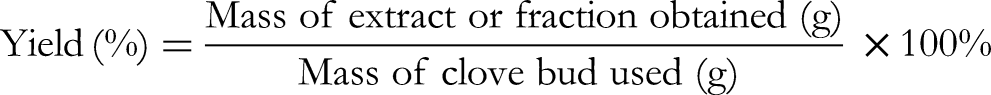Abstract
Keywords
Introduction
The global rise of antibiotic-resistant pathogens poses a severe public health challenge, necessitating alternative antimicrobial strategies.1–3 It is now widely recognized that misuse of antibiotics has driven bacteria to evolve resistance, and new infection control measures are urgently needed.4,5 In addition, the side effects of many synthetic antibacterial agents further highlight the need for safer and more effective alternatives.6–8 There is also growing interest in replacing chemical preservatives in food with natural alternatives, as several synthetic additives have been linked to potential health risks. 9 In this context, plant-derived phytochemicals have attracted significant attention due to their antimicrobial activity and potential role as resistance modulators. 10 Many spices and herbs are rich in bioactive compounds such as phenolics, alkaloids, and terpenes, which have demonstrated efficacy against antibiotic-resistant bacterial strains. 11 Among these, Syzygium aromaticum (commonly known as clove) has been widely recognized for its potent antibacterial and antifungal properties against both foodborne and clinical pathogens.5,10
Clove has a long history of use in traditional medicine and food preservation. Its essential oil contains up to 85%–90% eugenol, a phenolic compound recognized as safe by the WHO, along with synergistic minor components.12,13 These components are responsible for its broad-spectrum bioactivities, which have made clove oil useful in dental care, pharmaceuticals, and food applications.13,14 Importantly, unlike many plant-based compounds that are either volatile or poorly soluble, clove contains both volatile (eg, essential oils) and non-volatile (eg, polyphenols, glycosides) constituents, which contribute to its potent antimicrobial efficacy.5,14
To fully explore the antibacterial potential of clove, it is essential to consider how extraction methods influence the composition and activity of the resulting extracts. The choice of extraction technique can significantly affect the yield, diversity, and bioactivity of phytochemicals obtained from plant materials.15,16 Furthermore, limited research has been conducted on the bioactivity of specific solvent fractions, which may contain overlooked or synergistic antimicrobial agents.
In the present study, we investigated the antibacterial activity of S. aromaticum by employing two different extraction techniques:—simple maceration and hot Soxhlet extraction—using methanol as the solvent. The resulting crude extracts were analyzed for phytochemical content (Table 1) and evaluated for antimicrobial activity (Table 2). In addition, the Soxhlet extract was fractionated into different solvent-based fractions, which were subsequently tested for antibacterial efficacy (Figure 1e to h, Table 3). Clove essential oil was also included in the analysis (Table 4). This experimental design was chosen to uncover how extraction conditions affect yields of active compounds and to identify which fraction(s) confer the strongest antibacterial effects. Ultimately, these results will help understand which fraction in clove is most pharmacologically relevant and how the choice of extraction method influences bioactivity.

Workflow for Extraction of Clove Bud (Steps a-d) and Subsequent Fractionation of the Soxhlet Extract (Steps e-h): (a) Maceration Extraction; (b) Soxhlet Extraction; (c) Concentration of Crude Extracts and Fractions by Rotary Evaporation; (d) Essential-Oil Extraction Via Hydrodistillation; (e) n-Hexane Fraction (Upper Layer); (f) Dichloromethane Fraction (Lower Layer); (g) Ethyl Acetate Fraction (Lower Layer); (h) n-Butanol Fraction (Upper Layer).
Materials and Methods
Plant Material, Preparation, and Identification
Dried clove buds were commercially sourced from a certified supplier in Kombolcha City, Ethiopia. No permits were required, as the material was cultivated (non-wild) and not listed as endangered under Ethiopian law. Buds were selected for uniformity based on physical characterization (size, intact structure, absence of defects), then pulverized using a coffee mill and sieved to a particle size ≤1 mm. Botanical authentication was conducted by Wollo University Herbarium (voucher Sol 003/2024), with the specimen archived for verification.
Extraction Methods
Maceration Extraction
Clove bud powder (150.0 g) was placed in the conical flask. 1.5 L of methanol was then added and left to stand for 7 days. The mixture was swirled occasionally after covering the flask with foil, as shown in Figure 1a. After 7 days, the liquid was decanted, and Marc was pressed. The squeezed liquid was mixed with the supernatant. The supernatant was filtered with filter paper using gravity filtration and concentrated using a rotary evaporator at 41 °C. Concentrated extracts were transferred into vials and stored in a refrigerator at 4 °C.
Soxhlet Extraction
A powdered clove sample (30.0 g) was placed in a thimble inside a Soxhlet apparatus connected to a round-bottom flask containing 300 mL of methanol, as shown in Figure 1b. The solvent was heated to reflux for 6 h, allowing its vapors to condense and percolate through the sample. As the thimble filled, the solvent carrying the extracted phytochemicals returned to the flask through the siphon mechanism. This cycle repeated seven times. The extract was then concentrated via a rotary evaporator at 41 °C (Figure 1c). The entire process was repeated five times to extract from a total of 150.0 g of clove buds. The concentrated extracts were stored in vials at 4 °C.
Essential Oil Extraction
Hydrodistillation was performed at atmospheric pressure using a heat mantle and a Clevenger-style apparatus (Figure 1d). Dried clove buds (150 g) were placed in 400 mL of water within a double-neck round-bottom flask—one neck for gradual water addition, and one for vapor release—and heated to the local boiling point of approximately 95 °C (Kombolcha, elevation ∼1840 m) for 6 h. Throughout the extraction, a total of 1.5 L of water was incrementally added to sustain gentle boiling. The vapor was condensed through a borosilicate glass condenser, and the resulting distillate (1250 mL) was collected in a volumetric flask. After allowing the mixture to stand in a separatory funnel for 8 h, the essential oil layer was separated by volume measurement and stored at 4 °C.
Solvent Fractionation
The crude extract (25.0 g) was mixed with 150.0 mL of distilled water and stirred for 5 min. Then, it was transferred into a separatory funnel, and n-Hexane (125.0 mL) was added. The mixture was vigorously shaken and allowed to stand for 24 h (Figure 1e). The upper n-Hexane layer was separated. Similarly, the aqueous layer was sequentially fractionated in equal volumes of dichloromethane, ethyl acetate, and n-butanol, as shown in Figure 1(f, g, h)
Phytochemical Screening Tests
The following standard methods were employed to identify phytochemicals in the extracts, as shown in Figure 2.17–19

Phytochemical Screening of Soxhlet-Derived Clove Bud Extract Fractions:(i) n-Hexane Fraction; (j) Dichloromethane Fraction; (k) Ethyl Acetate Fraction; and (l) n-Butanol Fraction.
After phytochemical screening, assay validation was conducted to ensure specificity and sensitivity. Samples were tested alongside authentic standards (caffeine for alkaloids; quercetin for flavonoids) to confirm reagent performance. Methanol and water blanks were included to rule out false positives that might arise from impurities, or contamination from glassware. All tests were performed in triplicate to ensure reproducibility.
In Vitro Antibacterial Activity Test
Media Preparation
38 mg of Muller-Hinton Agar medium (MHA) powder was dissolved in 1 L of distilled water. The mixture was heated until completely dissolved, yielding a clear solution. The flask was tightly sealed and autoclaved at 121 °C for 15 min to ensure sterilization. After cooling to room temperature, the sterile MHA medium was poured into Petri dishes under a laminar flow hood to achieve a uniform depth of 4 mm. The plates were stored in a sterile refrigerator until inoculation. 11
Preparation of Working Solutions and Justification of Concentrations
Crude extracts and solvent fractions were dissolved in DMSO to create 0.5 g/mL stock solutions, followed by dilution to working concentrations of 0.5-10 mg/mL. Final doses applied in the assay ranged from 25 to 500 µg per well. These concentrations were guided by preliminary screening and are consistent with previously reported MIC values for S. aromaticum extracts and essential oil, which typically do not exceed 500 µg for clinically relevant bacteria.13,20,21 This range enabled clear assessment of concentration-dependent activity. A 0.85% saline solution was also prepared for bacterial suspension.
Inhibition Zone Determination
Fresh bacterial cultures were suspended in 5 mL of sterile 0.85% saline and adjusted to a 0.5 McFarland standard. The bacterial strains used—Staphylococcus aureus, Enterococcus faecalis (Gram-positive), and Escherichia coli, Klebsiella pneumoniae (Gram-negative)—were selected due to their clinical importance, frequent resistance profiles, and relevance to both hospital-acquired and foodborne infections.22,23 These widely studied pathogens also provide comparability with previous phytochemical antibacterial studies.24,25
Sterile cotton swabs were used to evenly streak MHA plates. Wells (6 mm) were aseptically created using a cork borer, and extract solutions (50 µL) or essential oils (10-50 µL) were added. 26 DMSO (50 µL) was the negative control, while ciprofloxacin (5 µg) was the positive control. Plates were left at room temperature for 2 h for diffusion before incubation at 37 °C for 24 h. 27 All tests were conducted in triplicate, and inhibition zones were measured in millimeters.
Statistical Analysis
The percentage yield of crude extracts and their solvent fractions was calculated as:
The mass of extract per well in antibacterial assays (µg) was determined by: m = C × V, where C is concentration (µg/µL) and V is volume added (µL).
All antibacterial assays—whether comparing Soxhlet versus maceration extracts, evaluating Soxhlet solvent-partitioned fractions, or testing essential oil volumes—followed an identical core workflow: triplicate inhibition-zone measurements were recorded and expressed as mean ± SEM; data were organized in GraphPad Prism 10.4.2 as grouped tables with two fixed factors; ordinary two-way ANOVAs assessed the main effects of treatment (extraction technique, fraction×concentration, or oil volume), bacterial strain, and their interaction; and, where interactions were significant, strain- or condition-specific post hoc comparisons were performed (Šidák or Tukey with family-wise α = 0.05). Across all analyses, no repeated-measures structure was specified, and statistical significance was uniformly set at P < .05.
Results
Extraction Yields of Clove Bud Extracts and Fractions
Clove bud extracts yielded 10% (hydrodistillation), 27% (maceration), and 32% (Soxhlet). For Soxhlet crude, n-hexane, dichloromethane, ethyl acetate, and n-butanol fractions yielded 25.9%, 15.7%, 15.2%, and 22.6%, respectively; maceration fractions yielded 17.3%, 12.5%, 16.3%, and 10.0%. Thus, all Soxhlet fraction yields exceeded those from maceration.
Phytoconstituent Comparison of Maceration and Soxhlet Extracts
Phytochemical screening of the extracts obtained by maceration and Soxhlet methods revealed the presence of five phytochemicals: steroids, triterpenoids, flavonoids, tannins, and phlobatannins in both methods. However, glycosides, alkaloids, carbohydrates, and anthraquinones were exclusively detected in the maceration extract, with no presence in the Soxhlet extract.
Phytochemical Screening Test Result of Crude Extracts and Soxhlet's Fraction.

Keys for Table-1 The ‘+’ sign stands for presence, and the ‘-’ sign stands for the absence of specific phytochemicals in extract and fractions of clove buds.
(HF = n-hexane, DCMF = dichloromethane, EAF = ethyl acetate, BF = n-butanol fractions).
Antibacterial Activity
Comparison of Antibacterial Activities of Soxhlet and Maceration Extracts
The results of antibacterial activity evaluation of extracts obtained by soxhlet and maceration revealed that both crude extracts succeeded in suppressing bacterial growth with variable potency, as depicted in Table 2.
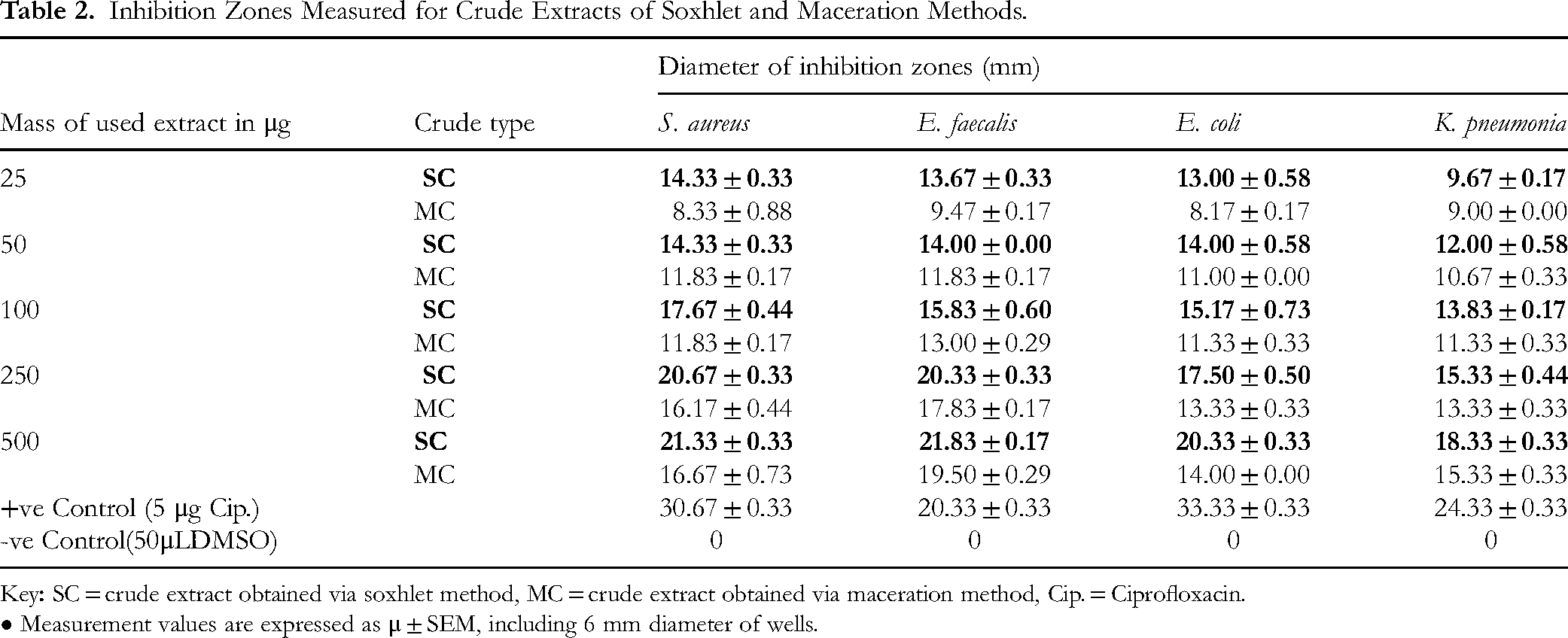
Inhibition Zones Measured for Crude Extracts of Soxhlet and Maceration Methods.
Key
• Measurement values are expressed as μ ± SEM, including 6 mm diameter of wells.
Two-way ANOVA revealed that both extraction technique and extract concentration significantly affected inhibition-zone diameters for S. aureus, E. coli, and E. faecalis, with significant technique × concentration interactions in all three (all P < .01). For K. pneumoniae, extraction technique and concentration were significant (P < .0001), but their interaction was not (P = .0683). Extraction technique, concentration, and interaction accounted for 36.3%, 58.3%, and 2.6% of the variance in S. aureus activity; 45.3%, 48.5%, and 2.9% in E. coli; and 13.4%, 84.4%, and 1.0% in E. faecalis. For K. pneumoniae, technique and concentration explained 9.1% and 86.6% of variance, respectively.
Post hoc Šidák tests showed that Soxhlet extracts produced significantly larger inhibition zones than maceration at all tested concentrations for each bacterium (eg, S. aureus at 25 µg: 14.33 ± 0.33 mm vs 8.33 ± 0.88 mm, P = .012; E. coli at 100 µg: 15.17 ± 0.73 vs 11.33 ± 0.33 mm, P < .001; E. faecalis at 250 µg: P < .001), with differences persisting up to 500 µg. In K. pneumoniae, no concentration-dependent interaction was observed despite overall technique and concentration effects.
Antibacterial Activities of Solvent Fractions of Soxhlet Extract
The methanolic crude extract of clove bud obtained via the soxhlet method inhibited all the tested bacteria better than any of its fractions, as depicted in Tables 2 and 3.
Inhibition Zones Were Measured for Solvent Fractions of the Extract Obtained via Soxhlet.

• HF = n-hexane, DCMF = dichloromethane, EAF = ethyl acetate BF = n-butanol fractions.
The most susceptible bacterium of soxhlet's crude fractions, particularly by n-butanol fraction, was E. faecalis, in which the resulted inhibition zone ranged from 8.67 ± 0.17 mm to 15.00 ± 0.29 mm for all used concentrations, followed by S. aureus (8.50 ± 0.29-14.67 ± 0.17 mm) and K. pneumonia (8.00 ± 0.29 mm-14.67 ± 0.44 mm) as shown in Table 3 and Figure 3. The least susceptible bacterium was E. coli, ranging from 8.50 ± 0.29-14.67 ± 0.44 mm inhibition diameter.
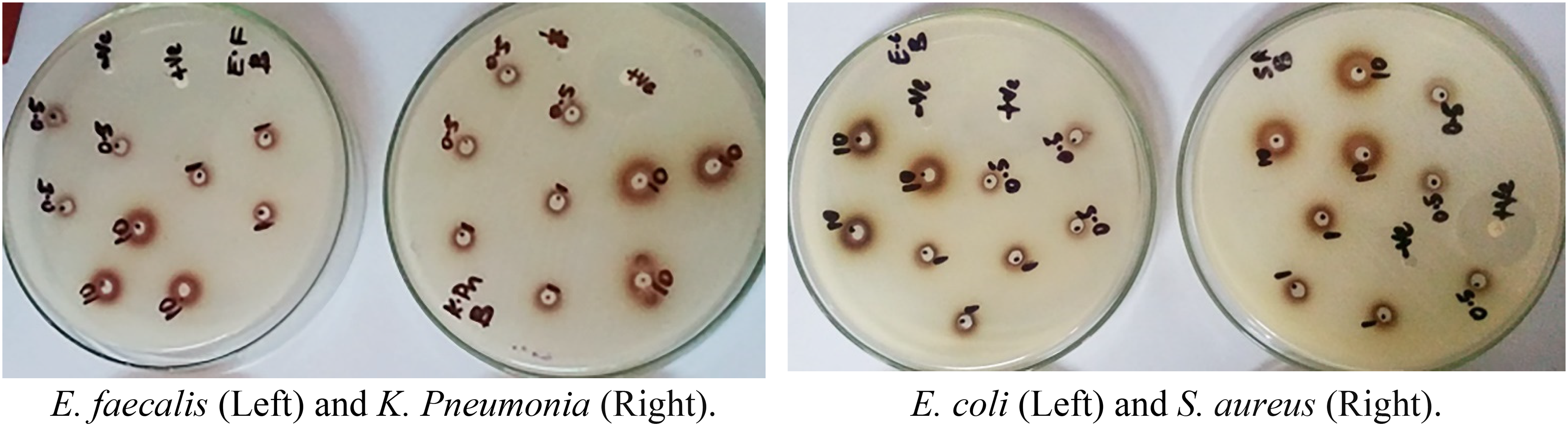
Antibacterial Activity of the n-Butanol Fraction Against Pathogenic Bacteria.
Among the four solvent fractions tested, only the n-butanol extract exhibited consistent, dose-dependent antibacterial activity across all concentrations and bacterial strains. Inhibition zones for n-butanol increased significantly between 25 µg and 50 µg (Δ = 1.00 mm, 95% CI [0.58, 1.42], P < .0001), between 25 µg and 500 µg (Δ = 6.33 mm, 95% CI [5.92, 6.75], P < .0001), and between 50 µg and 500 µg (Δ = 5.33 mm, 95% CI [4.92, 5.75], P < .0001). The ethyl acetate fraction showed measurable inhibition only at 500 µg, while hexane and dichloromethane fractions produced no detectable zones of inhibition at any concentration. A two-way ANOVA confirmed that the Fraction × Concentration interaction was the sole significant source of variation (F11,96 = 11.91, P < .0001), accounting for 50.16% of the total variance. Neither the bacterial strain main effect (F3,96 = 0.98, P = .4048; 1.13% variance) nor the Strain × Fraction/Concentration interaction (F33,96 = 0.95, P = .5583; 11.95% variance) reached significance, indicating that extract fraction and dose alone drive the observed antibacterial differences.
Antibacterial Activity of Clove Essential Oil
The essential oil exhibited antibacterial activity against Gram-positive and Gram-negative bacteria at the used concentrations (Table 4).
In Vitro Inhibition Results on Antibacterial Efficacy of Clove Essential Oil on bacteria.

The S. aromaticum oil demonstrated antibacterial activity against most tested organisms, with inhibition zones ranging from 7.67 ± 0.17 to 19.50 ± 0.29 mm. The most active effects were observed against S. aureus (19.50 ± 0.29 mm) and E. coli (15.83 ± 0.44 mm), as presented in Table 4. This result agreed with an earlier report that clove extract was effective against food-borne microbes. 28
The antibacterial activity of the oil varied significantly with both bacterial strain and oil volume, and their interaction was also highly significant (Two-Way ANOVA: strain × volume F6,24 = 49.17, P < .0001, 5.83% variance; strain F3,24 = 1228.36, P < .0001, 72.78%; volume F2,24 = 529.73, P < .0001, 20.92%). Because the interaction was significant, we examined dose-dependent effects within each strain. In S. aureus, inhibition increased stepwise with oil dose (10→25 µL: + 3.17 mm, P < .0001; 25→50 µL: + 1.33 mm, P = .0022). E. coli and E. faecalis exhibited similar dose–response increments (maximally +4.50 mm and +3.00 mm, respectively; all P < .0001). K. pneumoniae showed the largest shifts (+7.67 mm and +10.17 mm for 10→25 and 10→50 µL; P < .0001), confirming its high sensitivity to oil volume.
Discussion
This study compared maceration and Soxhlet extraction methods for S. aromaticum buds, examining phytochemical composition, antibacterial activity, and the potency of derived solvent fractions and essential oil. The findings reveal significant differences in chemical profiles and bioactivities depending on the extraction method, providing insights into optimal strategies for isolating antibacterial agents from clove.
Phytochemical Profiles of Maceration and Soxhlet Extracts
The maceration and Soxhlet extracts showed different chemical compositions (Table 1). The Soxhlet method extracted a larger amount of material because it continuously cycles hot solvent, but some heat-sensitive compounds were only found in the cold maceration extract. This is similar to what other studies have found, showing that long periods of heat can break down and change the structure of compounds that are not stable at high temperatures.29,30 The Soxhlet extract had a higher concentration of heat-stable compounds like phenolics and terpenoids, which is consistent with previous reports of clove extracts made using reflux conditions.31,32
Qualitative tests confirmed that both extracts contained steroids, triterpenoids, flavonoids, tannins, and phlobatannins. However, glycosides and alkaloids were only found in the maceration extract. This supports previous findings that using cold extraction helps to keep bioactive compounds from breaking down. 33 Thus, extraction method significantly influences the chemical composition of the extract. Maceration favors alkaloids and glycosides, while Soxhlet concentrates phenolics and terpenoids. Although qualitative assays are not highly specific, they demonstrate that both methods can complement each other for targeted extraction.
Antibacterial Activity of Crude Extracts
Both the maceration and Soxhlet extracts were able to inhibit the growth of Gram-positive and Gram-negative bacteria, and the effect increased as the concentration of the extracts increased (Table 2). However, the Soxhlet extract consistently produced larger zones of inhibition than the maceration extract across all four tested bacteria. This difference was statistically significant (P < .0001), confirming that using continuous hot solvent extraction helps to extract more antibacterial phytochemicals. The greater activity of the Soxhlet extracts probably comes from the fact that it yields more bioactive phenolics and that the heat might convert glycosides into more active aglycones.
The antibacterial effect increased with extract concentration (P < .0001) but leveled off after 250 µg, probably because the extract couldn't diffuse through the agar efficiently at higher concentrations. The significant interaction between the extraction method and the concentration for S. aureus, E. coli, and E. faecalis (P < .05) suggests that the Soxhlet extract has the biggest advantage at medium concentrations, whereas its uniform superiority against K. pneumoniae suggests differential cell-wall susceptibility. These findings agree with previous research on the strong antimicrobial effects of eugenol-rich clove extracts.14,24,34,35
The maceration extract also showed some antibacterial activity, 36 suggesting that the phytochemicals it contains, including alkaloids and glycosides, contribute to the overall effect. This supports earlier studies reporting broad-spectrum antibacterial effects of clove extracts.5,13,20 Overall, the extraction method plays a crucial role in modulating antibacterial potency.
Bioactivity of Soxhlet-Derived Solvent Fractions
Solvent fractionation of the Soxhlet extract showed clear differences in antibacterial activity among the fractions (Table 3, Figures 1 and 3). The n-butanol fraction exhibited the strongest and most consistent antibacterial activity against all tested bacterial. 37 The ethyl acetate fraction also showed good activity, but not as strong as the n-butanol. This indicates that solvents with medium polarity, like n-butanol, tend to concentrate polyphenolic compounds. These compounds, such as tannins, flavonoids, saponins, and anthraquinones, are known for their ability to fight microbes.31,38 The hexane and dichloromethane fractions, on the other hand, showed no antibacterial activity, likely due to insufficient concentrations of active compounds. The notable efficacy of the n-butanol fraction, even at low concentrations, suggests the presence of potent compounds that may act by disrupting cell membranes or inhibiting enzymes. Other studies similarly report strong antibacterial properties in n-butanol extracts of medicinal plants.5,13 Statistical analysis confirmed that the differences among fractions were significant (P < .05), and the type of bacteria didn't change this result.
These results suggest the presence of novel antibacterial phytochemicals in the n-butanol fraction, potentially including anthraquinones, phlobatannins, or saponins. Further studies are needed to isolate and characterize these components.
Antibacterial Efficacy of Clove Essential Oil
Clove essential oil exhibited the greatest antibacterial potency, achieving significant inhibition at substantially lower volumes (Table 4). Statistical analysis confirmed significant effects for both oil volume and bacterial strain (P < .0001), with differential sensitivity across bacteria. Notably, S. aureus and E. coli were inhibited at lower oil volumes, highlighting the oil's diverse action against different pathogens.
This high efficacy likely results from the oil's eugenol content (∼85%), which can penetrate and disrupt bacterial membranes,25,39 even in Gram-negative strains including E. coli and K. pneumoniae.13,40 Furthermore, clove oil can enhance the effectiveness of antibiotics, even against resistant bacteria, 29 suggesting its potential as a valuable addition to antibiotic treatments.
Despite its lower yield compared to crude extracts, the essential oil's stronger activity highlights the importance of concentrated active compounds. Eugenol, along with acetyl eugenol and β-caryophyllene, 20 appears central to the oil's bioactivity. These findings support clove essential oil as a promising natural antimicrobial agent.
Mechanistic Insights into Antibacterial Action
Eugenol, the major compound in clove oil, is widely reported to exert antibacterial effects, potentially by interacting with bacterial membranes and increasing their permeability.25,39 This may explain its effectiveness against both Gram-positive and Gram-negative bacteria observed in our study. It has also been suggested that eugenol interferes with bacterial energy metabolism and communication, possibly by affecting ATPase activity and virulence factors. Its hydroxyl group may bind to proteins, influencing enzyme function and cell stability.25,41
Other active compounds—especially those in the n-butanol fraction—such as tannins and flavonoids, may also contribute. Tannins are known to form complexes with membrane proteins and enzymes, potentially disrupting cell wall integrity and metabolic processes. 38 Flavonoids may insert into lipid bilayers or chelate metal ions, interfering with bacterial growth.42–45
The combined presence of these phytochemicals could lead to a broad antibacterial effect by targeting multiple pathways. Previous studies have also noted that S. aromaticum extracts can act synergistically with antibiotics like imipenem and amoxicillin–clavulanate. 24 Overall, the extract's activity likely involves several mechanisms, but further targeted studies are needed to confirm these pathways.
Limitations and Future Directions
Limitations of this study include the unknown harvest season of the commercially sourced clove buds, which may impact antibacterial efficacy due to seasonal variations in phytochemical content, and the use of agar diffusion, which can underestimate the activity of compounds with poor diffusion. Future research should address these limitations by standardizing the sourcing of plant material, investigating greener and more consistent extraction methods, and incorporating direct assays to evaluate antibacterial mechanisms—such as membrane integrity and enzyme activity—rather than relying solely on inferred effects. Further, the identification and characterization of individual active constituents will ultimately enable optimized S. aromaticum extracts for therapeutic use.
Conclusions
Syzygium aromaticum bud extracts prepared via the Soxhlet method demonstrated superior antibacterial effects against all evaluated pathogens (E. faecalis, S. aureus, K. pneumoniae, and E. coli) compared to maceration (P < .0001), with concentration-dependent effects and significant method × concentration interactions for three pathogens. Paradoxically, despite maceration extracting a broader range of phytochemicals, this disparity arises from Soxhlet's efficiency in enriching thermostable antimicrobials and heat-mediated conversion of glycosides to bioactive aglycones, while avoiding matrix interference. The
These results demonstrate how extraction method and solvent selection influence clove's phytochemical composition and antibacterial efficacy. The n-butanol fraction, in particular, provides a rich source of polar antimicrobials suitable for combating antibiotic-resistant pathogens and for use in natural preservatives or topical formulations. Future work should focus on isolating and characterizing the bioactive compounds in the n-butanol fraction, validating their therapeutic potential in vivo, and elucidating their mechanisms of microbial inhibition. By strategically optimizing extraction techniques and solvent systems, our findings offer a clear pathway for developing sustainable, clove-derived antibacterial agents.
Footnotes
Acknowledgements
The authors would like to acknowledge that this manuscript is based on the MSc thesis work of Solomon Getachew conducted at Wollo University. The authors are grateful to Wollo University for providing laboratory facilities and materials, and Amhara Public Health Institute for assisting with the antibacterial assay experiments. Special thanks are also extended to Dr Belete Adefris Legesse for his valuable guidance and insightful suggestions throughout the study.
Ethical Approval
No human or animal subjects were used in this study.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author's Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data for this study are available in the manuscript.