Abstract
Keywords
Introduction
For more than 2000 years, Scutellaria baicalensis has been one of the most widely used medicinal herbs in China and was officially included in the Chinese Pharmacopoeia in 2020. 1 S. baicalensis originated in East Asia and is extensively grown in European and American countries. Nevertheless, China is the primary source of S. baicalensis. 2 The principal active constituents of S. baicalensis include baicalin, baicalein, and wogonosides, with baicalin being a key compound derived from its roots. 3 S. baicalensis is widely distributed in northeast, north, central, and southwest China and is also common in vast areas north of the Yangtze River. 4 It is traditionally used for clearing heat, drying dampness, purging fire, and detoxification, treating conditions such as lung-heat cough, damp-heat diarrhea, sores, abscesses, swelling, and pain. Extracts from S. baicalensis exhibit diverse pharmacological activities, including effects on the nervous system, immune system, hepatoprotection, anticancer properties, antibacterial and antiviral actions, and antioxidative effects. 3
In recent years, many studies have explored the factors affecting the quality of S. baicalensis. The content of pharmacological substances in S. baicalensis varies depending on the germplasm. 5 Li et al used HPLC to analyze chemical components, demonstrating marked geographical differences in medicinal content. 6 Additionally, Georgieva et al, employing HPLC-electrochemical detection (HPLC-ECD), found variations in baicalin, baicalein, and wogonin content across 14 germplasm materials and nine growing regions. 7 Research indicates that microorganisms in the rhizosphere can affect the production of premium medicinal substances and enhance the concentration of active medicinal components. 8 Inorganic element accumulation in S. baicalensis is influenced by soil composition, with baicalin levels correlating with soil inorganic element content. 9 In addition, organic carbohydrates have a significant impact on the content of astragaloside. 10
Most current research focuses on a specific influencing factor, pharmacological action, and chemical composition, with few studies that comprehensively analyze various factors. Therefore, this study takes Scutellaria baicalensis samples from Shanxi, Hebei, and Inner Mongolia as the research object, measures the content of baicalin and total protein, analyzes their antibacterial effect, as well as the influence of climate factors, soil physicochemical properties, and soil microorganisms on the content of baicalin, explores the differences in effective components of Scutellaria baicalensis from different origins, and explores the potential influencing factors of authentic formation of Scutellaria baicalensis from multiple aspects.
Materials and Methods
Medicinal Herbs
In June 2023, five point sampling method was used to select Scutellaria baicalensis plants. After digging out the roots, the stem seedlings, fibrous roots, and soil were removed. When the plants were partially dried, the cork was removed and then dried completely. The soil shaking method is used to collect rhizosphere soil, which is mixed and placed in sterile bags. After labeling the information, it is brought back to the laboratory and a portion is stored in a −80°C freezer for subsequent determination of soil microbial diversity; Part of it is processed for the determination of soil physicochemical properties. Sample information of S. baicalensis is shown in Table 1. Professor Zhao Yutang from Chengde Medical College identified it as the dried root of S. baicalensis Georgi, a plant in the family Lamiaceae.
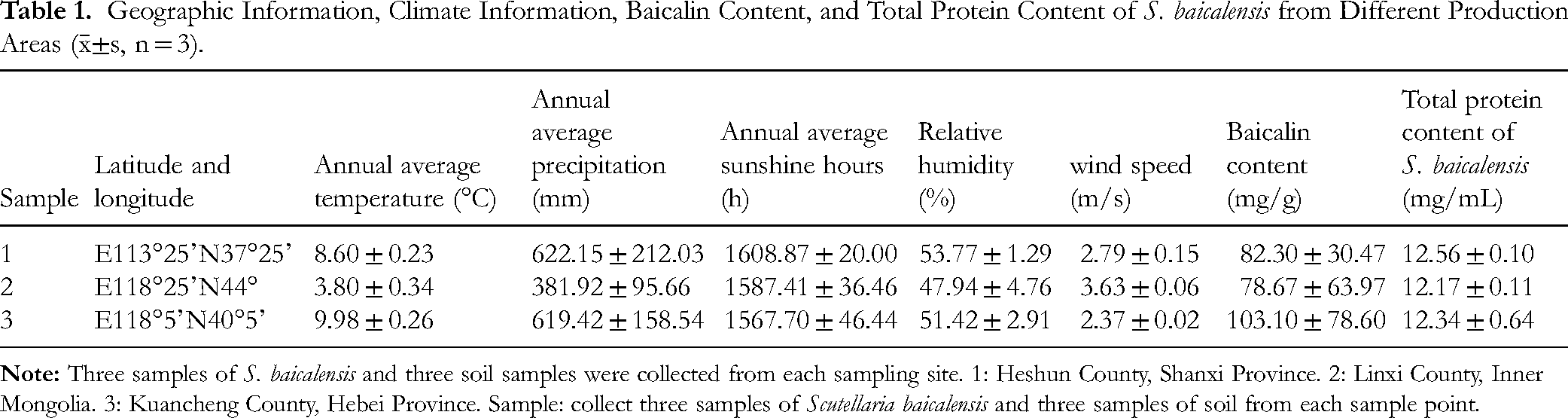
Geographic Information, Climate Information, Baicalin Content, and Total Protein Content of S. baicalensis from Different Production Areas (
Main Reagents and Instruments
Baicalin standard (C21PC1701B; Froming Pusitang of Beijing Jinming Biotechnology Company Limited; purity: 98.78%) was used. Carbinol (WXBB7259 V; Sigma-Aldrich) and acetonitrile (WXBB7245 V; Sigma-Aldrich) were included. A Shimadzu high-performance liquid chromatograph (LC-20AT; Froming Shimadzu Corporation of Japan) was employe. A low-temperature, high-speed centrifuge (5804R; Forming Eppendorf China Limited) was also used. Phosphoric acid (20190520; Froming Tianjin Chemical Reagent Supply and Marketing Company), a BCA protein concentration determination kit (P0012; Forming Beyotime Biotechnology), and a protein extraction kit (BC3720; Beijing Solaibao Technology Company Limited) were utilized. Gentamicin sulfate injection (Guangxi Nanning Baihui Pharmaceutical Group Co., LD, Sinopiate H45020250).
Determination of Baicalin Content in S. baicalensis from Different Origins
Preparation of Standard Solution
The appropriate baicalin reference substance was placed in a 10 mL container, dissolved in methanol, and quantified. The solution was then removed and appropriately diluted to obtain a standard solution of baicalin with a concentration of 64 μg/mL.
Sample Preparation
The product was dried in a cool place. After sieving, 0.3 g of the powder was placed in a conical flask equipped with a stopper. Forty milliliters of 70% ethanol was added, heated, and refluxed for 3 h. The solution was cooled, weighed, made up to volume with 70% ethanol, shaken thoroughly, and filtered. One milliliter of the filtrate was transferred to a 25 mL volumetric flask, diluted to the mark with methanol, and shaken well for subsequent analysis. The roots of Scutellaria baicalensis are placed in an oven at a constant temperature of 60°C and dried to a constant weight by blowing air. It is then crushed using a universal crusher, sieved through a 40 mesh sieve, and placed in a dry wide mouthed bottle for later use. Accurately weigh 0.3 g of powder and place it in a conical flask with a stopper. Add 40 mL of 70% ethanol, heat and reflux for 3 h, and let it cool naturally at room temperature before weighing. Make up for the reduced weight with 70% ethanol, shake well, and filter through a 0.22μm microporous membrane,. Measure 1 mL of filtered liquid and place it in a 25 mL volumetric flask. Add methanol to the mark and shake well for later use.
Chromatographic Condition
A C18 chromatographic column (150 mm × 4.6 mm, 5 μm) was used. The mobile phase consisted of methanol, water, and phosphoric acid solution (45:55:0.2). The flow rate was set to 0.8 mL/min, and detection was performed at a wavelength of 280 nm. The column temperature was maintained at 35°C.
Determination of Total Protein Content in S. baicalensis from Different Sampling Locations
Extraction of Total Protein was Performed According to the Per Kit (BC3720) Detection Method
Collect the leaves of Scutellaria baicalensis from different origins and store them in liquid nitrogen for future use. Take 1.0 g of sample, grind it in an ice bath, grind the sample with protein extraction buffer and transfer it completely to a 10 mL centrifuge tube. The amount of extraction buffer used is 3 times the fresh weight of the sample. Shake the ground sample well and let it stand at 4°C for 1 h to fully dissolve the protein. Shake the placed sample well, centrifuge at 4°C and 12000r/min for 30 min, transfer the supernatant to a 10 mL centrifuge tube, add pre cooled acetone at −20°C at a ratio of 1:2.5 (v: v), shake well, place at −20°C for 1 h, centrifuge at 4°C and 12000r/min for 30 min, remove the supernatant, and place the precipitate at −20°C for 20 min to completely evaporate acetone. Add the sample buffer to dissolve the precipitate (with a buffer volume of 300 μL per gram of sample). After the precipitate is fully dissolved, centrifuge at 4°C and 12000r/m for 5 min. Take the supernatant to obtain the protein solution to be tested, and store at −20°C.
BCA Protein Quantification was Performed According to the Detection Method of the Reagent Kit (P0012)
Determination of Antibacterial Effect of S. baicalensis on Staphylococcus aureus
Liquid Preparation
S. baicalensis powder was weighed and placed in a 500 mL round bottom flask. Distilled water (10 times the powder weight) was added, and the mixture was heated to reflux for extraction. The extract was filtered and concentrated to achieve a crude drug extract concentration of 1 g/mL. The final product was sealed and stored at 4°C for later use.
Bacteriostatic Experiment
The resuscitation strain was thawed in a 37 °C water bath and then inoculated into 1 mL glycerol bacteria and 100 mL LB liquid medium for overnight cultivation. For culture medium preparation and plating, 10 g of LB broth medium, 10 g of agar, and 500 mL sterile water were added to a conical flask and sterilized at 115 °C for 20 min in an autoclave. When the medium cooled to approximately 50 °C, the cultured bacterial solution was added at a 1:10 ratio, mixed well, and poured into plates. Approximately 1/3 of the plate height was filled, and the plates were allowed to dry and solidify. Filter paper was fully immersed in the test solution for 30 s and evenly placed on the plate. After incubation for approximately 14 h, the diameter of the inhibition zone, including the filter paper (6 mm), was measured. Using gentamicin sulfate injection as the positive control group and sterile water as the negative control group.
Physicochemical Analysis of Rhizosphere Soil of S. baicalensis from Different Sampling Locations
Determination of Soil pH
A 10.0 g soil sample was placed in a 50 mL beaker, and 25 mL of water was added. The mixture was shaken horizontally for 2 min and allowed to stand for 30 min at a controlled temperature of (25 ± 1)°C. A pH electrode was inserted into the sample, gently shaken until the reading stabilized, and the pH value was recorded.
Determination of Soil Nitrogen, Sulfur, and Carbon
The physical and chemical indicators of soil nitrogen, sulfur, and carbon were tested according to the following standards: the “National Standard of the People's Republic of China” (GB7173-87) for soil total nitrogen determination, the “Forestry Industry Standard of the People's Republic of China” (LY/T 1255-1999) for forest soil total sulfur determination, and the “Agricultural Industry Standard of the People's Republic of China” (NY/T1121.6-2006) for soil organic matter determination.
Detection of Soil Nitrate Nitrogen, Nitrite, and Ammonium Nitrogen
The contents of soil nitrate nitrogen, nitrite, and ammonium nitrogen were determined using reagent kits BC0045, BC1480, and BC1510, respectively.
Microbial Composition Detection of Rhizosphere Soil of S. baicalensis from Different Sampling Locations
DNA Extraction and PCR Amplification
Genomic DNA was extracted, and DNA concentration was detected using agarose gel electrophoresis and a NanoDrop 2000. PCR amplification was performed using barcode-specific primers to ensure accuracy and efficiency, based on the sequenced region.
Library Construction and Sequencing
Library construction, sequencing, electrophoresis detection of PCR products, magnetic bead purification, and second PCR amplification were carried out. The steps were repeated, quantified with Qubit, and equal amounts of samples were mixed for sequencing. The sequencing was completed by Shanghai Ouyi Biotechnology Company Limited.
Bioinformatics Analysis
Cutadapt software was used to trim primer sequences from the raw data. Using DADA2, raw data were filtered, denoised, concatenated, and subjected to quality control analysis following the default parameters of QIIME 2 (November 2020). Representative sequences and amplicon sequence variants (ASVs) were generated. The 16S rRNA sequences were annotated by comparing them with the Silva database (version 138) using the QIIME 2 Feature Classifier. This analysis was completedby Shanghai Ouyi Biomedical Technology Company Limited.
Statistical Analysis
All data were expressed as mean ± SD, and one-way ANOVA and correlation analysis were performed using SPSS23.0 software, with P < 0.05 indicating statistical significance.
Results
Analysis of Baicalin Content in S. baicalensis from Different Regions
This study measured the content of baicalin in Heshun County, Shanxi Province to be 82.30 ± 30.47 mg/ml, in Linxi County, Inner Mongolia to be 78.67 ± 63.97 mg/ml, and in Kuancheng County, Hebei Province to be 103.10 ± 78.60 mg/ml. Within the expected range of relevant literature.11,12 The results of baicalin content determination in S. baicalensis of different origins are shown in Table 1, with the highest levels observed in samples from Hebei. This finding highlights the potential influence of regional factors on baicalin accumulation, warranting further investigation into the underlying environmental and soil conditions.
Analysis of Total Protein Content in S. baicalensis from Different Regions
The total protein content of S. baicalensis from the three production areas showed no significant differences (Table 1). Leaves are directly exposed to the environment and are sensitive to environmental changes. 13 Measuring their protein content can evaluate the impact of environmental factors on plants, such as light, temperature, humidity, pollution, etc.14–17 Therefore, this study determined the protein content in Scutellaria baicalensis leaves. There are many factors that affect the protein content of Scutellaria baicalensis, which may be related to differences in Scutellaria baicalensis germplasm, climate factors, soil factors, and experimental control conditions in different regions.
Antibacterial Effect of S. baicalensis from Different Origins Against S. aureus
The antibacterial efficacy of S. baicalensis extracts from different regions was assessed against S. aureus (Table 2, Figure 1). This study evaluated the antibacterial effect of Scutellaria baicalensis extracts from different regions against Staphylococcus aureus (Table 2, Figure 1). The minimum inhibitory concentration of Scutellaria baicalensis solution against Staphylococcus aureus was measured to be 1.56 mg/ml, which is within the expected range in relevant literature.18,19 Results indicated that higher extract concentrations consistently yielded better antibacterial activity. At concentrations of 100, 50, 25, and 3.12 mg/mL, extracts from Hebei demonstrated the strongest antibacterial effect. For lower concentrations (12.5, 6.25, and 1.56 mg/mL), extracts from Shanxi were more effective. Overall, S. baicalensis from Hebei and Shanxi exhibited superior antibacterial properties compared to samples from Inner Mongolia. At the same time, the antibacterial diameter of the positive control group was 33.2 mm. When the concentration of the drug solution was 100 mg/mL, the antibacterial diameter was close to that of the positive control group, indicating that high concentration Scutellaria baicalensis solution has a good inhibitory effect on Staphylococcus aureus. In addition, the correlation analysis between baicalin content and antibacterial diameter showed a correlation coefficient of 0.568, indicating a positive correlation between the two.

Inhibition Zones of S. aureus by S. baicalensis Georgi from Different Regions. 1-1, 1-2, 2-1, 2-2, 3-1 and 3-2 are Samples of Scutellaria baicalensis from Shanxi Province (Drug Concentrations: 100 mg/ml, 50 mg/ml, 25 mg/ml, 12.5 mg/ml, 6.25 mg/ml, 3.12 mg/ml, 1.56 mg/ml, 0.78 mg/ml), 4-1, 4-2, 5-1, 5-2, 6-1, and 6-2 are Samples of Scutellaria baicalensis from Inner Mongolia (Drug Concentrations: 100 mg/ml, 50 mg/ml, 25 mg/ml, 12.5 mg/ml, 6.25 mg/ml, 3.12 mg/ml, 1.56 mg/ml, 0.78 mg/ml), 7-1, 7-2, 8-1, 8-2, 9-1 and 9-2 are Samples of Scutellaria baicalensis from Hebei Province (Drug Concentrations: 100 mg/ml, 50 mg/ml, 25 mg/ml, 12.5 mg/ml, 6.25 mg/ml, 3.12 mg/ml, 1.56 mg/ml, 0.78 mg/ml.).
Inhibitory Zone Diameter of S. aureus Inhibited by S.baicalensis Georgi from Different Places (

Correlation Between Baicalin Content and Climatic Factors
Five climate factors, namely annual average temperature (°C) (a), annual average precipitation (mm) (b), annual average sunshine hours (h) (c), relative humidity (%) (d), and wind speed (m/s) (e), were analyzed for their influence on baicalin content (Table 3). The climate factor data are listed in Table 1. The correlation analysis between baicalin and climatic factors showed that the baicalin content in S. baicalensis was positively correlated with annual average temperature, annual average precipitation, and relative humidity, and negatively correlated with annual average sunshine hours and wind speed.
Correlation Analysis Between Climatic Factors and Baicalin Content.

Correlation Between Baicalin Content and Rhizosphere Soil Properties
The influence of soil on scutellaria baicalensis is multifaceted. Alkaline and acidic soil will affect the absorption of trace elements such as calcium, phosphorus and magnesium by baicalin, thus affecting the growth of baicalin and the accumulation of effective components. At the same time, some functional groups in the molecular structure of baicalin are more sensitive to the pH of the environment, and the stability and chemical behavior of baicalin will change under different pH values. Some intermediate products of carbon metabolism are precursor substances for the synthesis of flavonoids. When the concentration of carbon dioxide in the plant growth environment is too low, the reduction of carbon fixation will lead to the reduction of precursor substances for the synthesis of baicalin, and ultimately reduce the yield of baicalin. Nitrogen can promote root growth and participate in the metabolic process. At the same time, appropriate nitrogen supply can improve the physiological state of scutellaria baicalensis and enhance the photosynthesis and nutrient absorption capacity of the plant. 20 Appropriate amount of nitrogen can enhance the efficiency of photosynthesis and carbon metabolism, and provide precursors for the synthesis of baicalin and other secondary metabolites. Ammonium nitrogen and nitrate nitrogen are different forms of nitrogen, which can affect the accumulation rate of secondary metabolites by regulating the expression of nitrogen assimilation genes, and then affect the synthesis of baicalin. 21
However, it should be noted that if excessive nitrogen fertilizer is applied, it may lead to adverse consequences such as reduced scutellaria yield. In the actual production, it is necessary to control the amount of nitrogen reasonably according to the specific situation in order to achieve the purpose of increasing the content and yield of baicalin. Sulfur can be involved in regulating the activity of many enzymes to maintain normal plant metabolism. Sulfur indirectly supports photosynthetic efficiency by maintaining chloroplast structural integrity and promoting chlorophyll precursor synthesis. In addition, secondary compounds produced by sulfur metabolism can improve plant resistance to diseases, pests, drought and other adversities. 22
Physicochemical analysis of rhizosphere soils (Table 4) showed that samples from Shanxi contained higher levels of total nitrogen, sulfur, carbon, nitrate nitrogen, and nitrite compared to soils from Hebei and Inner Mongolia. A correlation analysis (Table 5) suggested that baicalin content was positively influenced by total nitrogen levels within a certain range, indicating that nitrogen enrichment may enhance baicalin accumulation.
Physical and Chemical Properties of Rhizosphere Soil of S. baicalensis Georgi from Different Origins (
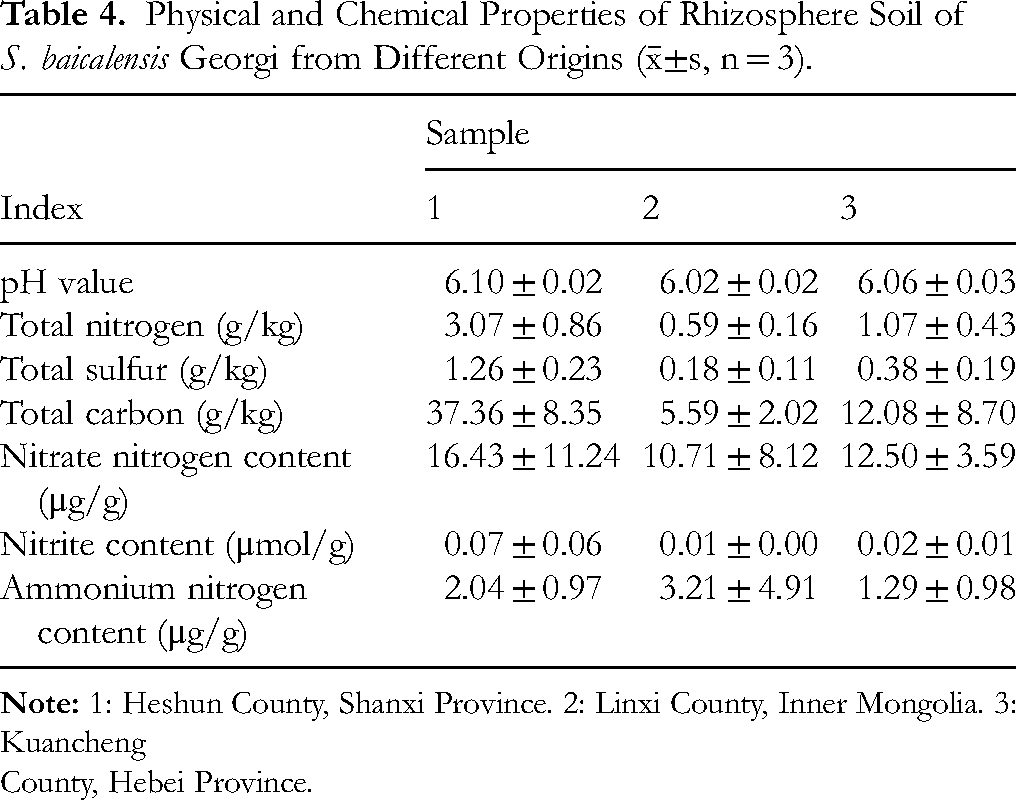
County, Hebei Province.
Correlation Analysis of Soil Physicochemical Factors and Baicalin Content.

Correlation Between Baicalin Content and Rhizosphere Soil Microorganisms
Microbial Community Composition and Diversity of Rhizosphere Soil of S. baicalensis from Different Origins
Rhizosphere microorganisms can enhance the resistance of medicinal plants to diseases, pests, and stress, promote the growth of medicinal plants and the development of medicinal parts, alleviate metabolic disorders of medicinal plants, and promote the synthesis and accumulation of medicinal substances. Rhizosphere soil microorganisms can provide various bioactive components for Scutellaria baicalensis. 23 and some rhizosphere microorganisms can induce gene expression in Scutellaria baicalensis and affect its metabolic process. 24
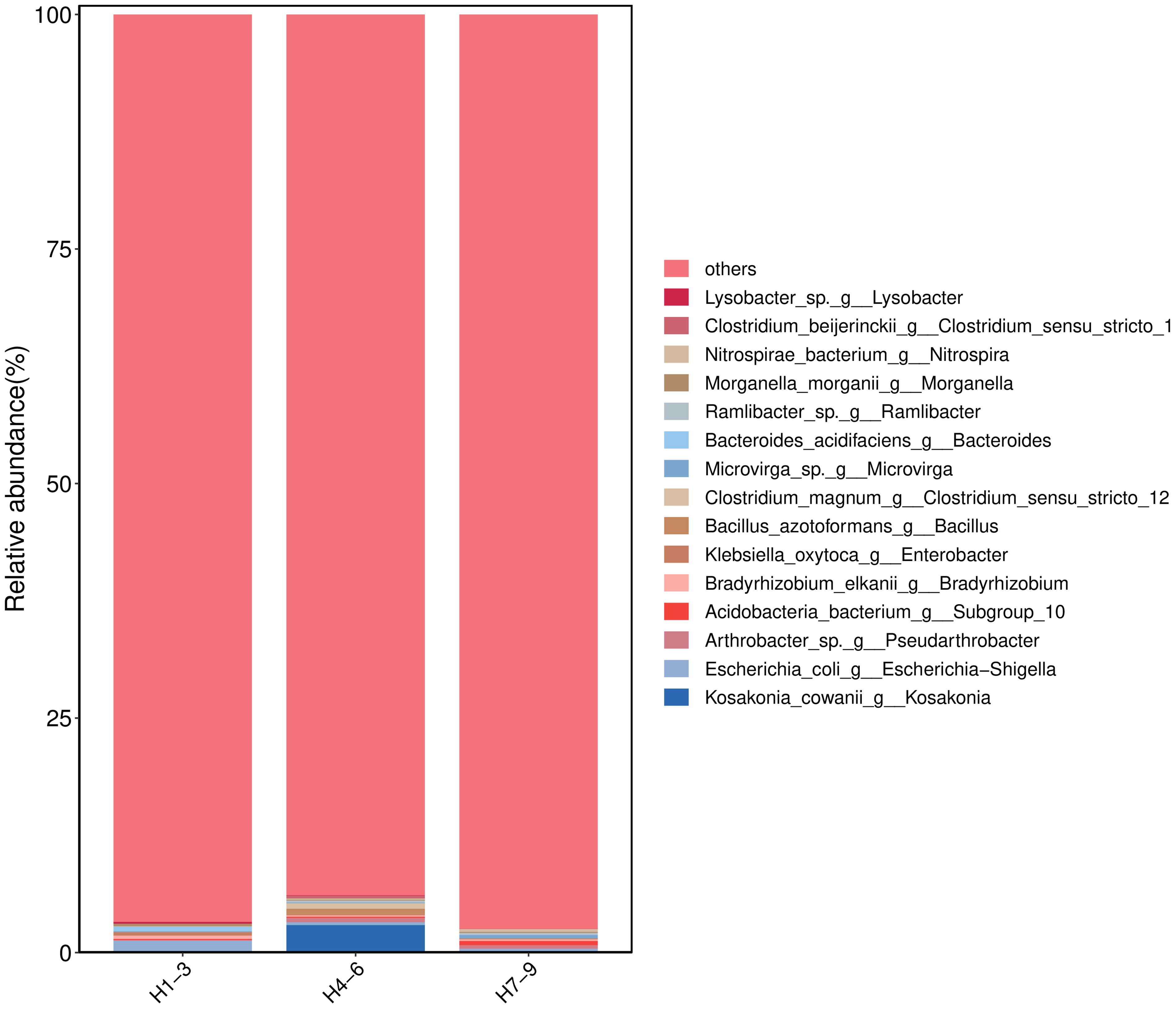
After high-throughput sequencing data quality control and deduplication, 19,437 ASVs were obtained from rhizosphere soil samples of S. baicalensis. These ASVs were classified into 36 phyla, 89 classes, 228 orders, 350 families, 636 genera, and 1281 species. Significant differences in microbial composition were observed across rhizosphere soils from different regions. The 15 most abundant genera were selected for analysis (Figure 2), with dominant genera including Sphingomonas, Enterobacter, TRA3-20, and Muribaculaceae. Further analysis of microbial community composition and correlations between regions was visualized using a Venn diagram (Figure 4). A total of 4776 ASVs were identified in Hebei rhizosphere soil, 4251 ASVs in Shanxi, and 3508 ASVs in Inner Mongolia, with 251 ASVs shared by all three regions. Common taxa included k Bacteria, p Proteobacteria, c Alphaproteobacteria, x Longiales, and f Longiaceae. At the species level (Figure 3), the rhizosphere soil from Inner Mongolia exhibited the highest bacterial species diversity, with Kosakonia comwanii (genus Kosakonia) being notably abundant. The terrestrial phase diagram (Figure 5) shows the relative abundance of microorganisms in the rhizosphere soil across different production areas, with Proteobacteria, Actinobacteria, and Bacteroidetes being particularly abundant in Shanxi, while Proteobacteria, Bacteroidetes, and Gemmatimonadota dominated in Hebei. In all regions, Proteobacteria and Actinobacteria were the most abundant phyla.

Histogram of Community Structure of Samples from Different Sampling Locations, the Figure Shows the Top 15 Species in Terms of Microbial Abundance in the Rhizosphere Soil of Scutellaria baicalensis from Different Origins, with Dominant Species Including Sphingomonas, Enterobacter, TRA3-20, Muribaculaceae, etc.
Histogram of Community Structure of Samples from Different Sampling Location. The Figure Shows the Microbial Species Level Identified by Species Selection at the Genus Level in the Rhizosphere Soil of Scutellaria baicalensis from Different Origins.

Using Venn Diagram to Demonstrate and Analyze the Correlation and Differences of Rhizosphere Soil Microbial Communities in Scutellaria baicalensis from Different Origins.

Using a Ternary Phase Diagram to Display the Relative Abundance of Microorganisms in Rhizosphere Soils from Different Origins.
Alpha diversity index (Table 6) revealed that the sequencing depth provided a comprehensive representation of microbial species, with a coverage index of 0.999. The high ACE and Chao1 indices indicated substantial microbial abundance in the samples, while the Simpson index (>0.98) confirmed high diversity. The Shannon index (>8) indicated that the microbial communities were diverse and evenly distributed across the samples. Principal coordinates analysis (PCoA) (Figure 6) showed clear clustering of samples from different regions, with Hebei samples more tightly clustered. A sample from Inner Mongolia exhibited greater dispersion. The first principal component (PC1) contributed 77.72% to the variation, while PC2 contributed 6%, and the total variation explained by the first two components was 83.72%. These findings demonstrate high microbial diversity and clear geographic differentiation across the rhizosphere soils of S. baicalensis.

Using PCoA to Display the Principal Coordinates Analysis of Samples from Different Regions, there is a Clear Clustering of Samples from Various Production Areas, with Hebei Samples Showing a Higher Degree of Clustering and Inner Mongolia Having a Dispersed Sample. The Contribution of PC1 is 77.72%, the Contribution of PC2 is 6%, and the Total Contribution of the Two Principal Components is 83.72%.
Alpha Diversity Index of Rhizosphere Soil microbiota from Various Origins.

Correlation of Rhizosphere Soil Microbial Communities in S. baicalensis from Different Habitats
At the order taxonomic level, the top 16 most abundant microorganisms across all samples were selected for differential analysis. The relative abundance of these microorganisms in each sample is shown in (Figure 7, 8). Solibacterales, Gammaproteobacteria, Desulfovibrionales, and Oscillospirales were found to be highly abundant in rhizosphere soil samples from Shanxi, but less so in those from Inner Mongolia and Hebei. Conversely, the abundance of P2-11E was higher in Inner Mongolia rhizosphere soil samples, with no significant presence in Hebei or Shanxi. Rhizosphere soil samples from Hebei exhibited greater abundances of Rhodothermales, EPR3968-O8a-Bc78, Chlamydiales, Latescibacterota, Babeliales, and Zavarziniales. To explore the relationships between microbial species, the top 50 most abundant species were selected for correlation analysis. The SC-I-84 species, belonging to Proteobacteria, was significantly correlated with several other microorganisms. It exhibited a negative correlation with Bacillus and a positive correlation with Ellin6067 and Puia. Nordella showed a negative correlation with Massilia and Pseudomonas, while it was positively correlated with Gaiella, MB-A2-108, and KF-JG30-B3.

Microbial Clustering Heatmaps of Samples from Different Regions, Correlation of Rhizosphere Soil Microbial Communities of Scutellaria baicalensis from Different Origins Classified by Order, and Selection of the Top 16 Microorganisms with Relative Abundance in All Samples for Differential Analysis.
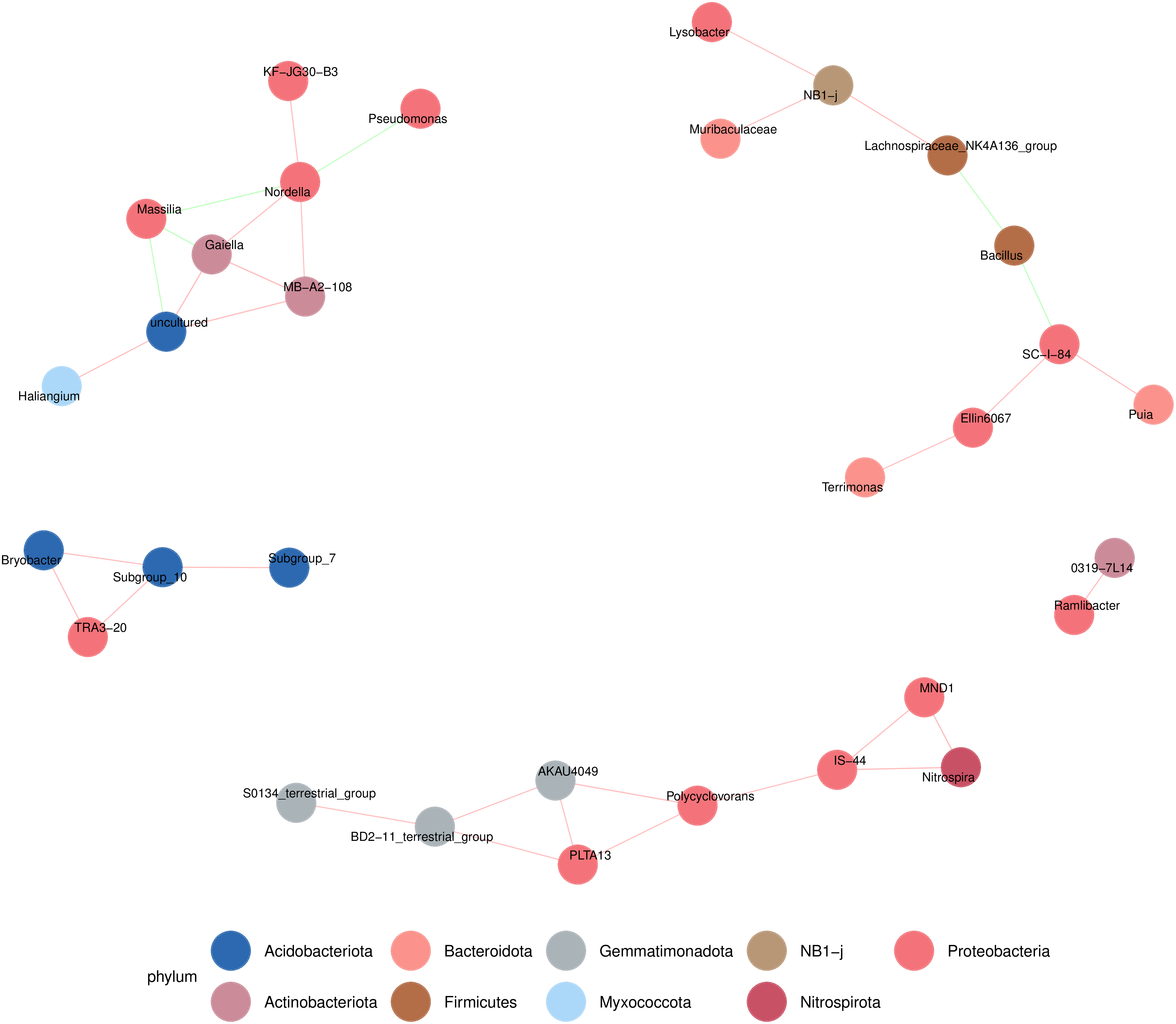
Differential Microbial Correlation Network of Samples from Different Regions.
Gemmatimonas can improve soil fertility and promote the growth of Scutellaria baicalensis by enhancing soil nutrient absorption, producing beneficial substances, and nitrogen fixation. On the other hand, competitive colonization can prevent pathogens from causing damage to crops, protect plants from infection, and produce beneficial metabolites to effectively inhibit or kill pathogenic microorganisms, thereby providing a relatively safe and healthy growth environment for them. 25
Deformable bacteria promote the development of Scutellaria baicalensis roots by secreting enzyme substances, helping the roots better absorb nutrients and promote their growth and development. At the same time, it can also improve soil structure, enhance soil permeability and water retention capacity, thereby providing better growth conditions for it. Research has shown that Ellin6055 is positively and negatively correlated with each active ingredient in Scutellaria baicalensis, and is an important genus that affects the accumulation of active compounds in Scutellaria baicalensis. It also exhibits varying degrees of correlation with soil physicochemical factors. 26
Actinobacteria are important decomposers in soil, capable of promoting the decay of animal and plant remains in the soil, thereby participating in the organic matter and nutrient cycling of the soil. It has the functions of decomposing organic matter, improving soil structure, fixing nitrogen, promoting plant growth, inhibiting pathogens, and maintaining soil ecological balance. 27
The genus Bifidobacterium can not only improve the nutrient absorption and utilization efficiency of Scutellaria baicalensis, inhibit the growth of pathogenic bacteria, but also improve land quality and promote the growth and development of Scutellaria baicalensis.
Ramlibacter bacteria can promote plant growth and nutrient absorption: enhancing plant disease and stress resistance, increasing soil permeability, 28 and reducing soil salinization can be used as excellent organic fertilizers.
The top 50 species with horizontal abundance in the results include Gemmatimonas, Proteobacteria, Actinobacteria, etc These genera are dominant in terms of species composition and have a closer relationship with soil, which can promote plant growth in different aspects. By elucidating the functions of these genera, the quality of cultivated Scutellaria baicalensis can be further ensured, and they can serve as biological nutrient bacteria or inhibitory antagonistic bacteria.
Correlation Between Rhizosphere Soil Microorganisms and Baicalin Content in S. baicalensis from Different Habitats
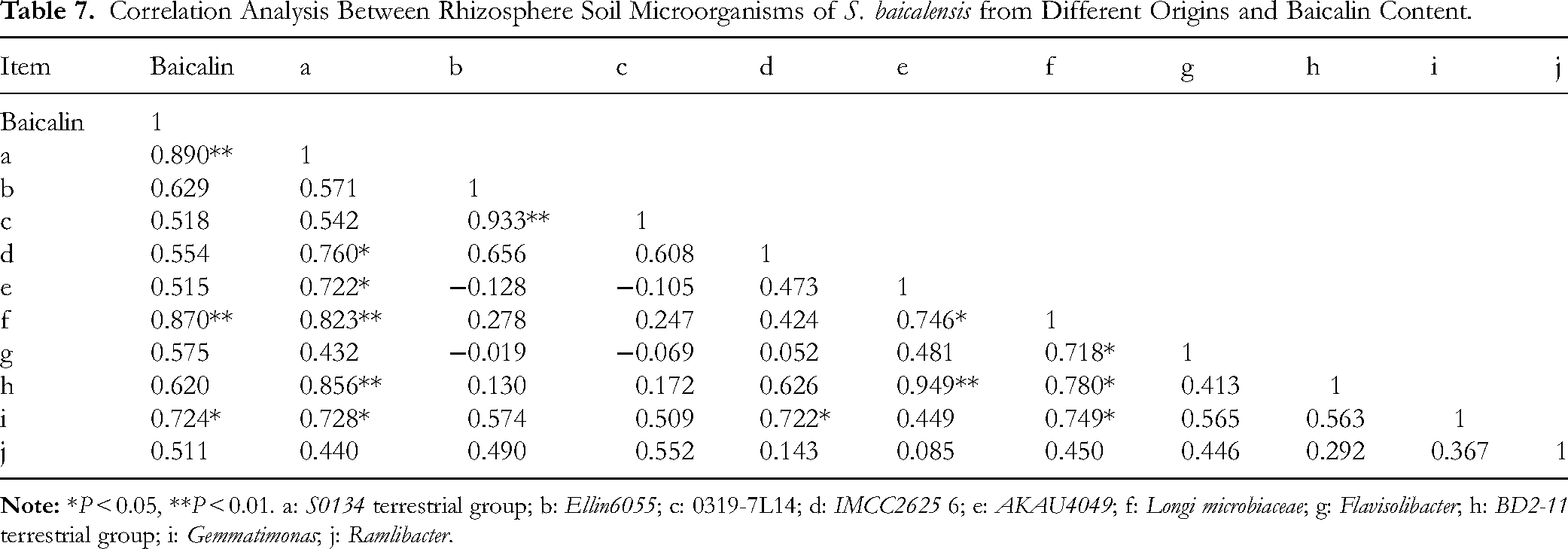
To analyze the influence of rhizosphere soil microorganisms on baicalin content, the top 50 microorganisms by relative abundance were selected for correlation analysis using SPSS software (version 23.0; Table 7). After screening, the top 10 microorganisms that correlated with baicalin content were identified. The results showed that the S0134 terrestrial group, Longimicrobiaceae, and Gemmatimonas, were significantly correlated with baicalin content. Additionally, Ellin6055, 0319-7L14, IMCC26256, AKAU4049, Flavisolibacter, the BD2-11 terrestrial group, and Ramlibacter were correlated with baicalin content.
Correlation Analysis Between Rhizosphere Soil Microorganisms of S. baicalensis from Different Origins and Baicalin Content.
Discussion
Environmental factors play crucial roles in the development of potent compounds from S. baicalensis. 29 This study focused on analyzing the correlation between environmental factors and the accumulation of baicalin in S. baicalensis from three distinct regions. The findings highlighted that S. baicalensis from Chengde, Hebei Province, exhibited superior baicalin content and quality compared to other regions. The soil in Chengde's Huangqin planting areas consists of shrub brown and coarse bone soil, while river areas feature sandy and sandy loam soil with strong nutrient retention and efficient water transport.10,29 High levels of Fe, phosphorus, and Sr in the soil of Chengde created ideal conditions for cultivating high-quality S. baicalensis. Moreover, sandy soil with a high Fe content and slightly alkaline pH is suitable for S. baicalensis. 30 Using HPLC fingerprinting, Wang et al analyzed the chemical profiles of Huangqin from various habitats, applying hierarchical clustering, principal component, and peak area analyses. The fingerprints of S. baicalensis from Chengde significantly differed from other regions. 31 These results are consistent with those of the present study. S. baicalensis is valued in traditional Chinese medicine for its anti-inflammatory, antioxidant, and antiallergenic properties, often used to treat fever, cough, allergic rhinitis, and wheezing. 32 The baicalin content positively correlates with its antibacterial effects, with Chengde samples showing superior efficacy at higher concentrations. The accumulation of baicalin in S. baicalensis closely relates to climate factors such as light, temperature, moisture, and nitrogen.33–37 This study found baicalin levels positively correlated with annual mean temperature, rainfall, and relative humidity, but negatively correlated with annual mean sunshine hours and wind speed. Notably, light conditions not only affect the flavonoid content, but also induce the transformation of several types of flavonoid components. 38 Additionally, different temperatures have varying effects on the accumulation of baicalin content in S. baicalensis, with 25 °C and 30 °C being more suitable for growth, with higher content observed as temperatures rise within this range. 39 In the present study, low rainfall in 19 producing areas was associated with reduced baicalin levels, with severe water deficiency inhibiting its accumulation. 40 Our results indicated that within a certain range, increasing the temperature and soil moisture could be beneficial to the accumulation of active components in S. baicalensis.
Nitrogen is an essential nutrient that limits plant growth and production, as it is a fundamental component of chlorophyll and proteins, directly linked to photosynthesis in plants. 41 Phosphorus and potassium are equally crucial nutrients for the growth and development of plants. 42 Different levels and and combinations of nitrogen, phosphorus, and potassium impact S. baicalensis growth and secondary metabolite production. Growth indicators and baicalin content were higher under combined fertilization treatments compared to single-element applications. 43 The yield of baicalin was significantly affected by nitrogen and phosphorus levels. Baicalin yield was notably influenced by nitrogen and phosphorus levels. As nitrogen and potassium fertilization rates increased, baicalin content initially increased and then decreased. At lower fertilization levels, baicalin content increased and then stabilized with higher phosphorus fertilization rates. 44 Nitric oxide is produced by nitric oxide synthase-like enzymes and plays an important role in the biosynthesis of baicalin, acting as part of the defense response of S. baicalensis plant cells to ultraviolet (UV)-B radiation. 45 In this study, the highest baicalin content was found in S. baicalensis from Hebei, where the total nitrogen level was moderate. A positive correlation between baicalin content and total nitrogen was observed, indicating that both high and low nitrogen levels can affect baicalin accumulation. Rhizosphere microorganisms can promote the growth of medicinal plants, the development of medicinal parts, and the synthesis and accumulation of medicinal substances. 46 In this study, the S0134 terrestrial group, Longimicrobiaceae, and Gemmatimonas showed a significant positive correlation with baicalin content, all belonging to Gemmatimonadetes. These microorganisms are closely related to the stability of the soil microbial community and soil phosphate accumulation. 47 They exhibit characteristics of anaerobic phosphorus release and aerobic phosphorus absorption, which help in phosphate accumulation. 48 When phosphate levels reach a critical point, they further support baicalin accumulation. 49 Therefore, the S0134 terrestrial group, Longimicrobiaceae, and Gemmatimonas may have influenced soil phosphate concentration, contributing to baicalin accumulation in S. baicalensis.
The production of high-quality S. baicalensis is influenced by multiple factors. Future interdisciplinary research combining traditional Chinese medicine, agriculture, plant genetics, and molecular biology can help explore the regulatory mechanisms between the ecological environment and the active components of S. baicalensis. This approach will improve cultivation practices and enhance the quality of S. baicalensis.
In summary, the formation of high-quality Scutellaria baicalensis is influenced by various factors. In future practice, the content of baicalin Scutellaria baicalensis in this area can be increased by selecting planting areas with suitable acidity and alkalinity, adjusting the ratio of nitrogen, phosphorus, potassium and other fertilizers moderately during cultivation, and adjusting the appropriate water and temperature. This article mainly studies the effects of climate, soil and other factors on baicalin, but does not conduct research on molecular mechanisms and plant genetic genes. In the future, based on this, emerging interdisciplinary fields such as traditional Chinese medicine, agriculture, plant genetics, and molecular biology can be combined to apply transcriptomics and genomics research methods to explore the transcription status and transcriptional regulation rules of genes in the secondary metabolism process of Scutellaria baicalensis, and to explore key genes in the biosynthesis pathways of various chemical components in Scutellaria baicalensis, To understand the impact mechanism of the pharmacological substance basis of Scutellaria baicalensis on germplasm and ecological environment, in order to achieve precise regulation of Scutellaria baicalensis cultivation.
By comparing the differences in baicalin content and antibacterial effects among different origins of Scutellaria baicalensis, this study explores the climatic and soil factors that affect the accumulation of baicalin, optimizes the cultivation methods of Scutellaria baicalensis, and provides theoretical basis for improving its quality. The method used is reliable, simple and feasible, and can provide data support for the improvement of its processing technology and the establishment of new processes. However, looking back at the entire experimental process, there are still shortcomings in this study, such as a small sample size and incomplete analysis of influencing factors. Therefore, further in-depth research will be conducted in the future.
Footnotes
Statement of Human and Animal Rights
All experimental data in this experiment do not involve animal or human rights issuesv.
Consent for Publication
All of the authors are aware of and agree to the content of the article and their being listed as a co-author of the article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hebei Natural Science Foundation (H2022406056).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
