified structures formed by cells at varying stages of differentiation. 29 These models can partially simulate transdermal absorption, serving as a critical method for validating the skincare mechanisms and effects of substances following cellular studies and prior to clinical trials.30,31 Additionally, 3D skin models provide valuable data to support the screening of new ingredients, formulation optimization, and safety evaluations. Clinical trials, on the other hand, provide the most accurate representation of a substance's effects on human skin. However, they are influenced by external variables such as individual differences, environmental conditions, and lifestyle factors. Moreover, clinical trials are costly and time-intensive, which can make it challenging to elucidate the underlying molecular mechanisms of efficacy.
Despite extensive research by scholars and experts on the effects and mechanisms of ginseng root extract on skin cells such as keratinocytes, fibroblasts, and melanocytes, most studies have been limited to cell line models. Currently, there is a relative scarcity of research validating these mechanisms at the tissue level. 32 Thus, it is essential to conduct in-depth investigations into the efficacy mechanisms of ginseng root extract at the primary cell level and to explore how these mechanisms are transmitted and regulated at the tissue level.
Materials and Methods
Preparation of PGRE
In this experiment, all ginseng used was more than four years old and harvested in the autumn of 2023 from Anseong-si, Gyeonggi-do, South Korea. The ginseng roots were cleaned using a surface washing system specifically designed for ginseng. After removing excess water, the roots were rolled for 30 min to disrupt the cell walls and then sliced into 1 cm pieces using a slicer (SM-3, Samjin Machinery, Gimpo-si, Gyeonggi-do, South Korea). For anaerobic aging, the sliced ginseng was placed in low-density polyethylene film and subjected to facultative aging for 21 days at 50 °C in an industrial drying oven (SHIDO-2250FG, Samheung Energy Co., Ltd Sejong-si, South Korea), followed by drying for 48 h at 60 °C to remove all moisture. The PGRE was prepared by soaking the slices in a 60% ethanol solution (product number: 1.00980, Sigma, St. Louis, USA) at 50 °C for 5 h. Subsequently, the extract was vacuum-filtered using No. 1 filter paper (11 μm, 1002-047, Whatman, Maidstone, Kent, UK) to remove solid precipitates. It was then concentrated and purified using a rotary evaporator (EXTREVA ASE, Thermo Fisher Scientific, Waltham, MA, USA) at 50 °C until dry. Finally, the resulting ginseng root extract (brown powder) was dissolved in dimethyl sulfoxide (DMSO), diluted in fresh culture media, and added to the cells for experimental use. 33 For the PGRE, in addition to the standard ginsenosides, we only controlled the content of Gypenoside XVII (GP-17), requiring that the final extract contain ≥3.2% GP-17, as quantified using HPLC (High Performance Liquid Chromatography) methods.
Cell Viability Assay
The human primary keratinocyte cells used in this experiment were purchased from Shaanxi Biocell Co., Ltd (Shaanxi, China), and all primary cells were within five passages. The cells were thawed and cultured until they reached approximately 60% confluence. They were then seeded into 96-well plates (93599, Corning, NY, USA) and incubated over-night in a 37 °C incubator with 5% CO2 (150I, Thermo Fisher Scientific, Waltham, MA, USA). The experiment included a zeroing group, a solvent control group, a positive control group, and a sample group. For the sample group, eight concentration gradients of the PGRE working solutions were prepared, with three replicate wells for each concentration (Table 1).
Concentration Gradients of PGRE.

Abbreviation: PGRE, Panax ginseng root extract.
Once the cells in the 96-well plates reached 50%–60% confluence, the treatments were administered. The zeroing group, which served as a reference for background readings, did not receive any cell seeding and was provided only with 200 μL of culture medium in each well. The solvent control group received 200 μL of culture medium in each well, serving as a baseline for comparison. The positive control group was treated with 200 μL of culture medium containing 10% DMSO (D4540-1L, Sigma, St. Louis, USA) to evaluate the response under known conditions. The sample group was administered 200 μL of culture medium containing the appropriate concentration of the test sample for efficacy assessment. After applying the treatments, the 96-well plates were placed back into the CO2 incubator (37 °C, 5% CO2) and incubated for 24 h.
After 24 h of incubation, the supernatant was removed, and MTT working solution (0.5 mg/mL, M5655, Sigma, St. Louis, USA) was added to each well. The plates were incubated at 37 °C in the dark for 4 h. After incubation, the supernatant was discarded, and 150 µL of DMSO was added to each well to dissolve the formazan crystals. The optical density (OD) values were measured at 490 nm using a microplate reader (Epoch, BioTek, Vermont, USA). Subsequently, the supernatant was removed again, and images were captured using an inverted microscope (CKX53, Olympus, Tokyo, Japan).
The relative cell viability (%) was calculated using the following formula: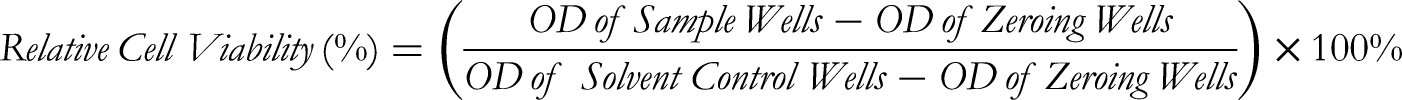
Anti-carbonylation Test Using Reactive Oxygen species (ROS)
This experiment follows the standard protocol outlined in T/SHRH 032-2020 Anti-Wrinkle and Firming Lifting Efficacy Test of Cosmetics – In Vitro Test Method for Reactive Oxygen Species (ROS) Inhibition with Keratinocytes. 34 All primary cells were within five passages. The procedure involved detecting changes in fluorescence intensity of the intracellular fluorescent dye DCFH-DA (H2DCFDA, 2',7'-Dichlorodihydrofluorescein diacetate) and measuring the ROS content in each test group to calculate the ROS inhibition rate of the sample groups.
Keratinocytes were seeded into a 6-well plate at a density of 2 mL per well (approximately 1.0-1.3 × 105 cells/mL) and incubated at 37 °C with 5% CO2 for 24 h. Once the cells reached 80%–90% confluence, the culture medium in each well was replaced with 2 mL of the following: blank control group (complete Dulbecco's Modified Eagle Medium, DMEM, C11995500BT, Gibco, NY, USA), positive control group (10 μM 4-Hydroxynonenal, 4-HNE, H914143 1 mg, Macklin, Shanghai, China), and sample group (50 ppm of raw PGRE + 10 μM 4-HNE). The cells were incubated at 37 °C with 5% CO2 for 24 h. After incubation, the supernatant was removed, and the wells were washed twice with phosphate-buffered saline (PBS). Subsequently, 2 mL of DCFH-DA (S0033M, Beyotim, Shanghai, China) and Hoechst 33342 (H6110-25 mg, Macklin, Shanghai, China) were added to each well and incubated for 20 min in the dark. The wells were then washed three times with PBS. The samples were observed under a fluorescence microscope (Eclipse Ni-U, Nikon, Tokyo, Japan) with excitation and emission wavelengths of 494 nm and 520 nm, respectively. Images were captured, and the fluorescence intensity of the images was analyzed using ImageJ 1.8.0 software (NIH, Bethesda, MD, USA). Three random fields were photographed for each group.
IL-1α and PGE2 Test Using Enzyme Linked Immunosorbent Assay (ELISA)
In a 6-well plate (3516, Corning, NY, USA), 0.9 mL of EpiGrowth culture medium (Biocell, Guangdong, China) was added to each well, and epidermal skin models (Biocell, Guangdong, China) were carefully transferred into the wells according to the test groups outlined in Table 2. For the negative control (NC), positive controls (PC1, PC2), and test groups, 25 μL of 0.1% sodium lauryl sulfate (SLS) solution (L6026, Sigma, St. Louis, USA) was applied to the surface of the skin models and incubated for 30 min. Following incubation, dexamethasone solutions (100129, NFDC, Beijing, China) at designated concentrations were applied to the PC1 group, while the test samples were evenly distributed on the surfaces of the skin models in the test groups. The models were then incubated in a CO2 incubator (37 °C, 5% CO2) for 24 h. After the 24-h incubation, the culture medium from each epidermal skin model was collected into centrifuge tubes (MCT150-C, AXGEN, California, USA). The collected samples designated for ELISA analysis were stored in a −80 °C freezer (MDF-U53V, SANYO, Tokyo, Japan) until further use. Detection and analysis were performed using ELISA kits, following the manufacturer's instructions. The IL-1α ELISA kit was sourced from Abcam (ab100560, Abcam, Cambridge, United Kingdom), and the PGE2 ELISA kit was obtained from ENZO (ADI-901001, ENZO, NY, USA).
Test Grouping and Conditions.

Positive control 1 is the positive control for assessing tissue morphology, while positive control 2 is the positive control for detecting IL-1α, PGE2, and NF-κB.
Abbreviations: PGRE, Panax ginseng root extract; SLS, sodium lauryl sulfate.
Epidermal Tissue Morphology Examination
The treatment and stimulation conditions for the skin models followed the procedures described in Section 2.4. After incubation, WY14643 solutions (C7081, Sigma, St. Louis, USA) at the specified concentrations were applied to the PC2 group, while the test samples were evenly distributed on the surfaces of the models in the test groups. At the conclusion of the incubation period, residual test substances on the model surfaces were rinsed off with sterile PBS (P1010, Solarbio, Beijing, China). Any remaining liquid, both inside and outside the models, was carefully removed using sterile swabs (BK02, HYNAUT, Qingdao, Shandong, China). The models were then fixed with 4% paraformal-dehyde (BL539A, Biosharp, Hefei, Anhui, China) for 24 h. Following fixation, the samples underwent hematoxylin-eosin (HE) staining for microscopic examination. Images were captured and analyzed using a microscope.
NF-κB Test Using Immunofluorescence (IF)
The treatment and stimulation conditions for the skin models were consistent with the procedures outlined in Section 2.4. After fixing the skin model with 4% paraformaldehyde (BL539A, Biosharp, Hefei, Anhui, China), the tissues were embedded, sectioned, and processed as follows: Paraffin sections were baked on a slide warmer at 70 °C for 4 h, followed by dewaxing in xylene (10023418, SINOPHARM, Shanghai, China) for two consecutive 10-min immersions. Rehydration was carried out sequentially in absolute ethanol, 95% ethanol, and 75% ethanol for 5 min each, with subsequent washing in PBS (P1010, Solarbio, Beijing, China) three times for 5 min each. Antigen retrieval was performed in 0.01M sodium citrate solution under high pressure, followed by cooling and washing three times with PBS. Endogenous peroxidase activity was blocked with 3% H2O2 (88597, Sigma, St. Louis, US) for 30 min at room temperature, then washed three times with PBS. Serum blocking was performed using serum from the same species as the secondary antibody, incubating at 37 °C for 1 h. Primary antibody incubation was conducted overnight at 4 °C, followed by washing three times with PBS. Secondary antibody incubation was performed at room temperature for 1 h, with subsequent washing three times with PBS. Nuclear staining was conducted by applying 100 μL of Hoechst33342 (H6110-25 mg, Macklin, Shanghai, China) working solution to each section, incubating at room temperature for 5 min, and washing three times with PBS. The sections were then blotted, mounted with anti-fade reagent, and imaged under a fluorescence microscope (DM2500, Leica, Wetzlar, Germany) at 20× magnification within 24 h.
Statistical Analyses
All values are presented as the mean ± standard deviation (SD), and the coefficient of variation (CV) was also calculated. These data were obtained from at least three independent replicate experiments. Statistical analysis was conducted using the t-test, with a p-value of < .05 considered statistically significant. All analyses were performed using SPSS version 27.0 (IBM Corporation, Armonk, NY, USA).
Results
Cytotoxicity Assessment of PGRE
Cell viability refers to the proportion of healthy cells within a tissue. High toxicity leads to a reduction in cell viability. This experiment aimed to determine the concentration threshold of the test substance that minimally impacts cells. Based on the MTT (Methyl Thiazolyl Tetrazolium) assay and morphological results (Figure 1), it was observed that when PGRE was applied to keratinocytes, the cell viabilities at concentrations of 25 μg/mL, 50 μg/mL, and 100 μg/mL were 90.05%, 85.73%, and 78.75%, respectively. At a concentration of 50 μg/mL, the extract did not exhibit significant cytotoxicity (cell viability >80%). However, at concentrations exceeding 200 μg/mL, most cells were unable to survive. Considering the results of the cell viability tests and the concentration range likely to be used in future clinical formulations, a concentration of 50 μg/mL was selected for subsequent experiments.

Cell Survival Rate of PGRE at various Concentrations. Values are Presented as the Mean (n = 3). Different Lowercase Letters (a–e) Indicate Significant Differences (p < .05) According to Duncan's Multiple Range Test. Abbreviation: PGRE, Panax ginseng root extract.
Additionally, the inter-group coefficient of variation (CV) for cell viability was analyzed at each concentration. The inter-group CVs for the control group and the groups treated with PGRE at concentrations of 3.125, 6.25, 12.5, 25, 50, 100, 200, and 400 μg/mL were 3.6%, 5.7%, 3.2%, 4.0%, 4.1%, 5.9%, 5.9%, 23.0%, and 19.1%, respectively. The inter-group CV for the 10% DMSO group was 78.8%. These results indicate that at a concentration of 100 μg/mL, the inter-group CV remained within a reasonable range, supporting the validity of the experimental results.
ROS Suppression Measured by DCFH-DA Assay
Figure 2 presents the results of parallel experiments conducted with a 50 μg/mL PGRE sample, a blank control, and a model control, along with an analysis of significant differences in the outcomes. The experimental findings reveal that the 50 μg/mL PGRE achieved a ROS inhibition rate of 51.6%. Compared to the positive control group, it significantly reduced the production of protein carbonylation products, demonstrating notable anti-carbonylation efficacy.

DCFH-DA Fluorescence Images Show the Inhibitory Effect of PGRE on Reactive Oxygen Species (ROS) in Keratinocytes. Green Fluorescence Represents ROS Levels, with Stronger Fluorescence Indicating Higher ROS Levels. Results of ROS Level and the ROS Inhibition Rate. ROS Level (%) = (Fluorescence Value of the Sample Test Group ÷ Fluorescence Value of the Model Control Group) × 100%; ROS Inhibition Rate (%) = [(Model Control-Sample) ÷ (Model Control-Blank Control)] × 100%. When Performing Statistical Analysis Using the t-test Method, Significance Compared to the BC Group is Indicated by # (p < .05). Abbreviation: PGRE, Panax ginseng root extract.
Figure 2 provides a visual comparison of fluorescence staining, where stronger green fluorescence indicates more severe carbonylation. As shown, the positive control group exhibits intense green fluorescence due to severe carbonylation, signifying a high level of protein carbonylation products. The coefficient of variation (CV) for ROS levels among the blank control group, positive control group, and experimental group were 12.1%, 7.2%, and 8.1%, respectively, all of which fall within a reasonable range, indicating the reliability of the experimental results.
Inhibition of IL-1α and PGE2 Secretion
The inhibitory effects of the sample on IL-1α and PGE2 are presented in Figure 3. At a concentration of 50 μg/mL, the PGRE significantly inhibited the inflammatory factor IL-1α, with an average inhibition rate of 34.17%. It also showed a notable inhibitory effect on the inflammatory mediator PGE2, achieving an average inhibition rate of 20.70%. Under stimulated conditions, the negative control group exhibited a significant increase in IL-1α and PGE2 levels compared to the blank control, confirming the effectiveness of the stimulation. The coefficients of variation (CVs) for IL-1α in the blank control group, negative control group, positive control group, and experimental group were 11.1%, 6.5%, 4.9%, and 3.9%, respectively, while the CVs for PGE2 were 2.9%, 3.0%, 1.4%, and 1.0%, respectively. All CV values were within a reasonable range, supporting the reliability of the experimental data.

Detection Results of IL-1α and PGE2 Levels. When Performing Statistical Analysis Using the t-Test Method, Significance Compared to the BC Group is Indicated by # (p < .05); Significance Compared to the NC Group is Indicated by * (p < .05).
Enhancement of Viable Epidermal Thickness via HE Staining
The measurement of viable epidermal thickness was conducted to evaluate the potential skin-damaging effects of PGRE and validate the safety profile of the tested concentration. A reduction in epidermal thickness typically indicates compromised barrier function or cellular damage, whereas maintenance or enhancement of thickness suggests tissue integrity and reparative capacity. This approach aligns with the study design to comprehensively assess both efficacy and safety.
Figure 4 shows the epidermal thickness observed in sections following HE staining, and the effect of the samples on the thickness of the viable epidermal layer. In Figure 4, the light pink area represents the epidermal layer, which reflects the integrity and strength of the skin barrier. The negative control group shows that under stimulation, epidermal thickness is significantly reduced, confirming the effectiveness of the stimulation conditions. Treatment with 50 μg/mL PGRE resulted in a significant increase in the thickness of the viable epidermal layer, achieving an enhancement rate of 22.91%, which surpasses the 14.61% enhancement rate observed with the positive control group treated with 50 μM WY14643. Notably, the absence of epidermal thinning in PGRE-treated groups, coupled with significant thickness recovery compared to the damaged negative control, confirms that PGRE does not induce skin injury. These results validate the rationality of the study design, which integrates epidermal thickness as a critical marker for both safety and barrier repair assessment.

HE Staining Images Show the Improvement Effect of PGRE on the Damage to the Viable Epidermal Layer. The Light Pink Areas Represent the Viable Epidermal Layer, and the Thicker These Areas are, the Better the Repair Effect. Immunofluorescence Staining Images Showing the Inhibition of Nuclear Transcription Factor NF-κB by PGRE. The More and Brighter the Green Fluorescence, the Higher the Amount of NF-κB. Results of the NF-κB Immunofluorescence Analysis. Integrated Optical Density (IOD), Which Reflects the Amount of NF-κB. When Performing Statistical Analysis Using the t-test Method, Significance Compared to the BC Group is Indicated by # (p < .05); Significance Compared to the NC Group is Indicated by * (p < .05). Abbreviation: PGRE, Panax ginseng root extract.
The coefficients of variation (CVs) for the viable epidermal cell layer in the blank control, negative control, positive control, and experimental groups were 1.1%, 3.0%, 2.0%, and 4.7%, respectively, all of which are within a reasonable range, ensuring the reliability of the data.
NF-κB Activation Suppression via Immunofluorescence
The effect of the samples on NF-κB levels is presented in Figure 4. Fluorescence staining was used to assess the inflammatory factor NF-κB, with higher fluorescence intensity indicating greater amounts of NF-κB. Figure 4 demonstrates that the stimulatory conditions led to a significant increase in fluorescence intensity in the negative control group, confirming the efficacy of the stimulation. Compared to the negative control group, treatment with 50 μg/mL of PGRE significantly reduced NF-κB levels, achieving an inhibition rate of 43.65%. The positive control group, treated with 100 μg/mL of dexamethasone, showed an even higher inhibition rate of 57.87%. The coefficient of variation (CV) for NF-κB levels was 20% for the BC group, 2.5% for the NC group, 10.8% for the PC group, and 9.9% for the experimental group. Except for the BC group, all CV values fall within an acceptable range. The high CV observed in the BC group can be attributed to the normalization process, where the Integrated Optical Density value of the BC group was set to 1 for comparison with other groups. The small mean value in the BC group amplified the CV, pushing it beyond the typical range. T-test revealed significant differences between the BC group and all other groups, validating the experimental results.
Discussion
External stimuli, including ultraviolet radiation, chemical irritants, and allergens, induce oxidative stress by generating excessive ROS, which disrupt cellular redox balance and promote protein carbonylation—a hallmark of skin aging and barrier dysfunction associated with dermal yellowing rather than melanin-mediated pigmentation. 41 In this study, PGRE (50 μg/mL), derived from four-year-old white ginseng roots harvested in Anseong-si, South Korea, demonstrated potent ROS scavenging activity (79.9% inhibition), effectively mitigating 4-HNE-induced protein carbonylation. The extract's anti-carbonylation efficacy aligns with its high content of gypenoside XVII (GP-17, ≥ 3.2%), a bioactive ginsenoside known for its antioxidant properties. 14 ROS overproduction further activates cyclooxygenase-2 (COX-2), a key enzyme in the prostaglandin E2 (PGE2) synthesis pathway. 42 Elevated PGE2 binds to EP receptors, activating protein kinase A (PKA) via cAMP signaling, which phosphorylates transient receptor potential vanilloid 1 (TRPV1).43–45 While Jang et al 45 demonstrated that red ginseng inhibits histamine-dependent itch via the H1R/TRPV1 pathway, our study focuses on PGRE's broader anti-inflammatory effects, independent of histaminergic pathways. Instead, our data suggest that SLS-induced irritation amplifies PGE2-mediated sensitization, driving calcium influx and inflammatory responses.46,47
SLS, a well-characterized skin irritant (not a sensitizer), activates Langerhans cells (LCs) in the epidermis. These dendritic cells internalize antigens, migrate to lymph nodes, and initiate immune responses by presenting antigens to T cells. 48 Activated LCs release cytokines (eg, IL-1α) and chemokines, amplifying inflammatory cascades. Our findings suggest that ginseng root extract, enriched with ginsenosides like Rb1 and Rg3, may suppress LC activation by reducing PGE2, a critical mediator of dendritic cell maturation and migration.46,47 Notably, the extract's ability to inhibit IL-1α (34.17%) and PGE2 (20.70%) highlights its dual role in mitigating both innate and adaptive immune responses.
IL-1α, a pro-inflammatory cytokine upregulated in our SLS-stimulated model, binds to its receptor to activate the nuclear factor kappa B (NF-κB) pathway.49,50 Activated NF-κB translocates to the nucleus, upregulating COX-2 expression and PGE2 synthesis. 51 This creates a self-amplifying loop, as NF-κB also exerts positive feedback on IL-1α transcription, further driving inflammation. 52 Our data reveal that ginseng root extract significantly suppressed NF-κB activation (43.65% inhibition), disrupting this vicious cycle. We propose that NF-κB serves as the central node in PGRE's multi-target efficacy, particularly through its regulation of inflammatory feedback loops. The suppression of NF-κB activation (43.65% inhibition) not only interrupts the hierarchical reinforcement of inflammation but also disrupts the self-amplifying cycle between IL-1α and COX-2/PGE2 pathways. This hypothesis is further supported by the observed reduction in IL-1α (34.17%) and PGE2 (20.70%) levels, which are downstream targets of NF-κB. We speculate that NF-κB's dual role in both initiating and sustaining inflammatory cascades makes it a critical mediator of PGRE's anti-inflammatory effects. This hypothesis is supported by prior studies showing that ginsenosides like Rd inhibit NF-κB nuclear translocation via SIRT1/AMPK (AMP-activated protein kinase) pathway activation, 53 while Rb1 suppresses TRPV1-mediated calcium influx. 54 However, this remains a preliminary conjecture, and future studies are required to elucidate the precise molecular interactions governing NF-κB's feedback mechanisms in the context of ginsenoside activity.
The anti-inflammatory and antioxidant effects of ginseng root extract are attributed to its bioactive ginsenosides, which target multiple nodes in the ROS/COX-2/PGE2/NF-κB axis. Ginsenoside Rb1 enhances mitochondrial antioxidant defenses to reduce ROS, 55 while Rh2 suppresses COX-2 expression and PGE2 synthesis with potency comparable to NSAIDs. 56 Rg3 blocks TRPV1 activation, attenuating calcium influx and neurogenic inflammation, 45 and Rd inhibits NF-κB nuclear translocation via SIRT1/AMPK pathway activation. 53 These mechanisms align with prior cellular studies,54,57 yet our use of the EpiKutis® epidermal model bridges a critical gap by validating transdermal efficacy. The extract increased viable epidermal thickness by 22.91%, confirming its tissue-level reparative effects—a finding unattainable in conventional monolayer cell cultures.28–31 For instance, while earlier studies demonstrated ginsenoside-mediated barrier repair in HaCaT cells, 58 our 3D model recapitulated the stratified epidermal architecture, enabling direct observation of SLS-induced damage reversal. This integration of 3D models underscores their value in cosmetic research, as they not only replicate human skin physiology but also validate transdermal delivery and tissue-level responses, overcoming the limitations of traditional 2D systems.29,30
Beyond its anti-inflammatory properties, ginseng root extract may hold untapped potential in anti-inflammatory and hyperpigmentation therapies. Ginsenosides like Rb2 inhibit melanogenesis by suppressing PGE2-mediated tyrosinase activity,59,60 while collagen synthesis may be enhanced through TGF-β signaling, as suggested by studies showing ginsenoside-induced filaggrin upregulation in keratinocytes. 58 Future studies should explore these pathways to expand the extract's applications in cosmetic formulations, particularly for addressing photoaging and uneven skin tone.
The multi-target efficacy of ginseng root extract is further supported by its non-saponin components, which synergistically contribute to its biological functions alongside the core ginsenosides (Table 3). For instance, polysaccharides regulate the skin microenvironment by enhancing immune responses, 35 volatile oils may indirectly support skin health through digestive system modulation, 36 while amino acids and peptides collaboratively promote cellular metabolism and repair.37,38 Additionally, flavonoids and phenolic acids reinforce the extract's antioxidant network, with flavonoids exhibiting anti-inflammatory properties 39 and phenolic acids strengthening antioxidant defenses via free radical scavenging. 40 Together with ginsenosides, these constituents form a complex bioactive network that multi-dimensionally supports the extract's comprehensive effects in antioxidant activity, anti-inflammatory responses, and barrier restoration.
The Main Components of ginseng Root Extract (Aside from Ginsenosides).

This study reveals a critical bidirectional interplay between inflammation and oxidative stress, centered on a self-reinforcing positive feedback loop coupling NF-κB signaling and ROS. Exogenous stimuli (eg, TNF-α) and endogenous ROS oxidatively modify critical residues in the IKK complex while inhibiting phosphatase activity, thereby promoting IκB degradation and nuclear translocation of NF-κB. 61 Significantly, activated NF-κB transcriptionally upregulates pro-inflammatory cytokines (TNF-α, IL-1β), inducible nitric oxide synthase, and NADPH oxidase (NOX) subunits, synergistically amplifying nitric oxide (NO) and superoxide (O2−) production. These species rapidly react to form peroxynitrite (ONOO−), a potent oxidant. 62 Newly generated ROS further activate the NF-κB pathway, while NF-κB-driven gene expression perpetuates ROS generation, establishing a vicious NF-κB/ROS circuit. Taken together, our data underscore PGRE's multi-target efficacy, with NF-κB regulation emerging as a central mechanism. By concurrently addressing oxidative stress (via ROS scavenging), inflammation (via NF-κB/IL-1α/PGE2 suppression), and barrier repair (via epidermal thickening), PGRE demonstrates comprehensive skincare benefits. This integrated approach aligns with the growing demand for natural ingredients that target multiple pathways in skin aging and inflammation.
Conclusions
This study demonstrates that PGRE at 50 μg/mL effectively mitigates oxidative stress, inflammation, and barrier dysfunction through integrated cellular and tissue-level mechanisms. In keratinocytes, PGRE reduced 4-HNE-induced ROS by 51.6% and suppressed inflammatory mediators (IL-1α: 34.17%; PGE2: 20.70%) and NF-κB activation (43.65%). Tissue analysis revealed a 22.91% enhancement in viable epidermal thickness, outperforming the positive control. Central to these effects is PGRE's modulation of NF-κB signaling, which disrupts inflammatory feedback loops and amplifies its multi-target efficacy. While these findings highlight PGRE's potential as a natural skincare ingredient, limitations include the single concentration tested and lack of in vivo validation. Future research should focus on dose-dependent responses, clinical trials, and mechanistic studies to unravel NF-κB's regulatory role in PGRE's anti-inflammatory and barrier-repair activities. This work provides a foundation for developing ginseng-based cosmetics targeting oxidative stress, inflammation, and skin aging.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251364582 - Supplemental material for Panax Ginseng Root Extract Exhibits Antioxidant and Anti-Inflammatory Properties by Diminishing Oxidative Stress Levels and Modulating the NF-κB Signaling Pathway at Both the Cellular and Tissue Levels in the Skin
Supplemental material, sj-docx-1-npx-10.1177_1934578X251364582 for Panax Ginseng Root Extract Exhibits Antioxidant and Anti-Inflammatory Properties by Diminishing Oxidative Stress Levels and Modulating the NF-κB Signaling Pathway at Both the Cellular and Tissue Levels in the Skin by Bo-Zheng Zhang, Hao-Yang Yu, Chao-Fan Tian, Min Yu, Yun-Ha Lee and Zhi Xiong in Natural Product Communications
Footnotes
Acknowledgements
The authors thank AMOREPACIFIC R&I Center for financial and sample support.
Authors Contributions
Conceptualization: B.-Z.Z.; Methodology: B.-Z.Z. and H.-Y.Y.; Software: C.-F.T.; Validation: B.-Z.Z., C.-F.T., and H.-Y.Y.; Formal analysis: B.-Z.Z.; Investigation: B.-Z.Z. and H.-Y.Y.; Resources: Z.X.; Data curation: C.-F.T.; Writing - original draft preparation: B.-Z.Z.; Writing - review and editing: B.-Z.Z., C.-F.T., and H.-Y.Y.; Visualization: C.-F.T.; Supervision: H.-Y.Y. and Z.X.; Project administration: Z.X.; Funding acquisition: Y.-H.L., Z.X., and M.Y.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by AMOREPACIFIC R&I Center. The APC was funded by Amorepacific (Shanghai) D&I Center Co., Ld
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
