Abstract
Background
Morphine use is often accompanied by the development of tolerance and addiction, with relapse being a hallmark of addictive behaviour. This study examined the potential of the naturally occurring molecule Carvacrol (CA) to mitigate morphine addiction and associated symptoms in mice.
Methods
Different behavioural paradigms, including, tail immersion, hot plate, jumping, diarrhoea score, weight loss, and conditioned place preference (CPP) were used. Biochemical analyses assessed antioxidant enzyme activities and nitric oxide levels, while RT-PCR and immunohistochemistry (IHC) were performed to study gene and protein expression changes, respectively.
Results
Findings revealed that CA dose-dependently suppressed tolerance, withdrawal symptoms, and reduced morphine-induced conditioned place preference (CPP). Furthermore, CA-treated animals exhibited a significant increase in the activities of Glutathione S-Transferase (GST), reduced glutathione (GSH), and catalase (CAT), along with a reduction in lipid peroxidase (LPO) levels in the brain compared to morphine-only treatment. The Immunohistochemistry (IHC) studies showed that CA inhibited nNOS expression, while mRNA analysis revealed that CA curtailed the expression of NR2B, a subunit of the NMDA receptor. These effects, along with reduced expression of proinflammatory mediators such as TNF-α and NF-κB, suggest that CA acts through antioxidant, anti-inflammatory, NR2B- and nNOS-dependent mechanisms to counter morphine-associated tolerance, dependence, and CPP.
Conclusions
CA may be an interesting natural agent that may be useful in the suppression of morphine analgesic tolerance, withdrawal symptoms, and place preference and beneficial in ameliorating morphine addiction.
Introduction
Morphine with a history spanning over centuries still remains a cornerstone worldwide for managing pain in clinical practice. Morphine might find its use in severe chronic intra-operative, post-operative, central, and cancer-related pains due to its efficacy, potency, rapid onset, and cost-effectiveness. However, the use of morphine is not without limitations. For instance, the emergence of antinociceptive tolerance is clinically evidenced by the reduction of total analgesia following repeated administration of morphine and severely limits its clinical use. 1 Hence, higher doses are needed to maintain the intended analgesic effect which may lead to respiratory depression and brain hypoxia.2,3 Another phenomenon associated with repeated morphine administration is physical and psychological dependence. 4 Symptoms such as anxiety, sneezing, anorexia, abdominal discomfort, and diarrhoea may frequently arise following cessation of morphine use.4,5 Other than physiological reasons, these unpleasant symptoms might contribute to increased drug-seeking behaviour, impaired judgment, and recurring preoccupation among various psychological implications that might precipitate as addiction.
Besides the use of morphine for pain relieving and medical benefits, this drug has the potential for misuse that might lead to opioid use disorder (OUD) and addiction. The perception of insufficient control of pain, relief from emotional distress, and recreational opioid use are among various reasons that might contribute to this disorder. 6 Drug misuse causes changes in the reward circuits, which are key neural networks involved in processing pleasure and reinforcement. These changes can lead to obsessive behaviour and cravings which are central features of addiction. 7 In particular, relapse to drug use is one of the major hindrances in the treatment of OUD which is triggered by drug reinstatement, linked cues, and environment, feelings, individuals, or stress.8–12 Current therapies for morphine addiction, such as opioid substitution treatments, while effective in reducing withdrawal symptoms and cravings, have significant limitations. These include the risk of developing dependence on the substitute drugs, adverse side effects, high relapse rates, and the limited ability to address the underlying neurochemical and behavioural changes associated with addiction. 13 Furthermore, these therapies often fail to adequately target the oxidative stress and inflammatory pathways that contribute to morphine-induced neuroadaptations. Consequently, there is a pressing need for novel therapeutic agents that can address these limitations by targeting multiple biochemical and behavioural aspects of addiction. Medicinal plants have historically served as a source of new drugs and novel pharmacologically active molecules. 14 A phenolic monoterpenoid Carvacrol (CA) found in essential oils of thyme (Thymus vulgaris), oregano (Origanum vulgare), wild bergamot (Citrus aurantiumbergamia), pepperwort (Lepidium flavum), and other plants is known for a wide range of biological activities. 15 Among others, the neuroprotective effects of CA are particularly notable and have been reported in various studies.16–20 Therefore, this study was undertaken to investigate the beneficial effects of this agent on morphine tolerance, dependence, and relapse to drug use through a variety of behaviour and biochemical paradigms.
Materials and Methods
Animals
Wild type male albino mice weighing 20-25 g were used for the experiments. All mice were randomly divided into groups for each experiment, with a minimum of n = 6 per group as follows: Group 1 (Saline treated), Group 2 (Morphine treated), Group 3 (Carvacrol 10 mg/kg + morphine) and Group 4 (Carvacrol 20 mg/kg + morphine). The animals were housed in groups with no more than 3 animals per cage under standard laboratory conditions, including a 12-h alternating light/dark cycle at a controlled temperature of 25 °C, with free access to food and water. These housing conditions were selected to minimize stress and ensure uniformity in environmental exposure across groups. All the investigational protocols were approved by the Research and Ethics Committee of the Institute of Pharmaceutical Sciences (Ref. No. REC/RIPS/2023/17) and adhered to the international guidelines for principles of animal care. A summary of experimental protocols including behavioural and biochemical paradigms have been presented in Figure 1.
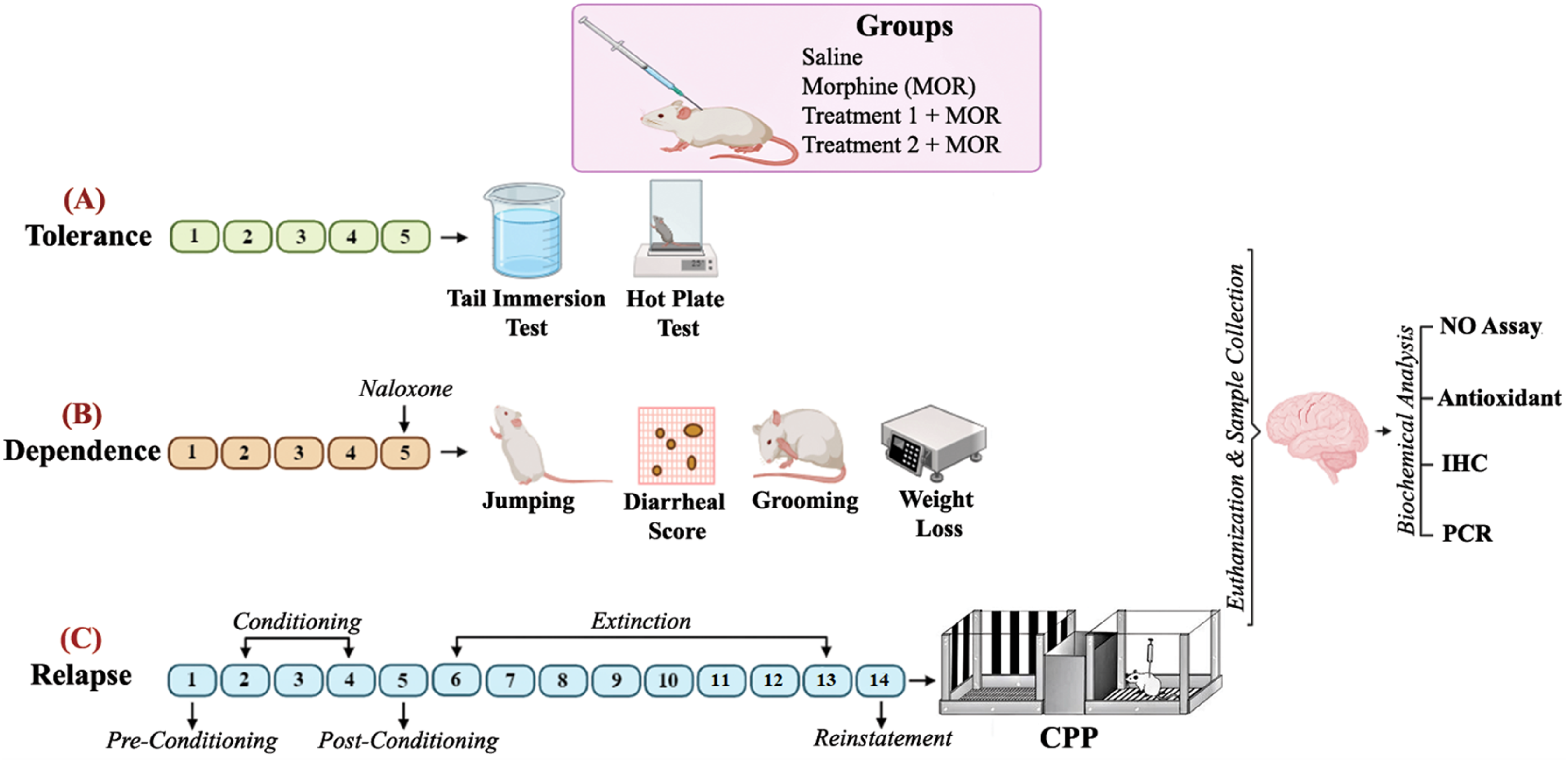
Experimental Protocol (A) Tolerance Model: in Tolerance Model, Tail Immersion and hot Plate Test Were Employed for Behaviour Analysis. (B) Dependence Model: Morphine Withdrawal Symptoms Were Analysed After Naloxone Injection (C) Relapse Model. Condition Place Preference Test was Used for Evaluating the Acquisition, Extinction, and Reinstatement.
Drug and Reagents
Carvacrol (#282197, PubChem ID: 24857025), 5, 5′-dithiobis (2-nitrobenzoic acid) (DTNB), Griess reagent, trichloroacetic acid (TCA) and N-(1-naphthyl) ethylenediamine dihydro chloride, ethanol, xylene, 3, 3-diaminobenzidine peroxidase (DAB), Avidin Biotin Complex (ABC) kit, 1-chloro-2, 4-dinitrobenzene (CDNB) and hydrogen peroxide (H2O2), were purchased from Sigma-Aldrich (USA, St. Louis, MO). Phosphorylated nuclear factor-kappa B (SC-27190), tumor necrosis factor-alpha (SC-52B83), inducible NOS (SC-7271) antibodies were purchased from Cruz Biotechnology, Dallas, Tx, United States, and neuronal NOS (18984-1-AP) antibody was purchased from Proteintech, Manchester, UK.
Induction of Morphine Tolerance
Tolerance to the antinociceptive effects of morphine was induced by repeated administration of three daily doses of morphine to mice (50 mg/kg, 50 mg/kg, and 75 mg/kg) (i.p.) for 4 consecutive days. On the fifth day, a single dose of 50 mg/kg was administered. 21 Each day, CA (10 mg/kg or 20 mg/kg) (i.p.) was administered 45 min prior to each injection of morphine to separate treatment groups. Behavioural studies including tail-immersion and hot-plate tests were conducted 60 min after the administration of the first dose of morphine. 22 Conducting the tests at 60 min ensured that measurements reflected the maximum pharmacological activity of morphine. 23 In the tail-immersion test, the animal's tail was placed in a beaker filled with water that had been heated to 52 °C and the response latency to tail withdrawal from heat was recorded. 21 To avoid injury to the animal, the cut-off time was set at 20 s. 24 Similarly, a hot plate test was conducted using a hot plate apparatus covered with a transparent glass cylinder, having a temperature set at 50 ± 2 °C. The time in seconds until the mice licked their hind paw or jumped to avoid pain was recorded with a cut-off time of 90 s. 21 The animals were closely monitored throughout the experimentation to promptly identify and prevent any signs of injury.
Induction of Morphine Dependence and Withdrawal
Mice were injected with three daily intraperitoneal doses of morphine (50 mg/kg, 50 mg/kg, and 75 mg/kg) (i.p.), for 4 days at 8 AM, 11 AM and 4 PM. On the fifth day, a single dose of 100 mg/kg of morphine was injected (i.p.) followed by naloxone administration (4 mg/kg) (i.p) after an hour. 25 For treatment groups, CA (10 mg/kg or 20 mg/kg) (i.p.) was given 45 min prior to each dose of morphine injection. Withdrawal symptoms including jumping, diarrhea, weight loss, and grooming were recorded. 25
Conditioned Place Preference
This test was performed to determine the effect of CA on morphine use relapse.26,27 The apparatus consisted of three equal-sized compartments: one with a red “net” floor, another with a black-dotted floor and black/white striped walls, and a central compartment that was kept plain. During the pre-conditioning phase, animals were allowed unrestricted access to all compartments for 15 min to assess baseline preferences, and those with strong preference for one compartment (>66% of the time) were excluded. In the conditioning phase that lasted for three days, animals received morphine (60 mg/kg) (i.p.) and were confined to the compartment for 30 min (morphine paired). The same animals were injected with saline at-least four hours later and confined to the other compartment (saline paired). To understand the effect of CA, the molecule was administered in the test groups (i.p.) at high and low doses (10 mg/kg and 20 mg/kg) 45 min prior to administering morphine to separate groups. A separate saline group served as the control, similarly confined and receiving only saline injections in each chamber at least four hours apart for three days. On the fifth day, no treatment was given and the post-conditioning test was conducted, allowing the animal free access to explore all compartments for 15 min, and the time spent in each was recorded. After, animals were not administered any treatments until any morphine-associated place preference for all groups was completely abolished and comparable with the saline-only group. Mice were evaluated in a drug-free state every 24 h until no preference was observed. Finally, in the reinstatement phase, treatment animals received a priming dose of morphine (1 mg/kg) with or without CA administration (45 min prior), and reinstatement of CPP was evaluated.
Tissue Collection and Biochemical Processing
Animals were sacrificed at the end of the study, brain samples were collected on an ice-cold glass plate and stored at −80 °C or 4% formalin solution for further processing. Selected samples were homogenized in 0.1 M sodium phosphate buffer (pH 7.4) having phenyl methyl sulfonyl fluoride as a protease inhibitor. Centrifugation was performed at 4000×g (sample) for 10 min at 4 °C and the supernatant obtained was further processed for biochemical assays. 28
Quantification of Nitrite in the Hippocampus
Nitric oxide (NO) levels were evaluated indirectly by measuring the concentration of nitrite in the mouse hippocampus using Griess reaction quantification method. 25 Then, animals were decapitated and their brains were isolated. The hippocampus tissues were then homogenized and after 10 min centrifuged (13 000 RCF) for 15 min. Finally, the supernatant was isolated and analyzed.
Antioxidant Assays
This assay was performed according to the previously described procedures with few modifications. 28 Reduced glutathione (GSH) was measured using 2-nitro-5-thiobenzoic acid, a yellowish by-product of GSH and DTNP oxidation. The absorbance was measured with a microplate reader at 412 nm and GSH was expressed as moles per mg of protein. For the Glutathione-S-Transferase (GST) assay, the formation of CDNB was used for measuring GST activity at an absorbance of 340 nm and results were expressed as CDNB conjugate formation per minute per milligram of protein. Catalase (CAT) activity was determined by the degradation of H2O2 with absorbance measured at 240 nm and expressed as moles of H2O2/min/mg protein. Finally, for lipid peroxidation (LPO), Malondialdehyde, a by-product of this reaction was used to determine the extent of lipid peroxidation in the tissue. Absorbance was measured at 532 nm and LPO was expressed as nmoles/min/mg protein TBARS.
Immunohistochemistry Analysis
Immunostaining was performed as reported previously. 29 Tissue samples were sectioned at a thickness of 5 µm using a microtome, and coronal sections were collected based on the stereotaxic atlas of the mouse brain. For the cortex, the analysis focused specifically on the prefrontal cortex (PFC). Within the hippocampus, regions of interest included the dentate gyrus (DG). The anatomical selection of these regions ensures the evaluation of zones most relevant to morphine-induced neuro-plastic changes and the effects of CA. Tissue samples were hydrated using xylene and graded alcohol in different serial dilutions and finally with distilled water followed by washing with PBS. For antigen recovery, Proteinase K was used. Tissues were then dipped in a 3% H2O2 solution. Blocking serum was applied and slides were left at room temperature for about 1 h. Primary antibodies (dilution 1: 100) were applied to the tissues and kept overnight at 4 °C. Then, a suitable secondary antibody was applied and slides were left for 2 h and washed using PBS. Afterward, ABC was applied and left for an hour. In the final step, the tissues were stained using a DAB solution. Mounting media and coverslips were applied to the slides and air-dried. Images (10x and 40x magnification) were obtained with Olympus microscope and ImageJ software was used for image processing.
Real-Time Reverse Transcription-PCR (RT-PCR) Analysis for Gene Expression of NR2B, nNOS, and iNOS
RNA was isolated from brain samples using TRIzol, as mentioned previously. 30 NanoDrop was used to assess RNA quality and quantity. Viva cDNA synthesis kit (Vivantis cDSK01-050) was used to convert RNA to cDNA. PCR was performed on Galaxy XP Thermal Cycler (BIOER, PRC) and 2X Amplifyme Universal qPCR mix according to the manufacturer's guidelines. Following are the sequences for the primers used: NR2B gene, (Forward) 5′-GCCATGAACGAGACTGACCC-3′, (Reverse) 5′-GCTTCCTGGTCCGTGTCATC-3′; nNOS gene, (Forward) 5′-ACC CAA CGT CAT TTC TGT CC-3′, (Reverse) 5′-AAG GTG GTC TCC AGG TGT GT-3; iNOS gene, (Forward): 5′-CAGAAGCAGAATGTGACCATC-3′, (Reverse)5′-CTTCTGGTCGATGTCATGA-3′.
Molecular Docking
The molecular docking study of CA was conducted to assess its binding affinity with inducible nitric oxide synthase (iNOS), neuronal nitric oxide synthase (nNOS), and the NR2B receptor using PyRx software integrated with AutoDock Vina. The structure of CA was obtained from PubChem (https://pubchem.ncbi.nlm.nih.gov/) and converted to PDB format using OpenBabel to ensure compatibility for docking studies. Ligand optimization was performed using the Universal Force Field (UFF) to minimize steric clashes and optimize the conformation. The target proteins—iNOS (PDB ID: 1DD7), nNOS (PDB ID: 6AV2), and NR2B (PDB ID: 5EWJ)—were retrieved from the Protein Data Bank (https://www.rcsb.org/) (Brown et al, 2022). During protein preparation, crystallographic water molecules were removed using AutoDockTools to avoid interference during ligand binding. The binding sites of the native inhibitors (1PM601 for iNOS, BY7803 for nNOS, and QEL503 for NR2B) were used to determine the grid box coordinates for defining the correct binding pocket, but the inhibitors themselves were not retained. Hydrogen atoms were added, and Gasteiger charges were assigned to ensure accurate modeling of electrostatic interactions. The docking simulations were carried out using PyRx, where the prepared ligand and protein structures were imported, and the docking grid was focused on the binding pockets defined by the native inhibitor binding sites. Default docking parameters were used to predict the binding affinities, reported in kcal/mol, which indicate the stability of the ligand-protein interactions.
Statistical Analysis
Data were expressed as mean ± SEM Statistical analysis was performed using GraphPad Prism software (Version 8). Data were analyzed using one-way and two-way ANOVA followed by post-hoc Tukey's test. Individual data points are displayed in all figures for increased transparency. A P-value of <.05 was considered statistically significant.
Results
Carvacrol Delays the Development of Morphine Antinociceptive Tolerance
Animals treated with morphine exhibited rapid emergence of tolerance to analgesic effects compared with controls during hot plate and tail immersion tests (Figure 2A and B). In contrast, CA at a dose of 20 mg/kg significantly delayed the development of morphine tolerance during these behavioural tests ie two-way ANOVA showing values of hot plate F(12, 100) = 74.13, P < .0001 and tail immersion test F(12, 100) = 292.1, P < .0001 and having Tukey's post-hoc test = P < .001. Interestingly, a lower dose of CA (10 mg/kg) was far less effective than the higher dose (20 mg/kg) and had little effect in delaying the emergence of antinociceptive tolerance.
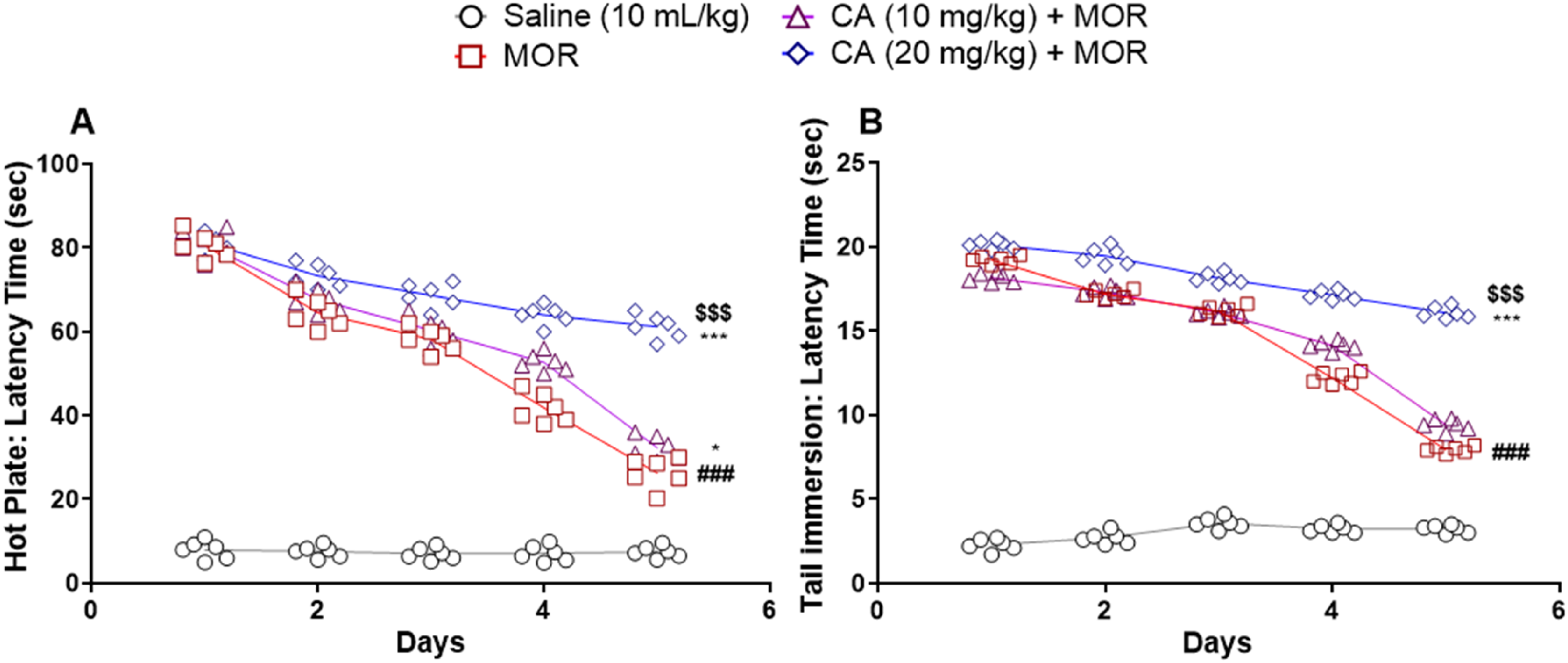
The Effect of Carvacrol (CA) on Tolerance to the Analgesic Effect of Morphine (MOR) in Mice (n = 6). For Induction of Tolerance, Animals Received MOR for 4 Consecutive Days (50, 50, and 75 mg/kg/day), and on the Fifth Day, Only a Single Dose of MOR 50 mg/kg was Injected. CA Was Injected Prior to MOR.
Carvacrol Mitigates Naloxone-Induced Morphine Withdrawal
Naloxone injection after five days of morphine administration precipitated the withdrawal symptoms as compared to the saline-treated animals (P < .001). In contrast, CA pre-treatment significantly caused a reduction in morphine withdrawal symptoms ie number of jumps F(3, 20) = 640.3, P < .0001 (Figure 3A), diarrhoea score F(3, 20) = 345.5, P < .0001 (Figure 3C), weight loss F(3, 20) = 1284, P < .0001 (Figure 3D), and grooming behaviour F(3, 20) = 509.6, P < .0001 (Figure 3B) as revealed by the one-way ANOVA results and these beneficial effects were especially pronounced at higher (Tukey's post-hoc = P < .001).

Effect of Carvacrol (CA) Pre-Treatment on Naloxone-Induced Withdrawal Symptoms in MOR-Dependent Mice. CA was Injected in Mice (n = 6) Prior to MOR Throughout the Experiment. Data is Presented as Mean ± SEM
Carvacrol Blunts the Acquisition While Accelerating the Extinction of Morphine-Conditioned Place Preference
In the pre-conditioning phase, no significant difference was observed between the animal groups (Figure 4A). However, during post-conditioning, morphine-treated mice spent significantly more time in the morphine (MOR)-paired compartment as compared to the control group (P < .001) (Figure 4A). Interestingly, CA dose-dependently decreased the time spent in MOR-paired compartment during this phase with two-way ANOVA F(3, 40) = 155.6, P < .0001), Tukey's post-hoc test P < .01 day (Figure 4A). In the extinction phase, CA treated animals dose-dependently exhibited a prompt reduction in preference towards the morphine-paired compartment from day 6 to 13 than animals injected with morphine only, with two-way ANOVA displaying results of F(21, 160) = 89.61, P < .0001 and with Tukey's post-hoc revealing no-significance among all groups on last day (Figure 4B). Interestingly, the effect of the higher dose of CA was significantly more pronounced than the low dose on most days during extinction. Finally, CA at 20 mg/kg exhibited minimum reinstatement of CPP than other treatments following priming dose of morphine, with one-way ANOVA results F(3, 20) = 579.8 P < .001 and Tukey's post-hoc test = P < .001 (Figure 4C).
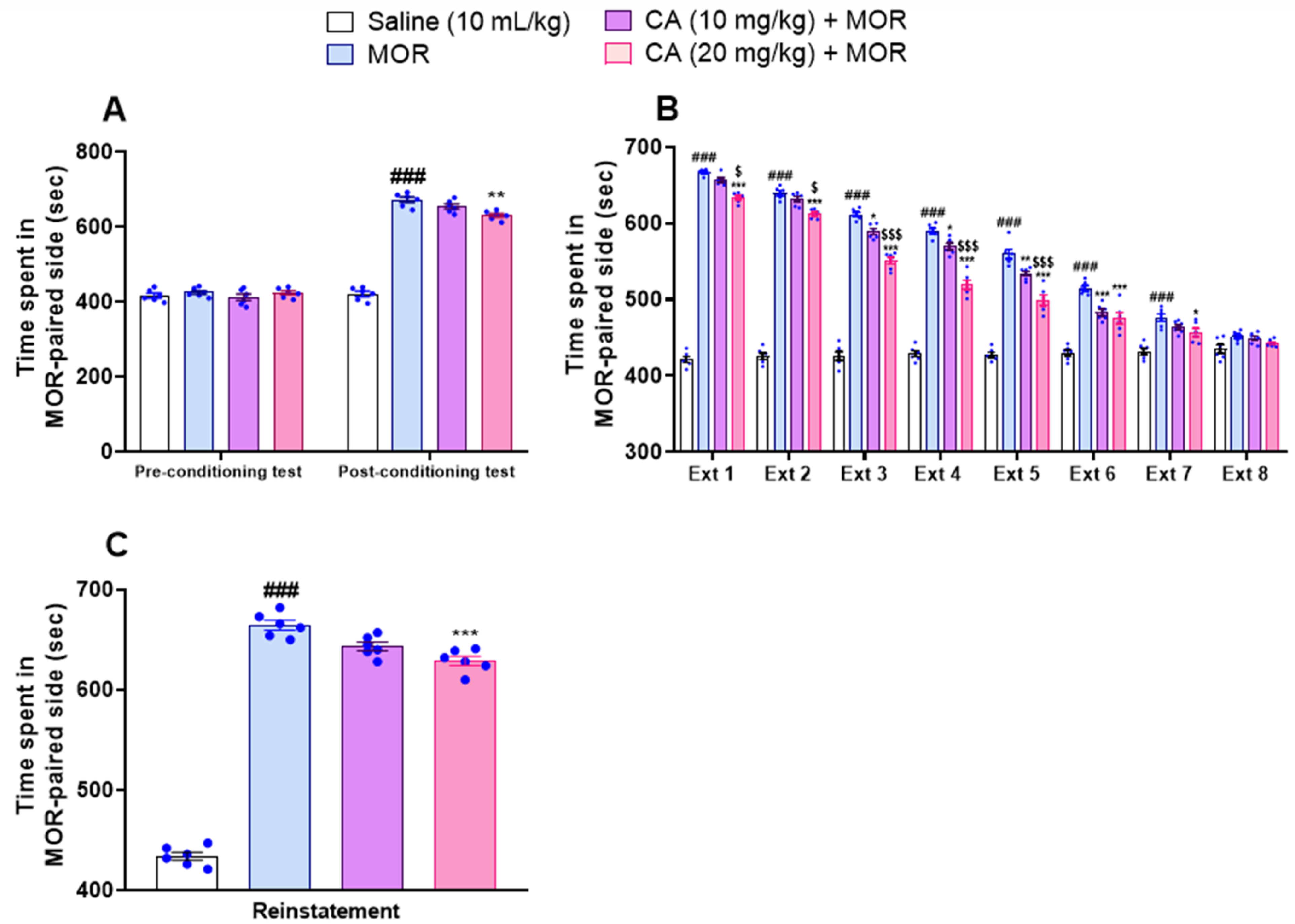
Effect of Carvacrol (CA) on the
Effect of Carvacrol on Morphine-Induced Oxidative Stress
Significant reductions in GST, GSH, and CAT were observed in the hippocampus of morphine-treated animals (Figure 5A to C) as compared to saline-treated groups (P < .001). In contrast, CA-treated animals exhibited significant restoration in these parameters with a notable effect of 20 mg/kg dose ie GST [F (3, 20) = 60.04, P < .0001] with Tukey's post-hoc test = P < .01, GSH [F (3, 20) = 130.0, P < .0001] with post-hoc Tukey's test = P < .001, and CAT F(3, 20) = 72.06) Tukey's post-hoc test = P < .01. Further, morphine-treated animals exhibited a significant increase in LPO which was curtailed after treatment with CA, dose-dependently F(3, 20) = 222.4, P < .0001 with Tukey's post-hoc test = P < .001 (Figure 5D). The effect of CA (20 mg/kg) was significantly greater than CA (10 mg/kg) on GST, GSH, and LPO levels.

Effect of Carvacrol (CA) Against
Carvacrol Curtails Nitrite Production in the Hippocampus of Morphine-Treated Mice
Nitrite assay in brain hippocampus was performed to evaluate the nitric oxide production. Morphine treatment caused highly significant increases in nitrite levels in mice hippocampus (P < .001) (Table 1). In contrast, CA dose-dependently caused a significant decrease in nitrite concentration determined using Griess reagent (Table 1).
Effect of Carvacrol (CA) on Nitrite Levels in the Hippocampus of MOR-Treated Mice.

Data were expressed as the mean ± S.E.M, and analyzed using One-way ANOVA (post-hoc Tukey's test). Where,###P < .001 versus saline and **P < .01 versus MOR.
Effect of Carvacrol on TNF-α and p-NF-κB
The beneficial effect of CA on crucial mediators of inflammation ie TNF-α, p-NF-κB was investigated. It was observed that morphine treatment resulted in significant gains in the expression of TNF-α in mouse cortex and hippocampus regions (P < .001) (Figure 6A and B). In contrast, only CA 20 mg/kg treated animals exhibited significant reductions in the expression of TNF-α than morphine-treated mice, with one-way ANOVA results F(3, 16) = 1.072, P < .0001 and Tukey's post-hoc test for Cortex: P < .001, for Hippocampus: P < .01) (Figure 6A and B). In addition, p-NF-κB expression was significantly increased in morphine-treated animals than saline-treated group (P < .001) (Figure 7A and B). However, CA-treated mice exhibited significant reductions in the expression of p-NF-κB than animals injected with morphine only, presenting one-ways ANOVA results F(3, 16) = 2.486, P < .0001 and Tukey's post-hoc test CA 20 mg/kg, Cortex: P < .01, Hippocampus: P < .01 (Figure 7A and B). Though greater in magnitude, the difference between low and high doses of CA was not statistically significant.
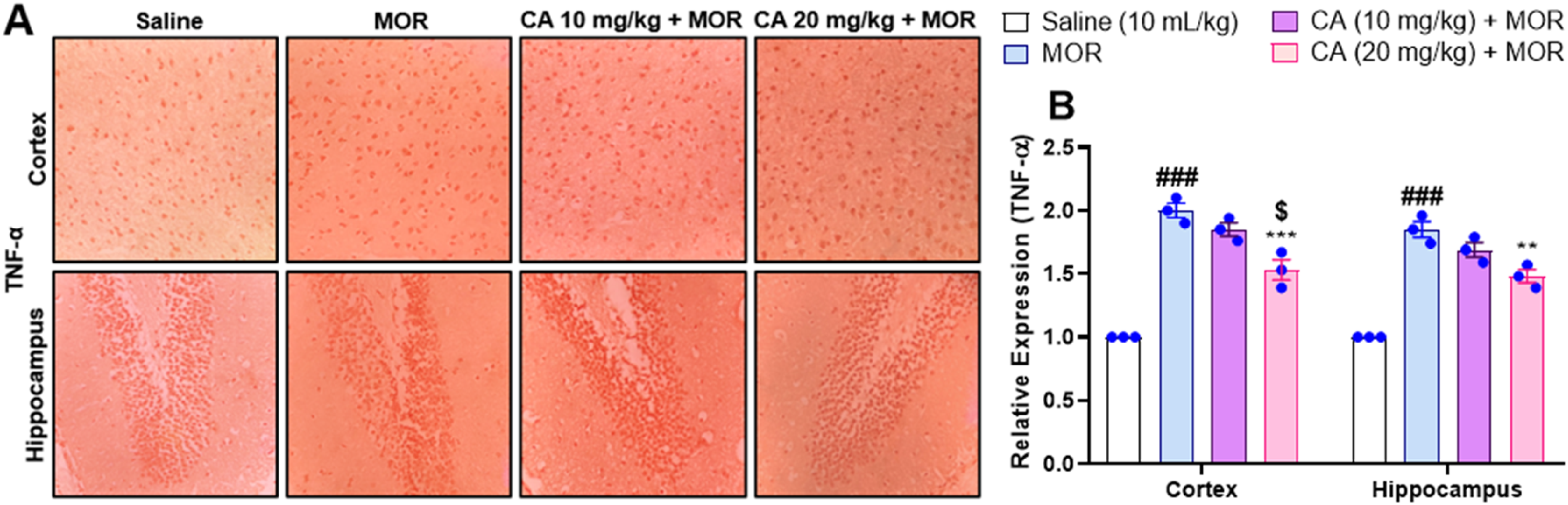
Effect of Carvacrol (CA) Against Tumor Necrosis Factor Alpha (TNF-α) Expression in Morphine (MOR) Treated Mice Brain Tissues Using Immunohistochemistry.

Effect of Carvacrol (CA) Against Nuclear Factor Kappa B (p-NF-κB) Expression in Morphine (MOR) Treated Mice Brain Tissues Using Immunohistochemistry Technique.
Carvacrol Mitigates the Protein Expression of nNOS in cortex and Hippocampus
Morphine administration resulted in a significant increase in nNOS protein expression in cortex and hippocampus regions of mice brains (P < .001) (Figure 8A and B). In contrast, CA administration caused a significant reduction in protein expression of nNOS in both these regions with one-way ANOVA F(3, 16) = 1.483, P < .0001 and Tukey's post-hoc test for CA 20 mg/kg, Cortex: P < .001, Hippocampus: P < .001 (Figure 8A and B). In particular, the effects of 20 mg/kg dose of CA were more pronounced than 10 mg/kg in animals treated with this agent (Figure 8A and B).

Effect of Carvacrol (CA) Against nNOS Expression in Morphine (MOR) Treated Mice Brain Tissues Using Immunohistochemistry Technique.
Carvacrol Curtails the Protein Expression of iNOS Only in the cortex at Higher Dose
Morphine treatment resulted in significant gains in the protein expression of iNOS in cortex and hippocampus regions (P < .001) (Figure 9A and B). Interestingly, CA at 10 mg/kg did not exert any significant effect on the iNOS in the cortex and hippocampus regions. Indeed, significant beneficial effects of CA on iNOS expression were only observed at higher doses ie 20 mg/kg in the cortex region but not in the hippocampus with one-way ANOVA showing values F(3, 16) = 3.356, P < .0001, Tukey's post-hoc test for Cortex: P < .01 (Figure 9A and B).
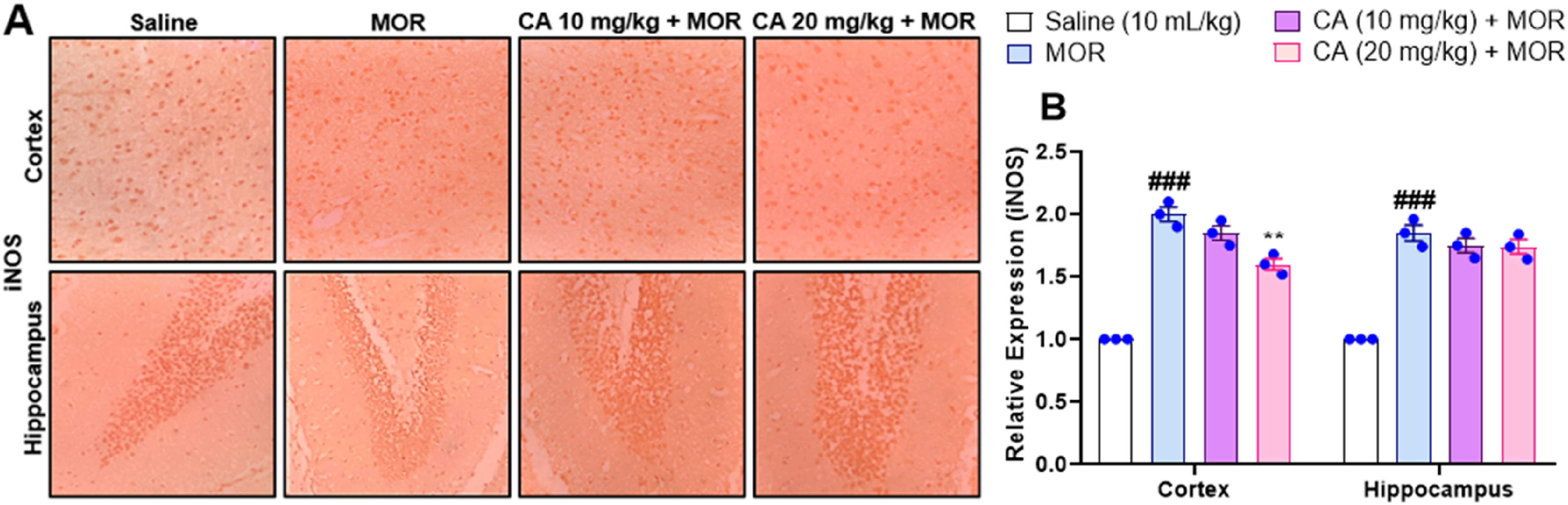
Effect of Carvacrol (CA) Against iNOS Expression in Morphine (MOR) Treated Mice Brain Tissues Using Immunohistochemistry Technique.
Effect of Carvacrol on mRNA Expression of nNOS and iNOS
Morphine treatment caused significant elevations in the mRNA expression of iNOS and nNOS, respectively (P < .001) (Figure 10A and B). However, CA-treated animals exhibited significantly reduced expression of nNOS in mice hippocampi, dose-dependently with one-way ANOVA F(3, 8) = 611.1, P < .0001 with Tukey's post-hoc test for CA 10 mg/kg, P < .01 and for CA 20 mg/kg, P < .001 (Figure 10B). In contrast, no significant effect of CA on iNOS mRNA expression was observed during our study at test doses of 10 mg/kg and 20 mg/kg with one-way ANOVA F (3, 8) = 107.6, P < .0001 and Tukey's post-hoc test showing no significance (Figure 10A).
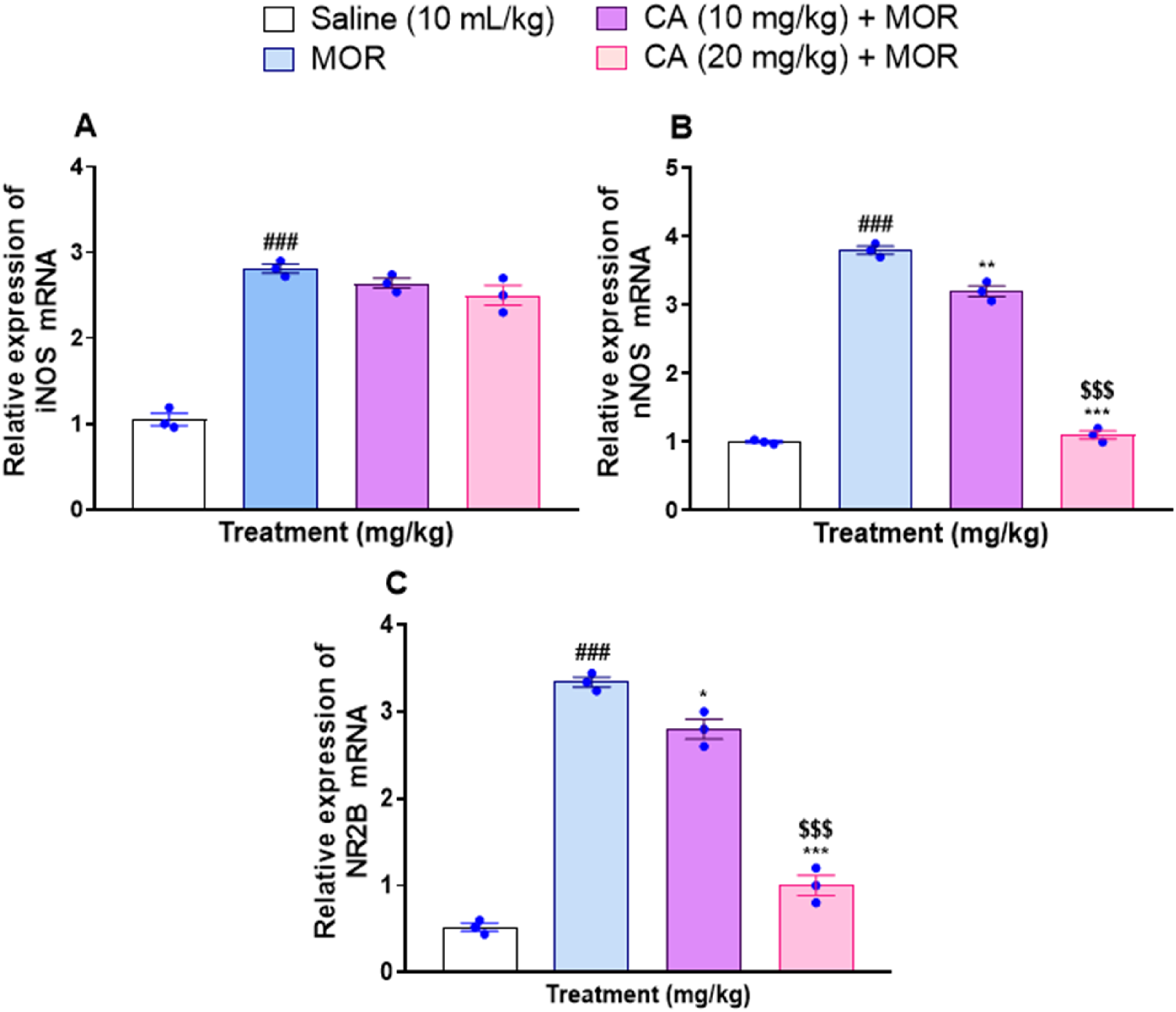
Gene Expression of
Carvacrol Inhibits the mRNA Expression of NR2B
The mRNA expression of NR2B in the hippocampi of morphine-only treated animals was significantly elevated than normal controls (P < .001) (Figure 10C). In contrast, CA-treated mice exhibited significantly less expression of NR2B, particularly at the higher dose of 20 mg/kg (P < .001) than the animals injected with morphine only, with one-way ANOVA F(3, 8) = 232.2, P < .0001 and Tukey's post-hoc test for CA 10 mg/kg: P < .05, for CA 20 mg/kg: P < .001 (Figure 10C).
Results of Molecular Docking Simulation
Carvacrol (CA) exhibited binding affinities of −5.6 kcal/mol for iNOS, −7 kcal/mol for nNOS, and −6.9 kcal/mol for the NR2B receptor. The negative binding affinity values signify spontaneous and energetically favourable interactions (Table 2). Notably, CA showed the highest binding affinity for nNOS (−7 kcal/mol), suggesting a stronger interaction compared to iNOS and NR2B. This selectivity may be beneficial for developing targeted inhibitors for neurological disorders, reducing the risk of non-specific inhibition of other nitric oxide synthases and associated side effects. Therefore, CA appears to be a promising candidate for selective inhibition of nNOS, particularly in therapeutic applications where modulation of neuronal nitric oxide is desired.
Summary of Probable Molecular Interactions of Carvacrol with the Target Proteins.

Discussion
Morphine addiction is still a significant challenge in treatment and social contexts due to the development of dependency, tolerance, behavior alterations, and relapse. Our results indicated that Carvacrol (CA) attenuated morphine tolerance, dependence, and the acquisition of CPP through a variety of biochemical mechanisms. CA was found to target nitric oxide, oxidative stress, TNF-α, p-NF-κB, and NR2B-dependent pathways in morphine addiction. These biochemical parameters were chosen for investigation based on previous reports signifying their role in the emergence of morphine dependence and tolerance.31–34
The role of nitric oxide in morphine-associated pathologies is widely known and the beneficial effects of NOS inhibition on morphine tolerance and dependence have been demonstrated in multiple previous studies. For instance, Majeed et al have shown that the administration of NO synthase (NOS) inhibitors L-NG-nitroarginine methyl ester (L-NAME) and NG-nitro-L-arginine attenuated the development of morphine tolerance and suppressed the development of dependence in Swiss mice. 35 NO is formed through the activity of NOS which includes neuronal (nNOS), inducible (iNOS), and endothelial (eNOS) isoforms. Based on their study in NOS-deficient mice, Heinzen et al have demonstrated that nNOS and not eNOS played a pivotal role in the development of morphine tolerance. 36 In addition, Romero et al have demonstrated that iNOS gene deletion partially prevented morphine tolerance while significantly suppressing withdrawal-induced hyperactivity. 37 During the current report, the favorable effects of CA involved greater inhibitory effects on nNOS than iNOS in the cortex and hippocampus regions. In addition, reduced nitrite levels indicating reduced NO synthesis were also observed during our study. Indeed, the inhibitory effects of CA on NO production have also been previously demonstrated. Hosseini et al have suggested that CA significantly reduced the NO production in the heart and aorta of LPS-injected rats. 38 Guimarães et al have further demonstrated that CA curtailed the LPS-induced production of NO by murine macrophages. 39 Another study by Li et al demonstrated that CA inhibited iNOS expression in middle cerebral artery occlusion rats. 40 In addition, inhibitory effects of CA on nNOS have also been demonstrated 41 which further corroborates our findings observed during the current study.
Oxidative stress and inflammatory mediators are known to play crucial roles in morphine dependence and tolerance. Indeed, several studies are available in which morphine treatments exhibited significantly increased levels of oxidants with weakened antioxidant defense mechanisms.42–48 In addition to oxidative stress, inflammatory mediators are also known to play their part. For instance, the effects of TNF-α in neuroinflammation and tolerance have been demonstrated.45,49 Interestingly, Ching-Hui et al revealed that a TNF-α antagonist, Etanercept, could suppress neuroinflammation and restore the antinociceptive effect of morphine. 50 Another inflammatory mediator NF-κB is a transcription factor that is involved in the expression of various gene targets including those involved in dependence like opioid receptors and neuropeptides. 51 Yang et al have demonstrated that IKK/NF-κB could be playing a critical role in the extinction of morphine CPP, thus representing an interesting target in overcoming morphine addiction. 52 Ghavim et al have suggested that Pioglitazone could prevent morphine antinociceptive tolerance that involved at least in part the suppression of neuroinflammation and NF-κB activity. 53 In the current investigation, CA-treated animals exhibited significant reductions in oxidative stress markers along with upregulation of antioxidant defenses. In addition, protein expressions of TNF-α and NF-κB were also attenuated in these animals compared with morphine treatment only. Indeed, these beneficial effects of CA on various parameters are in corroboration of earlier reports. Guimarães et al employed several bioassays and suggested that CA possessed significant anti-nociceptive and antioxidant activities. 54 Further, attenuating effects of CA on mechanical hypernociception and inflammatory response have also been described. 39 Together, these observations strengthen the probable application of CA in the mitigation of neurological oxidative and inflammatory stress caused by morphine and provide highly desired neuroprotection.
In addition to the mechanisms already described, CA-treated animals exhibited significant reductions in mRNA expression of NR2B. Ko et al have previously demonstrated that cingulate NR2B could contribute to morphine analgesic tolerance. 34 Li et al have further suggested that D2 receptor-NR2B could play a crucial role in morphine use disorders. 55 Ma et al have further suggested that the NMDA receptor having NR2B was crucial for morphine-induced reinstatement. 56 This was also demonstrated that NR2B subunit had more involvement with morphine related than natural reward. 57 Indeed, NR2B-containing NMDA receptors in the nucleus accumbens (NAc) and the dorsal hippocampus are crucial for reinstating rewarding responses to morphine. 56 Hence, the beneficial effects of CA observed during our study on the CPP might involve its effect on NR2B subunits of NMDA receptors that is also corroborated by various previous reports. Taken together, these observations suggest that CA possesses noteworthy biological activities that are of interest in morphine tolerance, dependence, and CPP. Further confirmatory studies in different conditions and larger samples are highly desirable.
Limitations
This study highlights the therapeutic potential of CA in mitigating morphine-induced tolerance, dependence, and conditioned place preference (CPP), but several limitations should be acknowledged. While acute phases of addiction are addressed, the absence of long-term studies on relapse and sustained abstinence limits the understanding of CA's lasting effects. Additionally, the use of male albino mice introduces potential variability, as genetic, sex-based, or environmental factors were not accounted for, affecting reproducibility. The study lacks pharmacokinetic data and analysis of inter-individual variability, which are essential for optimizing CA's dosing. The observed effects of CA on CPP expression may reflect changes in morphine-associated reward or alterations in memory. However, this study did not include tests of CA effects on non-drug rewards, such as sucrose, to distinguish these effects. Future investigations should explore CA's influence on memory and non-drug rewards to clarify its mechanism of action. These studies would provide further insights into the therapeutic potential of CA in mitigating addiction-related behaviors while delineating its broader neurobehavioral effects.
Conclusion
The results of the present study suggest that a naturally occurring compound CA may be beneficial in reducing morphine-induced tolerance and dependence, as it reduces NR2B and nNOS expression in mice brain tissues. Although previous studies have demonstrated the antioxidant, anti-inflammatory, and neuroprotective properties of CA, its role in addiction and relapse remains underexplored. This study is among the first to evaluate the effects of CA on morphine-induced behavioral changes across all phases of the addiction cycle. Additionally, the study elucidates the underlying mechanisms, including the inhibition of NMDA receptor NR2B subunit expression, suppression of nNOS, and modulation of proinflammatory markers (TNF-α and NF-κB). These findings highlight CA's potential as a therapeutic agent targeting both behavioral and molecular pathways in morphine addiction, offering a novel approach to addiction management.
Footnotes
Abbreviations
CRediT Authorship Contribution Statement:
Ismail Badshah, Neelum Gul Qazi, Maira Anwar: Writing – original draft, Investigation, Methodology, Formal analysis, Data Curation.
Sayed Muzzammil Masaud – Molecular Docking Studies.
Babar Murtaza and Muhammad Imran Khan: Supervision, Conceptualization, Resources, Writing – review & editing, Project administration.
Acknowledgment
BioRender software was used for some of the graphical portions of the manuscript.
Ethical Approval
All the investigational protocols were approved by the Research and Ethics Committee of the Institute of Pharmaceutical Sciences (Ref. No. REC/RIPS/2023/17) and were in accordance with the international guidelines for principles of animal care.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
