Abstract
Introduction
Cannabinoid receptors CB1 and CB2 are the primary endogenous receptors with which cannabinoids interact, inducing a variety of physiological and psychological effects. Although interactions with other receptors such as TRPV1 and GPCR55 have been recognized, these interactions may contribute significantly to cancer remediation by modulating internal cellular pathways. The main active constituents in the cannabis plant, cannabidiol (CBD) and tetrahydrocannabinol (THC), have been characterized as non-intoxicating with potential medical benefits or intoxicating with debated benefits, respectively. Nevertheless, cannabinoids have demonstrated efficacy as single-agent treatments for certain diseases.
Objectives
This study focuses on
Methods
The synthetic derivatives of THC and tetrahydrocannabivarin (THCV) were screened in
Results
The saturated products derived from THC and THCV have shown substantially lower IC50 values than the parent compounds and as compared to poly(ADP-ribose) polymerase (PARP) inhibitors, Olaparib and Veliparib.
Conclusion
The data collected suggest that these rare saturated cannabinoids may have promising activity comparable to or surpassing standard anticancer agents such as poly(ADP-ribose) polymerase (PARP) inhibitors.
Introduction
Cannabinoid receptors CB1 and CB2 are known to be expressed within the CNS (Central Nervous System) and PNS (Peripheral Nervous System) tissues. Their location and roles in expression during inflammation and disease, has prompted research to determine possible treatments using the CB1 and CB2 receptor pathways. Expression of these receptors has been observed during the mediation of certain cancer growth,
1
including but not limited to the TRPV channel (Transient receptor potential cation channel subfamily V member 1)
2
and GPCR55 (G-Protein Coupled receptor 55).
3
These other receptors are also expressed during the genesis of certain cancers and may play a role in the remediation of cancer.4,5 Several studies have shown anti-proliferative and pro-apoptotic properties of cannabinoids towards certain cancers

Structures of (9
THCV is a homolog of Δ9-THC, with the primary difference located in the alkyl chain on the C5 carbon with an alkyl chain of three carbon lengths rather than a five-carbon length alkyl chain similar to THC. THCV has been explored as an anti-obesity drug when taken in conjunction with metformin for reducing blood sugar set in early stage clinical, as well in murine models, through its effects of reducing appetite.11–13 Obesity is an established risk factor for pancreatic ductal adenocarcinoma (PDAC), contributing to both chronic inflammation and metabolic dysregulation.
13
THCV has previously shown anti-obesity effects via CB1 antagonism and CB2 agonism, suggesting its derivatives may offer dual benefit in PDAC through metabolic and oncologic pathways.
13
HHC, is an analog of Δ9-THC, the difference lying in the cyclohexene ring being hydrogenated to form the cyclohexyl ring, the lack of the double bond would assume the binding affinity to the cannabinoid receptors to be lessened.
13
HHC is determined to have novel status as a cannabinoid due to its prevalence being found in trace amounts in nature with limited to no data being accounted for. Although the cannabinoid was elucidated and synthesized in the 1940's by Adams, no clinical research or pre-clinical research has been done on the efficacy or effects of HHC. Although limited studies on toxicity and mechanisms on HHC have been published, there is still more work that needed to be done to contribute to the field of cannabinoid chemistry.14,15 HHCV is the hydrogenated derivative of THCV, following the path of HHC with no known clinical studies of note. Cannabinoids have been proposed for years with
The accepted mechanism of action according to literature for upregulation or downregulation signaling is not definitive, with the initial interaction and start of the cascades beginning with activation of the CB1 and CB2 receptors which belong to the G-protein coupled receptor (GPCR) family of proteins. 19 CB1 is the prominent subtype located within the CNS and are also expressed within the PNS. 20 The discovery of CB1 and its prominence within the CNS and PNS has garnered attention for possible treatment of neurodegenerative and neuropsychological disorders that can be treated through this avenue. 21 Although activation of CB1 receptors is also indicated with the psychotropic effects negatively associated with the use of psychoactive cannabinoids. 22 The CB2 receptor plays an integrative role within the brain, G.I (Gastro-Intestinal), PNS, and the immune system. 23 Unlike the CB1 receptors, the activation of the CB2 Receptors with cannabinoids, do not provide the psychotropic “high” that is associated with agonists of the CB1 receptor, which would be the more likely place to design compounds for better treatment. 24 CB2 plays a significant role in anti-inflammation and remediation of cancer growth.24,25 CB2 receptors are implicated in a variety of modulatory functions, including immune suppression, induction of apoptosis, and induction of cell migration.26–28 The CB1 receptors like the CB2 receptors can be allosterically modulated by synthetic ligands, in a positive and negative fashion. 29 THCV is shown to act as an antagonist of the CB1 receptor in small concentrations but can act as a partial agonist at larger concentrations. 30 HHC in animal models are known to bind to the CB1 receptor producing similar effects to THC. 15 HHCV has no known clinical data for receptor binding or physiological responses. Although it is presumed that HHC is a hydrogenated derivative with CB1 binding activity, and THCV is a CB2 agonist and in large amount a CB1 partial agonist, that HHCV its hydrogenated derivative would display similar agonistic effect.
Cell surface receptors, GPCRs can detect molecules on the cellular membranes and activate various upregulation or downregulation responses within the cell. 31 GPCR activation is mediated through bound agonists. Estimated 34% of approved drugs target the GPCR complex 32 in various target organs. The GPCR complex, is implicated in a variety of physiological processes, not limited to but including, regulation of immune system activity and inflammation, autonomic nervous system transmission, homeostasis modulation, and implicated in growth and metastasis of certain types of tumors. 33 The CB1 and CB2 receptor are class A serpentine GPCR that signals primarily through the adenylyl cyclase-inhibiting heterotrimeric G protein Gi and the ERK1/2 pathways. 31 Although they are not limited to these specific pathways, they are the most common pathways. Over 100 GPCR's have been expressed at the mRNA level within pancreatic adenocarcinoma tumors.33,34 Several GPCRs GPRC5A, F2R and F2RL1 are expressed in multiple PDAC (pancreatic ductal adenocarcinoma) cell types while other GPCRs are expressed in a specific setting within microenvironments of the cell.34,35 Although the research of targeting GPCRs in pancreatic cancers is relatively new, 36 Mutated KRAS (Kirsten rat sarcoma viral oncogene homolog) which is expressed in ∼70% of pancreatic adenocarcinoma tumors, show crosstalk with GPCR pathways, suggesting a role for GPCRs in modulating the impact of KRAS mutations. Hence, signaling by certain GPCRs may be oncogenic in pancreatic adenocarcinoma tumors, especially tumors with GNAS and KRAS mutations, making GPCRs a pertinent target.34–37
Due to the lack of early detection and the limited response to designed treatments, PDAC is considered to have a terrible prognosis, and is extremely aggressive with lethal malignancy and often detected after rapid metastasis. PDAC is the most common type of pancreatic neoplasm, and accounts for more than 90% of pancreatic cancer cases. 38 PDAC has an average 5-year survival rate of less than 10%. 39 The need for new treatments for pancreatic cancer is pertinent, as many of the on-market compounds are limited for the direct treatment of pancreatic cancer. The poor genomic and proteomic analysis of various tumors fails to distinguish the proper target and treatment plan. Aside from poor prognosis, tumor microenvironment is characterized by dense desmoplasia and extensive immunosuppression. Extensive desmoplasia results in various cell infiltration, vascularization, and hypoxia, preventing drugs to target such areas specifically.39,40 GPCR ligand targeting applies, as the membrane protein is present, also accounts for 20% of all cancers that contain a mutated GPCR or g-alpha subunits, making these targets increase specificity for PDAC. 41 Targeting this receptor using cannabinoids might prove to be a possible target of choice for therapeutic potential, as GPCRs mediate a broad range of autocrine and paracrine responses in cancer cells. They bind to a diverse group of ligands, including small peptides, lipids, and proteins (eg, chemokines). 42 The density of GPCRs on the cell surface is typically 103-104 receptors/cell, which should be adequate to ensure ample uptake of the targeted drug cargo.43,44
Methods
Chemicals, Reagents and Instruments
CBD (HPLC 99%) was purchased from GVB Biopharma (NV, USA). CBDV (HPLC 99%) was purchased from BayMedica (NV, USA). p-Toluene sulfonic acid, dichloromethane, palladium on carbon, hydrogen gas and MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] were purchased from Sigma-Aldrich, St. Louis, MO, USA. All cells were maintained in DMEM (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin and 100 μg/mL streptomycin in a 5% CO2 atmosphere at 37 °C. The cell lines have been tested and authenticated in a core facility of the Applied Genomics Technology Center at Wayne State University. NMR solvents (CD3CN) was purchased from Cambridge Isotope Laboratories, Andover, MA. Instruments used were the Shimadzu TQ8050 NX Mass Spectrometer, Shimadzu Nexis GC-2030 Gas Chromatograph, Bruker Avance II NMR 500 Mhz, Shimadzu AOC-20i Plus Autosampler, and Agilent 1100 series HPLC with Diode Array Detector was used to determine isomers using C18-RP column. MIA PaCa-2 (CRL-1420), HPAC (CRL-1703), ASPC-1 (CRL-1682) and Panc-1 (CRL-1469) cells were purchased from American Type Culture Collection (ATCC, Manassas, VA). Based on cell proliferation data, IC50 values were calculated using GraphPad Prism software using a nonlinear regression curve fit.
Synthesis of HHC and HHCV
The synthesis of HHC and HHCV is displayed in Figures 2 and 3. CBD was used as a starting material for the synthesis of HHC. In the first step, Δ8 THC was synthesized from CBD by reacting with
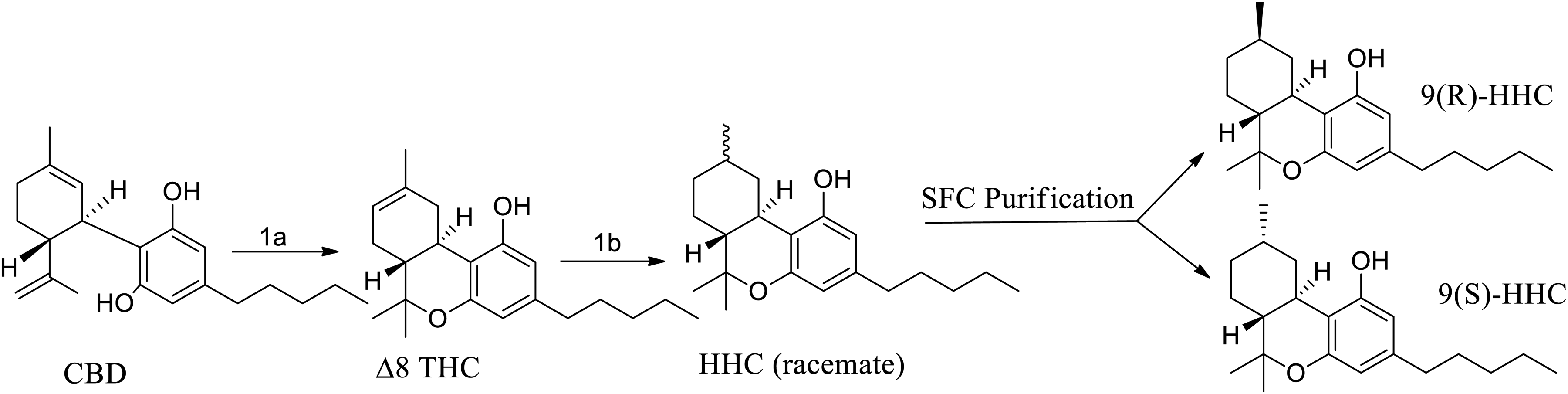
Reagents and Conditions: Reagents and Conditions: (1a)

Reagents and Conditions: (2a) DCM, Argon Purge 0 °C, TIBAL, 0 °C-rt, 20 h.; (1c) EtOH, Argon Purge, 1 h, rt., Pd/C, 1-5 bar, H2, 50 °C, 24 h.
Cannabidivarin (CBDV) was used as a starting material for the synthesis of tetrahydrocannabivarin (D9-THCV (SI, Figures 9, 10, 14). CBDV isolate was treated with tri-isobutylaluminum (TIBAL) to produce the Δ9 THCV as the desired product. Hydrogenation of Δ9 THCV leads to formation of a mixture of HHCVs comprised of the 9(S)- and 9(R)-HHCV diastereomers (SI Figures 11, 12, 15).
In-vitro Screening of THCV, HHC, HHCV, and PARP Inhibitors
Cell viability using MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay was performed by using the following method. 46 A total of 3000-8000 PDAC cells were seeded per well in 96-well plates. Following attachment, cells were treated with different compounds at varying doses as indicated for 72 h. Growth inhibition was determined by MTT assay. The MTT solution was added to the media at a final concentration of 0.8 mg/mL and cells were incubated at 37 °C for 2 h. After aspiration of the media, formazan crystals were dissolved in DMSO. Optical densities were measured at 570 nm using SynergyHT plate reader (BioTek, Winooski, WI, USA). To calculate IC50 values for all drugs, GraphPad Prism Software was used (GraphPad Software, San Diego, CA, USA).
Sanger Institute in-vitro Screening of Antineoplastics
Cells are seeded at an optimized density in medium with 5% or 10% FBS and 1% penicillin/streptomycin. The optimal cell number for each cell line is determined to ensure that it is in growth-phase at the end of the assay and to maximize the dynamic range of endpoint measurements. 24 h after plating, cells are treated with a dose titration of each compound, except for lines screened at MGH where drugging occurs the same day as plating. Following drugging, plates are returned to the incubator for assay at a 72-h time point. (Cell-lines screened at MGH are drugged the same day as plating). Cell viability is determined using either a DNA dye (Syto60) or metabolic assay (Resazurin or CellTiter-Glo). All screening plates are subject to stringent quality control measures.47,48
GDSC2 has been generated at the Wellcome Sanger Institute since 2015 following improvements to the screen design and assay. Compounds are stored in Storage Pods (Roylan Developments) providing a moisture-free, low oxygen environment, and protection from UV damage. Cells are seeded in 1536-well plates and an Echo555 Acoustic Dispenser (Labcyte) used to deliver compound doses. Promega CellTiter-Glo is used to measure cell viability at the assay endpoint. Drug treatments use a standard dose response format: 7-point dose curve incorporating a half-log dilution step (1000-fold range), 7-point dose curve with 2 × 2-fold dilutions followed by 4 × 4-fold dilutions (1024-fold range).
47
Dose-response curves are fitted using the non-linear mixed effects model47,48 incorporated in the GDSC IC50 package. All available replicates for an experimental combination (cell line + compound) are used to fit each curve and obtain IC50 and AUC estimates (previous editions of GDSC data have fitted a single dose response). Biomarker discovery uses the GDSCTools python package of Cokelaer et al
49
to run ANOVA for each dataset independently. All available replicates for an experimental combination (cell line + compound) are used to fit each curve and obtain IC50 estimates. Biomarker discovery uses the GDSCTools python package48,49 to run ANOVA for each dataset independently.
Experimental Section
Analysis of (R) and (S) HHC
HHC was dissolved in acetonitrile-d3 and 1H, 13C, COSY, HSQC, HMBC and NOESY data were acquired on a 500 MHz Bruker AVANCE II system at 25 °C. 1H, 13C, COSY, HSQC, HMBC data sets were analyzed to yield complete 1H and 13C peak assignment. Once the peaks were assigned, NOESY data were analyzed to yield stereochemistry information about the orientation of the hexanyl-methyl group. The instruments used were the Shimadzu TQ8050 NX Mass Spectrometer, Shimadzu Nexis GC-2030 Gas Chromatograph, Shimadzu AOC-20i Plus Autosampler, and Agilent 1100 series HPLC with Diode Array Detector was used to determine isomers using C18-RP column50.
Synthesis of HHC
HHC was synthesized according to literature procedure.
50
The crude oil was then dissolved in hexane and purified over silica (0% to 5% Ethyl Acetate). The fractions of interest were concentrated
Chiral Separation of (R) and (S) HHC
The diastereomers of HHC were separated by Supercritical Fluid Chromatography (SFC). SFC is used in industry primarily for separation of chiral molecules and uses the same columns as standard HPLC systems and CO2 as the mobile phase. HHC (5 g) was submitted for diastereomeric resolution. The analytical and preparative SFC methods described below were used for the purification. Analytical SFC Method: Column 4.6 × 100 mm Chiralpak AD-H from Chiral Technologies (West Chester, PA); CO2 Co-solvent (Solvent B) Isopropanol; Isocratic Method 25% Co-solvent at 4 mL/min System Pressure 125 bar; Column Temperature 40 °C; and Sample Diluent Isopropanol. Preparative SFC Method: Column 2.1 × 25.0 cm Chiralpak AD-H from Chiral Technologies (West Chester, PA); CO2 Co-solvent (Solvent B) Isopropanol; Isocratic Method 30% Co-solvent at 80 g/min; System Pressure 100 bar; Column Temperature 25 °C; and Sample Diluent Isopropanol. The HHC was purified using the preparative method described above. The collected fractions were dried in a rotary evaporator at 40 °C and transferred to the final container using isopropanol. The isolates were analyzed and recovery details for each isolate was calculated by SFC (UV220 nm) to be 4.6%:71.8%:23.5%, 50 additional data tables are provided MS and GC-MS of isomers listed in (SI Figures 16-18).
Synthesis of THCV
In a 20L reactor, under argon, CBDV (3 kg, 10.5 mol) was added and dichloromethane (DCM) (20L) set to stir for 1 h. To the solution was added dropwise Triisobutylaluminum 1 M solution in hexanes (2 L, 2 mol) over a period of 1 h. The reaction was stirred at room temperature overnight. HPLC with a diode array detector showed no starting material, only desired product Δ9-THCV. The reaction was quenched with water and extracted with DCM. The combined organic was washed with brine, dried (Na2SO4) and concentrated in vacuo to give dark red oil. The oil was purified via wiped film distillation to afford 2.55 kg of Δ9-THCV (85% of yield) HPLC spectrum shown in (SI Figure 14). The NMR of the final product matches with reported data. 51 1H NMR (500 MHz, CD3CN) 6.19 (1H), 6.14 (1H) 5.44 (1H), 3.26 (1H), 2.66 (1H), 2.43 (2H), 2.16 (1H), 1.85 (1H), 1.73 (1H), 1.68 (3H), 1.61 (2H), 1.34 (3H), 1.05 (3H), 0.93 (3H), 13C NMR (101 MHz, CD3CN) δ 155.2, 153.9, 141.4, 133.5, 118.7, 109.7, 108.3, 106.6, 75.4, 44.3, 36.5, 35.0, 30.8, 26.9, 26.2, 23.3, 22.0, 17.0, 12.5.
Synthesis of HHCV
HHCV was synthesized according to the general literature procedures. 45 The crude oil was then dissolved in hexane and purified over silica (0% to 5% Ethyl Acetate). The fractions of interest were concentrated in-vacuo and then distilled to afford a yellow oil, HPLC spectrum is shown in SI Figure 15. 1H NMR (500 MHz, C6D6) δ 6.36 (1H), 5.87 (1H), 4.95 (1H), 3.09 (1H), 2.53 (1H), 2.33 (2H), 1.69 (1H), 1.48 (4H), 1.29 (4H), 1.09 (1H), 0.98 (3H), 0.95 (1H), 0.90 (2H), 0.84 (3H), 0.74 (1H); 13C NMR (101 MHz, CD3CN) δ 155.84, 155.57, 142.63, 111.42, 110.71, 108.73, 77.67, 50.74, 49.92, 39.71, 38.30, 36.25, 33.18, 30.27, 28.69, 28.14, 24.75, 19.49, 14.49.
Results
Sharafi et al
52
have reported the potential use of cannabinoids such as CBD and THC for the Treatment of Pancreatic Cancer. Thus, we believe that the hydrogenated THC and THCV analogs, could serve as promising leads for better treatment outcome of patients diagnosed with pancreatic cancer. The use of rare cannabinoids in the treatment of pancreatic cancer has been revealed through
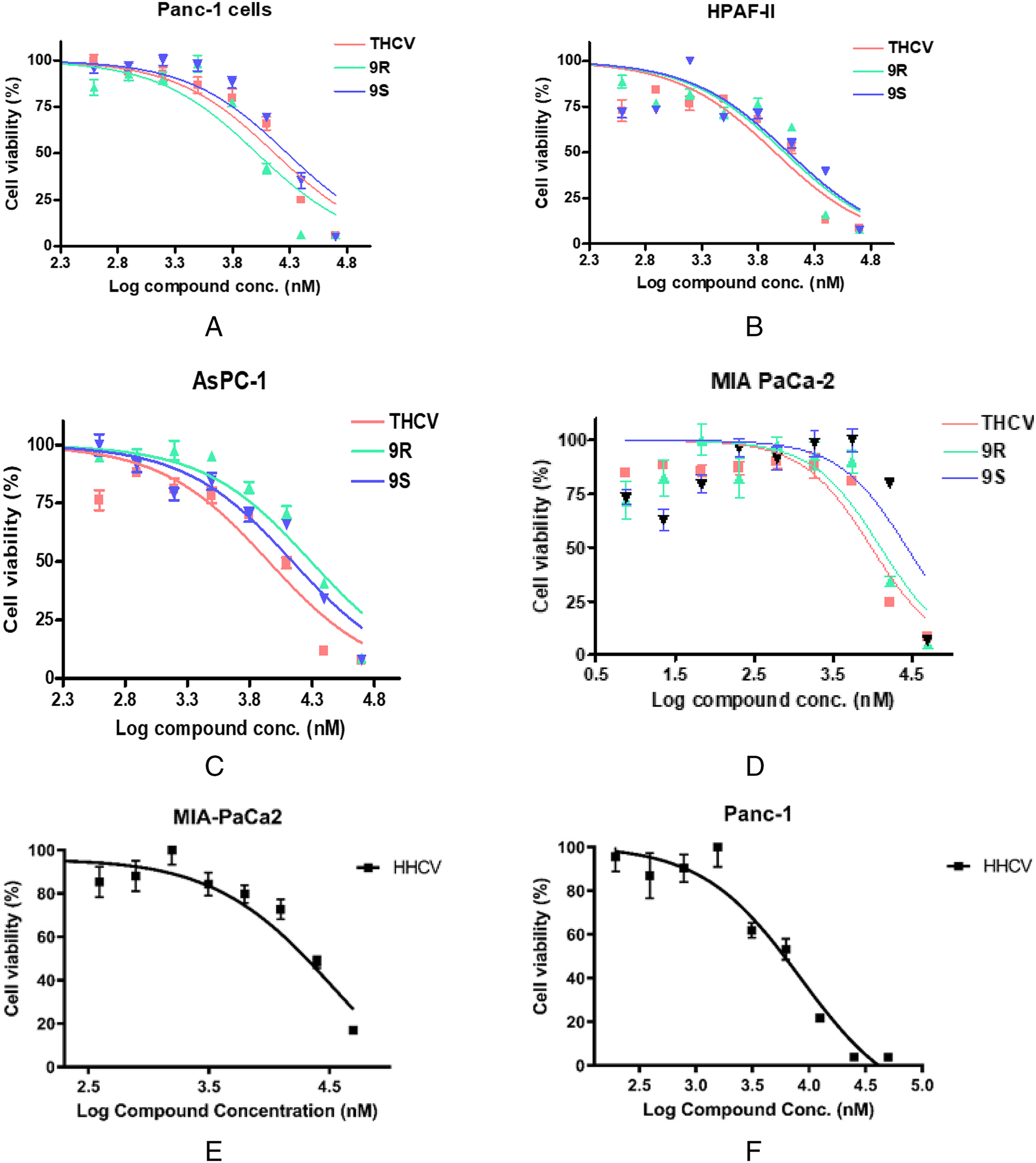
Effect of Cannabinoid Compounds THCV, (R) and (
Shown above in Figure 4C is THCV and HHC tested against the AsPC-1 cell line, and the HHC compound has slight variations within the IC50 values between the (R/S)-Isomers, but THCV shows a lower IC50 values across all three cell lines as referenced below in Table 1. Although the values are in micromolar concentration, the values are still low enough for viable data. MIA-PaCa2 cell line data shown above in Figure 4D, depicts the comparison of the various cannabinoids with the THCV providing a lower IC50 value compared to the full hydrogenated derivatives. In Figure 4E and F, the isolated graphs depict HHCV compared to PANC-1 and MIA-PaCa2 cells, cannabinoids scaffolding can be used to possibly help design drugs with lower IC50 values. Saturation of the olefin in THCV causes a positive impact in IC50 similar to the data seen in the review by Lovering et al 53
IC50 Values of Cannabinoid Compounds THCV, (9R) and (9S) HHC, and HHCV on the Proliferation of Panc-1, HPAF-II, AsPC-1 and MIA-PaCa2 Pancreatic Cancer Cells.
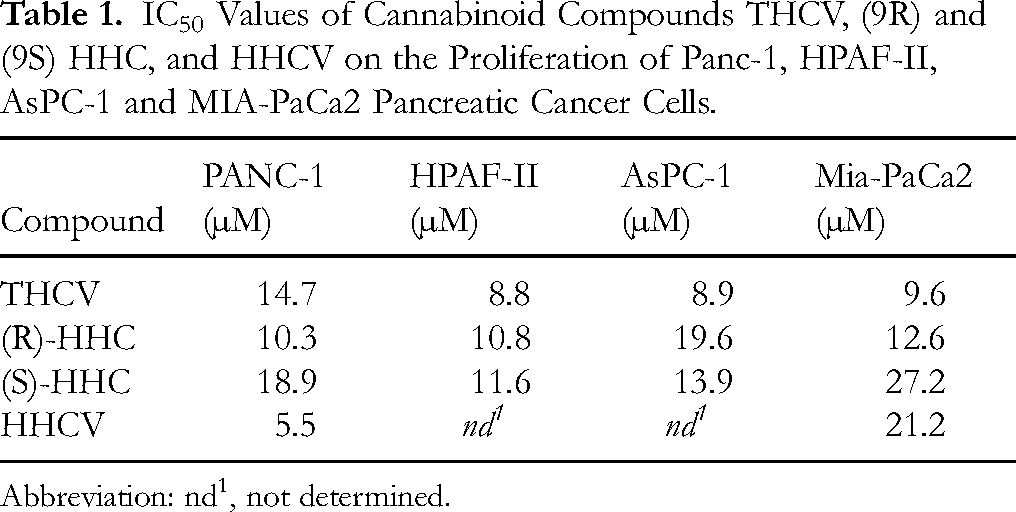
Abbreviation: nd1, not determined.
PARP is a type of nuclear enzyme that helps repair DNA damage in cells. 54 PARP inhibitors work by preventing cancer cells from repairing, allowing apoptosis to occur. 55 These drugs are a type of targeted therapy used to help treat cancers. The use of these inhibitors as a control to compare THCV, HHC, and HHCV against the inhibitors, shows efficacy towards the ability for the rare cannabinoids to be considered semi-efficient antineoplastics. As reported previously by Cohen, 56 a prototypical PARP inhibitor, PJ-34 57 has also demonstrated a better potency than marketed PARP inhibitors in our MTT assay of PDAC cell lines [IC50 values: 5.2 μM (Mia-Paca2), 2.1 μM (HPAF-II), 6.7 μM (AsPC-1) and 7.2 μM (PANC-1)]. The compiled data of the compounds tested on various cell lines was compared to PARP inhibitors currently on the market or in clinical trials shown in Figure 5 and IC50 values depicted below in Table 2, to provide more objective evidence towards the usage of HHC and THCV as possible pancreatic antineoplastic compounds.

Effect of PARP Inhibitors, Olaparib and Veliparib on Pancreatic Cancer Cell Lines.
IC50 Values of PARP Inhibitors on the Proliferation of HPAF-II, and MIA- PaCa2 Pancreatic Cancer Cell Lines.

Discussion
Limitations of this study include the outsourcing of biological testing and intrinsic sensitivity constraints of the MTT assay. While performed by a reputable institution, the assay can occasionally overestimate or underestimate cell viability. This is evidenced by observed deviations in colony formation and growth across replicates. Despite triplicate plating, minor variability remained, which could result from inherent assay noise, contamination, or minor impurities (although each compound was >95% pure). Additionally, the lack of mechanistic validation (eg, apoptotic markers, pathway modulation) limits the depth of our conclusions. Future studies using orthogonal assays (eg, Annexin V/PI staining, Western blotting) and 3D models will help refine the pharmacologic profile.
The data reported by Cohen 56 have shown that the prototypical PARP inhibitor, PJ34 57 demonstrated an excellent potency than marketed PARP inhibitors in PDAC cell lines especially PANC-1. Unlike other PARP inhibitors (Olaparib and Veliparib), the prototypical agent PJ34 which shares similarity of the tri-cyclic core of the THC scaffold, exclusively eradicated a variety of human cancer cells without affecting proliferating and non-proliferating healthy somatic cells. However, PJ34 has shown a promising anticancer activity in in vitro, and pancreatic tumor xenograft model, 56 its earlier development was discontinued primarily due to its mutagenic potential as observed in the Ames test. 58
The various PARP inhibitors that were tested on the same cell lines that THCV, HHC, and HHCV were tested on, especially HPAF-II, had generally a weaker response than the cannabinoids, which show a less efficacy towards the treatment of pancreatic cancer unless a higher dose is used which would entail other side effects that would be counterintuitive towards a treatment. As shown in Table 2, prototypical PARP inhibitor, PJ34 demonstrated a better potency than marketed PARP inhibitors in HPAF-II cell lines. 59 PARP inhibitors are relatively new to the market but have been used in the treatment of ovarian, fallopian tube, and peritoneal cancer. 59 Indications that PARP Inhibitors can be used in the treatment of but not limited to lung, pancreatic, prostate, and kidney and bladder cancer are still being researched.
Data derived from the Sanger Institute, 48 allowed us to compare our results to published information. The data derived from common antineoplastics that treat pancreatic cancer (Cisplatin, Gemcitabine, and Bortezomib), 48 were assayed against HPAF-II, MIA-PaCa2, and ASPC-1 PDAC cell lines from the Sanger Institute. The use of this data provides in-depth look into the effectiveness of these compounds as compared to the cannabinoids as shown below in Table 3.
Compiled Data from in-vitro Experiments and Sanger Institute Assayed Data, Comparing the IC50 Values of Tested PARP Inhibitors and Cannabinoid, with the IC50 Data of Anti-Cancer Agents Derived from GDSC2 Data Set from Sanger Institute. 48

Conclusion
THCV, HHC, and HHCV are presented as potential lead compounds for pancreatic cancer therapeutics. Although their IC50 values are not as potent as conventional cytotoxics, their efficacy in specific cell lines is comparable to FDA-approved antineoplastics. Supporting data from the Sanger Institute further suggest pharmacologic relevance. Nevertheless, the study has several limitations: IC50 values were determined via MTT alone; no in vivo or mechanistic validation has yet been performed; and minor impurities or variability in cell culture may influence outcomes. Ongoing SAR and analog development aim to improve potency and selectivity, with a focus on transitioning from micromolar to nanomolar efficacy. Future directions include evaluating 2D and 3D PDAC models, assessing key apoptotic and survival pathways, and combination studies with Gemcitabine and Abraxane in both in vitro and PDX systems.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251348390 - Supplemental material for Synthesis, Characterization, and in vitro Experiments of Saturated Cannabinoids on Pancreatic Ductal Adenocarcinoma Cell Lines (HPAF-II, MIA-PaCa2, ASPC-1, and PANC-1)
Supplemental material, sj-docx-1-npx-10.1177_1934578X251348390 for Synthesis, Characterization, and in vitro Experiments of Saturated Cannabinoids on Pancreatic Ductal Adenocarcinoma Cell Lines (HPAF-II, MIA-PaCa2, ASPC-1, and PANC-1) by Westley Cruces, Tesfay T. Tesfatsion, Giovanni A. Ramirez, Maite L. Docampo-Palacios, Arianna C. Collins, Kyle P. Ray and Prakash G. Jagtap in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
The Authors would like to acknowledge Jin Hong, PhD of Custom NMR Service for providing professional spectrum analysis as well as Asfar Azmi, PhD of Karmanos Cancer Institute for his assay work.
Ethical Considerations
Ethical approval is not applicable for this article.
Author Contributions
Conceptualization: PGJ, KPR, WC. Methodology: PGJ, KPR, WC. Investigation: GAR, ACC, TTT. Data Curation: GAR. Formal Analysis: GAR, PGJ, WC. Writing – Original Draft: GAR, TTT, PGJ, WC. Writing – Review and Editing: GAR, MLD, PGJ, WC. Supervision: KPR, WC. Project Administration: KPR, WC. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PGJ, KPR, WC are inventors on US 63/411,506, September 29, 2022 that cover these compounds. PGJ, KPR, WC are founders of Blackstone Therapeutics. KPR, WC are founders of Colorado Chromatography Labs. GAR, MLD, ACC, TTT are employed by Colorado Chromatography Labs.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplementary Information is available for this paper. 1H NMR, 13C NMR, LCMS and GCMS of the products is listed in ![]() . Within the supplementary information, methods and complete data analysis of data is provided from the Sanger Institute,
48
public data used for modeling and design of novel compounds.
. Within the supplementary information, methods and complete data analysis of data is provided from the Sanger Institute,
48
public data used for modeling and design of novel compounds.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
