Abstract
Objective/Background
The medicinal plant
Methods
Freeze-drying and oven-drying were compared for active ingredient preservation. Four solvents were tested for extraction efficiency and antioxidant properties. Phenolic stability was assessed under heating, varying pH, and simulated human digestion. UHPLC-ESI-Q-TOF-MS identified chemical constituents. Oil stability tests, acute oral toxicity studies, and animal experiments evaluated safety and potential applications.
Results
Freeze-dried fruit showed significantly higher active component levels than oven-dried (total carbohydrate: 781.35 ± 7.46 mg/g; total protein: 829.08 ± 9.69 mg/g; total phenolics: 67.52 ± 1.78 mg/g; total flavonoids: 90.45 ± 0.93 mg/g; total tannins: 62.12 ± 1.84 mg/g). Antioxidant assays (IC50 for DPPH: 56.63 ± 1.84 μg/mL; ABTS: 30.18 ± 0.96 μg/mL) confirmed superior bioactive compound preservation. Adding ethanol to the solvent improved extraction efficiency and antioxidant properties, with ethanol and 80% ethanol extracts from freeze-dried fruit performing equally well. Phenolics in the ethanol extract showed stability in heating, pH variation, and simulated digestion tests. HPLC-ESI-Q-TOF-MS identified 51 compounds, primarily flavonoids and phenolics. The ethanol extract stabilized sunflower oil and showed no acute oral toxicity. Animal experiments demonstrated significant hepatoprotective effects against D-galactosamine-induced liver damage in rats.
Conclusion
Freeze-drying is the preferred method for preserving active ingredients in
Keywords
Introduction
There are approximately 200 species of plants belonging to the genus

Morphology of
Plants of the
Experimental Section
Plant Materials
In October 2023, the fruit of
Two Drying Methods of R. ussuriensis Fruit
The
Phytochemical Composition Analysis
An analysis of phytochemical composition was performed on fifteen distinct chemical components, employing a previously validated methodology. 14 For a comprehensive understanding of the experimental methods, see the Supplemental Material for full details.
Production of Various Extracts from Oven-Dried R. ussuriensis Fruit (ORUF) and Freeze-Dried R. ussuriensis Fruit (FRUF)
The objective of this experiment was to explore how different solvents affect the efficiency of extracting active components from FRUF and ORUF. To achieve this, four different polar solvents were utilized: water, ethanol, 80% ethanol, and methanol. The extraction protocol was performed according to established methodology. 14 The detailed experimental procedure was as follows:
The collected samples of
Phytochemical Quantification Analysis
A quantitative analysis of phytochemicals was conducted to determine the contents of several compounds, such as total protein content (TProC), total phenolic content (TPheC), total flavonoid content (TFC), total carbohydrate content (TCC), total tannin content (TTanC), gallotannin content (GC), condensed tannin content (CTC), total phenolic acid content (TPAC) and total alkaloid content (TAC). The methods employed for this analysis were previously described in references.14-15 The detailed experimental procedure was as follows:
TCC
Briefly, 250 μL of FRUF or ORUF extract (0.02-0.5 mg/mL) in distilled water, 125 μL of phenol solution (5%), and 625 μL of H2SO4 were mixed in an Eppendorf tube and incubated for 30 min. Subsequently, 200 μL of the sample was pipetted from each Eppendorf tube onto a microplate. A calibration curve was produced based on glucose (0-200 mg/L) as a standard. The absorbance of the sample was recorded at 490 nm against a blank sample consisting of FRUF or ORUF extract with distilled water. The mean of three readings was used and TCC was expressed in milligrams of glucose equivalents (GE)/g of FRUF or ORUF extract.
TProC
Briefly, 200 μL of bicinchoninic acid working solution and 20 μL of FRUF or ORUF extract (0.1-2.0 mg/mL) in distilled water were mixed in a microplate and incubated at 37 °C for 30 min. A calibration curve was produced based on bovine serum albumin (0-500 mg/L) as a standard. The absorbance of the sample was recorded at 562 nm against a blank sample consisting of FRUF or ORUF extract with distilled water. The mean of three readings was used and TProC was expressed in milligrams of BSA equivalents (BSAE)/g of FRUF or ORUF extract.
TPheC
Briefly, 100 μL of FC reagent (1 M) and 200 μL of FRUF or ORUF extract (0.5-2.0 mg/mL) in distilled water were mixed in an Eppendorf tube and incubated for 5 min. Subsequently, 500 μL of Na2CO3 solution (20%) was added and allowed to stand at room temperature for 40 min in the dark (with mixing every 10 min). Subsequently, 200 μL of the sample was pipetted from each Eppendorf tube onto a microplate. A calibration curve was produced based on gallic acid (0-100 mg/L) as a standard. The absorbance of the sample was recorded at 750 nm against a blank sample consisting of FRUF or ORUF extract with distilled water and Na2CO3. The mean of three readings was used and TPheC was expressed in milligrams of gallic acid equivalents (GAE)/g of FRUF or ORUF extract.
TFC
Briefly, 100 μL of AlCl3 (2%) in methanol and 100 μL of FRUF or ORUF extract (0.5-5.0 mg/mL) in methanol were mixed in a microplate and incubated at room temperature for 10 min. A calibration curve was produced based on quercetin (0-100 mg/L) as a standard. The absorbance of the sample was recorded at 415 nm against a blank sample consisting of FRUF or ORUF extract with methanol. The mean of three readings was used and TFC was expressed in milligrams of quercetin equivalents (QE)/g of FRUF or ORUF extract.
TPAC
Briefly, 20 μL of FRUF or ORUF extract (0.2-2.0 mg/mL) in distilled water, 20 µL of Arnow reagent, 20 µL of HCl solution (0.1 M), 120 µL of distilled water and 20 µL of NaOH solution (1 M) were mixed in a microplate and recorded immediately at 490 nm against a blank sample (Arnow reagent was replaced with distilled water). A calibration curve was produced based on caffeic acid (0-100 mg/L) as a standard. The mean of three readings was used and TPAC was expressed in milligrams of caffeic acid equivalents (CAE)/g of FRUF or ORUF extract.
TTanC
Briefly, 200 μL of FC reagent (1 M) and 200 μL of FRUF or ORUF extract (0.2-2.0 mg/mL) in distilled water were mixed in an Eppendorf tube and incubated for 5 min. Subsequently, 100 μL of Na2CO3 solution (20%) and 1500 μL of distilled water were added and allowed to stand at room temperature for 30 min in the dark (with mixing every 10 min). Subsequently, 200 μL of the sample was pipetted from each Eppendorf tube onto a microplate. A calibration curve was produced based on tannic acid (0-200 mg/L) as a standard. The absorbance of the sample was recorded at 725 nm against a blank sample consisting of FRUF or ORUF extract with distilled water and Na2CO3. The mean of three readings was used and TTanC was expressed in milligrams of tannic acid equivalents (TAE)/g of FRUF or ORUF extract.
GC
Briefly, 875 µL of FRUF or ORUF extract (0.2-2.0 mg/mL) in methanol and 375 µL of saturated KIO3 solution were mixed in an Eppendorf tube and incubated at 15 °C for 120 min. A calibration curve was produced based on gallic acid (0-400 mg/L) as a standard. The absorbance of the sample was recorded at 550 nm against a blank sample (KIO3 was replaced with distilled water). The mean of three readings was used and GC was expressed in milligrams of GAE/g of FRUF or ORUF extract.
CTC
Briefly, 4 mg of phloroglucinol was added to 2 mL of FRUF or ORUF extract (0.5-10.0 mg/mL) in distilled water. Subsequently, 1 mL of HCl solution and 1 mL of formaldehyde solution were added and mixed in an Eppendorf tube and incubated at room temperature overnight. The precipitate was separated by filtration, the unprecipitated phenolics were measured in the filtrate according to the method of TPheC.
TAC
Berberine hydrochloride (1.24-12.36 mg/L) was used as a reference material to construct a standard curve. The absorbance was obtained at 420 nm against a blank sample of chloroform. The mean of three measurements was calculated. The total alkaloid content is expressed in milligrams of berberine hydrochloride equivalents (BHE)/g of FRUF or ORUF extract.
Assays for Antioxidant Capability
Multiple methods were employed to evaluate antioxidant capability, encompassing the 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), 1,1-diphenyl-2-picrylhydrazyl radical (DPPH), hydroxyl radical, superoxide radical, cupric ion reducing antioxidant capacity (CUPRAC), Ferric ion reducing antioxidant power (FRAP), metal chelating, hydrogen peroxide (H2O2), singlet oxygen,
DPPH Assay
Briefly, 100 µL of FRUF or ORUF extract (0.01-2.0 mg/mL) in methanol and 100 µL of DPPH in methanol (50 µM) were mixed in a microplate and allowed to stand at room temperature for 20 min in the dark. The absorbance of the sample was recorded at 515 nm. The Half-maximal inhibitory concentration (IC50) values were calculated and expressed as the mean ± standard deviation (SD) in μg/mL.
ABTS Assay
Briefly, 190 μL of diluted ABTS solution and 10 μL of FRUF or ORUF extract (0.5-5.0 mg/mL) in DMSO were mixed in a microplate and incubated for 20 min in the dark. The absorbance of the sample was recorded at 734 nm. The IC50 values were calculated and expressed as the mean ± SD in μg/mL.
Hydroxyl Radical Assay
Briefly, 50 µL of FRUF or ORUF extract (0.1-10.0 mg/mL) in DMSO, 50 µL of FeSO4 solution (3 mM) and 50 µL of H2O2 solution (3 mM) were mixed in a microplate and incubated for 10 min. After then 50 µL of salicylic acid solution (6 mM) was added and incubated at room temperature for 30 min in the dark. The absorbance of the sample was recorded at 492 nm. The IC50 values were calculated and expressed as the mean ± SD in μg/mL.
Superoxide Radical Assay
Briefly, 45 µL of FRUF or ORUF extract (0.1-10.0 mg/mL) in DMSO (10 mg/mL), 15 µL of NBT in DMSO (1 mg/mL) and 150 µL of NaOH in DMSO (50 μM) were mixed in a microplate and the absorbance of the sample was recorded immediately at 560 nm against a blank sample (NBT was replaced with DMSO). The scavenging activity was expressed as % scavenging rate and was calculated as follows: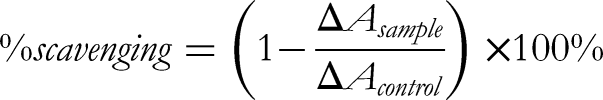
FRAP Assay
Briefly, 20 µL of FRUF or ORUF extract (0.16 mg/mL) in DMSO and 180 µL of FRAP reagent were mixed in a microplate and incubated at 37 °C for 30 min in the dark. A calibration curve was produced based on FeSO4 (0-600 mg/L) as a standard. The absorbance of the sample was recorded at 595 nm. Trolox was used as positive reference. The FRAP was expressed as the Trolox Equivalent Antioxidant Capacity (TEACFRAP).
CUPRAC Assay
Briefly, 20 µL of CuCl2 solution (100 mM), 50 µL of neocuproine in 96% ethanol (7.5 mM), 50 µL of NH4Ac solution, 20 µL of FRUF or ORUF extract (0.2-2.0 mg/mL) in DMSO, and 30 µL of distilled water were mixed in a microplate and incubated at 50 °C for 20 min. This mixture was allowed to stand at room temperature for 10 min. The absorbance of the sample was recorded at 450 nm. The CUPRAC was expressed as the Trolox Equivalent Antioxidant Capacity (TEACCUPRAC).
Iron Chelating Assay
Briefly, 50 µL of FRUF or ORUF extract (0.5-10.0 mg/mL) in methanol, 110 µL of ultra-pure water, and 20 µL of FeCl2 solution (0.5 mM) were mixed in a microplate and incubated for 5 min. Subsequently, 20 µL of ferrozine solution (2.5 mM) was added and incubated for 10 min. The absorbance was recorded at 562 nm against a blank sample (ferrozine solution was replaced with water). The IC50 values were calculated and expressed as the mean ± SD in μg/mL.
Copper Chelating Assay
Briefly, 40 µL of FRUF or ORUF extract (0.5-10.0 mg/mL) in ultra-pure water, 140 µL of acetic acid-sodium acetate buffer solution (pH 6.0, 50 mM), and 10 µL of CuSO4 solution (5 mM) were mixed in a microplate and incubated for 30 min. Subsequently, 10 µL of pyrocatechol violet solution (4 mM) was added and incubated for 30 min. The absorbance was recorded at 632 nm against a blank sample (pyrocatechol violet was replaced with water). The IC50 values were calculated and expressed as the mean ± SD in μg/mL.
H2O2 Assay
Briefly, 70 µL of phenol solution (pH 7.0, 12 mM, in 84 mM phosphate buffer (PBS)), 20 µL of 4-aminoantipyrine solution (pH 7.0, 0.5 mM, in 84 mM PBS), 32 μL of H2O2 solution (pH 7.0, 0.7 mM, in 84 mM PBS), 8 µL of horseradish peroxidise (EC 1.11.1.7) solution (pH 7.0, 1 U/mL, in 84 mM PBS) and 70 µL of FRUF or ORUF extract (0.5-10.0 mg/mL) (pH 7.0, in 84 mM PBS) were mixed in a microplate and the absorbance of the sample was recorded immediately at 504 nm against a blank sample (phenol solution was replaced with PBS). The IC50 values were calculated and expressed as the mean ± SD in μg/mL.
Singlet Oxygen Assay
Briefly, 40 µL of FRUF or ORUF extract (0.5-10.0 mg/mL) (pH 7.4, in 45 mM PBS), 50 µL of N,N-dimethyl-4-nitrosoaniline (pH 7.4, 0.2 mM, in 45 mM PBS), 20 μL of histidine solution (pH 7.4, 0.1 mM, in 45 mM PBS), 20 µL of NaClO solution (pH 7.4, 0.1 mM, in 45 mM PBS), 20 μL of H2O2 (pH 7.4, 0.1 mM, in 45 mM PBS) and 50 µL of PBS (pH 7.4, 45 mM) were mixed in a microplate and allowed to stand at room temperature for 40 min. The absorbance of the sample was recorded at 440 nm against a blank sample (FRUF or ORUF extract was replaced with PBS). The IC50 values were calculated and expressed as the mean ± SD in μg/mL.
β -Carotene Bleaching Assay
Briefly, 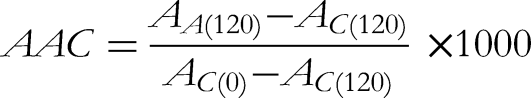
HClO Assay
HClO was freshly prepared by adjusting the pH of a 1% (v/v) of NaClO to 6.2 with 1% H2SO4. The concentration of HClO was determined by reading the absorbance at 235 nm and using the molar extinction coefficient of 100 M−1·cm−1. Briefly, 20 µL of FRUF or ORUF extract (10 mg/mL) aqueous solution, 20 µL of 150 mM taurine aqueous solution, 20 µL of 0.5 mM HClO solution and 140 μL of PBS (pH 7.4, 50 mM) were mixed in a microplate and incubated for 10 min. Subsequently, 2 µL of 2 M KI aqueous solution was added and mixed. The absorbance was recorded at 350 nm against a blank sample (taurine and HClO were replaced with water). IC50 values were calculated and expressed as the mean ± SD in μg/mL.
NO Assay
Briefly, 3 mL of FRUF or ORUF extract (1 mg/mL) in methanol and 3 mL of sodium nitroprusside solution (pH 7.4, 5 mM, in 0.1 M PBS) were mixed in an Eppendorf tube and incubated at 25 °C for 150 min. At intervals, 100 μL of the sample was pipetted from each Eppendorf tube onto a microplate containing 100 µL of Griess reagent. In the control group, FRUF or ORUF extract was replaced with methanol. The absorbance was recorded at 546 nm against a blank sample (Griess reagent was replaced with distilled water).
Investigations on the Stability of Ethanol Extract and 80% Ethanol Extract Derived from FRUF
The pH, thermal, and gastrointestinal tract model system stability of both the ethanol extract and 80% ethanol extract of FRUF, using methods previously outlined in. 14 The detailed experimental procedure was as follows:
pH Stability
The stability in acidic and basic environments was investigated using ethanol or 80% ethanol extract (2 mg/mL) of FRUF dissolved in deionized water with the pH adjusted to 1, 3, 5, 7, 9, or 11 using 1 M HCl or 1 M NaOH. The final concentration of ethanol or 80% ethanol extract was 50 mg/mL. After incubation at room temperature for 1 h, the pH of the mixture was adjusted to 7 and the TPheC and the ABTS scavenging abilities were examined.
Thermal Stability
To evaluate the thermal stability, ethanol or 80% ethanol extract (2 mg/mL) of FRUF dissolved in deionized water (50 mg/mL, pH 7) was placed in test tubes with screw caps. The test tubes were placed in a boiling water bath (100 °C). Samples were removed after 0, 15, 30, 60, 120, 180, and 240 min and cooled in an ice-water bath, and the TPheC and the ABTS scavenging abilities were examined.
Modeling of the Stability in the Gastrointestinal Tract
100 mL of ethanol or 80% ethanol extract (1 mg/mL) of FRUF in distilled water were mixed with 10 mL of PBS (pH 6.8, 10 mM) and incubated at 37 °C for 2 min (oral condition). Then 0.5 mL of 1 M HCl-KCl buffer (pH 1.5) and 5 mL of pepsin solution (pH 1.5, 32 U/mL in 1 M HCl-KCl buffer) were added to samples. The mixtures incubated at 37 °C for 60 min (stomach condition). Thereafter, 1 mL of 1 M NaHCO3 together with 1 mL of a mixture of bile and pancreatic juice (pH 8.2, 10 mg/mL of pancreatin, 14,600 U/mL of trypsin, and 13.5 mg/mL of bile extract in 10 mM PBS) was added to the mixture, and the pH was adjusted to 6.8. The mixtures were incubated at 37 °C for 3 h (duodenal condition). The results were used for the determination of TPheC and ABTS scavenging abilities of methanol extract during simulated gastrointestinal digestion and were taken at 0, 0.5, 1-4 h.
Ultra-High-Performance Liquid Chromatography-Mass Spectrometry (UHPLC-MS) Analysis
Ethanol extract of FRUF was analyzed using UHPLC (Agilent 1290 system) with Q-TOF-MS (Agilent 6545 system). A ZORBAX SB-C18 column (150 × 3.0 mm, 1.8 µm; Agilent) was used. The column temperature was set to 40 °C. The mobile phase was a mixture of 0.1% formic acid in water (solvent A) and a mixture of 0.1% formic acid in acetonitrile (solvent B) at a flow rate of 0.4 mL/min. Linear gradient elution was applied (0-1 min, 95% A; 1-30 min, 95-70% A; 30-50 min, 70-30% A; 50-56 min, 30-1% A; 56-60 min, 1% A). The extract was diluted to 1 mg/mL with methanol and filtered using a 0.22 µm membrane before use. The sample injection volume was 5 µL. The Q-TOF-MS was operated in positive-ion mode with scan range
The experimental conditions for UHPLC-MS were implemented according to established methodology, 14 guaranteeing the accuracy and dependability of the analysis. exhaustive details.
Oxidative Stability of Oils
The oxidative stability of extra virgin olive oil (EVOO) and cold-pressed sunflower oil (CPSO) was evaluated using established analytical techniques. 14 The detailed experimental procedure was as follows:
Extra virgin olive oil (EVOO) and cold-pressed sunflower oil (CPSO) were placed in separate flasks. Ethanol extract of FRUF was added to the EVOO and CPSO flasks at concentrations of 100 and 25 μg/g, respectively. To compare with the stabilizing effect of methanol extract, EVOO and CPSO were supplemented with synthetic antioxidants TBHQ and BHA at 200 μg/g. A control group was prepared without antioxidants. The flasks were left open and placed in an oil bath at 160 °C to simulate frying. Two samples from each category were removed from the flasks every 4 h for duplicate analysis. The oxidative stability of the oils was evaluated by measurement of the free acidity (percentage of oleic acid), peroxide values (milliequivalents of O2/kg oil), and ultraviolet absorption at 232 and 270 nm (K232 and K270).
Oral Acute Toxicity Study
Twenty adult Kunming mice (19-22 g) were acquired by Liaoning Changsheng Biotechnology Co., Ltd (animal license number SCXK (Liao) 2020–0001; Liaoning, China). Housed mice had free access to food and water under a 12 h light–dark cycle. All rats were reared adaptively for 3 d before starting the experiment. We followed the relevant policies in the Guidelines for the Use of Laboratory Animals developed by Tonghua Normal University. The Institutional Animal Care and Use Committee of Tonghua Normal University approved the experimental protocol (Ethic approval code: 20240037) and the reporting of this study conforms to ARRIVE 2.0 guidelines. 16
The mice were divided into two groups (n = 20) with five males and five females in each group. The mice in the healthy control group received vehicle treatment. The ethanol extract of the FRUF was dissolved in water to a final volume of 10 mL/kg mouse body weight (BW) and then administered to the mice in the FRUF group orally in a single dose of 2000 mg/kg ethanol extract of FRUF. The mice were then continuously observed for 1 h for behavioral changes and toxicity. Intermittent observations were made for next 6 h, and a final observation was conducted at 24 h. At this stage, the survival rate was calculated and we found that no mice died. All mice were euthanized using isoflurane. On the basis of the study results, two doses (150 and 300 mg/kg) were selected for further study.
Hepatoprotective Experiments
The rat liver protection study was performed according to established protocols, 14 comprising four key phases: animal selection, experimental procedure implementation, histopathological evaluation, and biochemical analysis. The specific experimental methodology is described below.
Animals
Adult male Wistar rats (170-200 g) were acquired by Liaoning Changsheng Biotechnology Co., Ltd (animal license number SCXK (Liao) 2020–0001; Liaoning, China). Housed rats had free access to food and water under a 12 h light–dark cycle. All rats were reared adaptively for 7 d before starting the experiment. We followed the relevant policies in the Guidelines for the Use of Laboratory Animals developed by Tonghua Normal University. The Institutional Animal Care and Use Committee of Tonghua Normal University approved the experimental protocol (Ethic approval code: 20240037) and the reporting of this study conforms to ARRIVE 2.0 guidelines. 16
Experimental Protocol
Forty rats were randomly assigned to five groups (
Histopathological Examination
The hepatic tissues of three rats were selected from each group, fixed with 4% paraformaldehyde, dehydrated and washed, and embedded in paraffin. After cutting into 4-µm thick sections, hematoxylin and eosin were used for staining. Observations were made using a light microscope. A digital camera was used to record histopathological changes.
Biochemical Analyses
Before anesthesia, each rat was weighed. The hepatic tissue weight and BW of each rat were used to calculate the viscera index (VI) as follows: VI = viscera weight (g)/BW (g) × 100%. For assessment of biochemical parameters related to liver function, serum samples were analyzed for albumin (ALB), γ-glutamyl transpeptidase (γ-GT), alanine aminotransferase (ALT), aspartate aminotransferase (AST), malondialdehyde (MDA) and catalase (CAT). Hepatic samples were analyzed for ALT, AST, MDA and CAT. All samples were analyzed using commercial kits according to the manufacturer's guidelines.
Statistical Analysis
Statistical analysis was performed to assess the significance of the data, which were expressed as mean ± standard error. One-way ANOVA was first conducted to identify significant differences among groups, followed by post-hoc LSD and Duncan tests for multiple comparisons. These tests were employed to determine intergroup differences in the pharmacological experiments, extraction rates, antioxidant activities, and content determinations. Statistical significance was defined at three levels:
Results
Qualitative Phytochemical Analysis
A primary aim of this research is to investigate how different drying techniques influence the composition of
Both sample sets contained proteins/amino acids, carbohydrates, organic acids, phenolics, tannins, flavonoids, alkaloids, anthraquinones, fats, and volatile oils. Quantitative analyses were performed for all detected phytochemicals except anthraquinones, for which quantification remains incomplete. Analytical results indicated the absence of coumarins, lactones, steroids, triterpenoids, saponins, cyanogenic glycosides, and cardiac glycosides in both samples; consequently, no quantitative analyses were conducted for these compounds.
Extraction Yields
The extraction rates of both dried samples, as illustrated in Table 1, followed a comparable trend; significantly, the 80% ethanol extract yielded the highest extraction efficiency. This may be due to its aqueous ethanol solution, which could result in a rich extract containing proteins, carbohydrates, phenolics, glycosides, and other compounds. The ethanol and water extracts followed closely behind, while the methanol extract had the lowest extraction rate.
Yields Obtained from Extracting FRUF and ORUF Using various Solvents.

Columns denoted by distinct superscripts signify a statistically significant difference (
Quantitative Phytochemical Analysis
TCC
The present investigation evaluated the TCC in various solvent extracts of FRUF, revealing a range of TCC from 623.84 ± 3.99 to 781.35 ± 7.46 mg of glucose equivalents (GE) per gram of extract. In contrast, the TCC of ORUF was found to lie between 574.97 ± 3.91 and 736.08 ± 2.00 mg GE/g extract, as detailed in Table 2. Notable differences were observed among the different groups, with the ethanol extract of FRUF demonstrating the highest TCC.
TCC, TProC, TPheC, TFC, TPAC, TTanC, GC, CTC and TAC of FRUF and ORUF Extracted with Various Solvents.

Columns denoted by distinct superscripts signify a statistically significant difference (
N.T. stands for not tested. The presented values represent the mean ± standard deviation obtained from three separate experiments. FRUF: freeze-dried
TProC
According to the research, the water extract of FRUF demonstrates a notable TProC of 829.08 ± 9.69 mg of bovine serum albumin equivalents (BSAE)/g extract. The water extract of ORUF exhibits a slightly lower TProC, yet it still attains a concentration of 822.05 ± 7.69 mg BSAE/g (Table 2).
TPheC
The experimental data in Table 2 indicates that the TPheC of different extracts varies between 47.97 ± 0.43 and 67.52 ± 1.78 mg gallic acid equivalents (GAE)/g extract. Among the tested extracts, the 80% ethanol extract of FRUF exhibited the highest content, followed by the methanol extract of FRUF, while the water extract of ORUF showed the lowest levels.
TFC
The TFC ranges from 23.77 ± 0.32 to 90.45 ± 0.93 mg quercetin equivalents (QE)/g extract (Table 2), among the tested extracts, the ethanol extract of FRUF exhibited the highest content, followed by the methanol extract of FRUF.
TPAC
As shown in Table 2, there is a minor variation in TPAC among the solvent extracts, with a relatively low concentration spanning from 8.25 ± 0.56 to 15.26 ± 0.89 mg caffeic acid equivalents (CAE)/g extract. The water extract of FRUF exhibited the highest content, followed by the water extract of ORUF.
TTanC, GC and CTC
To evaluate the tannin content in FRUF and ORUF extracted via various solvents, we analyzed key parameters such as TTanC, GC, and CTC, as shown in Table 2. The TTanC values ranged from 35.71 ± 0.20 to 62.12 ± 1.84 mg tannic acid equivalents (TAE)/g extract, with the highest TTanC recorded in the 80% ethanol extract of FRUF.
TAC
The experimental results revealed that only three of these extracts contained TAC, and the concentration was very low, ranging from 4.35 ± 0.01 to 6.50 ± 0.01 mg of BHE/g extract (Table 2). The methanol extract of FRUF exhibited the highest content.
In Vitro Antioxidant Capability
DPPH and ABTS
The ethanol extract of FRUF displayed robust scavenging capabilities in the DPPH assay, achieving an IC50 value of 56.63 ± 1.84 μg/mL. Likewise, the 80% ethanol extract exhibited notable scavenging activity in the ABTS assay, yielding an IC50 value of 30.18 ± 0.96 μg/mL (Table 3).
Assessing the Antioxidant Activity of Different Extracts from FRUF and ORUF Through DPPH, ABTS, Hydroxyl, and Superoxide Radicals.
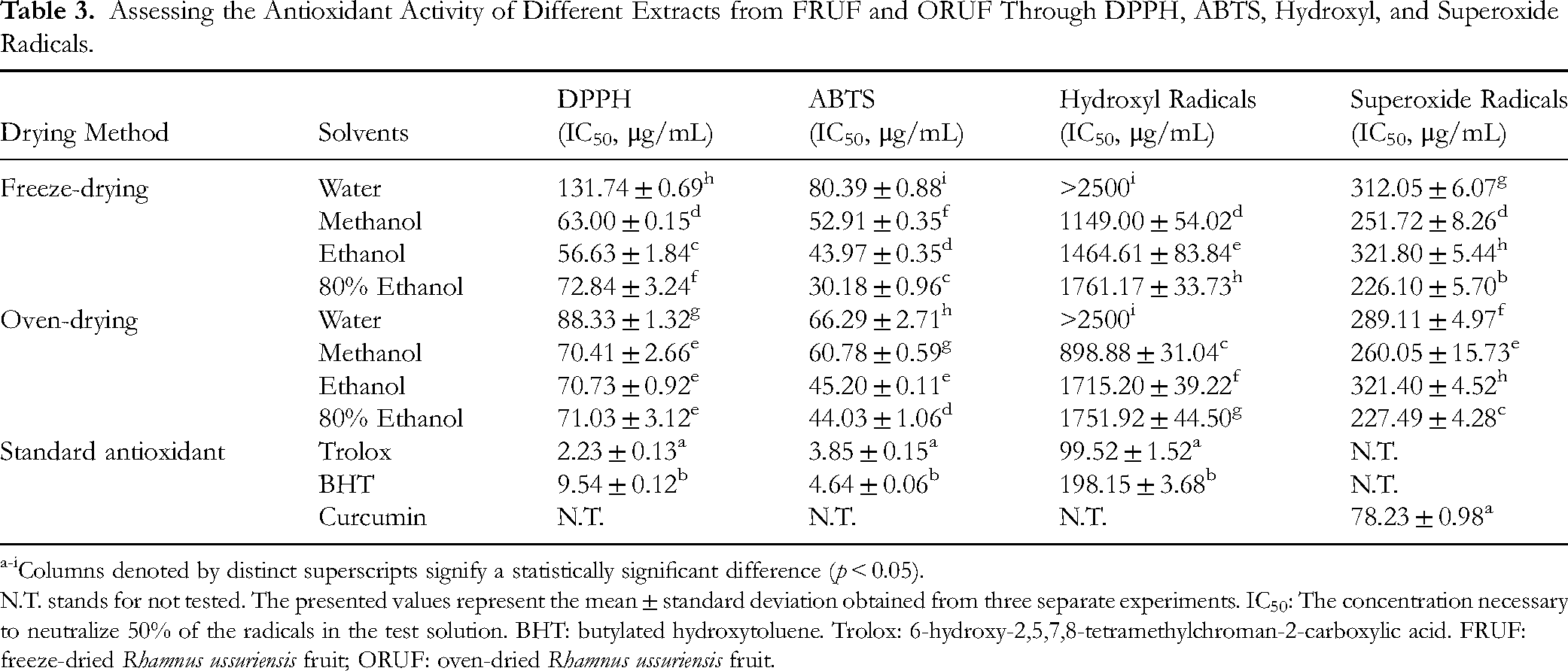
Columns denoted by distinct superscripts signify a statistically significant difference (
N.T. stands for not tested. The presented values represent the mean ± standard deviation obtained from three separate experiments. IC50: The concentration necessary to neutralize 50% of the radicals in the test solution. BHT: butylated hydroxytoluene. Trolox: 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid. FRUF: freeze-dried
Hydroxyl Radicals and Superoxide Radicals
This research indicates that the methanol extract of FRUF and ORUF possesses the greatest scavenging effect on hydroxyl radicals, whereas the 80% ethanol extract of these plants exhibits the highest scavenging activity against superoxide radicals, as detailed in Table 3.
FRAP and CUPRAC
The FRAP assay revealed that all extracts of FRUF and ORUF exhibited moderate ferric reducing ability. Additionally, the CUPRAC assay demonstrated that the 80% ethanol extract of FRUF and ORUF had the best cupric ion reducing ability (Table 4).
Assessing the Antioxidant Properties of various Extracts from FRUF and ORUF Through FRAP, CUPRAC, and Metal ion Chelating.

Columns denoted by distinct superscripts signify a statistically significant difference (
N.T. stands for not tested. TEAC: trolox equivalent antioxidant capacity. EDTANa2: ethylenediaminetetraacetic acid disodium salt. FRUF: freeze-dried
Metal Ion Chelation
This research has shown that the ethanol extract of FRUF has strong iron chelating properties. Furthermore, the water extract of ORUF exhibits significant copper chelating abilities (see Table 4).
H2O2 and Singlet Oxygen
Consequently, the effective removal of H2O2 is of utmost importance. Experimental results indicate that the ethanol extract of FRUF demonstrates notable H2O2 scavenging capabilities (IC50, 426.05 ± 15.16 μg/mL)(Table 5). Additionally, singlet oxygen, experimental results indicate that the 80% ethanol extract of FRUF exhibits notable singlet oxygen scavenging activity (IC50, 785.30 ± 4.89 μg/mL), rivaling the performance of the positive control, ferulic acid (Table 5).
Assessment of the Antioxidant Properties of various Extracts from FRUF and ORUF Through H2O2, Singlet Oxygen,

Columns denoted by distinct superscripts signify a statistically significant difference (
N.T. stands for not tested. BHA: butyl hydroxyanisole. FRUF: freeze-dried
β -Carotene Decolorization
This study found that the ethanol extract of FRUF exhibited comparable antioxidant activity (876.21 ± 22.38) to the positive controls BHT (878.26 ± 30.86;

Alterations in
HClO and NO
The HClO scavenging assay revealed that only the ethanol extract of FRUF displayed weak activity, while other extracts showed no detectable effects (Table 5). In contrast, the NO scavenging experiment demonstrated that multiple FRUF and ORUF extracts exhibited significant scavenging capacity. Specifically, the ethanol extract of FRUF exhibited the most significant effect, approaching the efficacy of the positive control, curcumin (Figure 3).

Alterations in Nitric Oxide Absorbance Were Observed When Exposed to various Extracts Derived from FRUF and ORUF. FRUF: Freeze-Dried
Stability Studies of Ethanol Extract and 80% Ethanol Extract of FRUF
This study evaluated the antioxidant capacity and stability of both the ethanol and 80% ethanol extracts of FRUF through three stability tests. The results of these tests are illustrated in Figures 4-6. These findings revealed that the TPheC and ABTS scavenging potential of the ethanol extract of FRUF maintained a relatively consistent level across varying pH values. In contrast, as the pH level increased, the TPheC content and ABTS scavenging activity of the 80% ethanol extract of FRUF initially rose but subsequently declined, suggesting its instability in both highly acidic and highly alkaline environments. This suggests that the phenolics in this extract may be phenolic glycosides, which are unstable and prone to hydrolysis in strong acid and alkali conditions.

Stability Assessments of the Ethanol Extract and 80% Ethanol Extract of FRUF at Different pH Levels Were Conducted Using TPheC (A) and ABTS (B) Assays. FRUF: Freeze-Dried

Thermal Stability Assessments of the Ethanol Extract and 80% Ethanol Extract of FRUF Were Conducted Using TPheC (A) and ABTS (B) Assays. FRUF: Freeze-Dried

Stability Assessments of the Ethanol Extract and 80% Ethanol Extract of FRUF in an in Vitro Simulation of the Human Digestive System Were Conducted Using TPheC (A) and ABTS (B) Assays. FRUF: Freeze-Dried
Within the initial 30 min of the heating process, notable variations were observed in both the TPheC and ABTS scavenging activity of both the ethanol and 80% ethanol extracts of FRUF. Significantly, the fluctuations were more pronounced in the 80% ethanol extract of FRUF. After this initial period, both parameters stabilized. However, the TPheC of the ethanol extract was consistently higher compared to that of the 80% ethanol extract.
During in vitro digestive system stability tests, both the TPheC of the ethanol extract and that of the 80% ethanol extract of FRUF maintained overall stability. However, the pure ethanol extract exhibited greater stability. It's worth noting that, under gastric conditions, the ABTS scavenging activity of the extracts significantly decreased, but it significantly rebounded under duodenal conditions. The 80% ethanol extract demonstrated a more pronounced fluctuation in activity. While ABTS is generally stable under acidic conditions, excessively acidic environments may compromise its stability, leading to decomposition or alteration that could reduce its efficacy as a radical scavenger.
UHPLC-MS Analysis
The chemical composition of the ethanol extract obtained from freeze-dried

Chemical Structures of the Compounds Identified in the Ethanol Extract of Freeze-Dried
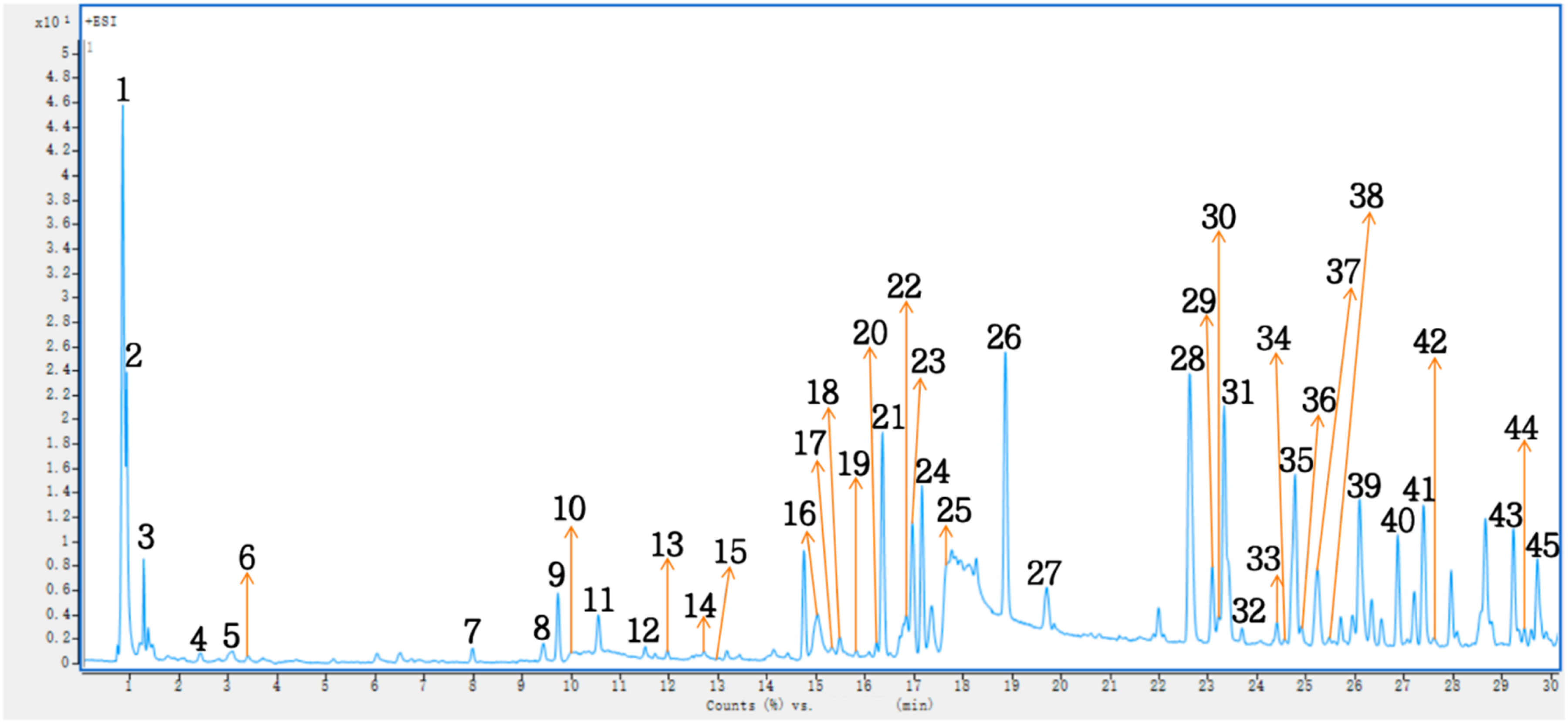
Positive-ion Mode UHPLC-MS Findings of Ethanol Extract of Freeze-dried
Identified Compounds in Ethanol Extract of Freeze-Dried Rhamnus ussuriensis Fruit.
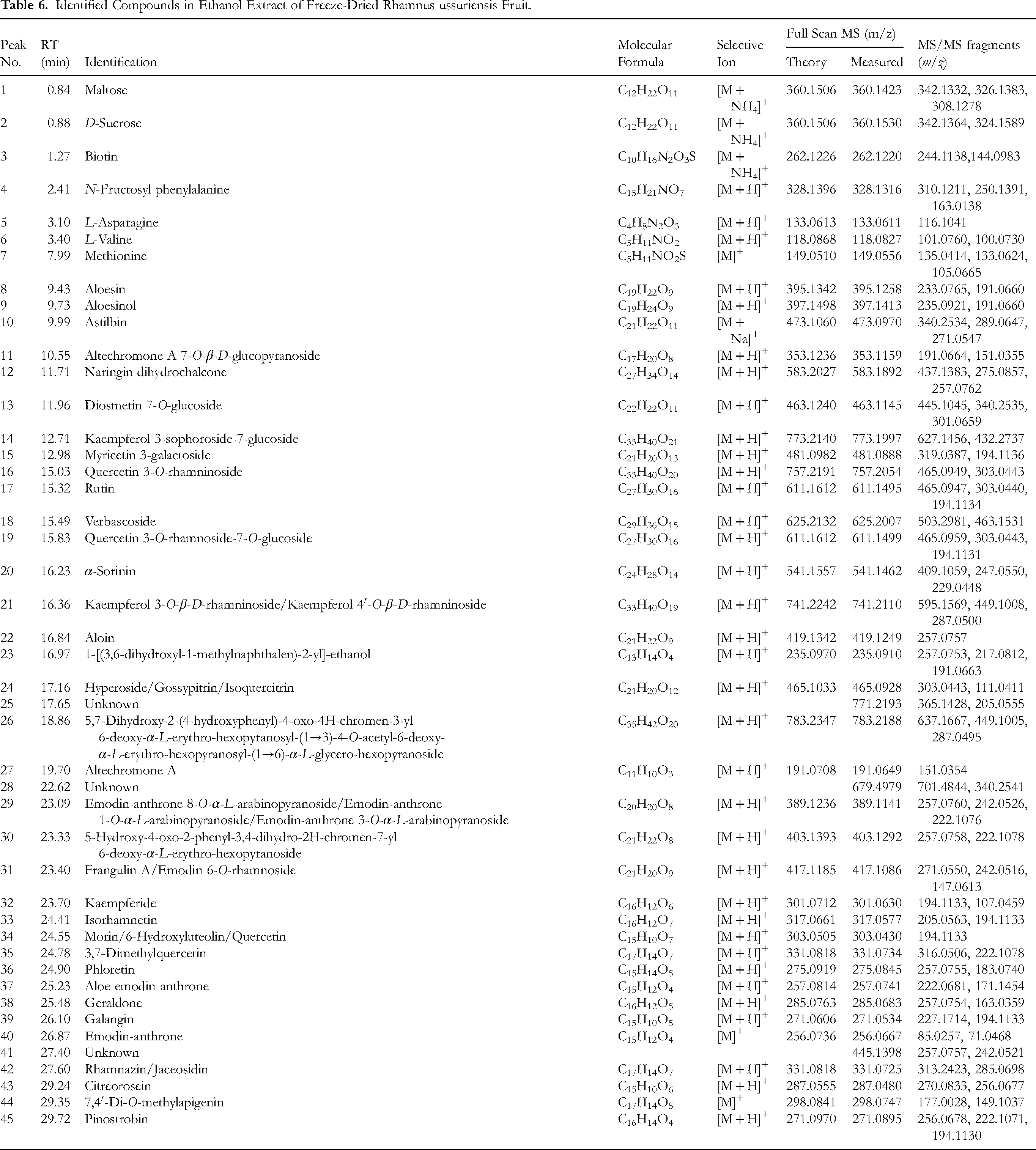
Peak 1 was conclusively identified as maltose by aligning its fragment ions, including
The
Peak 21, with an
Peak 31, which appeared at
Oxidative Stability Studies of Oils
The research indicates that introducing two doses of the FRUF ethanol extract into EVOO significantly decreased K270 values, outperforming BHA and TBHQ (Figure 9). However, for K232 values, peroxide levels, and acid values, the impact of these doses of the extract was minimal. Yet, it still demonstrated superior performance with significantly lower acidity compared to TBHQ (

Alterations in K232 and K270 Concentrations Were Observed in EVOO (A,C) and CPSO (B,D) Supplemented with BHA and TBHQ, as Well as Varying Amounts of the Ethanol Extract Derived from FRUF at 160 °C. BHA: Butyl Hydroxyanisole; TBHQ: Tertiary Butylhydroquinone; FRUF: Freeze-Dried

Alterations in the Levels of Acidity Values and Peroxide Values were Observed in EVOO (A,C) and CPSO (B,D) Supplemented with BHA and TBHQ, as Well as Varying Amounts of the Ethanol Extract Derived from FRUF at 160 °C. BHA: Butyl Hydroxyanisole; TBHQ: Tertiary Butylhydroquinone; FRUF: Freeze-Dried
Oral Acute Toxicity Study
An evaluation of the acute oral toxicity of the ethanol extract of FRUF was carried out using mice. In this study, a single dose of 2000 mg/kg of the extract was administered orally to twenty mice. Fortunately, all mice remained unaffected for 24 h, suggesting that the ethanol extract of FRUF lacks highly toxic components and poses no notable acute toxicity risk.
Hepatoprotective Activity
The outcomes indicated that both high and low doses of the ethanol extract evidently lowered the viscera index in rats compared to GII (

The Results of Treating Rats with Liver Injury Using the Ethanol Extract of FRUF on Their Hepatic Viscera Index are Presented, with Values Indicated as the Mean ± SEM (
A research found that rats in GII had significantly lower albumin levels compared to those in GI (

Impact of the Ethanol Extract of FRUF on Serum ALB (A) and
At the same time, rat serum levels of ALT, AST, and
The research outcomes unequivocally demonstrate that both liver tissue and serum ALT and AST levels in the model group were significantly elevated compared to the control group (

Impact of the Ethanol Extract of FRUF on Serum ALT (A), Liver Tissue ALT (B), serum AST (C), and Liver Tissue AST (D) Levels in Rats with Liver Injury. Data are Presented as Mean ± SEM (
After

The Influence of the Ethanol Extract of FRUF on Serum CAT Levels (A), Liver CAT Levels (B), Serum MDA Concentrations (C), and Liver MDA Levels (D) in Rats with Induced Liver Injury is Presented. Data Are Presented as Mean ± SEM (
After
The histopathological findings are illustrated in Figure 15. The control group exhibits a clear liver tissue structure, characterized by a normal cellular arrangement and absence of inflammatory cell infiltration in the vicinity of the portal vein (depicted in Figure 15A). Conversely, in GII, the liver tissue structure undergoes disruption, marked by the disappearance of hepatic cords and the presence of single-cell necrosis (no-tailed arrow) (Figure 15B). Additionally, there is a notable presence of inflammatory cells (long-tailed arrow), which becomes apparent under closer inspection using a 400x microscope, more prominent infiltration and necrosis of inflammatory cells were observed in GII (Figure 15C). It is worth noting that the low-dose group demonstrated considerable improvement in liver cell integrity, showing a relatively reduced count of inflammatory and necrotic cells in comparison to GII (as illustrated in Figure 15D). When compared to the GIII, the high-dose group demonstrated even better results, with reduced inflammation and necrotic cells (Figure 15E). It is worth mentioning that the GV showed the most pronounced therapeutic effect, with a substantial decrease in inflammation and necrotic cells, along with a relatively preserved liver cell structure (Figure 15F).

Histological Examination of Liver Tissue Sections from Various Groups at 200× Magnification was Conducted. The Single-cell Necrosis is Indicated by a No-tailed Arrow, While Inflammatory Cells are Marked by a Long-tailed Arrow. Histological Examination of Liver Sections in Different Groups (200× Magnification). (A): Control Group (200× Magnification); (B):
Discussions
Qualitative Phytochemical Analysis
However, the experimental results for alkaloids were inconsistent, yielding both negative and positive outcomes. Meanwhile, the Bertrad's reagent tests for both samples also showed inconsistent results, potentially due to the low alkaloid content in the samples. The precise content will require further determination through quantitative analysis. Regarding the four flavonoids tested in the two samples, three were positive and one was negative. This could be attributed to two factors: firstly, the presence of dark-colored components in the fruit samples, which hindered observation; secondly, the low flavonoid content in the sample combined with the low sensitivity of the experiment, making it challenging to detect positive results. However, given the three positive results, it is unlikely that the flavonoid content in the sample is low. Overall, while the different drying methods produced some variability in detection rate, the core profile of active ingredients remained largely consistent.
Extraction Yields
Upon analyzing different plant species, we observed a clear trend: extracts derived from polar solvents have often exhibited antioxidant properties.15,49-50 Consequently, this study employed four solvents—water, ethanol, 80% ethanol, and methanol—to extract FRUF and ORUF. The aim of the extraction process was to assess the content and antioxidant capabilities of various active compounds present in the two dried samples. Upon completion of the extraction, vacuum distillation was utilized to guarantee complete evaporation of all remaining solvents. Rigorous measures were taken to ensure thorough solvent removal, preventing any adverse effects on these experimental outcomes or their practical applications.
Quantitative Phytochemical Analysis
TCC
It is worth mentioning that the TCC in the water extract was relatively lower, potentially due to a decrease in the plant's carbohydrate content and a corresponding increase in glycoside content. Although water is highly effective as a solvent for isolating highly polar carbohydrates such as oligosaccharides and polysaccharides, glycoside compounds, including phenolic glycosides and flavonoid glycosides, may not dissolve readily in water because of their lower polarity. In contrast, organic solvents such as methanol or ethanol, which possess better solubility properties, could more effectively extract a broader spectrum of carbohydrate-related compounds, encompassing glycosides.
TProC
Plant-based protein provides a wide range of health advantages and constitutes a highly nutritious, easily digestible, and well-absorbed source for the human body. It demonstrates various health-promoting biological activities, including anti-inflammatory, antibacterial, antidiabetic, and antioxidant properties.
51
A study was conducted utilizing the method prescribed by AOAC International to determine the protein content in
TPheC
Phenolics and their derivatives are naturally occurring compounds found abundantly in medicinal plants, vegetables, and fruits, and constitute the primary antioxidants in the human diet. Numerous epidemiological studies have revealed their anti-cancer properties,
53
emphasizing their significant impact on health, especially for managing diabetes and cardiovascular diseases.
54
An experiment utilizing 80% methanol to extract different parts of
TFC
Flavonoids, prevalent in grains, fruits, vegetables, and medicinal plants, serve as quintessential representatives of polyphenolic compounds and play pivotal roles across diverse biological processes, possess multiple biological activities, and are vital in preventing metabolic diseases.
55
Studies have shown that 70% ethanol extracts from the leaves, stems, and fruits of 13 species of Rhamnaceae plants contain flavonoids.
10
Upon analyzing the results of this experiment, it was also discovered that various extracts of
TPAC
Phenolic acids are a type of organic acid that exhibit various biological activities and health benefits, widely found in nature. They exhibit a range of beneficial properties, including antibacterial, antioxidant, and anti-inflammatory effects. 56 The experimental results further revealed that R. ussuriensis fruit extracts contain moderate levels of phenolic acids.
TTanC, GC and CTC
Tannins constitute a widespread and diverse class of polyphenolic compounds that are abundant in plant material, are well-known for their antioxidant and antibacterial characteristics.57-58 Broadly speaking, these compounds could be categorized into two structural types: condensed tannins and hydrolyzable tannins, with gallotannin being a prominent example of the second type. It is noteworthy that both FRUF and ORUF extracts were abundant in GC, while CTC was undetectable in all four extracts of both FRUF and ORUF. These observations provide significant information for qualitative analyses. A previous study has reported that the water extract of freeze-dried
TAC
Alkaloids, nitrogen-containing compounds prevalent in plants, are renowned for their potent anti-inflammatory, anti-cancer, and detoxifying capabilities.
60
According to some literature, alkaloids have been detected in all extracts of
This study provides valuable insights into
In Vitro Antioxidant Capability
DPPH and ABTS
Currently, the most widely used and effective method for evaluating antioxidant activity involves assessing its ability to scavenge free radicals. A widely used method for assessing antioxidant activity involves examining stable free radicals like DPPH and ABTS, which can be determined using spectrophotometry and demonstrated by changes in color.
62
DPPH, being fat-soluble, and ABTS, being water-soluble, serve as excellent tools for screening and assessing natural antioxidants.63-64 Significantly, among the
Hydroxyl Radicals and Superoxide Radicals
Reactive oxygen species (ROS) are crucial in regulating various physiological functions of organisms. Oxidative stress arises when the production of ROS exceeds the capacity of the cellular defense system, serving as a fundamental principle in the pathophysiology of numerous diseases. 65 Within the body, hydroxyl radicals are a common type of ROS that can cause significant damage to biomolecules like proteins and nucleic acids. 66 Superoxide anion radicals, which are single electron reduction products of oxygen molecules, are another type of ROS produced in the body. 67 Both types of free radicals are closely associated with various inflammatory diseases, including chronic inflammation and cancer.65-66 Therefore, scavenging these two types of free radicals is essential for preventing these diseases.
The water extracts from both drying methods exhibited relatively high IC50 values (>2500 µg/mL), indicating limited hydroxyl radical scavenging capacity
FRAP and CUPRAC
Assessing the antioxidant potential of a sample is crucial in the search for therapeutic agents, often entailing the evaluation of its capacity to reduce copper and iron ions. Two prevalent methods for this evaluation are the FRAP and CUPRAC assays.
70
The FRAP assay operates in acidic conditions, while the CUPRAC assay operates under neutral conditions, which mirror the physiological environment more closely. Both methods are renowned for their economy and simplicity.
71
A study has shown that two flavonoids found in
Metal Ion Chelation
Assessing a sample's capacity to chelate metal ions is essential for gaining insights into its antioxidant potential.
73
The existence of iron and copper ions could accelerate the Fenton reaction, thereby elevating oxidative challenge within living systems. By chelating these metal ions, their reactivity is decreased and their harmful effects are negated.
74
The results highlight the encouraging prospects of
H2O2 and Singlet Oxygen
H2O2 plays a crucial role in the aging process of plants, functioning as a signal that accelerates aging in various species. It accomplishes this by inhibiting the activity of antioxidant enzymes, which leads to an excessive buildup of ROS, causing oxidative stress and cellular damage.
76
In humans, the buildup of H2O2 is closely associated with the advancement of diseases like Parkinson's, Alzheimer's and cerebrovascular accidents.
77
Furthermore, two flavonoids found in
Additionally, singlet oxygen, another type of ROS, plays a pivotal role in disease initiation, progression, and the aging process. 78 The results support further targeted experiments on singlet oxygen scavenging particularly tailored for the plant genus.
β -Carotene Decolorization
The
HClO and NO
HClO, a weakly acidic yet highly oxidizing disinfectant, functions as a swiftly reactive molecule produced by the human immune system in reaction to the detection of pathogens or potentially damaging stimuli. This property renders HClO highly effective in disinfection and sterilization. However, in certain contexts, it may also be linked to oxidative processes within organisms.
81
NO serves as a vital signaling molecule within biological cells, overseeing a wide range of cellular functions and engaging in the physiological mechanisms of the cardiovascular and immune systems, while also adjusting biological signals. However, high levels of NO can cause oxidative stress, leading to various human diseases.
82
Among the results of inhibiting NO production from extracts of thirteen species of
The use of an oven for sample drying presents both significant advantages and notable limitations that must be acknowledged. One of its primary benefits is its ability to efficiently and rapidly eliminate moisture from samples, particularly suited for batch processing, which drastically enhances work efficiency. Additionally, oven equipment is relatively straightforward, user-friendly, and cost-effective. The control over temperature and time parameters is straightforward, leading to a highly repeatable and consistent drying process, making it an excellent choice for standardized operations in both laboratory and industrial settings. A key disadvantage of oven drying arises when the sample contains antioxidant components such as flavonoids and phenolics, as excessively high temperatures can degrade these active ingredients, leading to loss of biological activity and nutritional value. 83 A report confirm that drying temperature significantly impacts plant components—for instance, the same water extract dried at 60 °C yields 118 more degradation-derived compounds than at 40 °C, 84 highlighting the sensitivity of thermolabile phytochemicals to thermal processing. To optimize the balance between drying efficiency and thermolabile component protection, the drying temperature was maintained at 50 °C based on literature evidence 84 and empirical optimization. This temperature regime was selected to simultaneously ensure: 1) effective moisture removal, 2) maximal retention of antioxidant constituents, and 3) preservation of functional properties.
In recent years, freeze-drying technology has emerged as a promising alternative with numerous advantages. Firstly, since it operates under vacuum conditions, it can safeguard easily oxidizable components and maintain their original chemical structure. 85 Secondly, the dried sample is lightweight, porous, and sponge-like, enabling it to quickly and fully dissolve upon rehydration, almost instantaneously restoring its original shape. This characteristic is advantageous for subsequent chemical analysis and applications. Finally, freeze-drying can remove over 95% of moisture, allowing the dried product to be stored for extended periods without deterioration, making it suitable for long-term storage and transportation. Despite its benefits, freeze-drying also has its disadvantages. Firstly, the equipment required for freeze-drying is complex and consumes a significant amount of energy, leading to relatively high costs. Secondly, when dealing with a large number of samples, the freeze-drying process can be time-consuming, potentially impacting experimental efficiency.
Given these considerations, this experimental design incorporated two drying methods: freeze-drying and oven-drying at 50 °C. This dual methodology offers an intriguing and significant investigation into how various drying methods influence the chemical composition and antioxidant properties of
Our previous extraction studies employed eight solvents, including four non-polar solvents: ethyl ether, ethyl acetate, dichloromethane, and
The quantified antioxidant compounds—TPheC, TFC, and TTanC—showed strong correlations with antioxidant activity: 1) DPPH/ABTS scavenging: The ethanol extract of FRUF exhibited the highest TPheC and TFC, corresponding to its superior DPPH and ABTS radical scavenging capacities, indicating that phenolics and flavonoids are key contributors to free radical neutralization. 2) FRAP/CUPRAC assays: The 80% ethanol extract's high TTanC aligned with its significant reducing power, suggesting tannins play a role in electron transfer mechanisms. 3) metal chelation: The ethanol extract's effective iron chelation correlated with its flavonoid content, consistent with the known metal-chelating properties of flavonoid glycosides such as rutin and quercetin derivatives. These findings demonstrate that the antioxidant efficacy of
The extensive experiments yielded two crucial findings that greatly enhance the knowledge of the antioxidant characteristics of
This research has not only provided valuable insights into the antioxidant properties of
Stability Studies of Ethanol Extract and 80% Ethanol Extract of FRUF
Based on these findings, the TPheC of the ethanol extract of FRUF is higher, and its active ingredients are more stable, which is expected to preserve its efficacy across various physiological conditions. These findings establish a basis for the storage, processing, and potential utilization of these extracts in pharmaceuticals and foods, leading to the selection of the ethanol extract for further in-depth investigation.
UHPLC-MS Analysis
The meticulous chemical profiling of the ethanol extract provides valuable insights into its diverse compound composition. Remarkably, many of the identified compounds have previously been isolated from the
Based on the UHPLC-MS findings and antioxidant assays, the structural features of key compounds were closely linked to their radical scavenging capacity. Flavonoid glycosides, such as quercetin derivatives and kaempferol glycosides, exhibited strong DPPH and ABTS scavenging activities due to their phenolic hydroxyl groups and conjugated π-electron systems. The catechol (3′,4′-dihydroxy) groups in quercetin derivatives enhanced hydrogen donation and electron delocalization, 86 while glycosylation at the 3-position (eg, rutin, hyperoside) stabilized the flavonoid radical intermediate, prolonging antioxidant activity. 87 Phenolic acids and anthraquinones, including aloesin and emodin-anthrone, showed moderate activity in metal chelation and H2O2 scavenging, as their carbonyl and hydroxyl groups facilitated redox reactions, with the anthraquinone core in emodin derivatives enabling resonance stabilization of radicals. 88 Significantly, the methoxy group in kaempferide reduced activity compared to quercetin analogs, underscoring the importance of free hydroxyl groups for radical neutralization. Overall, the structure–activity relationships revealed that hydroxylation pattern, glycosylation, and conjugation were critical determinants of antioxidant efficacy.
Oxidative Stability Studies of Oils
When oil is subjected to high temperatures, its chemical stability undergoes severe deterioration, resulting in a substantial acceleration of the oxidation process. In this process, high temperatures stimulate unsaturated bonds in oil molecules to generate free radicals, which trigger chain reactions and lead to the formation of polymers. This not only alters the fundamental properties of the oil but may also generate various compounds harmful to human health, such as carbonyl compounds and epoxy groups, which possess differing levels of toxicity and pose various health risks. 89 Consequently, adding antioxidants to oils can effectively enhance their stability. Nevertheless, employing synthetic antioxidants involves specific hazards.90-91 As a result, the food industry has a pressing demand for natural antioxidants. The peroxide and acid values are commonly used to gauge the initial oxidation level of oils. Furthermore, the K232 and K270 indices are widely adopted to evaluate the primary and secondary oxidation stages of oils, respectively. 92 These metrics are crucial for monitoring oil oxidation, thereby safeguarding the safety and quality of fried foods.
These findings suggest that the ethanol extract of FRUF is suitable for use in CPSO, which contains polyunsaturated fatty acids with multiple double bonds that can lead to instability. However, it is not suitable for EVOO, which is a representative of monounsaturated fatty acids. Further research is necessary to consider its use as an oil stabilizer.
Oral Acute Toxicity Study
According to a prior study, it was announced that
Hepatoprotective Activity
The liver, which is crucial for many biological processes, is prone to injuries that can lead to liver diseases.
Serum albumin levels serve as a crucial indicator of liver function. As liver function declines, the synthesis of albumin decreases, leading to reduced levels of serum albumin. 95 Elevated ALT and AST levels serve as indicators of liver damage. 96 The decrease in liver enzyme levels implies a mitigation of liver damage and inflammation. 97 These findings are akin to those observed with silymarin treatment, with the high-dose extract exhibiting a more pronounced effect.
These results indicate its therapeutic potential for liver-related diseases. A notable study limitation involves the incomplete assessment of key antioxidant enzymes, including superoxide dismutase, peroxidase, and glutathione peroxidase. These enzymatic biomarkers will be prioritized in subsequent investigations to comprehensively evaluate the antioxidant defense system.
The ethanol extract of FRUF exhibits dual antioxidant efficacy, demonstrated through both hepatoprotective effects in biological systems and stabilization of CPSO in lipid matrices.
Conclusions
This study systematically evaluated the effects of different drying methods (freeze-drying vs oven-drying) on the chemical composition and antioxidant properties of
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251349455 - Supplemental material for Screening the Extracts from Two Drying Methods of Rhamnus ussuriensis J.J. Vassil.: Assessment According to Active Ingredient Content, Antioxidant Potency, and Hepatoprotective Effects in Rats
Supplemental material, sj-doc-1-npx-10.1177_1934578X251349455 for Screening the Extracts from Two Drying Methods of
Footnotes
Abbreviations
Acknowledgements
We thank professor Junlin Yu, from Tonghua Normal University, collected and identified plants for us.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Development Plan Project of Jilin Province, China [grant number 20250601025RC]; and the independently selected topic project for basic scientific research of the Education Department of Liaoning Province, China [grant number LJ212410163034].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
