Abstract
Background
Piper chaba is a common plant in Bangladesh. It is enriched with many important biologically active compounds and popular for traditional healing.
Method
The methanol extract of the root of P. chaba was fractionated into n-hexane and ethyl acetate soluble fractions. The ethyl acetate fraction was subjected to various chromatographic techniques to isolate the pure compounds. The structures of the compounds were elucidated with the aid of UV, IR, NMR, including 1H, 13C, DEPT-135, COSY, HSQC, HMBC, spectroscopy, and LC-MS. The isolated compounds were subjected to cytotoxicity assay using HeLa cell lines.
Result
β-sitosterol (
Conclusion
To the best of our knowledge, this is the first report of isolation of the halogenated chalcone derivatives
Introduction
The genus Piper belongs to the Piperaceae family comprising about 1000 species and are native throughout the tropics and subtropics, with a few species in Eurasia. 1 The herb P. chaba Haunter (syn. of P. retrofractum Vahl and P. officinarum (Miq.) C. DC. locally known as Chui Jhal, a climbing, glabrous plant is available in various parts of India and Malay Islands. It grows abundantly in Bangladesh in Khulna division mainly in the Satkhira–Bagerhatt area. Its root is used as a spice in the Indian subcontinent. Different parts of this plant like stems, leaves, fruits, bark, and roots are known to be anti-flatulent, gastro-protective, appetizing, expectorant, antitussive and anti-fungal from the ancient time.2,3 Recent research findings showed that P. chaba reveals diverse biological activities4,5 including, anticancer6-8 and immunomodulatory 9 effects. Previous phytochemical investigation on this plant provided various types of promising bioactive compounds such as piperchabamides A-D, piperine, piperanine (Δα,β-dihydropiperine), pipernonaline, dehydropipernonaline, piperlonguminine, retrofractamide B (pipercide), guineensine, N-isobutyl-(2E,4E)-octadecadienamide, N-isobutyl-(2E,4E,14Z)-eicosatrienamide, methyl piperate, etc 10
In this study with P. chava, the known ergosterol peroxide (
Methods
General Experimental Procedures
In the separation process, silica gel 60 (Merck, 230-400 mesh) and F254 (Merck) were used for column chromatography and thin layer chromatography (TLC), respectively.
Instruments
The IR spectra were recorded with IR200 spectrophotometer (Thermo Electron Corporation, Waltham, MA). The NMR spectra were acquired in CDCl3 on an AVANCE 400 MHz instrument (Bruker Corporation, Switzerland) and the chemical shifts were referenced with the residual non-deuterated solvent signal. The ESI-MS spectra were recorded on a 6400 series triple quadrupole-time-of-flight mass spectrometer (Agilent, Palo Alto, CA).
Collection of the Plant Material
The roots of P. chaba were collected from Katalbari Upazila in the district of Kurigram, Bangladesh on May 2024 and was identified by Prof. Mohammad Zashim Uddin, Department of Botany, University of Dhaka. A voucher specimen (DUSH 10828) for this collection has been maintained at herbarium of the department. The collected roots were air dried and crushed into coarse powder and stored in airtight bottle.
Extraction and Isolation
The dried powder (about 568 g) of P. chaba was taken in a clean, round bottomed flask and extracted with methanol at room temperature and atmospheric pressure. The extract was concentrated to dry mass (18.74 g) using a rotary evaporator under reduced pressure. The concentrated extract was then partitioned in a separatory funnel by using n-hexane and ethyl acetate. The extracts were concentrated separately to dry mass with a rotary evaporator at 40 °C. Based on the thin layer chromatography (TLC) pattern, the ethyl acetate (5.42 g) fraction was subjected to column chromatography over silica gel and the column was eluted with n- hexane, ethyl acetate and methanol mixtures of increasing polarities (% ethyl acetate in n-hexane: 0, 1, 2, 3, 4, 5, 7, 8, 9, 10, 12, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90 and 100; % methanol in ethyl acetate: 0, 1, 2, 3, 5 and 10) to give total of 33 fractions, each 100 ml. All the column fractions were examined by TLC under UV light and by spraying with vanillin-sulphuric acid reagent. Depending on the TLC behavior, some fractions were combined in a beaker, and all were again marked as B1-B13. Among these, fraction B4 (225 mg) (eluted with 7-8% ethyl acetate in n-hexane) showed four spots on TLC plate with mild tailing. This fraction was again subjected to column chromatography over silica gel using n- hexane and ethyl acetate mixtures of increasing polarities (% ethyl acetate in n-hexane: 0, 1, 3, 5, 7, 8, 10, 12, 15, 20, and 30) to give a total of 11 fractions, each 100 ml. Each of the fractions was monitored by TLC and the fractions of similar behaviors were combined and marked as T1-T6. The fractions T3 (eluted with 7–8% ethyl acetate in n-hexane in the sub-column) and T4 (eluted with 10–12% ethyl acetate in n-hexane in the sub-column) appeared to contain single spot on TLC plate. The isolated solids were marked as compound
Cytotoxicity Assay
HeLa, a human cervical carcinoma cell line, was maintained in DMEM (1% penicillin-streptomycin (1:1) and 0.2% gentamycin) and 10% fetal bovine serum (FBS). Cells (2.0 × 104/100 μl) were seeded onto 96-well plate and incubated for 24 h at 37 °C with 5% CO2. 1 mg/ml of stock of compound in 1% DMSO in water was prepared. After incubation, 25 μl of the stock solution was added to each well. Then the plate was placed in carbon dioxide incubator at 37 °C with 5% CO2. After 24 h, cytotoxicity was examined under an inverted light microscope. Triplicate wells were used for each sample.11,12
Statistical Analysis
Three replicates (n = 3) of each sample were used and the values were calculated as mean ± standard deviation. Microsoft Excel 2010 software was used for this purpose.
Results
β-Sitosterol (1a ) with a Trace of Stigmasterol (1b )
White crystalline solid; IR (KBr) υmax 3238, 2958, 2933, 2864, 1454 cm−1; 1H-NMR (400 MHz, CDCl3): δ 5.35 (1H, br. s, 6-H), 5.33 (1H, dd, 23-H of
Piperine (2 )
White crystalline solid; IR (KBr) υmax 2993, 2855, 1636, 1549, 1193, 922 cm−1; 1H-NMR (400 MHz, CDCl3): δ 7.36 (1H, d, H-3), 6.95 (1H, d, H-2’), 6.86 (1H, d, H-6’), 5.95 (2H, s, -OCH2O-), 3.56 (2H, br. s, H-6”), 1.54 (2H, m, H-3”), 1.64 (2H, m, H-5”), 1.57 (2H, m, H-4”); 13C-NMR (100 MHz, CDCl3): 165.4 (C-1), 119.7 (C-2), 142.8 (C-3), 125.3 (C-4), 138.3 (C-5), 131.0 (C-1’), 105.6 (C-2’), 148.2 (C-3’), 148.1 (C-4’), 108.4 (C-5’), 122.5 (C-6’), 101.2 -OCH2O-), 45.3 (C-2”), 26.1 (C-3”), 75.3 (C-4”), 25.3 (C-5”), 46.9 (C-6”).
Ergosterol peroxide (3 )
White crystalline solid; IR (KBr) υmax. 3178, 3032, 1711, 1490, 1247 cm−1; 1H-NMR (400 MHz, CDCl3): δ 6.49 (1H, d, H-7), 6.31 (1H, d, H-6), 5.21 (1H, dd, H-22), 5.13 (1H, dd, H-23), 3.96 (1H, m, H-3), 0.99 (3H, d, H-21), 0.911 (3H, d, H-28), 0.87 (3H, s, H-19), 0.83 (3H, s, H-18), 0.80 (3H, d, H-27), 0.79 (3H, d, H-26); 13C-NMR (100 MHz, CDCl3): 135.41 (C-7), 135.21 (C-22), 132.31 (C-23), 130.76 (C-6), 82.17 (C-8), 79.44 (C-5), 66.49 (C-3), 56.19 (C-17), 51.68 (C-9), 51.07 (C-14), 44.56 (C-13), 42.77 (C-24), 39.75 (C-20), 39.34 (C-12), 37.96 (C-4), 36.92 (C-10), 34.69 (C-1), 33.07 (C-25), 30.11 (C-2), 28.66 (C-16), 23.4 (C-15), 20.88 (C-11), 20.63 (C-21), 19.96 (C-27), 19.65 (C-26), 18.19 (C-19), 17.57 (C-28), 12.88 (C-18).
(E)-1-(4-chlorophenyl)-3-(4-methylphenyl)-prop-2-en-1-one (4 )
Pale yellow solid; m.p. 156–158 °C (lit.
13
155-157 °C) IR (KBr) υmax 3100–3000, 1658, 1375–1331, 1275–1222, 738, 705–667 cm−1. 1H and 13C NMR data (
1H NMR (400 MHz) and 13C NMR (100 MHz) Spectral Data of Compounds

(E)-1-(4-chlorophenyl)-3-(4-methoxyphenyl)-prop-2-en-1-one (5 )
Pale yellow solid; m.p. 117–119 °C (lit.
13
116-118 °C); IR (KBr) υmax 3110–3000, 1711, 1400–1350, 1300–1217, 740 cm−1. 1H and 13C-NMR data (
Cytotoxicity Assay
Table 2 represents the cytotoxicity of the isolated compounds (
Cytotoxicity of the Isolated Compounds (

Discussion
β-sitosterol (

Structures of Compounds Isolated from P. chaba Roots.
Compound
The 1H NMR spectrum (Supplementary figure S10) of the compound
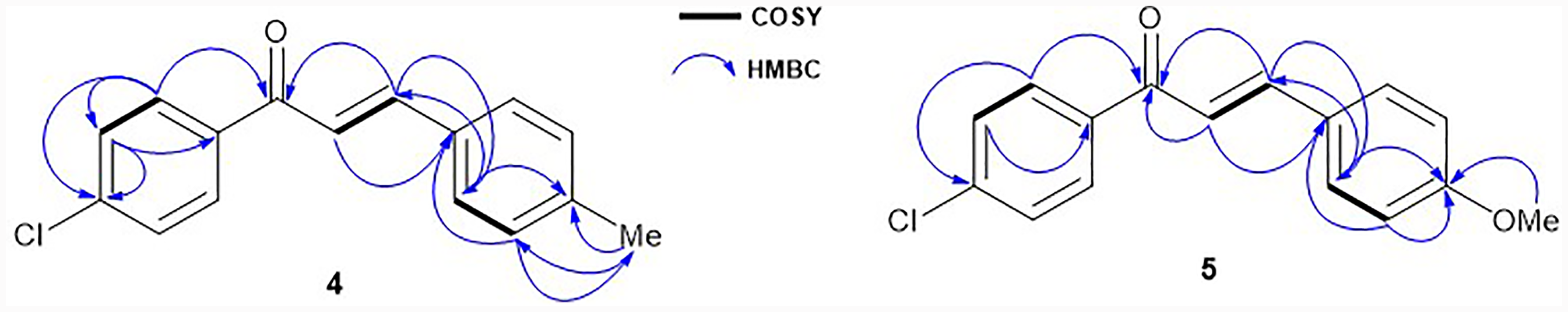
1H-1H COSY and HMBC Correlations of Compounds
Compound
The 1H NMR spectrum (Supplementary figures S17) of the compound
The 1H and 13C NMR spectral data of compound
The survival rate of HeLa Cells was less than 5% for the
Conclusions
In the present study with the root of P. chava, the well-known ergosterol peroxide (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251334718 - Supplemental material for Chlorinated Chalcones, Steroids and Piperine from the Roots of Piper chaba H. Growing in Bangladesh and Their Cytotoxicity on HeLa Cell Lines
Supplemental material, sj-docx-1-npx-10.1177_1934578X251334718 for Chlorinated Chalcones, Steroids and Piperine from the Roots of Piper chaba H. Growing in Bangladesh and Their Cytotoxicity on HeLa Cell Lines by Anath Chandra Roy, Md. Hasanur Rahman, Mohammad Rashedul Haque, Mohammad Sharifur Rahman, Tofail Ahmad Chowdhury and Mohammad Abdur Rashid in Natural Product Communications
Footnotes
Acknowledgments
The authors thank the members of the Division of Natural Products at Honam National Institute of Biological Resources for their preparations.
Ethical Considerations
Ethical Approval is not applicable for this article.
Author Contributions/CRediT
ACR performed the experiments, MHR provided methodology and supervision, MRH reviewed the manuscript, MSR conducted writing and data analysis, TAC designed and supervised the study, MAR conducted supervision, data analysis and reviewing. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data either generated or analyzed during this study are included in this published article or available from the corresponding author upon reasonable request.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
