Abstract
Objectives
This study aims to develop and validate a reliable procedure for the determination of cadmium, lead, arsenic, and mercury in herbal medicines using atomic absorption spectroscopy. The method was designed to achieve complete mineralization, minimize analyte loss, and reduce analysis time while ensuring accurate quantification of heavy metals in complex matrices. Given the potential toxicity of heavy metals, ensuring accurate quantification is crucial for quality control and regulatory compliance.
Methods
A microwave-assisted digestion technique using 65% nitric acid was employed for sample preparation. The analytical method was optimized using Design Expert 12 software to maximize recovery efficiency. Linearity, accuracy, precision, limit of detection (LOD), and limit of quantification (LOQ) were validated according to the ICH Q2(R1) guideline.
Results
Optimization results indicated that the recovery rates for Cd, Pb, As, and Hg were all approximately 90%, confirming the method's robustness and suitability for accurate heavy metal quantification in herbal medicine products. The method demonstrated great linearity (r > 0.995), with limits of detection (LOD) of 0.1-10 ng/ml and limits of quantification (LOQ) of 0.3-33.3 ng/ml. The relative standard deviation (RSD) was below 5%, confirming precision. The validated procedure was successfully applied to herbal medicine products.
Conclusion
The developed procedure provides a robust, accurate, and validated approach for the quantification of heavy metals in herbal medicines. Its successful application to real samples highlights its potential for routine quality control and regulatory assessment.
Keywords
Introduction
Herbal medicines hold a place of paramount importance in the healthcare systems of many nations, especially in Asia and Africa. According to published studies, nearly 80% of the global population relies on herbal medicine as a primary healthcare approach. 1 This reliance is not merely due to cultural and historical practices but also to the significant therapeutic potential of these natural remedies.
Numerous phytochemical constituents in herbal medicine such as alkaloids, glycosides, flavonoids, tannins, gums, oils, resins, and their derived metabolites, exhibit various pharmacological activities within the human system.2,3 Each of these compounds uniquely contributes to the therapeutic efficacy of herbal medicines. For instance, flavonoids are known for their antioxidant properties, which help protect cells from damage caused by free radicals. Alkaloids are known for their analgesic and antibacterial properties.4,5 Other phenolic compounds, such as tannins, phenolic acids, and lignans, also contribute to various biological activities. 6 Tannins are well-known for their antioxidant, antimicrobial, and wound-healing effects, while phenolic acids, such as caffeic and ferulic acids, possess strong anti-inflammatory and anticancer properties. 7 Lignans, found in seeds, grains, and vegetables, are recognized for their antioxidant and anticancer effects as well. 8 Glycosides, particularly cardiac glycosides, are crucial in managing heart conditions. 9
The diverse pharmacological activities of these compounds underscore the potential of herbal medicines in treating various ailments, ranging from minor infections to chronic diseases. Moreover, from the accessibility point of view, herbal medications offer a distinct advantage. They are not only available in drug stores but can also be found in many other retail outlets, including grocery stores and markets, making them readily accessible to a broad population. 1
Despite the benefits of herbal medicines, their increased use also raises concerns about safety, particularly regarding heavy metal contamination. 10 Heavy metals such as cadmium, lead, arsenic, and mercury are toxic and pose serious health risks when present in medicinal products. 11 These metals can enter herbal medicines through various pathways, including environmental contamination, cultivation practices, and during processing and storage. 12 Chronic exposure to heavy metals can lead to severe health issues, including neurological damage, kidney failure, and an increased risk of cancer. 11
Ensuring the safety of herbal medicines is thus imperative, and reliable methods for detecting and quantifying heavy metals in these products are crucial for the safety of public health. Atomic Absorption Spectroscopy (AAS) has become one of the most commonly utilized techniques in analytical chemistry for this application. AAS offers sufficient sensitivity for detecting most metals and metalloids and is relatively free from interferences. There are two basic atomization techniques in AAS: flame atomic absorption spectroscopy (FAAS) and electrothermal atomic absorption spectroscopy (ETAAS). FAAS is preferred when sufficient analyte is present in the sample due to its rapidity and simplicity. In contrast, ETAAS, though requiring more operator skill and being less rapid, provides superior limits of detection. 13
Traditional methods for sample digestion, such as open-vessel wet ashing, exhibit several disadvantages, including time-consuming, high risk of analyte contamination and loss, and incomplete mineralization of certain matrices. To address these challenges, digestion methods involving elevated temperatures and/or pressures are considered more effective. This method uses high pressure and temperature to achieve complete digestion of samples. The use of pressurized closed-vessel microwave systems minimizes contamination and loss of volatile elements, offering a more efficient and effective digestion process. Using TFM/PTFE digestion vessels allows for operation at elevated temperatures (up to 350 °C) and high pressures (up to 160 bar). This enables the thorough breakdown of organic matrix components with HNO3 as the sole digestion agent. 14
Although microwave-assisted sample preparation methods have been explored in previous studies, these have not been specifically applied to herbal medicines.15-18 Our study builds on these advancements but focuses on optimizing the sample preparation process to ensure that the method is more accessible, quicker, and less complex, while still providing effective quantification of heavy metals within permissible limits. A study by Jing Sun and colleagues (2024) developed a vortex-assisted combined with deep eutectic solvent-based liquid phase microextraction (VA-LPME-DES), which involves vortexing and centrifugation, followed by cooling to solidify the extract. 19 While this process is effective, it can be time-consuming and involves multiple steps. Our method aims to streamline the process by reducing the time and complexity involved in sample preparation, offering a more practical approach without compromising the accuracy of heavy metal quantification. Furthermore, a sensor-based method developed by Lin Zhang et al (2024) uses a metal-organic framework (MOF) (UiO66-NH2) and multi-walled carbon nanotubes (MWCNTs) for the simultaneous detection of Cd2+, Pb2+, and Hg2+ in water and herbal medicines. 20 However, this approach requires the use of complex metal-containing materials, which can increase the risk of environmental contamination. In contrast, our method avoids the use of such materials, making it more environmentally friendly and suitable for routine analysis.
Our focus on the Vietnamese market is particularly relevant, as there is a growing demand for efficient methods to detect heavy metals in herbal medicines, which are often inadequately regulated. The proposed method involves the quantification of cadmium, lead, arsenic, and mercury. Sample preparation is carried out using a microwave-assisted digestion technique with 65% nitric acid. The study explores several parameters to optimize the recovery rates of these heavy metals, ensuring accurate and reproducible results. Finally, the established method was applied to analyze some herbal medicine products in Vietnam's market to confirm the method's applicability.
These included four commercial formulations with distinct therapeutic uses and herbal compositions. Formulation A consists of Extractum radix polysciacis spissum and Extractum folii ginkgo siccus in a 5:1 ratio. This formulation is traditionally used to tonify qi and blood and is indicated for the prevention and treatment of conditions such as memory impairment, mental stress, headaches, dizziness, and insomnia. For Formulation B, it contains the following ingredients: Tuber Stephaniae glabrae, Folium Nelumbinis nuciferae, Herba Passiflorae foetidae, Folium Erythrinae variegatae, Herba Mimosae pudicae. This formulation is used for conditions such as insomnia or delayed sleep onset, as well as nervous exhaustion. It serves as an alternative to Diazepam in cases where patients develop a tolerance to the medication. The formulation C contains three main ingredients: Herba Phyllanthi urinariae, Radix Codonopsis pilosulae, Herba Adenosmatis caerulei. Formulation C supports liver health, detoxification, and spleen strengthening while enhancing blood circulation. It helps alleviate allergies, urticaria, skin rashes, heat rashes, and boils. Additionally, it aids in the treatment of hepatitis-related symptoms such as fatigue, jaundice, loss of appetite, indigestion, constipation, liver pain, and impaired liver function caused by excessive alcohol consumption or prolonged use of pharmaceutical drugs. Formulation D contains: Radix Angelicae sinensis, Herba Leonuri japonica, Radix Achyranthis bidentatae, Radix Rehmanniae glutinosae praeparata, Radix Paeoniae, Rhizoma Ligustici wallichii. Formulation D supports blood circulation and treats blood deficiency and stasis. It helps alleviate symptoms of cerebral and peripheral circulatory insufficiency, such as fatigue, headaches, dizziness, insomnia, memory decline, muscle pain, and limb numbness. Additionally, it aids in preventing and managing atherosclerosis, vascular occlusion, and strokes.
Materials and Methods
Chemicals and Reagents
All standard solutions of the heavy metals used in this study were acquired from Merck (Germany). Specifically, the standard solutions of cadmium, lead, arsenic, and mercury were obtained for calibration and validation purposes.
Nitric acid (65%) and hydrogen peroxide (30%), essential for the microwave-assisted digestion process, as well as the magnesium matrix modifier and palladium matrix modifier used to enhance the precision and accuracy of the AAS measurements, were also procured from Merck (Germany).
Ultra-pure water used in all solution preparations, dilutions, and digestion processes was prepared using Water Pro PS Polishing Systems (Labconco, USA).
A total of 4 commercial formulations (coded: A, B, C and D) were obtained from Vietnam's market.
Instruments
The digestion of herbal medicine samples was performed using a Multiwave PRO Microwave Reaction System (Anton Paar, Austria). Heavy metals determination was carried out with a PinAAcle 900T AA Spectrometer (PerkinElmer, USA). Hollow cathode lamps for cadmium and electrodeless discharge lamps for lead, arsenic, and mercury (PerkinElmer, USA) were used as the radiation sources.
Preparation of Standard Solutions, Blank Sample Matrix, and Spiked Samples
The standard solutions for each heavy metal were prepared by diluting the respective stock standard solutions (1000 ppm) with 0.2% nitric acid (HNO3).
To create a blank sample matrix, several commercial formulations from the Vietnamese market were examined. The formulation that showed no detectable heavy metals was selected as the blank matrix, ensuring it mimics the actual sample's properties while serving as a baseline for spiked sample analysis. 21
Appropriate amounts of the standard solutions were added to the blank sample matrix to prepare the spiked samples. 21 These spiked samples were then stored in clean, labeled polyethylene bottles at room temperature for further analyses.
Preparation of Herbal Medicine Samples
Finely ground herbal medicine samples were accurately weighed and transferred into reaction vessels. Specific amounts of 65% HNO3 and 30% hydrogen peroxide (H2O2) were added. The reaction vessels were then placed into a rotor and inserted into a Multiwave PRO microwave digestion device (Anton Paar) for digestion, employing the program recommended by the manufacturer. After digestion, the solutions were transferred into 50 ml volumetric flasks, diluted to the mark with 0.2% HNO3, and mixed thoroughly. 22 Different conditions, such as the amounts of sample, HNO3, and H2O2, were investigated.
Optimization of the Microwave-Assisted Digestion Process
The spiked samples were prepared, analyzed, and assessed by the recovery rates of each heavy metal to optimize the sample preparation process. The independent variables X1 (sample weight in g), X2 (HNO3 65% volume in ml), and X3 (H2O2 30% volume in ml) were selected for the response surface methodology design. 23
Twelve independent combinations and three replicates at the center point, previously determined through a one-factor-at-a-time approach, were used to analyze the responses. Randomization of the experimental order helped mitigate the impact of unforeseen variations in the observed responses.24,25
The response surfaces were modeled using least-squares regression of a second-order polynomial equation (Equation 1):
Method Validation Approach
The developed method was validated based on the ICH Q2(R1) guideline (International Conference on Harmonization of Technical Requirements for registration of pharmaceuticals for human use, 2005). 26
Specificity
Each sample type, including 0.2% HNO3, blank sample, blank sample matrix, standard solution, and spiked sample, was measured in triplicate. Criteria included verifying minimal or negligible absorbance at the typical analytical wavelength for 0.2% HNO3, blank, and matrix samples. The standard solution and spiked sample were assessed for absorbance at wavelengths specific to the elements analyzed.
Linearity
The linearity testing was conducted by describing the absorbance as a function of anlytical concentration by linear regression analysis within the appropriate range. Microsoft Office Excel was employed to calculate the statistical results.
Detection Limit and Quantitation Limit
The detection limit (LOD) of the analytical method was established by comparing measured signals from samples with known low concentrations of analyte to those of blank sample matrix. The LOD was defined as the lowest concentration at which the analyte can be detected with reliability.
From the LOD, the quantitation limit (LOQ) of the method was inferred using the formula: LOQ =
Accuracy
The accuracy of the developed analytical method was evaluated by spiking blank sample matrix with standard solutions of heavy metals at different concentrations, covering lower, medium, and higher levels at the allowable concentration limits on heavy metals. The concentrations of heavy metals recovered were compared to the known added concentrations.
Precision
The precision of the analytical method was evaluated based on its repeatability. Six spiked sample solutions were prepared at the allowable concentration limits of heavy metals in traditional medicines. Precision was evaluated by computing the relative standard deviation (RSD%).
Application of the Method
The spiked samples with the concentrations of 50%, 80%, 100%, 120%, and 150% of the limit of heavy metals in conventional literature were analyzed by AAS to produce the calibration curves.
Optimal conditions were applied to quantify heavy metals in herbal medicine products available in the Vietnam's market. Preparation of each sample was carried out as described previously. The content of each heavy metal in each heral medicine product was calculated according to the following formula:
Statistical Analysis
All experiments were conducted at least in triplicates, and data were expressed as mean and relative standard deviation (RSD). Pearson or Spearman correlation analysis and simple linear regression were conducted using Microsoft Excel to assess the relationship between variables. Statistical data for the optimization of the wet-digestion process were performed using Design Expert 12 software.
A P-value of <.05 was considered statistically significant. Graphical representations of data were created using Design Expert 12 to visually interpret the results.
Results
Method Optimization
According to the principle that sample weight provides sufficient analytical concentration without overloading the digestion vessel, and that a balanced volume of 65% HNO3 ensures complete digestion while minimizing the risk of sample loss, and an appropriate amount of 30% H2O2 achieves efficient oxidation of organic matter without compromising analytical stability, these factors were investigated to study their effect on the recovery rates of heavy metals.
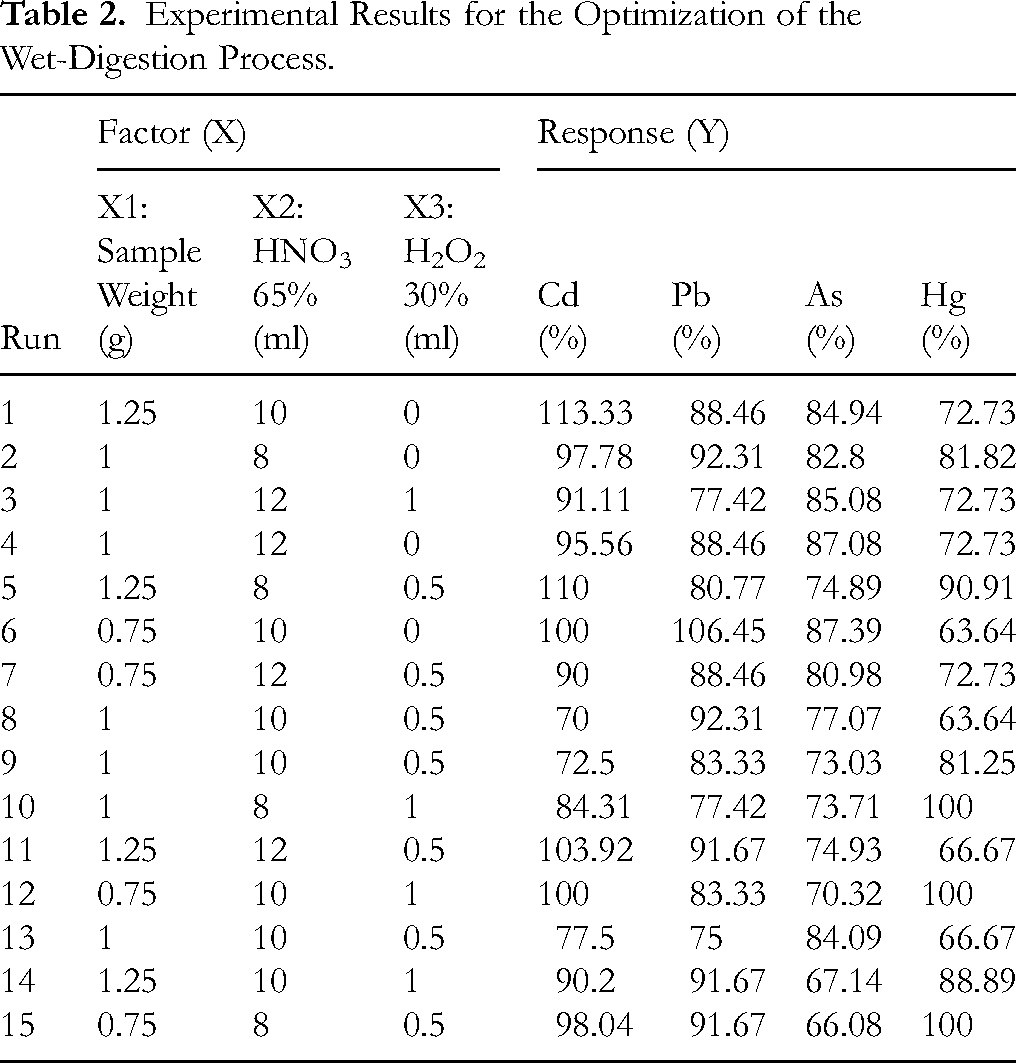
Guided by our previous in-house experiments, the key variables and their appropriate ranges for this investigation were identified, as presented in Table 1. The experimental results to support the RSM design is presented in Table 2.
Factors and Responses for Experimental Design.

Experimental Results for the Optimization of the Wet-Digestion Process.
The response data were analyzed using the Design Expert 12.0 software to obtain the appropriate equation for the prediction. The modeling statistical data and the prediction equations are presented in Table 3.
Statistical Data for the Optimization of the wet-Digestion Process (ns: not Significant).

A P-value less than .05 indicates that the model statistically explains the data, rejecting the null hypothesis of no relationship. Conversely, a lack-of-fit P-value greater than .05 suggests the model adequately fits the data without significant unexplained variation. Statistical analysis confirms the model's adequacy for navigating the design space.
The overall effects of the process variables on the target responses are described in Table 4. Based on these results, the optimal conditions for the wet digestion process are presented in Table 5.
Graphical Results. Part A: Joint Graphical Analysis: the Relationships Between all Variables Involved in 3D Space. The Binary Interactions Between the Remaining two Variables are Visualized by Placing one variable at the Center of the Experimental Range. Part B: Evaluating Model Fit: Two Essential Graphical Tools Assess the Model's Accuracy. First, we Compare Predicted and Observed Responses to see how Well the Model Simulates Real-World Changes. Second, we Examine the Distribution of Residuals (Differences Between Predicted and Observed Values) Across Each variable to Identify any Potential Biases.

Predicted Solution for the Optimization of the wet Digestion Process.

To confirm the model's predictive performance, the average results of the additional confirmation runs at the optimal setting have to be within their respective 95% prediction intervals, as presented in Table 6.
The Comparison of Experimental and Predicted Values of Responses Under Optimal Condition (n = 6).

Method Validation
Specificity
Specificity testing results are summarized in Table 7. The 0.2% HNO3, blank, and matrix samples showed negligible absorbance at the specific wavelengths corresponding to the targeted elements. In contrast, the standard solution and spiked sample exhibited significant absorbance at these wavelengths. This indicates that the method can selectively detect the heavy metals of interest without interference from matrix components or contaminants present in the blank samples. Therefore, the specificity of the method was confirmed.
Specificity Data. The Standard Solution and Spiked Sample Were Prepared at Concentration of 10 ppb for Cd and Hg, and 100 ppb for Pb and As.

Linearity
The linearity of the method was evaluated for cadmium, lead, arsenic, and mercury within specified concentration ranges. Absorbance readings were plotted against the concentration of each heavy metal to generate the calibration curves. The linear ranges were as follows: 5.0 to 15.0 ng/ml for cadmium and mercury, and 50.0 to 100.0 ng/ml for lead and arsenic. Within these concentration ranges, the regression equations of each analyte exhibited appropriate correlation coefficients (r ≥ 0.998 for cadmium and lead, r ≥ 0.995 for arsenic and mercury).
To evaluate the statistical significance of linearity for each analyte, an analysis of variance (ANOVA) was conducted. The results indicated no significant deviation from linearity (P < .05), as the calculated F values were lower than the critical F values. Therefore, the proposed method's linearity was validated within the chosen concentration ranges Table 8.
Linearity Assessment Results.

LOD and LOQ
For cadmium and lead, the LODs were determined to be 0.5 ng/ml and 10.0 ng/ml, respectively, while the LOQs were 1.7 ng/ml and 33.3 ng/ml. For arsenic and mercury, the LODs were 1.0 ng/ml and 0.1 ng/ml, with corresponding LOQs of 3.3 ng/ml and 0.3 ng/ml.
Accuracy
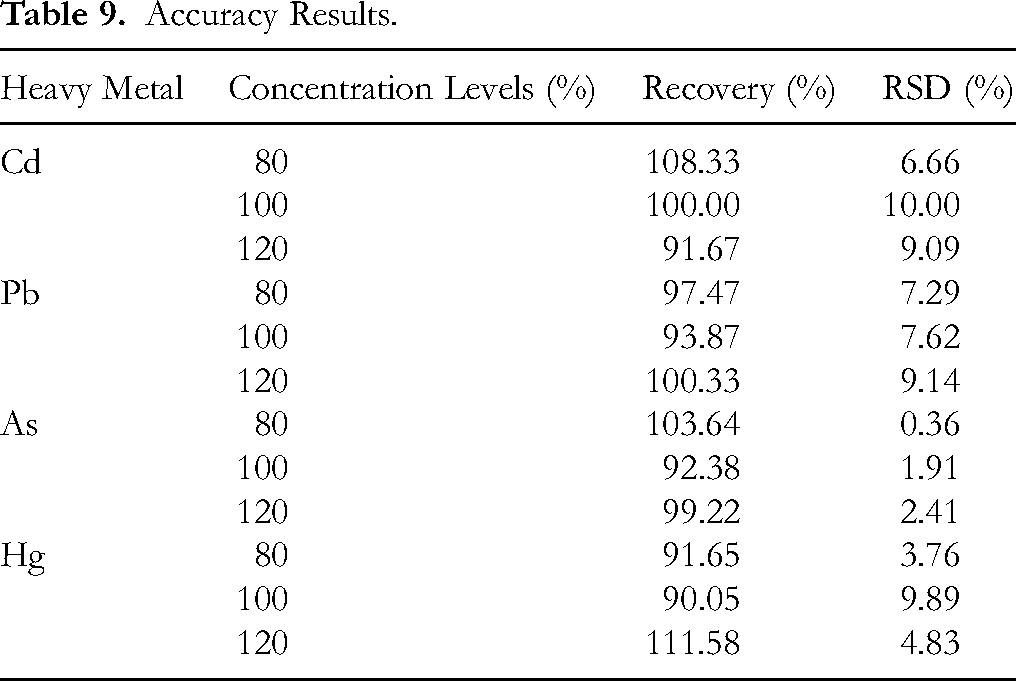
For cadmium, the recovery rates ranged from 91.67% to 108.33% across all concentration levels. For lead, the recovery rates ranged from 93.87% to 100.33%. Arsenic showed recovery rates between 92.38% and 103.64%, while mercury exhibited recovery rates from 90.05% to 111.58%. In all cases, the RSD did not exceed 10%. The results are represented in Table 9.
Accuracy Results.
Precision
For cadmium, the repeatability showed an RSD% of 3.23%. Lead exhibited an RSD% of 6.19%. Arsenic demonstrated an RSD% of 2.16%, and mercury showed an RSD% of 6.67%. These RSD% values indicate the high repeatability of the method.
Application to Herbal Medicine Products in Vietnam
The optimized method was applied to quantify heavy metals in various herbal medicine products available in the Vietnam's market. Four commercial formulations, labeled A, B, C, and D, were obtained and analyzed using the developed procedure. Each sample was prepared by accurately weighing a specified amount of the product and following the established digestion and dilution protocols.
For cadmium, the concentrations ranged from 0.15 to 0.22 µg/g across the different formulations. Lead concentrations varied from 0.00 to 3.70 µg/g, while arsenic levels were between 0.00 and 0.69 µg/g. Mercury was found in concentrations ranging from 0.09 to 1.44 µg/g.
Discussion
Based on the data presented in Table 4, at lower levels, increasing the sample weight, the volume of HNO3, and the volume of H2O2 led to a decrease in the extraction efficiency of cadmium. However, at higher levels of HNO3 and H2O2 volumes, increasing their volumes did not cause significant changes, while increasing the sample weight at these higher levels gradually improved cadmium extraction efficiency.
For lead, increasing the volume of H2O2 clearly decreased the extraction efficiency, while varying the volume of HNO3 had no significant impact on lead extraction. Increasing the sample weight led to a decrease in extraction efficiency, but at higher sample weights, further increases did not significantly affect the extraction.
For arsenic and mercury, the sample weight did not have a noticeable effect on extraction efficiency at any concentration. Increasing the volume of HNO3 improved arsenic extraction efficiency but reduced mercury extraction efficiency. Conversely, increasing the volume of H2O2 showed the opposite trend, where arsenic extraction decreased while mercury extraction increased.
Heavy metals such as cadmium, lead, arsenic, and mercury are known to pose serious health risks, even at trace levels. Lead exposure has been associated with neurotoxicity, impaired cognitive development in children, and renal dysfunction. 27 Cadmium accumulates primarily in the kidneys and bones, potentially leading to nephrotoxicity and skeletal damage. 28 Arsenic toxicity is a severe condition that may arise from chronic exposure through contaminated water, food, or air. 29 Clinical manifestations include gastrointestinal disturbances, dermal lesions, neuropathy, and an increased risk of various cancers.29,30 Mercury, particularly in its organic forms, can adversely affect the central nervous system, and is especially harmful to pregnant women and young children. 31 Given these significant health hazards, the accurate quantification and monitoring of these elements in herbal medicinal products are imperative. Herbal medicines, in particular, are among the product types most vulnerable to heavy metal contamination due to environmental pollution, agricultural practices, and inadequate processing control. Therefore, ensuring the safety, quality, and regulatory compliance of these products requires a reliable analytical approach.
The use of microwave-assisted sample digestion significantly advanced the determination of heavy metals in herbal medicines. This method offered rapid and efficient decomposition of samples using 65% nitric acid, ensuring thorough digestion while minimizing sample loss. Optimization of sample weight and reagent volumes was crucial in achieving high recovery rates for cadmium, lead, arsenic, and mercury. The method's robustness was evident in its ability to handle diverse matrices typical of herbal medicines, resulting in reliable quantification.
The toxic concentration limits for heavy metals in commercial formulations, as specified by the ASEAN Guidelines on Limits of Contaminants for Traditional Medicines, are as follows: cadmium (Cd) ≤ 0.3 ppm, lead (Pb) ≤ 10.0 ppm, arsenic (As) ≤ 5.0 ppm, and mercury (Hg) ≤ 0.5 ppm. 32
In our study, the analyzed commercial formulations were tested for cadmium, lead, arsenic, and mercury levels. The results indicate that all tested formulations contained heavy metal levels within the ASEAN permissible limits, except for mercury. Specifically, one out of the four herbal medicines analyzed exceeded the regulatory limit for mercury (1.4 ppm).
Higher mercury concentrations were observed in formulations containing Herba Phyllanthi urinariae, Radix Codonopsis pilosulae, and Herba Adenosmatis caerulei. This may suggest a possible link between these herbal ingredients and mercury contamination, which could be due to factors such as soil composition, environmental exposure during cultivation, or processing methods. Further investigation is needed to determine whether these specific ingredients inherently accumulate mercury or if contamination arises from external sources.
These findings highlight the importance of continuous monitoring and quality control in herbal medicine formulations to ensure consumer safety and regulatory compliance.
Limitation
The method was validated using a specific set of herbal medicine samples, and its applicability to a wider range of herbal formulations requires further investigation. While microwave-assisted digestion enhances analyte recovery, matrix effects from complex herbal compositions may still pose challenges in certain cases. The use of Flow Injection Analysis System (FIAS) could further improve the method's sensitivity, making it more suitable for detecting heavy metals at ultra-trace levels. Additionally, the validation process could be strengthened by incorporating intermediate precision testing to ensure method reproducibility across different conditions. In the optimization phase, factors such as digestion temperature and processing time were not extensively studied, as including these parameters would have significantly increased the number of experiments. Instead, the study focused on optimizing three key factors to achieve an efficient and practical analytical procedure.
Conclusion
The development of an analytical procedure for the determination of heavy metals in herbal medicines using atomic absorption spectroscopy was motivated by the critical need to ensure the safety and quality of these products. Herbal medicines, often derived from natural sources, can inadvertently contain harmful levels of heavy metals, posing serious health risks to consumers.
This study successfully addressed these concerns by establishing a robust method that offers several benefits. Firstly, the use of microwave-assisted sample digestion enhanced the efficiency and accuracy of heavy metal quantification. Optimization efforts focused on achieving high recovery efficiencies for cadmium, lead, arsenic, and mercury, which are essential for reliable quantification in complex matrices. Analytical validation demonstrated the method's strong performance with regard to linearity, accuracy, precision (RSD < 10%), and sensitivity (LOD and LOQ). Furthermore, the application of this developed procedure to quantify heavy metals in various herbal medicinal products from the Vietnamese market affirmed the method's applicability.
Footnotes
Acknowledgements
Our special thanks go to Pham Ngoc Thach University of Medicine for providing the equipment for this research.
Ethical Considerations
No human or animal subjects were involved in this study.
Author Contributions
All authors of this manuscript have contributed substantially to the research and preparation of this work. They collectively participated in the conception and design of the study, the acquisition of data, and the analysis and interpretation of results. Each author actively contributed to drafting the manuscript and revising it critically for important intellectual content. Furthermore, all authors have reviewed and provided final approval for the version to be published, and they agree to be accountable for all aspects of the work. This authorship complies with the guidelines established by the International Committee of Medical Journal Editors (ICMJE), ensuring that each author has met the criteria for authorship by making significant contributions to the conception, execution, or interpretation of the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
No human or animal subjects were involved in this study.
Statement of Informed Consent
No human or animal subjects were involved in this study.


