Abstract
Background
Mangifera zeylanica, an endemic plant in Sri Lanka, is traditionally utilized in local medicine for the treatment of various ailments. Traditionally, M. zeylanica has been employed to treat a range of ailments, including infections, inflammations, and certain chronic conditions. This study investigates the antimicrobial potential of methanolic and aqueous extracts of M. zeylanica bark from two regions in Sri Lanka, isolate active fractions, and develop a topical formulation.
Method
Plant bark was reflux-extracted to n-hexane, methanol, and water separately. The antimicrobial efficacy was assessed against standard bacterial strains, Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, and Candida albicans, and clinically isolated strains of S. aureus and MRSA. Extracts with the highest Zone of Inhibition (ZOI) were separated through column chromatography and evaluated. Cytotoxicity was determined using the brine shrimp assay. The most active and least toxic fraction was used to develop a topical antimicrobial formulation, which was evaluated for activity, stability, and physiochemical characteristics. GC-MS and FTIR spectroscopy were used to analyze the chemical composition of the selected active fraction.
Results
The methanolic inner extracts (GMI and RMI) showed the highest ZOI of 20.67 ± 0.29 mm against S. aureus. RMI and GMI fraction 01 exhibited the highest ZOI against S. aureus and MRSA, 20.00 ± 0.25 mm and 18.25 ± 0.58 mm, respectively. RMI fraction 01, with the lowest toxicity, was chosen for the topical formulation, targeting S. aureus and MRSA with a therapeutic range of 2.08 ± 0.90 to 26.80 mg/mL. This demonstrated stability and antimicrobial activity over six months. Phytochemical analysis of RMI 01 revealed allo-aromadendrene (70.48%) as the major compound.
Conclusion
This study underscores the significant antimicrobial potential of methanolic extracts of M. zeylanica bark, particularly fraction RMI 01, for developing effective topical antimicrobial formulations and suggests that the active compound, allo-aromadendrene, may contribute to the therapeutic properties.
Introduction
Antibiotic resistance has emerged as a critical global challenge, significantly complicating the treatment of infections caused by pathogenic bacteria and fungi.1,2 These infections are a leading cause of death worldwide, underscoring the urgent need for effective strategies to combat antimicrobial resistance.3,4 As a result, there is a growing focus in pharmaceutical research on developing new dosage forms that can effectively address this pressing issue. 5 Medicinal plants have been utilized to treat a variety of ailments.6-8 Recent studies have demonstrated their potential to exhibit antimicrobial activity against resistant strains of bacteria and fungi. This brings the novel approaches to develop drugs to address the problem of antimicrobial resistance.
Mangifera species belong to the Anacardiaceae family, comprising at least 40 species distributed in Malaysia, Sri Lanka, India, Philippines, and the tropics of the world. 9 The edible varieties commonly known as Mango are a popular fruit, some with high commercial value. Different parts of this plant, including its bark, seeds, and flowers, are employed in traditional medical practices to treat a range of ailments.10,11 Studies investigating the biological activities of Mangifera species have reported antimicrobial, antioxidant, and anticancer activities.12,13 While many studies report the biological activities of Mangifera indica, other mango species have also been reported with similar biological activities. Mangifera zeylanica, commonly known as “Etamba,” is a wild mango species endemic to Sri Lanka. The plant bears edible fruits and is widely located in the intermediate and wet zone forests in Sri Lanka. 14 Various parts of the Etamba plant, including the bark and leaves, are traditionally used in herbal medicine and are believed to possess anti-inflammatory and anticancer properties.10,15,16
These medicinal plants may contain varying concentrations of active compounds depending on the climatic conditions, harvest time, and processing methods. This leads to inconsistent potency and unpredictable therapeutic effects. Additionally, the direct use of medicinal plants can expose patients to harmful contaminants such as pesticides, heavy metals, or microbial pathogens. Furthermore, some medicinal plants contain compounds that can cause adverse effects.17,18 Therefore, formulating these natural extracts into standardized dosage forms ensures consistent potency, safety, and efficacy. This approach enables precise dosing, better patient compliance, and improved therapeutic outcomes. Moreover, formulations can be tailored to target specific sites of infection or inflammation, thereby maximizing the therapeutic benefits of the active compounds. 19
Topical formulations play a significant role in delivering therapeutic agents directly to the site of infection or inflammation. These formulations can enhance the local concentration of the drug, reduce systemic side effects, and improve patient compliance. Developing effective topical formulations involves ensuring that the active ingredients are stable, penetrate the skin effectively, and maintain their antimicrobial activity. 20 Furthermore, the delivery of drugs through the skin is highly effective due to its easy accessibility, extensive surface area, and significant interaction with the body's circulatory and lymphatic systems. 21 Therefore, the present work was undertaken to investigate the antimicrobial properties of n-hexane, methanol, and aqueous extracts of the inner and outer bark of M. zeylanica against Candida albicans, Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa. Further, the bioactive compounds exhibiting potential antimicrobial properties were isolated using column chromatography, and a topical cream formulation was developed from active fractions. These active fractions were analyzed through GC-MS analysis and FTIR spectroscopy.
Methodology
Collection of the Plant Material
M. zeylanica stem bark was collected into polythene zip bags from two randomly selected areas belonging to the wet zone, Rambukkana, Kegalle District and Neluwa, Galle District of Sri Lanka. The zip bags were sealed and transported to the laboratory and stored till extraction. The plants were authenticated at Bandaranayaka Memorial Ayurvedic Research Institute, Nawinna, Maharagama, and voucher specimens were deposited in their collection (Acc No 3004).
Preparation of the Extracts
The stem bark of the plants was separated into inner bark and outer bark, which were washed separately using distilled water. Then, the bark samples were air-dried and coarsely powdered. A 20 g portion of each powdered sample was reflux-extracted for 60 min using 200 mL of n-hexane, methanol, and water separately. Then the extract was filtered through Whatman No 01 filter paper to obtain the filtrate into a sterile flask. The n-hexane and methanol extracts were rotary evaporated, and the water extract was freeze-dried to yield the solid crude extracts. The percentage yield was calculated, and the plant extracts with the highest yield were chosen for further investigation. In total, 12 extracts were prepared from the two plant specimens using n-hexane, methanol, and water from the outer and inner bark, as summarized in Supplementary Table 1, adapted from Gunawardana et al, 2023. 22
ZOI of the Plant Extracts Against the Tested Microorganisms.

*Significant p < 0.05 compared to the results of positive control of the assay. Results are presented as mean ± standard deviation.
Antimicrobial Assay of the Crude Extract/Separated Fractions
Test Microorganisms
S. aureus (ATCC 25923), P. aeruginosa (ATCC 27953), E. coli (ATCC 25932), and C. albicans (ATCC 10231) were used to determine the antimicrobial activity of the plant extracts. Standard bacterial cultures and characterized clinical isolates of S. aureus (S. aureusb 01, S. aureusb 02, S. aureusb 03, S. aureusb 04 and S. aureusb 05), MRSA (MRSAb 01, MRSAb 02, MRSAb 03, MRSAb 04, MRSAb 05), P. aeruginosa and C. albicans were obtained from a local institute.
Preparation of the Plant Extract/ Separated Fractions
The crude powder/ separated fractions (100 mg) were weighed and transferred into a sterile test tube. Then, 1 mL of sterile distilled water was added and stirred to obtain a concentration of 100 mg/mL (A). Subsequently, a series of two-fold dilutions were prepared to obtain 50 mg/mL (B), 25 mg/mL (C), and 12.5 mg/mL (D) in sterile distilled water.
Agar Well Diffusion Assay
The antimicrobial activity of the crude extracts and isolated fractions was assayed by the agar well diffusion method. Eighteen-hour (18 h) old pure cultures were used to prepare microbial cell suspension with a turbidity of 0.5 McFarland standard. Each bacterial suspension was inoculated on Mueller-Hinton agar plates using the spread plate method. The plates were then allowed to dry for 5 minutes. Four wells were prepared using a sterile cork borer (8 mm in diameter), and the agar plug was removed, and the bottom of the well was sealed using a drop of Mueller-Hinton Agar. Subsequently, 150 µL of each plant extract (A, B, C, and D) were added to wells separately. Antimicrobial discs, gentamicin (10 µg/mL) for bacteria, ketoconazole (10 µg/mL) for C. albicans, and vancomycin (30 μg/mL) for MRSA were used as the positive controls. The plates were incubated at 37 °C overnight. On the following day, the sensitivity of the test organisms to each extract was determined by measuring the diameters of the zone of inhibition (ZOI), and the average ZOI was calculated. Sterile distilled water was used as the negative control. 23
Development of the Thin Layer Chromatography (TLC) Profile
The TLC profiles of the most bioactive extracts from M. zeylanica were obtained using various combinations of solvent systems in different proportions. A measured quantity of 0.01 g of each extract was dissolved in 1 mL of methanol. Approximately 10 μL of each solution was spotted on precoated aluminum silica gel G25 TLC plates. The solvent system was developed to achieve the best resolution (Chloroform:Methanol:n-Hexane:Formic acid, 7:3:2:1).
13
After development, the TLC plates were observed under a fluorescence lamp at a wavelength of 254 nm to determine the solvent front and the spots. The Retention factor (Rf) value was calculated using the following standard Equation 1.
Bioactivity-Guided Fractionation
Based on the results of the well diffusion assay, selected extracts with higher antimicrobial activity were subjected to bioactivity-guided fractionation using column chromatography. A column of 30 cm length having a diameter of 15 mm was packed with silica gel of mesh size 200-400 μm as the stationary phase, and ethyl acetate was used as the mobile phase. A slurry containing 0.5 g of extract was loaded onto the packed column. The fractions were eluted using a combination of ethyl acetate and methanol in different ratios. Eluents were collected separately from the bottom of the column at 10 mL intervals. Then, the collected fractions were subjected to TLC analysis. The fractions with similar profiles were pooled, and their antimicrobial activity was assayed by the agar well diffusion method.
Minimum Inhibitory Concentrations (MIC) and Minimum Bactericidal Concentrations (MBC)
Microbial cell suspensions of reference microbial strains and clinical isolates equal to 0.5 MacFarland turbidity standard were prepared. Ten microliters (10 μL) of this suspension were added to 9990 μL of sterile saline (0.9% NaCl w/v) to prepare the final inoculum. Then 100 μL from the inoculum and 100 μL of each dilution of separated fractions (A, B, C, D) were added to 96 well plates and incubated for 24 h at 37 °C. A 337.5 mg of resazurin powder was dissolved in 50 mL of sterile distilled water and vortexed the solution for 1 h to ensure homogeneity. 24 Subsequently, 50 μL of the resazurin solution was added into each well, and the plate was incubated again for 2 h. The viability of bacteria was recorded visually based on the color change from blue to pink. The lowest concentration at which color change was observed was recorded as the MIC value. MIC was determined in triplicates, and the average value was calculated for each test material against selected bacterial species.25,26 To determine the MBC, 10 μL from each well was transferred to Muller Hinton agar plates and incubated at 37 °C for 24 h. The lowest concentration of extracts showed the complete absence of visible growths on the Muller Hinton agar plate was taken as the MBC value.27,28
Determination of Toxicity of the Separated Fractions Through Brine Shrimp Lethality Assay
Artificial seawater (commercial marine salt 27.0 g/L) of pH between 7.9–8.1 was prepared. Brine shrimp eggs (0.1 g) were added to a conical flask containing 250 mL of artificial seawater and constantly aerated for 24 h in the presence of a light source. Active nauplii hatched were collected after 24 h.
A 100 mg of each fraction which exhibited potent antimicrobial activity was measured and dissolved in artificial sea water and prepared a stock solution of 100 mg/mL. A double dilution series of 50, 25, 12.5, 6.25, 3.13, 1.56, 0.78, 0.39, 0.19, 0.09, 0.05 mg/mL was prepared using artificial seawater. Then, 5 mL of these solutions were transferred to McCartney bottles, and 20 active nauplii were placed in each McCartney bottle. Test samples were kept for 24 h under light, and survivors were counted using a stereo microscope. The percentage of mortality in each concentration was calculated using equation 2 below, and the LC50 value was calculated.29,30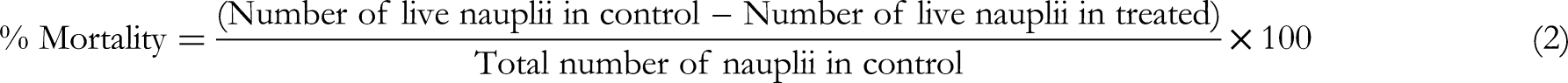
Calculation of the Therapeutic Range of the Antimicrobial Cream Formulation
Depending on the results obtained for MIC and MBC values and LC50 value, the therapeutic range was determined. Then three doses were selected, representing the high, middle, and lower ranges in the therapeutic range calculated.
GC-MS Analysis of the Separated Fractions with the Highest Antimicrobial Activity
The separated fractions which showed the highest antimicrobial activity were selected, and the chemical composition of the selected fraction was analyzed using Gas Chromatography-mass Mass Spectrometry (GC-MS) analysis on a HP-5MS 5% Phenyl Methyl column with a nominal length of 30.0 m, a diameter of 250.00 μm, and film thickness of 0.25 μm. The initial flow rate was 1.0 mL/min, and helium was used as the carrier gas. The initial column temperature started from 60 °C, and the temperature increased by 4 °C per minute to 220 °C. The total GC running time was 55.00 min. The components of the separated fraction were identified by comparison of their mass spectra with those of the spectrometer database using the Wiley W9N11 library. The identifications were confirmed by comparison of the fragmentation pattern and their retention indices with those reported in the literature.31,32
FTIR Analysis of the Separated Fraction
The Fourier-transform infrared (FTIR) analysis was performed using an ATR-FTIR spectrometer (Thermo Scientific – Nicolet iS10). FTIR spectra of solid methanolic extract (RMI Fraction 01) was obtained. The recording range of the spectrum was 525-4000 cm−1 at 16 scans per minute with a resolution of 4 cm−1 in transmission mode. 33
Formulation of Antimicrobial Cream
The required amount of three selected concentrations of active fractions was calculated and measured to prepare 30 mL of the formulation. The extracts were dissolved in sterile distilled water by stirring on a magnetic stirrer for 15 min. The oil phase was prepared by initially heating stearic acid and cetostearyl alcohol at 70 °C while constantly stirring at 300 rpm. Then, tween 80 and virgin coconut oil (VCO) were added to this mixture and mixed for another 10 min until the mixture cooled to 40-50 °C. Then, the water phase was added to the oil phase dropwise on a magnetic stirrer at 300 rpm. The resultant mixture was further stirred for another 20 min at 300 rpm. While stirring, triethanolamine (TEA), phenoxyethanol, glycerol, and fragrance were added dropwise, and then the mixture was homogenized at 10000 rpm for 5 min. 34
Evaluation of the Antimicrobial Activity of the Developed Formulations
The antimicrobial activity of the developed formulations was evaluated using agar well diffusion assay by adding a weight of 0.29 g of each cream to the wells using 3 cc/mL tuberculin syringes separately. 35 Then, gentamicin and vancomycin standards were also placed on respective plates and incubated at 37 °C overnight. The assay was done in triplicates. On the following day sensitivity of the test organisms to each extract was determined by measuring the diameters of the ZOI.
Stability Evaluation of the Topical Antimicrobial Formulation
Physical Appearance
The stability evaluation of the cream formulation, which was stored at room temperature (27 ± 2 °C), was carried out for 180 days. Visual observations on color, texture, and phase separation were taken on the first,15th, 30th, 45th, 60th, 75th, 90th, 105th, 120th, 135th,150th, 165th, and 180th days.
Characterization of the Formulations
The tests were carried out according to the specifications of Sri Lanka Standard (SLS) 743:2014 (specification for skin creams and lotions).
pH Determination of the Creams
Five grams (5.00 g) of the cream was dispersed in 45 mL of water, and the pH of the suspension was determined at 27 ± 2 °C using the pH meter (Thermo Scientific Orion Star A211). 36
Viscosity Determination of the Emulsions
The viscosity was measured using Brookfield viscometer. The freshly prepared formulation was kept at room temperature (28 °C) for 2 h before the measurements. Spindle number 28 was used for all measurements. Formulations were loaded to the small spindle adaptor of the viscometer, and the viscosity was measured at 10 rpm. Viscosity and the relevant torque values were recorded.
Thermal Stability Evaluation of Creams
A 20 mm wide and 5 mm thick strip of cream was applied on the internal wall of a small glass beaker. The beaker was placed in a humidity chamber for 8 h and observed for any separation of oil on removal.
Determination of Non-Volatile Matter
Approximately 5 mg of the sample was heated at 105 ± 2 °C for 2 h, and the weight of the residue was measured. The non-volatile matter percentage was calculated using the following equation 3.
Where,m1 – the mass of the dish with the sample before heating in grams, m2 – the mass of the dish with the sample after heating in grams, m3 – the mass of the empty dish in grams
Determination of the Water Content of the Formulation
Approximately 10 g of the cream sample was subjected to distillation by refluxing. Then, the apparatus was allowed to cool and separate the water and solvent layers. Once the two layers were separated, the volume of water was measured. The water content was calculated using the following equation 4.
Where; V = volume of water collected in the receiving tube in milliliters, d = density of water at room temperature, m = mass of the test sample in grams
Determination of the Peroxide Value
A 10 g portion of the cream was warmed in a water bath and separated the oil layer by centrifuging at 3500 rpm. The oil was dissolved in chloroform. Glacial acetic (15 mL) acid and fresh saturated aqueous potassium iodide (KI) solution (1 mL) were added and shaken well for 1 min. This mixture was then kept in a dark, cool place for 5 min. To the resulting mixture, 75 mL of distilled water was added and stirred vigorously. Then, a few drops of iodine were added to it, and the mixture was titrated with 0.002 M sodium thiosulphate solution. The peroxide value was calculated using the following equation 5,
Where; V – volume in mL of sodium thiosulphate solution used for titration, VO – volume in mL of reagent blank determination, n – normality of the thiosulphate solution, m – mass in g of oil used
Determination of the Spreadability of the Formulation
The spreadability of the formulations was determined by measuring the spreading diameter of 0.5 g of the cream sample on a pre-marked circle of 2 cm diameter between two horizontal glass plates. Then, 500 g weight was placed on the upper glass plate for 5 min. The average diameter was taken after triplicate measurements.
37
The spreadability was calculated using the following equation 6.
Where; S = Spreadability (cm2), m = Weight applied on upper glass plate (g), L = Diameter (cm), t = time taken for the glass plate to move the specified length (s)
Statistical Analysis
All the experiments were triplicated, and results were recorded as mean ± standard deviation. Data were statistically analyzed by Analysis of Variance (ANOVA) followed by Tukey's pot-hoc test at 0.05 significance level. LC50 values were calculated using the GraphPad Prism 9.0 software.
Results
Yield Percentage of the Plant Extracts
According to the study done by Gunawardana et al, 2023, 22 the inner and outer bark extracts of M. zeylanica using methanol and water have yielded the highest amounts, whereas extraction with n-hexane produced the lowest yield percentage. Consequently, methanol and water extracts were chosen for further testing.
Antimicrobial Activity of the Plant Extracts
ZOI were observed against S. aureus, C. albicans, and P. aeruginosa, while no ZOI was observed for E. coli. The highest ZOI for 3 selected organisms were seen with RMI, GMI, and RWI extracts, and these were used for further investigation.
The highest ZOI against C. albicans was observed in the presence of RMI. Both RMI and GMI extracts exhibited the highest antimicrobial activity against S. aureus. The highest ZOI against P. aeruginosa was observed for extract RWI (Figure 1). Interestingly, the RMI fraction at 100 mg/mL concentration showed comparable antimicrobial activity (p < 0.05) against S. aureus and P. aeruginosa compared to the positive control (Table 1).

ZOI Observed (A) 100 mg/mL, (B) 50 mg/mL, (C) 25 mg/mL, (D) 12.5 mg/mL, Different Concentrations of the (i) RMI Against C. albicans (ii) GMI and (iii) RMI Against S. aureus (iv) RWI Against P. aeruginosa.
Bioactivity-Guided Fractionation
Bioactivity-guided fractionation was performed on the extracts RMI, GMI, and RWI, and each extract was separated and pooled into three fractions based on Rf value. According to the previous study by Gunawardana et al, 2023, 22 all 03 extracts, were separated into 2 fractions where, fraction 01 had a Rf range of 0.6–0.8, and fraction 02 had a Rf range of 0.2–0.4.
Antimicrobial Activity of the Separated Fractions
RMI fractions 01 and 02 at 100 mg/mL resulted in a ZOI of 10.67 ± 0.76 mm against reference strains of C. albicans. However, no ZOI were observed against 5 clinical isolates of C. albicans. Further, no antimicrobial activity against P. aeruginosa was observed in the presence of separated fractions of RWI.
RMI Fraction 01 and GMI Fraction 01 (100 mg/mL) had the highest ZOI of 20.00 ± 0.25 mm and 18.25 ± 0.43 mm, respectively, against the reference strain of S. aureus, and both fractions were active against MRSA and clinical isolates of S. aureus. MIC and MBC for fraction 01 of GMI and RMI against standard strains of S. aureus and clinical isolates of S. aureus and MRSA 01 are depicted in Table 2. The lowest MIC for RMI fraction 01 and GMI fraction 01 were 2.08 ± 0.90 mg/mL and 12.50 ± 0.00 mg/mL, respectively.
MIC and MBC Concentrations of the Selected Fractions Against S. aureus and MRSA.

Brine Shrimp Assay for Detection of the Toxicity of the Separated Fractions
The LC50 values obtained for GMI fraction 01 and RMI fraction 01 were 3.11 mg/mL and 13.40 mg/mL, respectively (Figure 2). RMI fraction 01 had lower toxicity and the highest antimicrobial activity when compared to GMI fraction 01, and therefore, RMI fraction 01 was selected for the development of the formulation.

Percentage Mortality of the Separated Fractions GMI 01 and RMI 01 Against Log Drug Concentrations.
Determination of the Therapeutic Range of the Fractions
Based on the MIC and MBC values, the lowest therapeutic value was 2.08 ± 0.90 mg/mL and depending on the toxicity assay the LC50 was 13. 40 mg/mL. Therefore, the maximum toxicity of the separated fractions will be at the concentration 26.80 mg/mL. This means the administration of doses above 2.08 ± 0.90 mg/mL up to 26.80 mg/mL can provide the therapeutic activity to the patients. Thus, 2.08 ± 0.90 mg/mL to 26.80 mg/mL can be identified as the therapeutic range of the separated fraction 01 of RMI against S. aureus and MRSA. (Supplementary Figure 1).
Depending on the therapeutic range of the three doses, A (20 mg/mL), B (13.40 mg/mL), and C (10 mg/mL) were selected for the development of the formulation.
Antimicrobial Activity of the Formulation
Agar well diffusion assay for the formulation, which contained the RMI fraction 01 as the active ingredient without the preservative, demonstrated antimicrobial activity against S. aureus and MRSA at 20 mg/mL, 13.40 mg/mL, and 10.00 mg/mL. The antimicrobial activity of the developed formulation against reference strains and clinical isolates of S. aureus was found to be significant (p < 0.05) compared to the gentamicin standard at all test concentrations (Table 3). Similarly, the developed formulation demonstrated antimicrobial activity against different strains of MRSA was found to be significant (p < 0.05) compared to the vancomycin standard at all test concentration (Table 4). The formulation without the active ingredient (negative control/ placebo) had no ZOI (Figure 3).
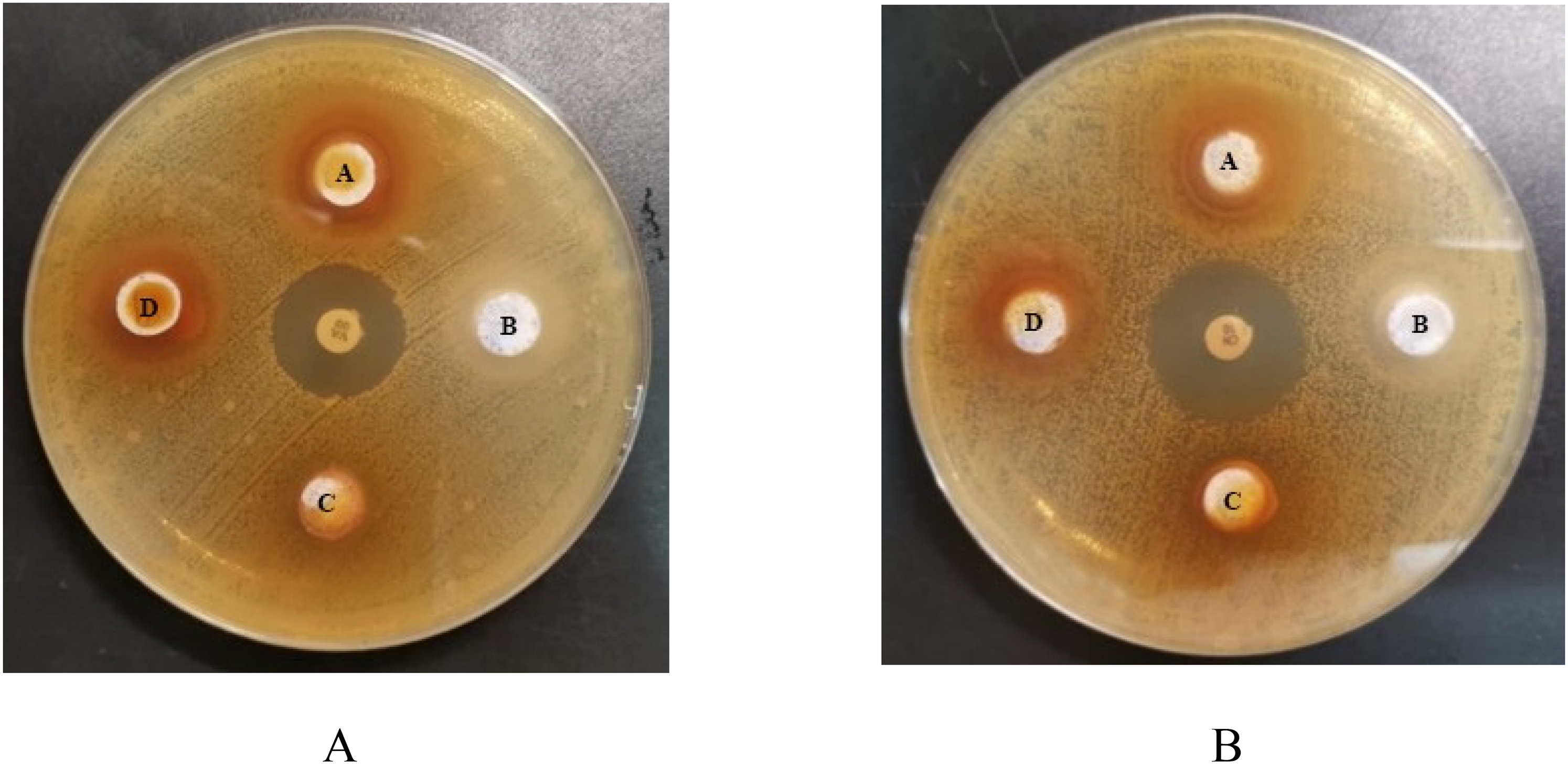
ZOI Shown by the Formulations Containing RMI Fraction 01 Against (A) MRSA and (B) S. aureus.
ZOI of Developed Formulations Against Reference Strain of S. aureusa and Clinical Isolates of S. aureusb.

*Significant p < 0.05 compared to the results of positive control of the assay. Results are presented as mean ± standard deviation.
ZOI of Developed Formulations Against MRSA.
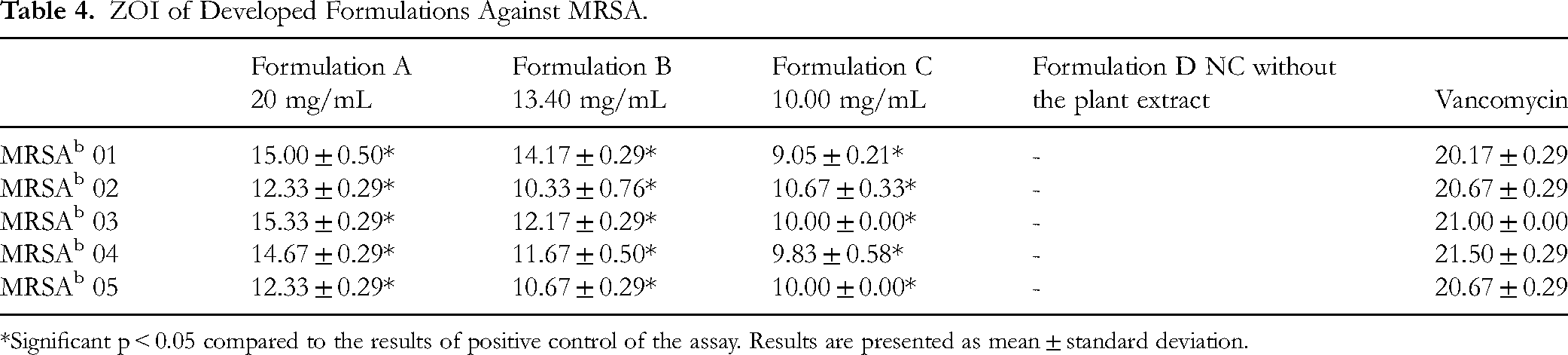
*Significant p < 0.05 compared to the results of positive control of the assay. Results are presented as mean ± standard deviation.
Stability of the Antimicrobial Formulation
The formulation was found to be stable with no phase separation, change in color, or texture for a period of 180 days.
Characterization of the Formulation
The results of characterization are shown in Table 5. The tests were carried out according to SLS 743:2014, and all the parameters were within the reference ranges. The peroxide value of the developed formulation was 0 milliequivalents/kg. This indicates that the formulation does not contain reactive oxygen contents, and the formulation was within the specified range. Further, no separation of oil was observed in the formulation on removal from the humidity chamber. Thus, the formulation was considered as thermostable.
Tested Parameters of the Antimicrobial Formulation.

GC-MS Analysis of RMI Fraction 01
Figure 4 depicts the GC-MS chromatograph of RMI fraction 01. Table 6 presents the GC-MS analysis results and the identified compounds in the extracts. Based on abundance, the major compound present in the RMI fraction 01 is allo-aromadendrene at 70.477%.
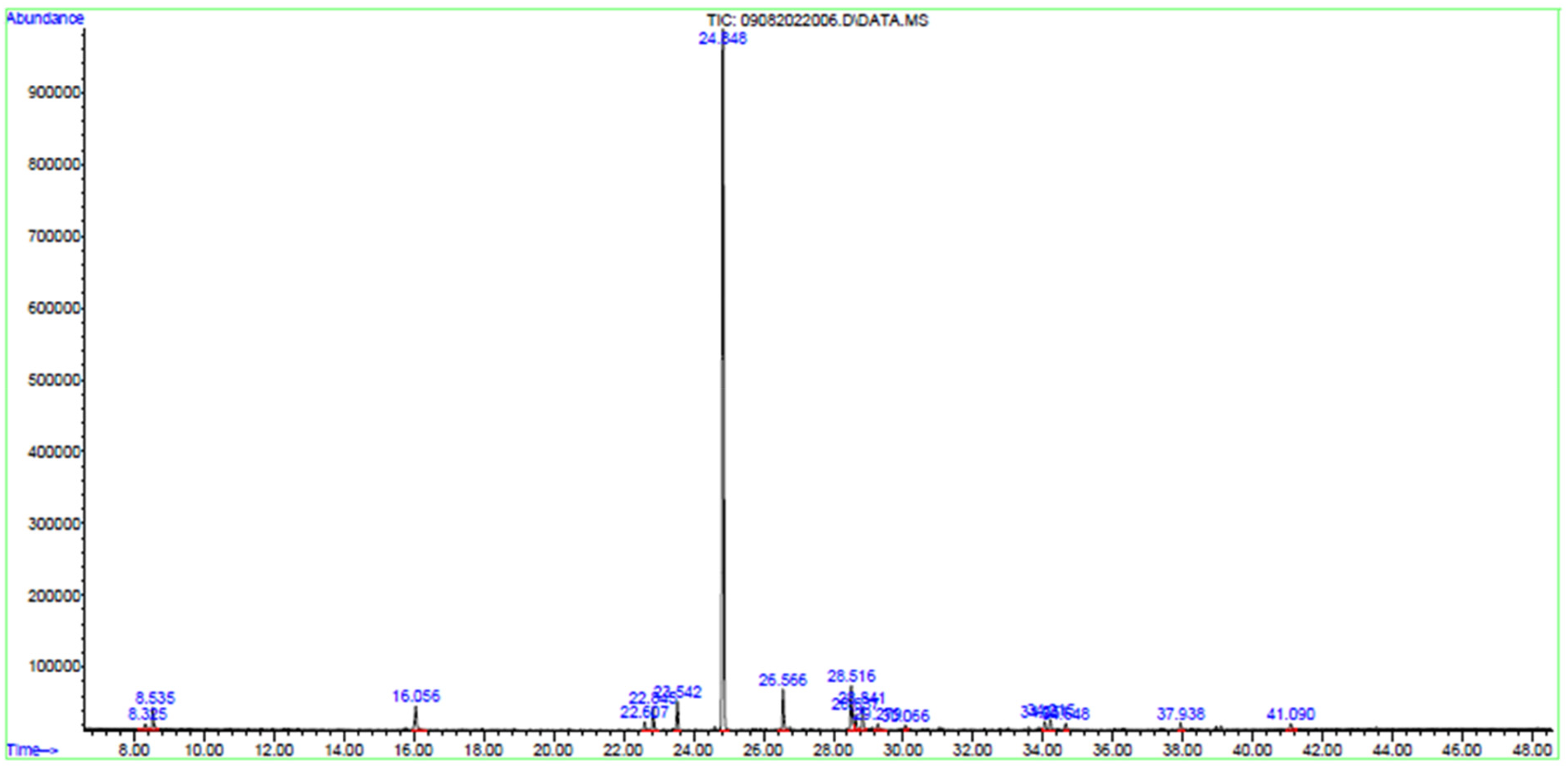
Chromatogram of the Bioactive Compounds Present in RMI Fraction 01.
Biologically Active Chemical Compounds of RMI Fraction 01 Detected from GC-MS.

FTIR Analysis of RMI Fraction 01
Figure 5 presents the FTIR spectrum of the RMI fraction 01. It displays the absorption peaks at specific wavenumbers, which can be used as a tool to characterize the sample. A broad peak at 3217 cm−1 suggests the presence of O-H stretching vibrations, which can be due to the presence of 2,4-di-tert-butylpheno and trifluoroacetic acid. Narrow peaks at 2922 cm−1, 1455 cm−1, and 1251 cm−1 correspond to C-H vibrations usually found in aliphatic hydrocarbon chains like decane, dodecane, teradecane, and hexadecane. The peak at 1614 cm−1 may indicate the presence of C = C stretching vibrations, characteristic of aromatic compounds or conjugated double bonds in constituent molecules like allo-aromadendrene, β-caryophyllene, caryophyllene oxide, and 2,4-di-tert-butylphenol.
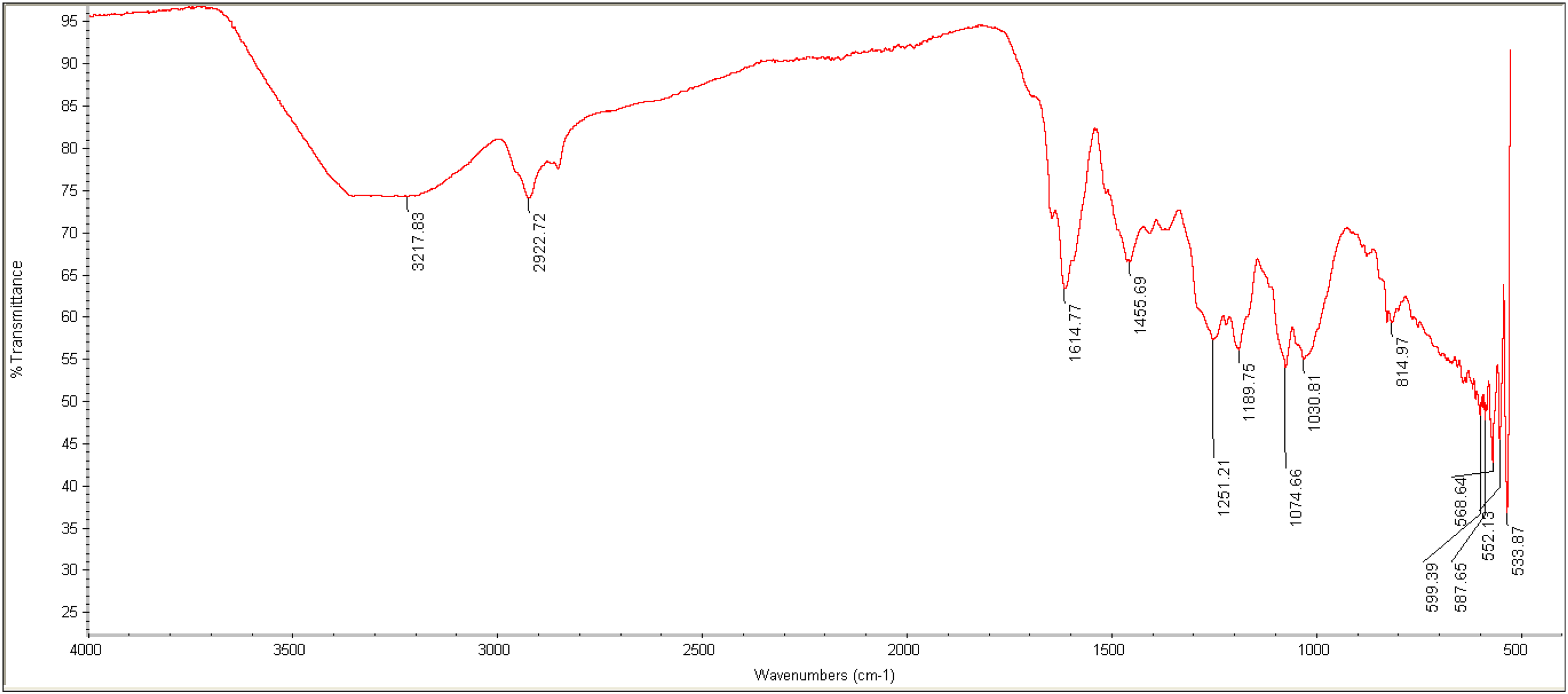
The FTIR Spectrum of the RMI Fraction 01.
Discussion
Investigation of endemic plants for their pharmacological properties may lead to novel therapeutics, environmental sustainability, and conservation. M. zeylanica, which is endemic to Sri Lanka, has been used in ayurvedic treatment of diarrhoea, vomiting, fevers, hiccups, oral diseases, bladder diseases, and uterus ailments. 38 This plant is known for being the tallest among the mango species and is under threat since it is not popular for domestic or commercial cultivation.
M. zeylanica stem bark has been traditionally used for managing a range of health conditions. In order to study pharmacological properties, n-hexane extracts were prepared to collect non-polar/lipophilic compounds, while polar/hydrophilic compounds were extracted into water and methanol. The phytochemical composition of different parts of the same plant may differ, and therefore, it is important to select the correct solvent for the extraction of the phytochemicals for further investigations. 39 A study by Ediriweera et al, 2016, 11 reported the anticancer activity of n-hexane extract of the seed kernel of M. zeylanica. However, in the present study, n-hexane extract resulted in a very low yield percentage, whereas methanol and water extracts resulted in a high yield percentage, enabling further investigation. Similarly, a study done in Zimbabwe reported water extract of M. indica stem bark has given a high percentage yield (13.67%), followed by methanol (10.33%), ethyl acetate (12.33%), and n-hexane (0.92%). This indicates that highly polar solvents are favored for extraction efficiency. 40
Although zones of inhibition against S. aureus and C. albicans were observed in the crude inner bark extracts of methanol and against P. aeruginosa in extracts of the inner bark of water, following fractionation the zones of inhibition were only observed against S. aureus and MRSA. The susceptibility of organisms to various phytochemicals can be affected by the composition of the microbial cell wall. The presence of alkaloids and saponins in aqueous extracts of the stem bark of M. indica have been attributed to antimicrobial activity against E. coli, 41 while the presence of flavonoids, (poly)phenols, and terpenoids have shown antimicrobial activity against S. aureus and C. albicans.42,43 According to the studies, phenolic acid has shown antibacterial activity against Gram-positive bacteria, while dihydrochelerythrine and dihydrosanguinarine alkaloid constituents have shown antimicrobial activity against Gram-positive and Gram-negative bacteria and C. albicans.44,45 A study done by da Silva et al, 2017, 46 has shown that the antifungal activity of eugenol, a monoterpenoid against C. albicans, may act through the production of free radicals. Flavonoids demonstrate antimicrobial activity against Gram-positive bacteria by disrupting nucleic acid metabolism, impairing cytoplasmic membrane function, and hindering energy metabolism. Additionally, flavonoids prevent bacterial attachment and biofilm formation, inhibit the porin channels on the cell membrane, alter membrane permeability, and reduce pathogenicity. 47
It was interesting to observe that although the separated fractions exhibited antimicrobial activity against reference strains of C. albicans, it was inactive against the clinical isolates. Further, there was no activity against P. aeruginosa and E. coli. Clinical isolates represent the heterogeneous spectrum of pathogens causing real-world infections. Clinical isolates may have developed various characteristics to survive in the complex and challenging environment of the human body, thereby demonstrating differing antimicrobial susceptibility. 48 The crude extracts contain a complex cocktail of phytochemicals, which can act synergistically to provide better antimicrobial activity compared to the activity of isolated phytochemicals. 49
Evaluating the cytotoxicity of a compound with therapeutic potential is a critical step in the early stages of drug discovery and development. The brine shrimp assay is a simple, cost-effective method to screen the toxicity in the laboratory compared to animal studies and cell culture. 50 A key outcome of employing the brine shrimp assay is the calculation of the lethal concentration (LC50). This enables the initial screening of safer and more promising compounds for further investigation. A study done by Parra et al, 2001, suggests there is a good correlation between LC50 value obtained from brine shrimp assay and LD50 value obtained from in vivo toxicity assay in mice with a correlation coefficient of r = 0.85. 51 Thus, in this study brine shrimp assay was used to calculate the therapeutic index along with the results of MIC and MBC. When evaluating the cytotoxicity of substances, a higher LC50 value signifies that a relatively higher concentration of the substance is required to induce cytotoxic effects in cells or organisms. This, in turn, implies that the substance has a lower potential to harm living cells or organisms and is, therefore, considered less cytotoxic. Moreover, a higher LC50 implies that a drug can be administered in a wider dose range before reaching potentially lethal toxicity levels. Thus, RMI fraction 01 was considered the least toxic and highest active fraction, and it was selected for the development of the antimicrobial formulation.
The formulation developed from RMI fraction 01 demonstrated consistent antimicrobial activity against S. aureus and MRSA at various concentrations, the highest concentration giving the highest ZOI. Specifically, the antimicrobial activity against reference strains and clinical isolates of S. aureus and MRSA was significantly higher (p < 0.05) compared to the positive controls at all test concentrations. This reinforces its potential as a treatment option in light of increasing antibiotic resistance. When preparing a formulation, standardization is essential to ensure safety, efficacy, and quality. Reference standards sourced from the British Pharmacopoeia (BP) and the SLS 743:2014 outline the methods for evaluating the stability and quality of an antimicrobial formulation over a period of six months under typical storage conditions. The BP provides an array of monographs and guidelines, serving as a gold standard for assessing the quality of pharmaceuticals. Moreover, the incorporation of SLS 743:2014, which specifically addresses the specifications for skin creams and lotions, attests to our commitment to local regulatory compliance. According to the present study, the stability tests conducted over a period of 180 days confirmed that the formulation maintained its integrity without any phase separation or changes in color or texture, indicating its suitability for long-term storage and use. Further, the viscosity, spreadability, pH, peroxide value, content of heavy metal, water, and non-volatile matter were within the specified range of SLS 743:2014.
Many pharmaceutical dosage forms of herbal products are designed today by incorporating isolated bioactive compounds rather than crude extracts. This can improve efficacy and reduce unwanted pharmacological actions. 52 Characterization analyses, including GC-MS and FTIR, provided valuable information regarding the chemical composition of RMI fraction 01. The major compound identified by GC-MS analysis, allo-aromadendrene, is a sesquiterpene, which suggests that it may play a significant role in the observed antimicrobial activity. FTIR analysis further revealed functional groups indicative of various phytochemicals. Allo-aromadendrene is characterized by a fusion of a cyclopropane ring to a hydroazulene skeleton 53 and has therapeutic value. Allo-aromadendrene obtained from the plant extracts of Onychopetalum amazonicum (Annonaceae) and Dianella ensifolia have exhibited good antimicrobial activity against Staphylococcus epidermidis and E. coli. 54
Conclusion
The present study underscores the potential of M. zeylanica as a source of natural antimicrobial agents against resistant strains of bacteria. Identification of the major compound allo-aromadendrene within the active fraction holds promising implications for the potential use of this compound as a future therapeutic agent in combating various microbial infections. The separated fractions, when formulated as a cream, also showed antimicrobial activity. Further, the formulation exhibited acceptable chemical and physical properties, indicating its ability and compatibility as a topical formulation. However, comprehensive studies are necessary to explore the therapeutic potential, safety, and effectiveness of compound allo-aromadendrene in clinical settings and develop the formulation further, incorporating the active ingredient.
Limitations
The reflux extraction process can degrade the thermolabile compounds due to the heat supplied during the process. This can impact the extraction efficiency and result in the loss of sensitive compounds that are susceptible to degradation at high temperatures. The use of GC-MS only may have limited the detection of certain phytochemicals, particularly those that are non-volatile or thermally labile. The inclusion of LC-MS could have provided a more comprehensive phytochemical profile, potentially identifying additional bioactive compounds responsible for the antimicrobial activity, which is a limitation of the present study. Further, NMR spectroscopy was not conducted in the present study for the structural elucidation of the compounds. The plant bark collected only from two districts was analyzed in the present study. This restricted geographical coverage may limit the detection of the phytochemical composition of plant materials that may vary based on environmental factors such as climate, soil type, altitude, and seasonal changes.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251346319 - Supplemental material for Development of Antimicrobial Formulation and Phytochemical Analysis of Mangifera zeylanica Bark Extracts
Supplemental material, sj-docx-1-npx-10.1177_1934578X251346319 for Development of Antimicrobial Formulation and Phytochemical Analysis of Mangifera zeylanica Bark Extracts by Shehara Lakchani Abeysinghe Gunawardana, Malitha Aravinda Siriwardhene and Narayana Mudalige Sathkumara Sirimuthu, Chinthika Gunasekara in Natural Product Communications
Footnotes
Abbreviations
Acknowledgement
Center for Nanocomposite Research, Faculty of Applied Sciences, University of Sri Jayewardenepura, Sri Lanka and Center for Plant Materials and Herbal Products Research, University of Sri Jayewardenepura, Sri Lanka
ORCID iDs
Ethical Considerations
Ethical approval to report this case was obtained from the Ethics Review Committee, Faculty of Medical Sciences, University of Sri Jayewardenepura (Approval number 40/19).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial assistance by University of Sri Jayewardenepura Research grant ASP/01/RE/MED/2018/72.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used and analyzed during the current study are all provided in the manuscript.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
