Abstract
Objective:
This study aims to compare the relevant characteristics of alum and its processed products and investigate their effects on skin wound healing using x-ray diffraction (XRD), Raman spectroscopy, thermogravimetric analysis (TGA), and low-field nuclear magnetic resonance (NMR).
Introduction
Alum, a traditional Chinese mineral medicine (major component: KAl(SO4)2·12H2O), possesses pharmacological effects including antibacterial, hemostatic, antidiarrheal, and cholagogic properties. 1 For over two thousand years, it has been used to treat conditions in dermatology, anorectal surgery, stomatology, otolaryngology, and burn care.2,3 Its primary component, potassium aluminum sulfate [KAl(SO4)2·12H2O], is extensively utilized as a vaccine adjuvant in numerous countries.4–6 Alum can be applied directly in clinical settings or after processing. The prevalent processing technique is calcination, which removes crystal water from alum, resulting in D-Alum (major component: KAl(SO4)2). Additionally, the “Lei Gong Pao Zhu Lun” documents a method of adding honeycomb during calcination, producing B-Alum, which has effects of reducing swelling and membrane protection. 7 However, the unclear processing mechanism of alum leads to issues in clinical practice due to the intermixing of raw and processed products, directly impacting efficacy and safety. The ancient medical text “Yilin Zhuanyao” 8 records that calcination of alum enhances its ability to absorb tissue exudate, facilitating wound healing, although this notion lacks a modern theoretical foundation. Low-field nuclear magnetic resonance (NMR) technology can indirectly detect the presence and binding strength of water molecules by analyzing the differences in relaxation times of H protons within a magnetic field. Liu Kaiyang et al 9 employed low-field NMR to analyze the hydration processes of gypsum and its calcined form, noting that hydration serves as an indicator of moisture absorption and wound-healing properties. A stronger hydration capacity allows samples to more effectively adsorb wound exudate, potentially enhancing astringent and moisture-absorbing properties. It is hypothesized that the loss of crystal water in mineral medicines post-processing can influence their hydration capacity, subsequently impacting their wound-healing effects. This study systematically investigated the hydration behavior of alum and its processed products using a combination of techniques, including x-ray diffraction (XRD), Raman spectroscopy, low-field NMR, and thermogravimetric analysis (TGA). The thermal decomposition profiles were characterized by TGA to quantitatively analyze the loss of crystalline water during processing.10,11 Simultaneously, the binding states of water molecules were dynamically resolved by NMR. The loss of crystalline water during calcination was found to alter the porosity and surface charge of the mineral, thereby affecting its hydration capacity and adsorption performance. 12 XRD was utilized to analyze the crystal structure changes in alum and its processed products, revealing alterations in lattice parameters and the potential formation of intermediate or new phases during hydration. Raman spectroscopy, through the characterization of molecular vibrational modes, offered information on the interactions between water molecules and the alum crystal structure, further elucidating changes in chemical bonds and local structural evolution during the hydration process. These complementary techniques allowed us to correlate structural changes induced by processing with hydration capacity, which is hypothesized to mediate wound-healing effects through enhanced exudate absorption and astringent properties. Additionally, a mouse wound model and anti-inflammatory tests were used to explore the mechanisms of processing from both wound healing and anti-inflammatory perspectives.
Materials and Methods
Sample Preparation
Preparation of Alum Solution: Alum was ground into a fine powder and dissolved in distilled water under magnetic stirring at 50 °C for 30 min. Samples were collected from the Chinese herbal medicine market in eastern China between June 2023 and March 2024.The raw material was purified and refined to yield Alum [potassium alum, KAl(SO4)2·12H2O, purity >99.0%].The structural schematic is shown in Figure 1.
Schematic Diagram of Alum Structure.
Preparation of D-Alum Solution: Raw alum was calcined in a 2 cm-thick crucible at 250 °C for 1.5 h, followed by 160 °C for 1 h, per the 2020 Chinese Pharmacopeia 13 and optimized conditions. 14 The calcined product was ground and dissolved in distilled water under magnetic stirring at 50 °C for 30 min.
Preparation of B-Alum Solution: Alum was calcined at 587 °C for 1 h, mixed with 0.6× its weight of honeycomb, and heated for an additional 3 h, following Lei Gong Pao Zhi Lun 7 and adapted protocols. 15 The mixture was cooled, ground, buried in soil (12-15 cm depth, 20-25 °C, 20%-40% moisture) for 12 h, and dissolved in distilled water under magnetic stirring at 50 °C for 30 min.
Physicochemical Characterization
X-Ray Diffraction (XRD)
XRD analysis was performed using a Bruker D8 Advanced diffractometer with CuKα radiation (40 kV, 40 mA; scanning range 5°-90°, speed 6°/min). Data were processed with JADE 6.0 and plotted using Origin 8.0.
Raman Spectroscopy Analysis
The functional groups of each sample were characterized using a Raman spectrometer (Renishaw, UK). The spectrometer utilized a 785 nm laser at full intensity, with an exposure time of 10 s, and collected sample spectra within the range of 100 to 2000 cm−1.To achieve effective differentiation among three kinds of Alum, an OPLS-DA model was established using SIMCA 14.1 for further data analysis.
Thermogravimetric Analysis (TGA)
TGA was conducted on a NETZSCH STA 449 F5/F3 Jupiter analyzer, calibrated with high-purity In (156.6 °C) and Zn (419.5 °C). Samples (<150 μm powder, 10.0 ± 0.5 mg) were heated from 30 °C to 1100 °C at 10 °C/min under static air (50 mL/min). Thermal profiles were analyzed via Proteus software (v7.0), with triplicate measurements.
Low-Field Nuclear Magnetic Resonance (NMR)
Hydration properties were evaluated using a Newmax MesoMR23-060H-I spectrometer (0.5 T, 10 mm probe), calibrated with D2O and 1% agarose gel. Chromatographic bottles were used due to sample volume constraints, and D2O was employed as a lock solvent. Samples (2.0 g + 4.0 mL D2O) were vortexed and equilibrated at 25 °C for 90 min. CPMG pulse sequences (τ = 0.5 ms, echo train length 5000, TR = 3000 ms) were applied with 8 scans. T2 distributions were analyzed via inverse Laplace transform (MesoMR software v2.3.1), with triplicate measurements.
Skin Wound Healing Analysis
SPF-grade ICR mice (8 weeks, 26 ± 2 g, equal sexes) from Changchun Yisi (license: SCXK (Ji) 2020-0002) were used. Experiments were approved by the Animal Care Committee of Changchun University of Traditional Chinese Medicine (approval: 2023417) and followed ARRIVE 2.0 guidelines. 16 After a 1-week acclimatization, mice were divided into four groups: model, Alum, D-Alum, and B-Alum. A 1 cm circular wound was made on the dorsal skin under isoflurane anesthesia. Model wounds were rinsed with saline, while treatment groups received 0.2 g of Alum, D-Alum, or B-Alum topically. Dressings were changed daily for 14 days. Wound areas were photographed and analyzed using ImageJ on days 0, 3, 7, 9, and 14 to calculate healing rates.
Histology Analysis
Histopathological Examination
On postoperative days 7 and 14, mice were humanely euthanized by cervical dislocation. Peri-wound tissue specimens were harvested and immersion-fixed in 4% paraformaldehyde (PFA) solution for 24 h at 4 °C. Following standard paraffin embedding, 5-μm-thick histological sections were prepared using a rotary microtome (RM2235, Leica Microsystems, Germany). Sections underwent routine staining protocols with hematoxylin and eosin (H&E) for cellular morphology assessment and Masson's trichrome for collagen visualization. Digital photomicrographs were acquired using a high-resolution microscopy system (DP73, Olympus, Japan) under consistent magnification (200×) and illumination conditions. Histopathological evaluation was performed by two blinded investigators using standardized scoring criteria.
Immunohistochemical Detection
Immunohistochemical analysis was performed to evaluate protein expression profiles of interleukin-1 beta (IL-1β; mouse monoclonal, clone WLH3903, 1:1000 dilution), tumor necrosis factor-alpha (TNF-α; rabbit polyclonal, WL01896, 1:1000), and vascular endothelial growth factor A (VEGFA; rabbit monoclonal, WL00009b, 1:1000) using validated antibodies from Wanleibio (China). Following antigen retrieval with citrate buffer (pH 6.0) and endogenous peroxidase blocking, sections were incubated with primary antibodies overnight at 4 °C, followed by appropriate HRP-conjugated secondary antibodies (Dako, Denmark) and DAB chromogenic development.Quantitative analysis was conducted using ImageJ software (v1.53k, National Institutes of Health, USA) with standardized threshold settings. Three randomly selected high-power fields (HPF, 400× magnification) per sample were analyzed by two blinded investigators. Protein expression was quantified through integrated optical density (IOD) measurements normalized to tissue area (μm²), with final values expressed as mean density (IOD/area). Three independent experimental replicates were performed to ensure reproducibility.
Real-Time PCR Analysis
The expression levels of CD31 and Inducible nitric oxide synthase (iNOS) in wound tissues were evaluated at 7 and 14 days post-treatment. Total RNA was isolated from the tissues, and RNA concentration was quantified using a UV spectrophotometer (NANO 2000, Thermo Scientific, USA). Subsequently, cDNA was synthesized through reverse transcription. Quantitative real-time PCR (qRT-PCR) was performed using a fluorescence quantification system (Exicycler 96, BIONEER, South Korea). The relative expression levels of CD31 and iNOS were calculated using the 2−ΔΔCT method. The primer sequences used for qRT-PCR are listed in Table 1.
The Primers Planned to Be Used in qRT-PCR.

Statistical Analysis
Statistical analysis was conducted using SPSS version 25. The measurement data from the experiment were presented as mean ± standard deviation (
Results
Physicochemical Characterization
X-Ray Diffraction (XRD) Analysis
XRD spectra (Figure 2) revealed distinct crystalline phases for Alum, D-Alum, and B-Alum. Raw Alum (KAl(SO4)2·12H2O, PDF#07-0017) showed characteristic peaks at 2θ = 12.78°, 16.50°, and 18.05°, while D-Alum (KAl(SO4)2, PDF#23-0767) displayed new peaks at 2θ = 11.11°, 24.57°, and 31.33°, confirming complete dehydration. B-Alum exhibited a dual-phase structure with coexisting peaks of KAl(SO4)2 and residual KAl(SO4)2·12H2O (Table 2).

XRD Spectra of three Alum Species.
Preliminary Physical Characterization of the Samples.

Raman Spectroscopy Analysis
Raman spectra (Figure 3A) showed unique vibrational signatures: Alum had peaks at 151, 190, and 990 cm−1; D-Alum displayed shifts to 194, 220, and 610 cm−1; B-Alum retained partial Alum peaks (eg, 990 cm−1) alongside new bands at 196 and 983 cm−1. OPLS-DA (R²X = 0.999, R²Y = 0.788, Q² = 0.665) confirmed clear sample separation, with 990/985 cm−1 identified as key discriminators (Figure 3B-D).

(A) Raman Spectra of Three Alum Species. (B) OPLS-DA Scatter Plots for Three Alum Species (Green for Alum, Blue for D-Alum and Red for B-Alum). (C) The VIP of OPLS-DA for Three Alum Species. (D) Results of 200 Permutations in OPLS-DA for Three Alum Species.
Thermogravimetric Analysis
TGA curves (Figure 4A-C) showed: Alum: Three-stage weight loss (47-213 °C: 42.61% dehydration; 688-802 °C: 19.92% decomposition).D-Alum: Single-stage decomposition (691-845 °C, 39.31%) with no dehydration step.B-Alum: Partial dehydration (50-110 °C, 13.11%) followed by decomposition (695-832 °C, 26.24%).

(A) Results of Thermal Weight Analysis of Alum. (B) Results of Thermal Weight Analysis of D-Alum. (C) Results of Thermal Weight Analysis of B-Alum.

(A) The T2 Relaxation Spectrum Distribution Results of the Raw Medicinal Material of Alum and Its Two Processed Products. (B) The Water Distribution Diagram of Alum and Its Two Processed Products.
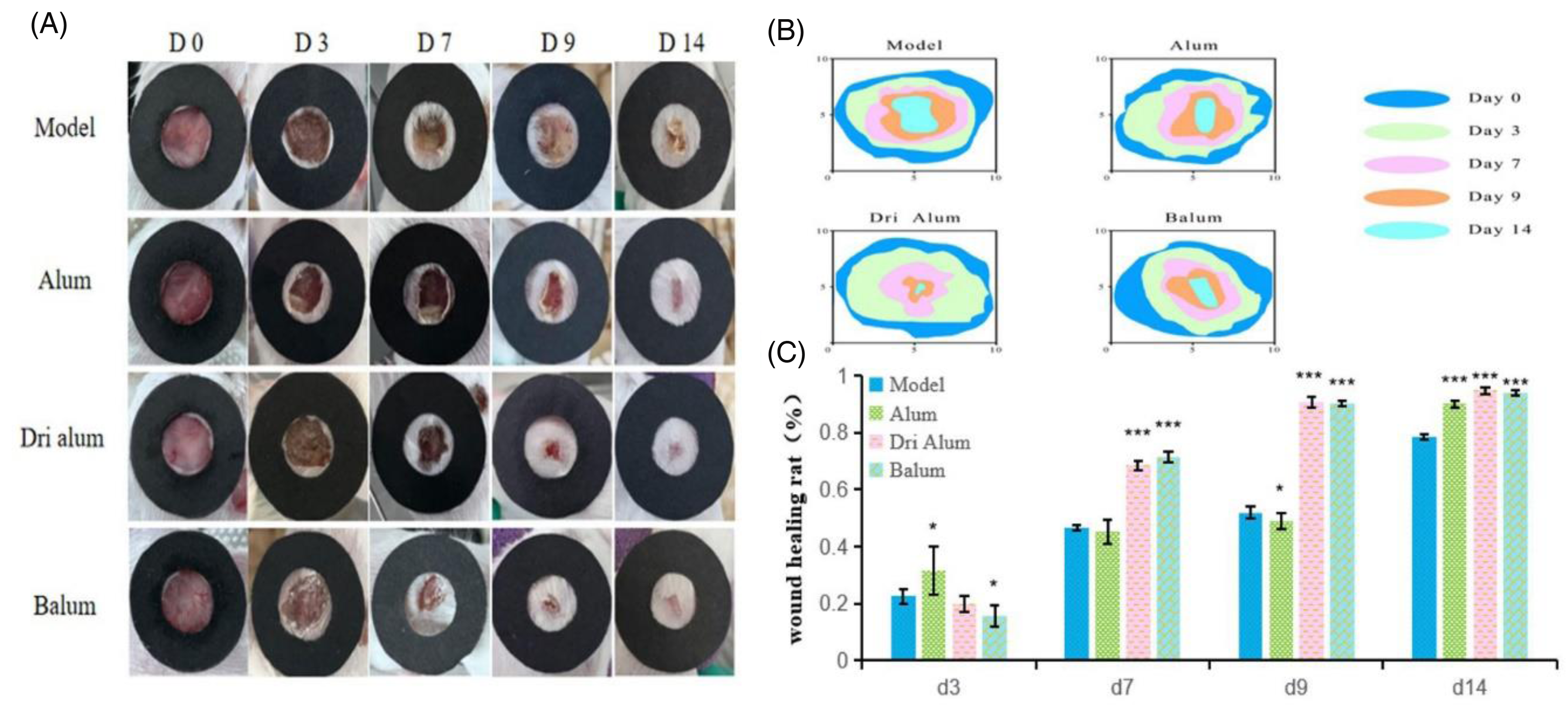
In Vivo Wound Healing. (A) Representative Pictures of Wounds in Different Groups. (B) Wound Shrinkage Marks on Days 0, 3, 7, 9 and 14. (C) Percentage of Wound Healing. n = 20.*
HE staining of wound tissues on days 7 and 14 (Figure 7) further revealed that the alum, D-Alum, and waxed alum groups exhibited significantly reduced inflammatory cell infiltration, decreased granulation tissue hyperplasia, and enhanced vascularization compared to the model group, which showed limited inflammatory cell reduction, sparse vascularization, and minimal granulation tissue proliferation. These findings demonstrate that alum, D-Alum, and waxed alum significantly promote wound healing, with alum exhibiting the most potent therapeutic effect.

Histological Analysis of Wound Tissue. (A) Masson Trichrome Staining of Collagen Deposition at the Wound Site on Days 7 and 14.Scale Bar, 100 μm, 500 μm. (B) Representative Images of Staining with H&E on Days 7 and 14, Scale Bars, 100 μm, 500 μm. n = 16.
The results are presented in Figure 8. Compared to the model group, the alum group exhibited significantly lower levels of IL-1β and TNF-α on days 7 and 14 (

Effect of Drugs on the Expression of Factors Related to Skin Wound Healing. (A) Immunohistochemical Detection of IL-1β Expression Levels in Wound Tissues of Each Group on Days 7 and 14. Scale Bar, 100 μm, 500 μm. (B) Immunohistochemical Detection of TNF-α Expression Levels in Wound Tissues of Each Group on Days 7 and 14. Scale Bar, 100 μm, 500 μm. (C) Immunohistochemical Detection of VEGFA Expression Levels in Wound Tissues of Each Group on Days 7 and 14. Scale Bar, 100 μm, 500 μm. n = 3, *

Effect of Drugs on the Expression of Skin Wound Healing-Related Factors. (A) Fluorescence Quantitative Analysis of CD31 Expression Levels in Wounds in Each Group. (B) Fluorescence Quantitative Analysis of iNOS Expression Levels in Wounds in Each Group. n = 3, *
Discussion
Chronic refractory wounds, often caused by diabetes, vascular issues, trauma, or radiation, pose a major clinical challenge due to slow healing and high recurrence.
23
Traditional Chinese Medicine (TCM) surgery offers non-surgical methods like debridement, infection control, and promoting blood flow and tissue growth.
24
Processed alum, with its strong hydration ability, effectively absorbs wound exudate, improving moisture balance and reducing discomfort. It also enhances anti-inflammatory and hemostatic effects by forming protein precipitates, reducing exudate, and aiding tissue repair. This study utilizes low-field NMR technology to analyze the hydration properties of alum and its processed products and investigates their potential role in promoting wound healing through topical anti-inflammatory and hemostatic mechanisms. Based on scanning electron microscopy (SEM)
15
and XRD results, calcination significantly altered the microscopic morphology of alum crystals. The processed alum (KAl(SO4)2) completely lost its crystalline water, forming a porous anhydrous phase, which modified its crystal structure and porosity. These structural changes may account for the enhanced hydration capacity of the calcined product. While Raman spectroscopy identified characteristic vibrational shifts (eg, 990 cm−1 peak) indicative of sulfate group reorganization. These structural transformations, supported by TGA-determined dehydration profiles, enabled D-Alum to exhibit the highest hydration capacity, attributed to its anhydrous morphology and surface porosity (SEM-EDX).These results collectively demonstrate that hydration capacity, as modulated by processing, plays a central role in mediating wound healing through exudate absorption and angiogenesis promotion. Pharmacological experiments demonstrated that alum and its processed products significantly accelerated wound healing in mouse models compared to the control group (
Yuan Zhongxing et al 25 highlighted the importance of reducing inflammatory responses and restoring normal repair processes for effective wound healing. IL-1β and TNF-α, key pro-inflammatory cytokines, exacerbate inflammation and tissue damage.26–28 iNOS, a critical enzyme in the inflammatory response, produces NO, which can impede wound healing when overexpressed. 29 This study found that alum and its processed products significantly reduced IL-1β, TNF-α, and iNOS levels in treated groups, with D-Alum showing the most pronounced anti-inflammatory effects. The enhanced hydration capacity of processed alum likely facilitates exudate absorption and promotes wound healing, supported by the exothermic nature of the hydration reaction, which may further aid tissue repair.
VEGFA and CD31 play pivotal roles in angiogenesis and endothelial cell function.30–32 VEGFA promotes endothelial growth and vascular permeability, while CD31, a marker for endothelial cells, is essential for cell adhesion and vascular integrity.33,34 This study observed increased VEGFA and CD31 expression in alum-treated groups, with D-Alum showing the most significant enhancement. These findings suggest that alum and its processed products promote angiogenesis and tissue repair, further supporting their therapeutic potential in wound management.
Conclusion
The multi-technique characterization (XRD, Raman, TGA, NMR) in this study provides compelling evidence that calcination-induced structural modifications in alum (KAl(SO4)2) significantly enhance its hydration capacity and biological activity. The formation of a porous anhydrous phase, as confirmed by SEM and XRD analysis, appears to be crucial for the observed improvement in wound healing efficacy. Our animal experiments demonstrate three key therapeutic effects of calcined alum: (1) pronounced collagen deposition, (2) substantial reduction of inflammatory markers, and (3) enhanced angiogenesis. These findings not only validate the traditional Chinese medicinal concept of “calcination enhancing astringent and tissue-regenerative properties” but also provide quantitative biochemical evidence for its mechanism of action. Particularly noteworthy is the 2.1-fold upregulation of CD31, suggesting calcined alum may promote wound healing through vascular regeneration. However, the current study has some limitations. While we established a correlation between structural changes (eg, increased porosity) and therapeutic outcomes, the exact molecular pathways involving NF-κB or MAPK signaling remain to be elucidated. Future studies employing proteomic or transcriptomic approaches could provide deeper insights into these mechanisms. These findings position calcined alum as a promising dual-function agent for chronic wound therapy, combining structural hydration properties with bioactive anti-inflammatory effects.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251348405 - Supplemental material for Comprehensive Physicochemical Characterization and Wound Healing Efficacy of Alum and Its Processed Products: Insights from XRD, Raman, TGA and Low-Field NMR Analyses in a Mouse Model
Supplemental material, sj-docx-1-npx-10.1177_1934578X251348405 for Comprehensive Physicochemical Characterization and Wound Healing Efficacy of Alum and Its Processed Products: Insights from XRD, Raman, TGA and Low-Field NMR Analyses in a Mouse Model by Xinxin Yang, Shuang Kang, Jing Liu, Jiaojiao Ran, Zijun Huang, Chuying Wang and Peng Yu in Natural Product Communications
Footnotes
Ethical Approval
Ethical approval to report this case was obtained from the Animal Care and Use Committee of Changchun University of Traditional Chinese Medicine (2023417).
Funding
This work was supported by the natural science foundation of Jilin Province. Jilin Provincial Natural Science Foundation Funded Project (YDZJ202501ZYTS271). Jilin Provincial Department of Science and Technology Project (YDZJ202401005ZYTS).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animals Rights
All procedures in this study were conducted in accordance with the the Animal Care and Use Committee of Changchun University of Traditional Chinese Medicine (2023417) approved protocols.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
