Abstract
Introduction
The bioactive compounds derived from plants have long been esteemed for their remarkable health benefits and versatile applications across the pharmaceutical, cosmetic, and food industries. Among these, polyphenols and flavonoids stand out due to their potent antioxidant, anti-inflammatory, and antimicrobial properties.
Additionally, research has revealed its choleretic, antidiabetic, anti-inflammatory, analgesic, and anticancer potential, further broadening its therapeutic applications.6–8 The diverse bioactivities of
Polyphenols and flavonoids in
Various extraction techniques have been employed to isolate bioactive compounds from
Despite its therapeutic potential, systematic optimization of its bioactive compound extraction remains underexplored. Optimization of bioactive extraction processes is crucial to leverage the full potential of plant-derived compounds. By refining extraction parameters, it becomes possible to maximize yields, enhance compound purity, and improve the cost-efficiency of downstream applications. Response Surface Methodology (RSM) is a particularly effective approach for this purpose, allowing researchers to evaluate and optimize the interplay of multiple variables simultaneously.
Response Surface Methodology (RSM) is a powerful statistical and mathematical approach used to design, enhance, and optimize processes. Originally described by Box and Wilson in 1951, RSM has become essential in evaluating the effects of multiple process variables and their interactions on response variables. This technique is particularly useful in optimizing complex processes, including extraction technology, as it reduces the number of experimental trials needed while providing precise and effective results. RSM involves designing experiments, building predictive models, and determining optimal conditions, making it a valuable approach for studying relationships between dependent and independent variables. By focusing on the most significant variables, RSM enhances the efficiency and accuracy of the optimization process, making it less laborious and time-consuming compared to traditional methods. This methodology has proven effective and versatile across numerous fields, showcasing its capability to optimize various processes. In several studies, RSM was applied to optimize polyphenols and flavonoids extraction from various sources such as Compositae, 16 alfalfa, 17 white turmeric, 18 apple, 19 tamarillo fruit, 20 and stinging nettle. 21
The extraction of bioactive compounds such as polyphenols and flavonoids from plant matrices is a widely researched area due to their significant medicinal and cosmetic applications.
This study employs Response Surface Methodology (RSM), a robust statistical tool for process optimization, to extract polyphenols and flavonoids from
Materials and Methods
Materials
Healthy green
Extraction of Polyphenols and Flavonoids
The collected
Analytical Methods
Analysis of Total Phenolics
Total phenolic content (TPC) was determined following the ISO 14502-1:2005 method using the Folin-Ciocalteu reagent, with minor modifications. Gallic acid was the standard chemical for phenolic compounds to make the calibration curve. Specifically, 2.5 mL of daily prepared Folin-Ciocalteu reagent (diluted ten-fold with distilled water) was added to 1 mL of the liquid sample. The mixture was then placed in an ultrasonic bath at room temperature for 2 min, followed by the addition of 2 mL of 7.5% (w/w) sodium bicarbonate. After incubating for 60 min, the absorbance was measured at 765 nm using a Jenway 7305 UV/Visible Spectrophotometer. The results were expressed as gallic acid equivalents (GAE) per gram of dry weight (DW).
Analysis of Total Flavonoids
Total flavonoid content (TFC) was assessed using the aluminum chloride colorimetric method
22
with slight adjustments. Quercetin served as the standard for the calibration curve. In this procedure, 0.5 mL of the liquid sample was mixed with 0.1 mL of 10% aluminum chloride, 3 mL of 98% ethanol, and 1.5 mL of 1 M potassium acetate. The mixture was then incubated at 45 °C for 45 min. Following incubation, the absorbance was recorded at 410 nm using a Jenway 7305 UV/Visible Spectrophotometer. The results were reported as quercetin equivalents (QE) per gram of dry weight (DW).
Experimental Design and Statistical Analysis
RSM was used to determine the optimum condition for polyphenols and flavonoids extraction of 
Here,
To determine the statistical significance and the initial range of the extraction variables, a Regular Two-Level Factorial Design (2n−1) was employed with four factors: extraction temperature, extraction duration, solvent-solid ratio, and solvent composition. Subsequently, a three-level, three-factor Box–Behnken design was chosen to evaluate the combined effects of three independent variables: extraction temperature (
Independent Variables and their Coded and Actual Values used for Optimization.
The Box–Behnken Design and Experiment Data for Polyphenols and Flavonoids Extraction from

Results
Preliminary Experiments
The results from the (2n−1) fractional factorial design revealed that extraction temperature, solvent-solid ratio, and solvent composition are statistically significant parameters based on their evaluated levels. Following preliminary experiments, extraction duration was deemed statistically insignificant and thus excluded from further analysis. Additionally, to enhance the experimental design, the range of the low and high values for the parameters was expanded, leading to the implementation of a more refined Box-Behnken design using RSM.
Statistical Analysis
In this study, the RSM was applied to optimize the extraction conditions for TPC and TFC from
ANOVA Table Showing the Variables as Linear, Interaction, and Quadratic Terms on TPC Response and Coefficients for the Prediction Models.

Similarly, the model for TFC exhibited an R² value of 0.9854 and an adjusted R² of 0.9691, suggesting that 98.54% of the variation in TFC is attributable to the experimental factors, thereby showcasing an excellent fit between the model and the actual data (Table 4). The comparison of R² and adjusted R² values for the models showed minimal variation, indicating that non-significant terms were successfully excluded from the model. This consistency suggests that the final models are well-calibrated and only include significant factors.
ANOVA Table Showing the Variables as Linear, Interaction, and Quadratic Terms on TFC Response and Coefficients for the Prediction Models.

F-value and p-value served as a tool to assess the significance of each coefficient. Variables become more significant as the absolute F-value increases and the p-value decreases. The significance of the models was further supported by the p-values and F-values obtained from the ANOVA. The model for TPC showed a p-value of 0.0003 and an F-value of 16.16, indicating that the model is statistically significant and that the independent variables have a substantial impact on the TPC response. For TFC, the model exhibited an even higher level of significance, with a p-value of less than 0.0001 and an F-value of 60.16. These results highlight the robustness of the regression equations and their ability to predict the outcomes effectively.
After excluding the influence of non-significant factors related to the process variables, the equations for the fitted models are presented in Table 5.
Response Surface Models after Eliminating Non-Significant Variables.

Total Phenolic Content
Figure 1 depicts the changes in TPC in relation to extraction temperature, solid-to-solvent ratio, and solvent composition.
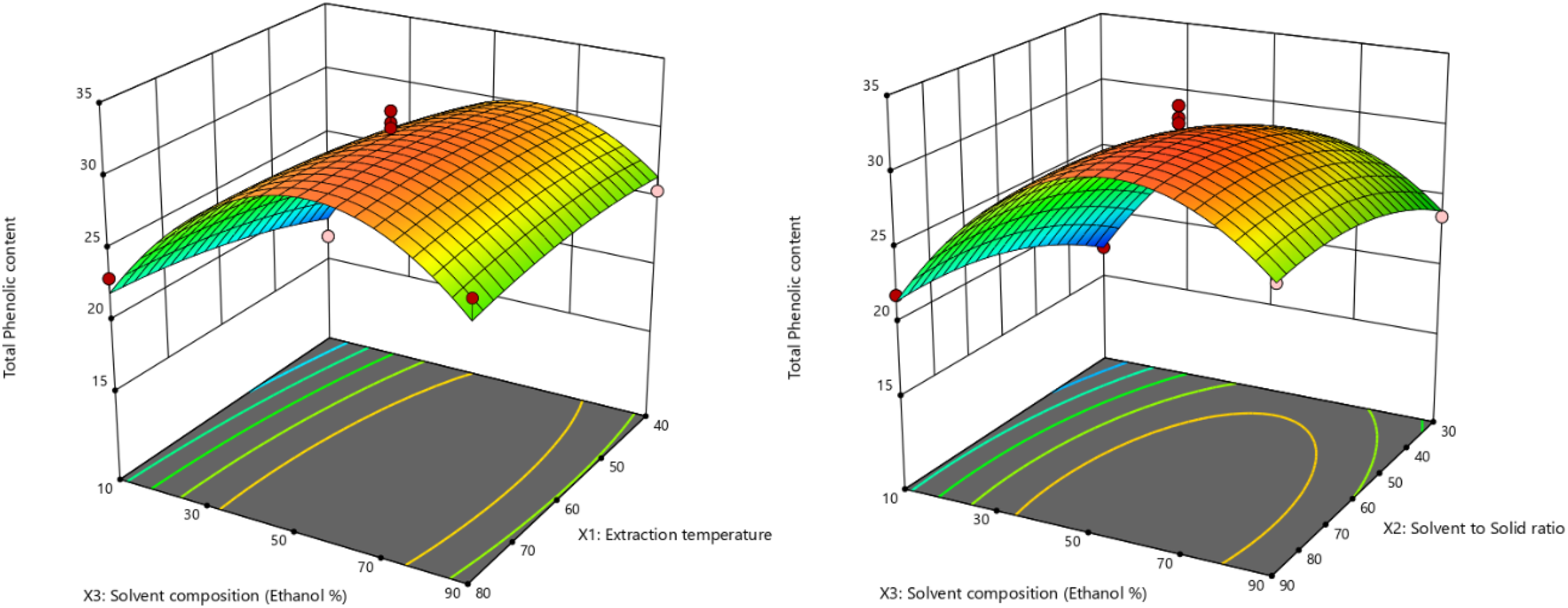
Response surface for the effect of extraction temperature and solvent composition at solvent to solid ratio = 60 mL/g (left) and solvent to solid ratio and solvent composition at extraction temperature = 60 °C (right) on TPC.
The ANOVA results for TPC indicate that among the three factors studied—extraction temperature (X1), solvent to solid ratio (X2), and solvent composition (X3)—the solvent composition had the most significant impact on TPC, with a p-value of 0.0006. This is consistent with the fundamental principles of solvent extraction, where the polarity of the solvent plays a crucial role in determining the solubility and subsequent extraction efficiency of phenolic compounds.
The solvent to solid ratio was also a critical factor, with a significant p-value of 0.0121. The quadratic effect of solvent composition (X32) on TPC is highly significant (p < 0.0001), indicating a non-linear relationship between solvent concentration and phenolic extraction. In contrast, the quadratic effects of extraction temperature (X12) and solid-to-solvent ratio (X22) are not significant for TPC (p = 0.3912 and p = 0.0893, respectively).
Total Flavonoid Content
Similarly, for TFC, the ANOVA results also highlighted the solvent composition (X3) as the most significant factor, with an extremely low p-value (<0.0001) (Figure 2).

Response surface for the effect of extraction temperature and solvent composition at solvent to solid ratio = 60 mL/g (left) and solvent to solid ratio and solvent composition at extraction temperature = 60 °C (right) on TFC.
The solvent to solid ratio (X2) was also a significant factor (p-value = 0.0039), underscoring its importance in the extraction process. The extraction temperature (X1) also did not significantly influence TFC (p-value = 0.2092). Regarding quadratic effects, none of the quadratic terms—extraction temperature (X12), solid-to-solvent ratio (X22), and solvent composition (X32)—showed significant p-values for TFC (p = 0.6783, p = 0.0618, and p = 0.1640, respectively).
Optimization and Verification
Through comprehensive numerical and graphical optimization using the Design-Expert software, the ideal conditions for maximizing TPC and TFC were determined. The optimal extraction parameters for achieving peak TPC and TFC are extraction temperature of 56 °C, solvent-to-solid ratio of 90 mL/g, and solvent composition of 75% ethanol. Under these conditions, the highest TPC and TFC values were recorded at 30.6117 mg GAE/g DW and 47.9087 mg QE/g DW, respectively. The optimal extraction region is visually represented in Figure 3, illustrating the efficacy of these parameters in maximizing both TPC and TFC.
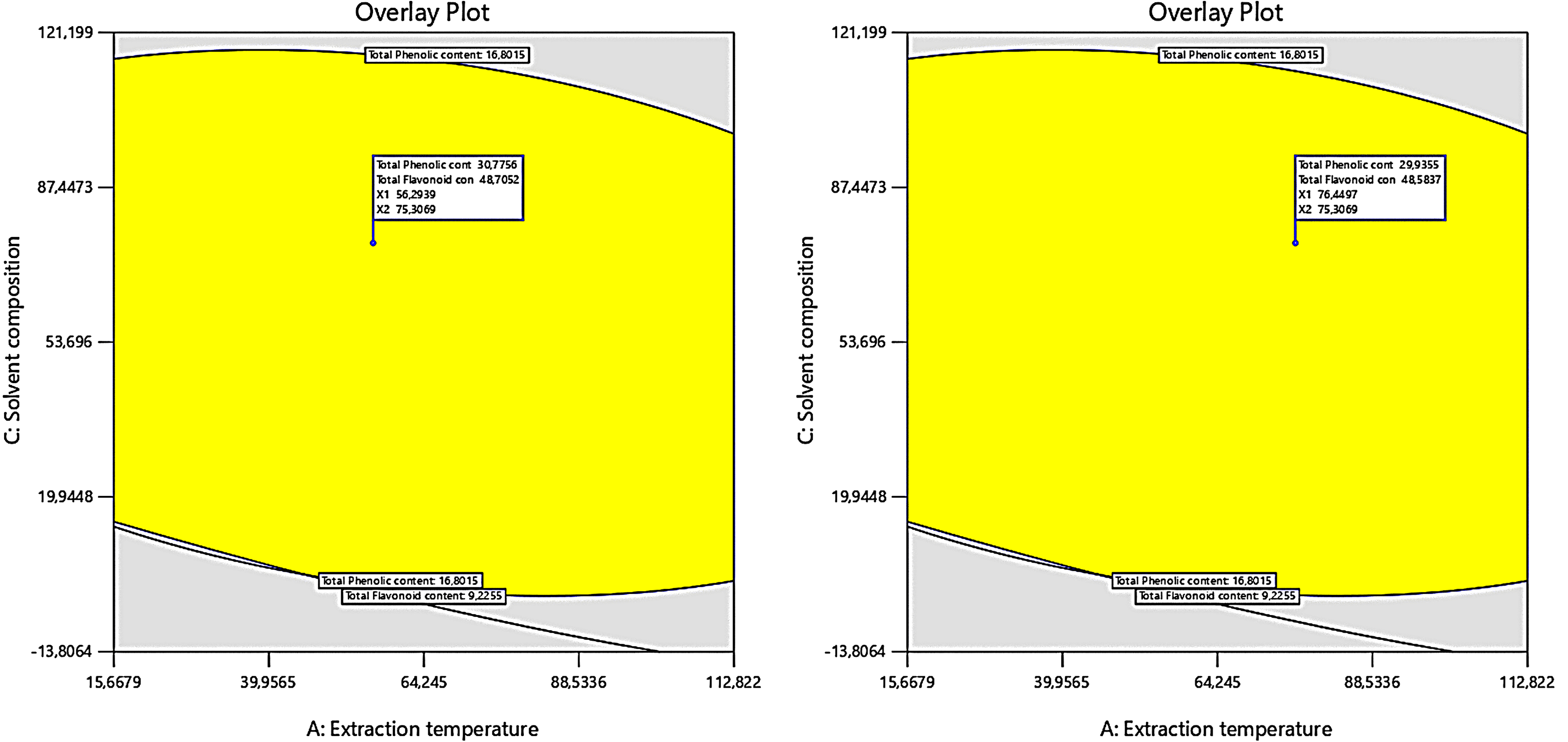
Response surface for the optimum region, obtained by overlaying contour plots of TPC and TFC.
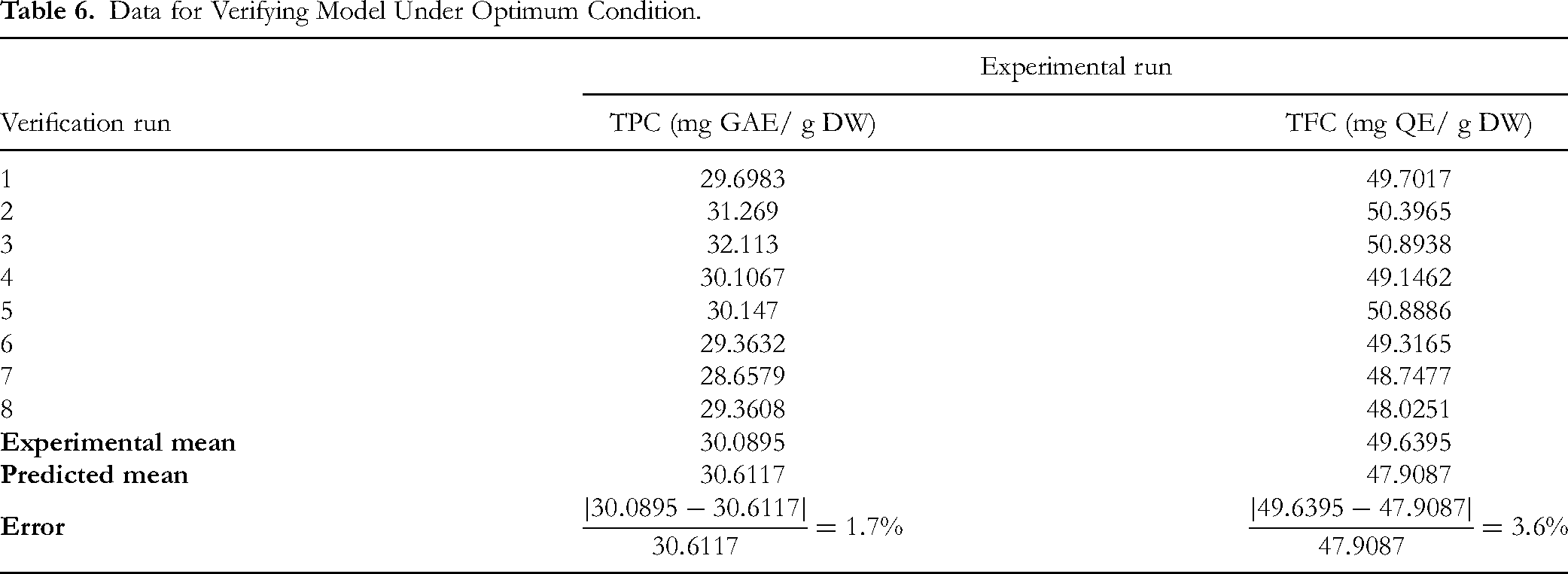
The efficacy of the models in predicting the optimal response values was assessed through the extraction of polyphenols and flavonoids under the conditions determined by the RSM optimization, including extraction temperature of 56 °C, solvent-to-solid ratio of 90 mL/g, and solvent composition of 75% ethanol. As shown in Table 6, the observed values for TPC and TFC are closely aligned with the predicted values, with no significant differences at the 5% significance level. These findings confirm a high degree of concordance between experimental and predicted results, demonstrating that the models used are both accurate and reliable.
Data for Verifying Model Under Optimum Condition.
Discussion
Examining the lack of fit is essential for assessing the adequacy and reliability of the models. A significant lack of fit typically indicates that the model fails to accurately represent the data within the experimental domain, particularly at points not included in the regression analysis. However, in this study, the lack of fit was not significant for any of the variables (p > 0.05), implying that the proposed models accurately predicted the related responses. This non-significant lack of fit confirms that the models provide a reliable representation of the experimental data, ensuring that the response surface models can be effectively used for optimization purposes.
Additionally, the coefficient of variation (CV) was calculated to assess the precision and reliability of the experimental data. The coefficient of variation (CV) measures the extent of variability relative to the mean of the data set. Generally, a low CV signifies minimal variation around the mean, which contributes to the development of a robust and reliable response model. The low CV values observed in the proposed models suggest that the experiments were conducted with high precision and consistency. The CV for TPC was 6.01%, indicating low variability and high precision in the measurements. For TFC, the CV was slightly higher at 7.39%, but still within an acceptable range, suggesting that the experimental results are consistent and reproducible.
Overall, the high R² values, significant p-values and F-values, and low CV percentages underscore the efficacy of RSM in optimizing extraction conditions. These findings are consistent with other studies that have utilized RSM to maximize the recovery of bioactive compounds, thereby enhancing the potential health benefits of plant extracts like those from
Total Phenolic Content
An optimal solvent to solid ratio is essential for maximizing TPC, as it ensures that there is sufficient solvent available to dissolve the phenolic compounds released during extraction. The outcome is rooted in mass transfer principles, where the concentration gradient between the solid and the solvent is regarded as the primary driving force for mass transfer. A lower solvent-to-solid ratio can lead to a more concentrated extract, but it also risks insufficient solvent availability and can limit extraction efficiency. Conversely, a higher ratio might result in a diluted extract, where the phenolic compounds are not fully utilized. Therefore, finding the right balance in the solvent-to-solid ratio is key to maximizing TPC. Belwal et al
23
also reported that increasing the ratio of extraction solvent to solid resulted in higher yields of phenolic compounds from
However, the extraction temperature did not show a significant effect on TPC (p-value = 0.1910), suggesting that within the studied range, temperature was not a limiting factor for phenolic extraction. This might be due to the thermal stability of the phenolic compounds in
In terms of interactions, TPC is not significant, indicating that the relationship between these factors and TPC is primarily linear within the studied range. This suggests that the optimization of TPC in
The quadratic effect of solvent composition on TPC indicates a non-linear relationship between solvent concentration and phenolic extraction. This suggests that moderate solvent concentrations are optimal for maximizing TPC, while both very low and very high concentrations may be less effective. Interestingly, it was observed that a 50% ethanol concentration resulted in higher TPC compared to a 90% ethanol concentration. This is attributed to the intermediate solvent polarity, such as 50% ethanol, which is more effective in solubilizing a broader range of phenolic compounds, which are not as efficiently extracted by either highly polar (water) or less polar (90% ethanol) solvents. The quadratic effect indicates that the solvent's polarity must be finely tuned to achieve maximum phenolic extraction. However, when the ethanol concentration was further increased to 70%, the yield began to decline. In the research of Ciric et al, 24 polyphenols were extracted from gallic and the quadratic effects of methanol concentration were also presented as the TPC was at optimum at around 70% methanol. Their study revealed a negative quadratic impact of ethanol concentration on TPC, with the optimal solvent mixture identified as 63% ethanol in water.
In contrast, the quadratic effects of extraction temperature and solid-to-solvent ratio are not significant for TPC, which points to a more straightforward linear relationship between these factors and TPC, with the primary focus on optimizing solvent composition.
Overall, the significant quadratic effect of solvent composition highlights the sensitivity of phenolic compounds to changes in solvent polarity. Achieving the highest TPC requires precise adjustment of solvent composition to balance the solubility and stability of phenolic compounds, while the solid-to-solvent ratio plays a secondary but still important role.
Total Flavonoid Content
The influence of solvent polarity is particularly important for flavonoid extraction, as flavonoids are often polar compounds that require a solvent of matching polarity for efficient extraction. Polar solvents are generally more effective in dissolving flavonoid compounds due to their ability to interact with the hydroxyl groups present in these compounds. This interaction facilitates the breakdown of plant cell walls, thereby enhancing the release and solubilization of flavonoid compounds into the solvent and maximizing the yield of flavonoids in the extraction process from
The solvent to solid ratio was a significant factor in the extraction process. This observation supports the idea that a higher solvent-to-solid ratio improves extraction efficiency by offering a greater volume of solvent relative to the solid material. This larger solvent volume enhances the dissolution and extraction of flavonoids, leading to an increased flavonoid content in the final extracts. A well-balanced solvent to solid ratio ensures that there is enough solvent to interact with and extract the flavonoids from the plant material. As with TPC, this ratio needs to be optimized to avoid either solvent saturation or dilution, both of which can reduce extraction efficiency.
The extraction temperature also did not significantly influence TFC. This indicates that similar to phenolics, the flavonoid compounds in
The interaction effects between the variables did not significantly affect TFC, with all interaction terms showing non-significant p-values. Similarly, the quadratic effects of the factors were not significant, except for the solvent composition's quadratic term in TPC, which suggests that the primary effects of these factors are linear. This reinforces the idea that the extraction of flavonoids from
The extraction temperature, solid-to-solvent ratio, and solvent composition showed significant p-values for TFC. This suggests that TFC extraction is primarily influenced by the linear effects of the factors, with no significant non-linear behavior observed in the response surface. Nevertheless, other studies have highlighted that solvent composition (ethanol or methanol concentration) can exert a negative quadratic effect on TFC, similar to the pattern observed for TPC. Researchers, such as, Amina et al 25 and Daghaghele et al 26 indicated that beyond a certain concentration, increases in ethanol or methanol can lead to a decrease in TFC, reflecting a complex interplay between solvent polarity and flavonoid solubility. This observed difference may be attributed to the varying chemical structures and compositions of flavonoid compounds from different sources, which can influence their solubility and extraction dynamics in different solvent environments.
In summary, for TFC, the linear effects of solvent composition and solid-to-solvent ratio are the most significant, with solvent composition being the predominant factor. The lack of significant interaction and quadratic effects implies that the optimization of flavonoid extraction can be achieved through a focus on these linear factors, without the need for complex interactions or non-linear adjustments.
Optimization and Verification
The findings of this study demonstrate the critical influence of extraction parameters on the yield of polyphenols and flavonoids from
The present study is critically compared with those reported by Subuki et al
13
and Yeong et al,
27
which also employed advanced optimization techniques for the extraction of bioactive compounds from
In contrast, the novelty of this study lies in its application of RSM to optimize solvent-based extraction parameters, providing a balance between yield, process simplicity, and industrial scalability. Unlike SFE, which requires high-pressure systems, or MAE, which necessitates specialized microwave equipment, the methodology employed here offers a cost-effective and accessible alternative. Furthermore, this work highlights the interaction effects of key variables such as solvent composition and temperature, offering deeper insights into the optimization process. This level of detail is absent in both Subuki et al and Yeong et al, where the studies focused more narrowly on specific technological advantages rather than a holistic optimization framework.
This study contributes to the broader field of natural product extraction by demonstrating that solvent-based methods, when optimized using RSM, can achieve comparable or superior yields to advanced techniques like SFE and MAE. By providing a comprehensive understanding of the interplay between extraction parameters, this work lays a foundation for further research into efficient extraction methods for
These findings indicate that the condensed extract, rich in polyphenols and flavonoids, holds significant potential for various industries, particularly cosmetics, pharmaceuticals, and functional skincare formulations. The extract can be incorporated into cosmetic bases such as creams, lotions, or serums, where its antioxidant, anti-inflammatory, and antimicrobial properties may contribute to skin health, aiding in the treatment of conditions such as eczema, fungal infections, and inflammation. Additionally, its potential antimicrobial activity suggests applications in natural preservative systems, offering a sustainable alternative to synthetic preservatives. These industrial applications highlight the broader relevance of
While this study provides valuable insights into the optimization of polyphenol and flavonoid extraction from
Conclusions
This study demonstrates the effectiveness of RSM in optimizing the extraction conditions for polyphenols and flavonoids from
Results from the present study highlighted the importance of precise parameter control in the extraction process to achieve optimal yields of bioactive compounds, which are pivotal for their therapeutic applications. Furthermore, the observed quadratic effect of solvent composition on TPC and the linear effect on TFC provides critical insights into the interaction of extraction variables and their impact on the efficacy of the extraction process. Additionally, the solvent-to-solid ratio also a critical factor in the extraction process that affects the yield of both polyphenols and flavonoids.
This research not only contributes to the understanding of optimal extraction techniques for
Footnotes
Acknowledgements
We acknowledge the support from Ho Chi Minh City University of Technology and Nguyen Tat Thanh University for this study.
Consent to Participate
There are no human participants in this article and informed consent is not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
