Abstract
Background
Moringa oleifera has long been cultivated in various parts of the world and has proven beneficial properties and applications. This study aimed to explore the potential of non-destructive techniques (Raman and infrared spectroscopy) to determine the qualitative metabolic composition of Moringa oleifera leaves.
Methods
Gas chromatography was used to study the extract's chemical composition. Infrared, Raman, and fluorescence spectroscopy were used to analyze the leaf extract's optical properties and chemical groups.
Results
Selective fluorescence excitation at different wavelengths was employed to obtain the excitation-emission matrices of the ethanol and methanol extracts of the plant. The excitation-emission matrices reveal two distinct regions: • The first was in the 450–600 nm range. • The second was in the 650–775 nm range.
Infrared spectroscopy identified peaks in the 3450–3200 cm−1 region, associated with hydroxyl groups, and around 2900 cm−1, linked to aliphatic C-H and CH2 vibrations, characteristic of fatty acids in the leaves of Moringa oleifera. The peak at 1625 cm−1 is related to the C = O stretching vibrations (lignin structures). Peaks coresponding to aromatic rings and others were also observed. Raman spectroscopy detected vibrations of methyl and methylene groups, C-C stretching in acyclic residues, C-C aldehyde vibrations, etc.
Conclusion
The results demonstrated that using these three techniques in tandem provides excellent results for the qualitative composition of plants. Excitation-emission matrices can be used in future studies to obtain fingerprints of plant extracts from different regions of the world by excitation in the blue part of the visible spectrum.
Introduction
Moringa oleifera thrives in tropical and subtropical regions of the world but is believed to have originated from Afghanistan, Bangladesh, India, and Pakistan. 1 All the parts of the plant were usable. The bark is used to treat ulcers, 2 hypertension, 3 toothaches, 4 wound healing, and skin infections. 5 Its roots are used in cases of helminthiasis, 6 paralysis, 7 kidney stones, 8 and liver diseases. 9 The leaves are used to alleviate insomnia 10 and wound healing, 11 and are also utilized in treating dysentery 12 and colitis. 13 The flowers serve as an aphrodisiac and aid in spleen enlargement. 14 The plant is rich in bioactive compounds such as phenolic acids, saponins, tannins, amino acids, proteins, tocopherols, carbohydrates, and unsaturated fatty acids. 15 It contains a plethora of vitamins (A, B1, B2, B3, B7, C, D, E, and K) and minerals such as potassium, calcium, iron, magnesium, phosphorus, and zinc. 16
The use of Moringa oleifera in various industries is growing rapidly. In the food industry, it is incorporated into functional foods and dietary supplements due to its high nutritional content. In the pharmaceutical sector, its extracts are investigated for potential therapeutic effects, including cancer prevention, cardiovascular health, and diabetes management. Furthermore, its bioactive components contribute to cosmetic formulations, promoting skin hydration and anti-aging effects.
The standard HPLC technique was used for extracts of Moringa Oleifera in ethanol, methanol and water to evaluate the content of phenolic and flavonoid compounds.17,18 Also Gas Chromatography and Mass Spectroscopy (GC-MS) has been used to measure the contents of phytocomponents in plant methanolic extracts of Moringa oliefera whole leaf19,20 in parallel with FTIR spectra measurements in the range 500 cm−1 to 4000 cm−1 to reveal the presence of functional groups (–OH, –CH3, =C-H, -CO-OH, C = O, C = C, Ar-OH, CH2). The FTIR technique has been used to reveal the contents of the mentioned groups in both extracts and powder of Moringa leaves.21-23
Various analytical techniques are most commonly used to determination the elemental and metabolic composition of Moringa oleifera. Plant samples are acid-decomposed to determine the concentration levels of their elemental composition. Various extracts are subjected to chromatographic analyses to determine the metabolic profile. All of these techniques require time and consumables. Recently, researchers have been focusing on investigating the possibilities of various non-destructive techniques, as well as their combination, for assessing the metabolic profile of the plant. For example, it has been found that Fourier transform infrared spectroscopy (FT-IR) combined with an appropriate regression analysis method can be used to predict phenolic compounds. 24 El Orche and co-authors use the same approach to detect counterfeit products containing Moringa. 25 In order to optimize the time for harvesting the best quality leaves of Moringa oleifera, energy-dispersive x-ray fluorescence spectrometry was used, this study highlights that there are seasonal variations in the concentrations of different elements and the amounts of bioactive compounds. 26 The potential of non-destructive and rapid spectroscopic methods has been demonstrated for determining the phytochemical and elemental profile of food waste to assess their quality and in an effective way for their utilization in nutritious diets. 27 The LIBS (Laser Induced Breakdown Spectroscopy) technique was applied 28 to evaluate the contents of different elements (C, Fe, Mg, Ca, K, O, Na, H, S and N) in aqueous extracts which exhibit distinct emission peaks from 247.83 nm for carbon to 868.028 nm for nitrogen.
The analysis of the literature review suggests that non-invasive techniques have the potential for qualitative evaluation of extracts as an alternative to expensive and time-consuming methods such as liquid and gas chromatography.
However, to our knowledge no studies have been performed on the characteristic fluorescence spectra of Moringa oleifera powder or extracts.
As evidenced by the literature each of the spectral methods used is characterized by dufferent complexiti and provides specific information on the content of elements, functional groups or ingtedients in powders or different extracts Moringa oleifera, the most widely used being the HPLC and the FT-IR nethods.
The objectives of this research are:
To present a comparison of the different spectral methods outlining their paricular capabilities.To investigate the potential of non-destructive spectroscopic techniques (Raman and infrared spectroscopy) for qualitative analysis of the metabolic composition of Moringa oleifera leaves. To provide an alternative, rapid and economical method for qualitative assessment of the composition of plant extracts from the leaves of the plant compared to traditional methods of gas and liquid chromatography.
Materials and Methods
Plant Material
The drying procedure was carried out at a regulated temperature of 40 °C in a laboratory dryer for 4 min. The pharmacopoeia requires a moisture percentage of 12%, however the final moisture content was less than 10%. A laboratory homogenizer (Bosch, Germany) was used to finely grind the dried leaves under controlled laboratory settings. After passing through a 0.5 mm mesh screen, the powder was kept in dry plastic containers until it could be analyzed. The selected drying temperature is optimal for preserving compounds such as polyphenols, flavonoids, and vitamins. Maintaining moisture levels below 10% ensures the absence of microbial growth, mold development, and rapid oxidation. Similar parameters were adopted by Iwansyah et al. 29
Extract Preparation
Extracts were prepared by steeping 1 g of ground dry leaves of Moringa oleifera in 10 mL of methanol or 50% or 70% ethanol (Merck, Darmstadt, Germany). The samples were vortexed (Vortex, Ika, Vortex 3, Germany) for 30 s and left in the dark at room temperature for 72 h. The resulting extracts were filtered through a filter paper and stored at 4 °C. The extraction and derivative procedures concirning GC-MS analisys were carried out following the methods outlined in the previous work (cite). 30
Chemical Component Analysis of Dried Moringa Oleifera Leaves
The analysis was conducted using a 7890A gas chromatograph (Agilent Technologies) coupled with a 5975C mass-selective detector (Agilent Technologies). Separation was performed on a 30 m × 0.25 mm (i.d.) DB-5 ms silica-fused capillary column with a 0.25 µm poly(dimethylsiloxane) coating as the stationary phase. Helium served as the carrier gas at a flow rate of 1.0 mL/min. The injector and transfer line temperatures were maintained at 250 °C.
The oven temperature program began at 100 °C for 2 min, followed by an increase of 15 °C/min to 180 °C, then 5 °C/min to 300 °C, with a final hold for 10 min. The injection volume was 1 µL, split mode (10:1). The mass spectrometer operated in electron impact (EI) mode at 70 eV, scanning a mass range of 50–550 m/z.
Retention indices (RI) were calculated by injecting a mixture of aliphatic hydrocarbons (C10–C40) from Sigma under the same temperature program. AMDIS software (Automated Mass Spectral Deconvolution and Identification System, NIST, Gaithersburg, MD, USA), version 2.73, was used with a standard n-hydrocarbon calibration mixture to determine RIs. Compound identification was achieved by comparing the RIs and spectral data with references from a custom reference library, the Golm Metabolome Database (http://csbdb.mpimp-golm.mpg.de/csbdb/gmd/gmd.html, accessed September 1 2024), and the NIST’08 database (National Institute of Standards and Technology, USA).The methodology for the quantitative assessment of polar components is described in. 31
Fluorescence Measurements
Fluorescence spectroscopy of the leaves and their extracts with various organic solvents was conducted using an Ocean Optics QE65000 fiber-optic spectrophotometer with a broadband Energetiq laser-driven light source (190-2500 nm) in combination with an Ocean Optics MonoScan 2000 fiber optic monochromator. The measurement scheme and methodology are detailed by Nikolova et al, 2023. 32 A similar measurement setup was used for different extracts, the excitation range being the same. The samples are excited at wavelengths ranging from 220 nm to 720 nm, increasing in steps of 10 nm, with a spectral bandwidth of approximately 15 nm. The sample is illuminated by a 800 μm core quartz polymer fiber, and the fluorescence is collected by a same size receiving fiber at a 45° angle relative to the excitation one to minimize reflected and scattered light. The integration time was 5 s.
Infrared Spectroscopy
Fourier transform infrared (FTIR) spectra were obtained using a Nicolet Magna-IR 560 FTIR spectrophotometer (Thermo Scientific Co., Waltham, MA, USA). The frequency range was 4000-400 cm−1 . The number of scans was set to 32 per sample with an accuracy of 10 cm−1 to reduce background noise generated by the instrument. Moringa oleifera leaves were mixed with KBr and pressed into a translucent circular thin film prior to analysis.
Raman Spectroscopy
Raman spectroscopy was performed using a Thermo Scientific instrument with a laser line at 1064 nm. We used a laser power of 50 mW, providing sufficient signal without damaging the sample and without increasing the overall fluorescence background. To avoid thermal degradation of the sample, the optimal exposure time was found to be 5 ms. At this exposure time, a good signal-to-noise ratio was achieved. A total of 50 scans were performed, ensuring reliable and reproducible results
Statistical Research
Using statistical tools, data from triplicate studies were handled in MS Office Excel 2010 program to ascertain the maximum estimation error at significant levels of p < 0.05 and standard deviation (±SD).
Results
The results of the gas chromatography analysis are presented in Table 1. RI is the calculated retention index and RT is the retention time.
Polar Metabolites in Moringa Oleifera Leaf.
Authors sought rapid and non-destructive methods related to applied photonics. Figure 1 shows the 3D emission-excitation matrix and its topographical 2D projection.

Excitation-Emission Matrices of Crushed Moringa Oleifera Leaves.
As extracts from organic solvents are often used in pharmaceutical technologies the fluorescence spectra of ethanol and methanol extracts are shown in Figures 2, 3, and 4.

Excitation-Emission Matrices of 70% Ethanol Extract from Crushed Moringa Oleifera Leaves.

Excitation-Emission Matrices of 50% Ethanol Extract from Crushed Moringa Oleifera Leaves.
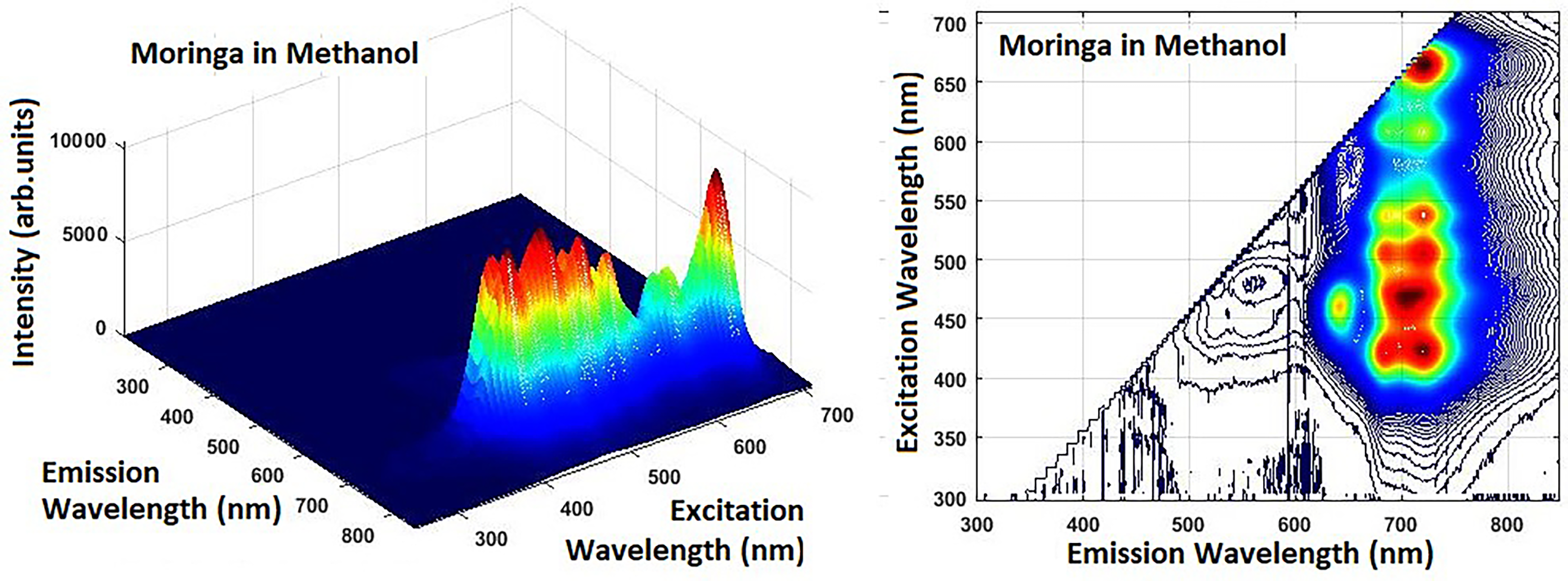
Excitation-Emission Matrices of Methanol Extracts of Crushed Moringa Oleifera Leaves.
Other methods in applied photonics include IR and Raman spectroscopy, which are used for the rapid screening of the quality of medicinal plants and their extracts. The Raman and IR spectra of Moringa oleifera leaves are shown in Figures 5 and 6, respectively. Similar results have been reported by Dzagli. 33

Infrared Spectrum of Moringa Oleifera Leaves.

Raman Spectra of Leaves From Moringa Oleifera.
Discussion
Based on the results obtained we now consider first, the significance of each of the ingredients identified and second, of the capabilities and advantages of the different analytical methods used.
High levels of glutamic acid (4.77 mg/g dw), aspartic acid (6.21 mg/g dw), glycerol (5.63 mg/g dw), glucitol (12.90 mg/g dw), and phenylalanine (2.44 mg/g dw) were found in Table 1. These are important for a number of cases and applications which we consider as follows.
Glutamic acid, a precursor of the neurotransmitter glutamate, plays a key role in the central nervous system. Therefore, extracts from Moringa oleifera leaves could be beneficial for improving brain function and memory as well as for treating certain neurological disorders such as epilepsy and neurodegenerative diseases.34,35
Aspartic acid is a non-essential amino acid, existing in two isoforms: L-aspartic acid (L-Asp), the biologically active form, and D-aspartic acid (D-Asp), which is found in smaller amounts and is mainly involved in neurological development and endocrine functions.
Phenylalanine is an essential amino acid that plays a crucial role in protein synthesis and serves as a precursor for several important neurotransmitters, including dopamine, norepinephrine, and epinephrine. Due to its role in the production of these mood-regulating chemicals, phenylalanine has been studied for its potential antidepressant effects. 36 It is thought to enhance cognitive function, improve alertness, and help alleviate symptoms of depression by increasing neurotransmitter availability in the brain. Therefore, the inclusion of Moringa oleifera leaves or their extracts in dietary supplements may contribute to improved mood and overall mental well-being, as these leaves are a natural source of phenylalanine. In addition to its potential mood-enhancing properties, Moringa oleifera leaves contain various bioactive compounds that can be beneficial for cosmetic applications. Glycerol (glycerin) and glucitol (sorbitol) are sugar alcohols that act as humectants, meaning they help retain moisture by attracting water to the skin. These compounds also exhibit osmotic properties, which can help maintain skin hydration and improve texture. Given the presence of these moisturizing agents, extracts from Moringa oleifera leaves could be valuable in the formulation of skin care products, offering natural hydration and skin-protecting benefits. 37
In our study we present possibly for the first time the detailed fluorescence spectra of dried leaves from Moringa oleifera as well as from their 50% and 70% ethanol extracts and methanol extract from them. The resulting three dimensional excitation-emission matrices contribute to the easy identification of phytochemical compounds and represent fingerprints of the studied samples. The use of solvents with different concentrations and polarity allows for better characterization of the biologically active substances contained in the leaves.
Two main regions of the fluorescence maxima were distinctly observed in the 3D fluorescence spectra of Moringa Oleifera leaves, (Figure 1):
The first was in the 450 nm - 550 nm range; The second was in the 650 nm -775 nm range.
The first of the two regions corresponds to blue-green and the second to red fluorescence, respectively.
The lower intensity peaks in the blue-green range (400-550 nm) are associated with phenolic compounds. The intensity of the emission peaks of these compounds varies depending on the type of solvent. They are most intense where the extraction of polar compounds is effective. This can be seen in Figure 3. The 50% ethanol extract is characterized by a relatively strong fluorescent signal in a narrow spectral range. It lacks the secondary fluorescent maxima of the 70% ethanol extract, probably due to a mixture of fluorophores. The methanol extract gives a more complex fluorescence profile as it extracts flavonoids and phenolic compounds. In all samples, chlorophyll fluorescence is the most pronounced. However, in the methanol extract, fluorescence in the blue region (350-450 nm) is observed and is associated with aromatic amino acids tryptophan and tyrosine, or various phenolic compounds. Weak fluorescent maxima are observed in the range (500-550 nm) due to carotenoid pigments.
The two fluorescence maxima in the second range were approximately around 729 nm and 650 nm. One of them is associated with emissions from chlorophyll b (670 nm −790 nm), and the other is related to emissions from chlorophyll a (650 nm).
The choice of solvents is crucial for the qualitative analysis of the extracts and their intended application. For example, phenolic compounds are key in evaluating the biological activities of the extracts, such as antioxidant and anti-inflammatory activities. The simultaneous use of excitation-emission, infrared (IR), and Raman spectroscopy allows for more accurate quantitative detection of components. Fluorescence spectroscopy enables the qualitative detection of pigments, phenols, flavonoids, and amino acids. For assessing the medicinal properties of Moringa oleifera, vibrational motions in the molecules reflected in Raman spectra are crucial. The interaction between molecules is explained by the functional groups detected in the IR spectra which were used to identify the active components.
The functional groups established through IR spectroscopy were used to identify the active components. The obtained IR spectra (Figure 5) exhibit two characteristic regions. One provides information about functional groups, while the other is associated with the unique fingerprint of leaf samples from the plant. The region that provides information about functional groups, focusing on stretching vibrations, ranges from 4000 cm−1 to 1450 cm−1.
Peaks in the range of (3450-3200) cm−1 are associated with the presence of hydroxyl groups (OH), related to the stretching and vibrations of hydroxyl groups found in fatty acids or protein structures. 38
The band around 2900 cm−1 is associated with CH2 vibrations, characteristic of fatty acids in the leaves of Moringa oleifera. 39 The peak reported in the literature is also associated with C-H stretching, attributed to kaempferol.40,41 Similar regions in the IR spectrum of Moringa oleifera leaves have been identified by Sharma Priyanka. 42 Peaks around 1550 cm−1 are linked to benzoic acid and rutin (C = C aromatic stretching), while the peak near 1630 cm−1 corresponds to the benzene ring skeleton.41,43,44
A detailed analysis of the spectrum (Figure 5) in the fingerprint region (from 1450 cm−1 to 500 cm−1) allows for the following conclusions:
45
The maximum around 1230 cm−1 confirms the presence of hydroxyl groups in phenolic and alcoholic compounds. The peak around 1400 cm−1 is associated with N–O symmetric stretching in an aliphatic nitrogen-containing compound. The maximum intensity around 1050 cm−1 is attributed to C = C bonds or C-C stretching in aromatic rings.
Other authors remark that, absorption peaks at (1425-1400) cm−1 correspond to C = C aromatic ring compounds, 46 serving as markers for phenols and flavonoids. The peak at 1050 cm−1 is associated with the C-O stretching of ester groups. 47
Despite the poor signal-to-noise ratio, some characteristic vibrational modes could be easily distinguished by using the Raman spectrometry (Figure 6). The stretching vibrations of the N-H bond in amino acids and = C-H in close proximity to the C = C double bond were positioned at 3309 cm−1 and 3131 cm−1, respectively. The most intense band with a maximum at 2929 cm−1 is a complex and arises from C-H stretching in the methyl and methylene groups. The peak at 2716 cm−1 might arise from C-C in the aldehyde. 48 The next mode, observed at 1972cm−1 arises from the symmetric stretching vibration of the C = C bond. The characteristic C = O bond in the lignins was found at 1680 cm−1, while the symmetric C = C vibration in aromatic residues was positioned at approximately 1601 cm−1, and the characteristic stretching vibration of C-N-O in the bond was found at 1531 cm−1. The next lower frequency modes positioned at 1448 cm−1 and 1410 cm−1 were due to the bending vibration of CH2 and CH3 residues and bending of the O-H bond, respectively. Symmetric stretching of C-C in acyclic residues is observed at approximately 1369 cm−1, while the mode at 1311 cm−1 corresponds to the bending vibration of the O-H bond in phenols. The band at 1162 cm−1 arises from the asymmetric stretching vibration of the C-O-C bond. 49
Raman microscopic images of Moringa olifeira leaves were taken immediately prior to spectrum scanning and shown on the Figure 7. This combination of the Raman analysis technique and traditional light microscop

Raman Microscopy of Moringa Oleifera Leaves.
The obtained images were well focused in the center, while the peripheries were blurry. This was due to the uneven leaf distribution on the sample holder surface. In addition, the images appear light green and quite bright, suggesting a strong fluorescence property.
Conclusions
Combined nondestructive optical measurements, including infrared, Raman, and 3D excitation emission fluorescence spectra, provide detailed information on the individual characteristics of Moringa leasves over a broad spectral range.
Moringa oleifera leaves contain significant bioactive compounds detectable using nondestructive spectroscopic techniques. Different spectrum areas matching to chlorophyll a and b, polyphenols, and carotenoids are revealed using excitation-emission fluorescence matrices. Using infrared spectroscopy, vibrations particular to flavonoids, phenolic acids, and fatty acids present in Moringa oleifera leaves are identified. Raman spectra confirmed the existence of these functional groups, therefore demonstrating the value of spectral approaches for plant research. The three complementary techniques can be used for quality control in producing herbal supplements
Footnotes
Ethical Considerations
This study did not involve experiments on humans or animals and does not require approval from the Ethics Committee of the Medical University – Varna.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, G.G. and K.N.; methodology, A.G.; validation, T.E., I.D. and A.G.; formal analysis, K.N.; investigation, O.K., D.N; resources, A.G.; data curation, S.F.; writing—original draft preparation, K.N.; writing—review and editing, G.G.; visualization, S.F.; supervision, K.N.; project administration, K.N.; funding acquisition, K.N. All authors have read and agreed to the published version of the manuscript.”
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Special thanks to the European Union-Next Generation EU for the financial support provided for the publication of the paper and of project № BG-RRP-2.004-0009-C02.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data are included in the paper and its supporting information files.Conflict of Interest declaration: The authors have declared that no competing interests exist
The financial support of the European Union-Next Generation EU for the publication of the paper under project № BG-RRP-2.004-0009-C02 is acknowledged.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable
