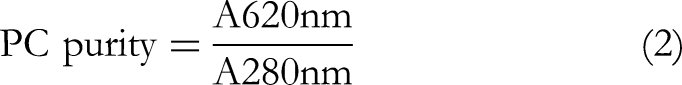Abstract
Keywords
Introduction
Phycocyanin (PC) is a naturally occurring phycobiliprotein derived from cyanobacteria such as Spirulina platensis.1,2 It is a light-harvesting pigment in photosynthesis (Figure 1) and has gained considerable scientific attention due to its diverse biological activities, including antioxidant, anti-inflammatory, and anticancer properties.1–3 The therapeutic potential of PC is linked to its unique chemical structure, comprising an open-chain tetrapyrrole chromophore covalently bound to a protein, which imparts both bioactivity and water solubility.2,4 These attributes make PC a promising candidate for pharmaceutical and nutraceutical applications. Among its various biological activities, PC has demonstrated significant potential in mitigating oxidative stress and modulating cellular pathways involved in chronic diseases.4–6 Its antioxidant properties help neutralize reactive oxygen species (ROS), which are key contributors to cellular damage and aging.2,5 Moreover, PC has been reported to exhibit anti-inflammatory effects by inhibiting pro-inflammatory cytokines and enzymes, suggesting its role in managing inflammatory-related disorders.1,3

Chemical Structure of C-phycocyanin Extracted from Spirulina sp. 7 .
One emerging area of research focuses onthe antiglycating properties of PC. Protein glycation, a non-enzymatic reaction between reducing sugars such as glucose and amino groups in proteins, forms advanced glycation end products (AGEs).6,8 The accumulation of AGEs is implicated in the pathogenesis of chronic disorders, including diabetes, neurodegenerative diseases, and cardiovascular complications. AGEs cause structural and functional alterations in proteins, contributing to cellular dysfunction, inflammation, and tissue damage.8–10 Targeting glycation processes with natural inhibitors like PC could offer preventive and therapeutic strategies for these conditions.8,11
Bovine serum albumin (BSA), a globular protein commonly used as a model for studying protein modifications, is particularly suited for investigating glycation mechanisms due to its well-characterized structure and functional similarity to human serum albumin(HAS)s.12–16 BSA has been extensively used in glycation studies due to its known amino acid residues susceptible to glycation, providing a reliable model for understanding protein-sugar interactions. Exploring the interaction between PC and BSA could enhance our understanding of the antiglycating potential of PC and its mode of action at the molecular level.13,17 Fluorescence quenching studies, employing spectroscopic techniques such as steady-state and time-resolved fluorescence, provide crucial insights into the binding dynamics.13,18 The Stern-Volmer equation can determine the quenching constant, indicating the presence of dynamic or static quenching mechanisms.17,19 Moreover, spectral shifts in fluorescence emission peaks upon PC binding can indicate conformational changes in BSA.12,16 A redshift may suggest increased polarity around aromatic residues like tryptophan, while fluorescence quenching points toward complex formation. These studies enable the characterization of interaction forces such as hydrogen bonding, van der Waals forces, and hydrophobic interactions.12,13,17,18
This study focuses on the extraction of PC from Spirulina platensis, the characterization of its interaction with BSA, and the evaluation of its antiglycation properties through glucose-induced glycation. By investigating these aspects, the research aims to provide deeper insights into the therapeutic potential of PC as a natural inhibitor of protein glycation, contributing to the development of novel interventions for AGE-related diseases. While BSA serves as a well-established model for glycation studies, future investigations will extend these findings to HSA to evaluate the pharmaceutical relevance of PC's antiglycating properties in human systems.
Material and Methods
Materials
The axenic culture of Spirulina platensis PCC 7345 used in this study was kindly donated by Dr Vani B from BITS Pilani, Hyderabad. The BG11 growth medium, which was essential for cultivating the microalgae, was procured from Himedia Laboratories, based in Mumbai, India. All other chemical reagents and materials used throughout the experiment were sourced from SD Fine Chemicals, Mumbai, India.
Methods
Culture Growth and Biomass Collection
Spirulina platensis PCC 7345 was cultured for 14 days in St. BG11 medium under continuous illumination and constant stirring at 100 rpm. Following the incubation period, the cell biomass was collected by centrifugation at 10 000 rpm for 10 min at 10 °C. The resulting biomass was then washed three times with distilled water to remove any residual medium.19,20
PC Extraction and Quantification
A 5 g portion of the washed biomass was added to 1 M sodium phosphate buffer and subjected to three freeze-thaw cycles. The mixture was centrifuged at 10 000 rpm for 10 min at 10 °C.19,20 The supernatant was analyzed spectrophotometrically employing a Shimadzu UV 1800 spectrophotometer to determine PC yield and purity using the following equations
21
:
Interaction Studies
Preparation of Solutions
PC was dissolved in phosphate-buffered saline (PBS, pH 7.4) at a specific concentration, then filtered through a 0.45 µm membrane to remove any particulates. BSA was dissolved in PBS at a concentration of 1 mg/mL and kept at 4 °C until use. Various molar ratios of BSA and PC were prepared by mixing equal volumes of BSA (1 mg/mL) with PC at different concentrations (0-10 uM). The mixtures were incubated at 37 °C for 1 h to allow interaction and equilibration. Control samples of BSA and PC alone were also prepared under identical conditions.12–14
Fluorescence Quenching Study
Fluorescence quenching studies were carried out using a titration technique. BSAat a concentration of 0.3 mg/mL was titrated with varying amounts of PC in the range of 0 to 10 μM, with an excitation wavelength of 280 nm. The emission spectra were collected between 300 and 500 nm at different temperatures, with a slit width of 5 nm, using an Agilent Cary Eclipse spectrofluorometer (Model No. G-9800AA, Agilent Technologies, Victoria, Australia). The temperatures were controlled at 25, 30, and 37 °C using a Cary single-cell Peltier accessory (Type-SPVF-1X0, Agilent Technologies, Victoria, Australia). The inner filter effect on PC fluorescence was corrected according to the formula provided.12–17:
Stern-Volmer Plot Analysis
The quenching of BSA fluorescence by PC was analyzed using the Stern-Volmer equation12–18:
A Stern-Volmer plot was generated by plotting against it to determine the quenching constant. The linearity of the plot indicated the quenching mechanism and binding affinity.12,16
Glycation Assays
Sample Preparation
Reaction mixtures containing BSA (10 mg/mL) and D-glucose (100 mg/mL) were prepared in phosphate-buffered saline (PBS, pH 7.4). Sodium azide (3 mM) was added to prevent microbial contamination. Samples were divided into experimental groups: BSA + glucose (glycated), BSA + glucose + PC (treated), BSA + PC (control), and BSA alone (control). Mixtures were incubated at 37 °C for four weeks with periodic sampling every week.20,22–25
Browning Assay
Aliquots of reaction mixtures (0.1 mL) were collected weekly. Absorbance was measured at 420 nm using a UV-Vis spectrophotometer. Increased absorbance indicated a Maillard reaction.
Fructosamine Assay
The samples (10 µL) were incubated with 0.5 mL of NBT reagent (0.25 mM in 100 mM carbonate buffer, pH 10.4) and allowed to react at 37 °C for 15 min. Absorbance was then measured at 530 nm using a UV-Vis spectrophotometer. To quantifyfructosamine content, 1-deoxy-1-morpholino-fructose (1-DMF) was used as the reference standard.
Protein Carbonyl Content Assay
Samples (0.1 mL) were combined with 0.1 mL of 10 mM DNPH in 2 N HCl and incubated in the dark for 1 h with occasional mixing. Proteins were precipitated by adding 20% TCA and washed with a mixture of ethanol and ethyl acetate (1:1). The precipitates were then dissolved in 6 M guanidine hydrochloride, and absorbance was measured at 370 nm. The carbonyl content of each sample was calculated and expressed as nmol of carbonyl per milligram of protein, using the extinction coefficient for 2,4-dinitrophenylhydrazine (DNPH) (ɛ = 22 000 M−1cm−1), as detailed in the literature. 26
Total AGE Formation Assay
The fluorescence intensity of total AGEs was measured at excitation/emission wavelengths of 370/440 nm. Triplicate readings were taken for each sample. 23
Individual Fluorescent AGEs
To analyze the intrinsic fluorescence of specific AGEs including argpyrimidine, pentosidine, vesperlysine, and crossline, samples were prepared at a concentration of 1.5 mg/mL in a 50 mM sodium phosphate buffer (pH 7.4). Emission spectra were recorded within the 300-600 nm (Table 1) wavelength range using an Agilent Cary Eclipse spectrofluorometer.25–29
Excitation and Emission Wavelengths of Different Fluorescent AGEs.

Statistical Analysis
Data analysis was carried out using GraphPad Prism version 9. A two-way ANOVA was performed to assess statistical significance. Data are presented as the mean ± standard deviation (SD) with a sample size of n = 3. Tukey's multiple comparisons test was used to compare differences between treatments. Statistical significance was considered at the P < .05 level compared to the positive control group.20,22–25
Results
Determination of Concentration and Purity of PC
The PC extracted from Spirulina platensis PCC 7345 had a concentration of 0.032 mg/mL. Its purity, expressed as the absorbance ratio and assessed using spectrophotometric analysis, was determined to be 0.2454, indicating the presence of other pigments and molecules in the crude extract.
Fluorescence Quenching
Stern-Volmer Plot
BSA displays intrinsic fluorescence attributed to its aromatic amino acids, mainly tryptophan, with additional contributions from tyrosine and phenylalanine.12,17 The fluorescence emitted by tryptophan is highly responsive to changes in its surrounding environment, making it a valuable tool for investigating protein interactions, structural rearrangements, and complex formations. This characteristic helps better understand the binding behavior of small molecules with proteins like BSA.14–16
The interaction between PC and BSA can be evaluated using fluorescence spectroscopy, where PC functions as a quencher. When excited at specific wavelengths, BSA emits fluorescence,12–18 and the intensity of this emission decreases upon PC binding (Figure 2). This reduction in fluorescence intensity indicates the formation of a complex between PC and BSA.

Fluorescence Spectra of BSA-PC Interaction [Graph of intensity (a.u.) versus Wavelength (nm) for 0-10 µM of PC; BSA – Bovine Serum Albumin; PC – Phycocyanin].
Fluorescence quenching experiments were conducted at temperatures of 25, 30, and 37 °C to study the interaction between PC and BSA. These measurements helped examine how temperature influences the binding process and the underlying quenching mechanism (Figure 3). Quenching can occur through two main mechanisms: dynamic quenching, involving collision-based interactions between PC and BSA and static quenching, where a stable, non-fluorescent complex forms.19–21 The quenching behavior between PC and BSA was analyzed using the Stern-Volmer equation. This approach facilitated the determination of the quenching constant and offered valuable insights into the mechanism driving the interaction.12,13,21

Stern-Volmer Plot Illustrating the Interaction between PC and BSA at various Temperatures. [Linear Regression plot of F0/FF_0/FF0/F versus [PC] for PC-BSA Interaction; PC – Phycocyanin, BSA – Bovine Serum Albumin].
A decline in BSA fluorescence intensity after PC addition indicates changes in the microenvironment around tryptophan residues, likely due to increased hydrophobic interactions.13,22 These findings offer valuable information on the binding dynamics, thermodynamic properties, and molecular interactions between PC and BSA.12–22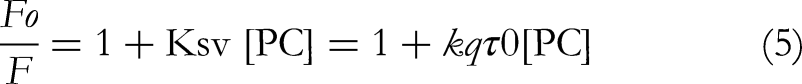
Here, [PC] denotes the concentration of the quencher phycocyanin. The Stern-Volmer quenching constant (
The Stern-Volmer constant (Ksv) was determined using the Stern-Volmer equation by applying linear regression to the plot of Fo/F versus [PC] (Figure 3). Additionally, the calculated quenching rate constant (kq) further validated the quenching mechanism based on the relevant equation.17,22
Here, τ0 represents the average lifetime of the molecule in the absence of a quencher.
The interaction between the PC–BSA complex was assessed by calculating the Kq value, which ranged from 1016 M−1 s−1 at the different temperatures (Table 2). Importantly, the calculated Kq values were significantly higher than the maximum value for scatter collision quenching, which is 2.0 × 10 M−1 s−1. This indicates that the quenching mechanism in the PC–BSA system is more efficient than typical collision-based processes. Furthermore, both the Stern-Volmer constant (Ksv) and the quenching rate constant (Kq) showed a clear increase with temperature, as presented in Table 2, suggesting that the interaction between PC and BSA intensifies at higher temperatures.12–16
Values of the Stern–Volmer Constant (Ksv) and Biomolecular Rate Constant (kq) for the PC-BSA Interaction.

Adding PC to a solution containing BSA led to a small redshift in the maximum emission wavelength (Figure 2). This shift suggests a change in the structure of the protein, specifically in the hydrophobic subdomain IIA. The interaction between the PC and the fluorophore likely creates a more hydrophobic environment, inducing this structural alteration. To further understand the nature of this interaction, the effect of temperature was investigated (Table 3). As temperature increases, the rate of molecular diffusion also increases, leading to more frequent collisions between the quencher (PC) and the fluorophore. In the case of dynamic quenching, this increased collision frequency should result in higher quenching constants.12–17,22
Binding Affinity and Stoichiometry of the PC and BSA Interaction
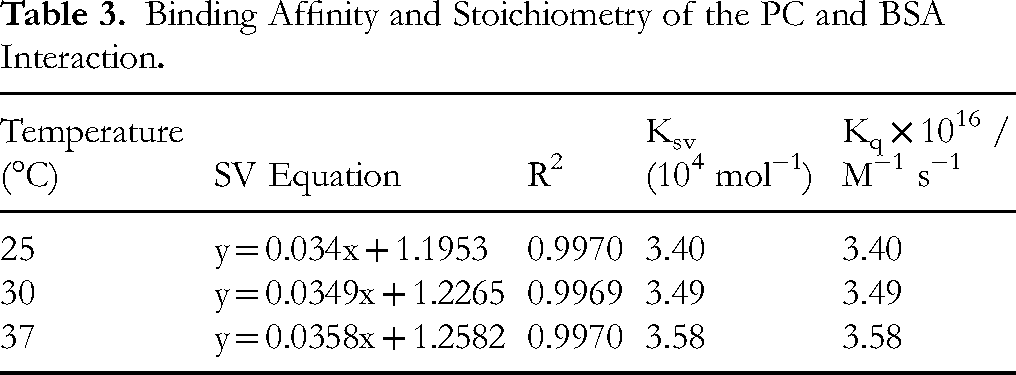
The observed increase in Ksv values with temperature confirms that dynamic quenching is the dominant mechanism. Additionally, the Kq values, which exceed the theoretical maximum for static quenching, further support this conclusion.12,13,17,18 The combined evidence from the redshift, temperature dependence of quenching, and high Kq values strongly suggests that the interaction between PC and BSA is primarily dynamic in nature, involving diffusion-controlled collisions between the molecules.12–18
Modified Stern-Volmer Plot
The binding interaction between PC and BSA was explored through a fluorescence quenching experiment, with the results analyzed using a modified Stern-Volmer plot.13–17 A plot of log [(Fo – F)/F] versus log [PC] yielded a linear relationship, indicating a single binding site for PC on the BSA molecule (Figure 4). The slope of the line reflects the number of binding sites (n), which was estimated to be around 1. The Y-intercept of the line represents the logarithm of the binding constant (Ka), allowing for the calculation of the dissociation constant (Kd) and the free energy change (ΔG) of the binding interaction.12,15

Modified Stern-Volmer Plot Illustrating the Interaction between PC and BSA [Graph of log ({Fo/F}/F) versus log [PC] for PC-BSA Interaction, Indicating a Single Binding Site; PC – Phycocyanin, BSA – Bovine Serum Albumin.].
The linear regression coefficient (R²) value confirmed the validity of the assumptions made in the derivation of the equations used to analyze the data. This indicates that the experimental data (Table 4) closely follows the theoretical model for a single-site binding interaction.12–15
Thermodynamic Parameters and Binding Characteristics of the PC–BSA Interaction.

The free energy change (ΔG) was calculated using the equation:
A negative value of ΔG indicates a spontaneous binding process, and the magnitude of ΔG provides information about the strength of the interaction. By quantifying the interaction's binding affinity and thermodynamic properties, this analysis provides valuable insights into the molecular forces driving the binding between PC and BSA.12–22
Antiglycation Studies
Browning
The inhibition of browning, a key indicator of glycation, was monitored by measuring absorbance at 420 nm (Figure 5). Over the course of 4 weeks, the absorbance of the BSA-glucose (BSA + G) control was 1.77 at week 4, while the BSA + G + PC system showed a significantly lower absorbance of 0.612, indicating a reduction in browning. This suggests that PC can effectively inhibit the formation of AGEs, a process associated with browning.

Weekly Analysis of Reduction in Browning [Graph of Absorbance versus Days; AG – Aminoguanidine, BSA – Bovine Serum Albumin, G – Glucose, PC – Phycocyanin].
When compared to aminoguanidine (AG), a well-known antiglycation agent, PC showed a reduction in browning, but AG exhibited more potent inhibition. On day 7, AG inhibited browning by 40.00%, whereas PC achieved only 31.02% inhibition. By day 28, AG reached 64.33% inhibition, while PC demonstrated 54.57% inhibition.
Protein-Carbonyl Content
The protein carbonyl content, a marker of oxidative damage from glycation, was measured throughout the study (Figure 6). At day 0, the carbonyl content of the BSA + G system was 0.243 μmol/mg. With the addition of PC, the carbonyl content increased slightly to 0.254 μmol/mg, while AG resulted in 0.237 μmol/mg, which reflects minor fluctuations due to the initiation of glycation reactions.

Weekly Analysis of Protein-Carbonyl Content [Graph of Protein-Carbonyl Content versus Days; AG – Aminoguanidine, BSA – Bovine Serum Albumin, G – Glucose, PC – Phycocyanin].
By day 28, the protein carbonyl content in the BSA + G control was 2.544 μmol/mg. Both PC and AG reduced protein carbonyl levels, with PC showing 2.207 μmol/mg and AG displaying 1.983 μmol/mg, indicating significant protection against oxidative damage. AG, however, exhibited more effective suppression, with 23.91% inhibition at day 28, compared to 17.45% for PC.
Fructosamine Content
Over time, the reduction in fructosamine content highlights superior antiglycation activity of AG compared to PC22,25 (Figure 7). On day 7, AG showed a percent reduction of 7.16%, surpassing 4.29% of PC. This difference widened as the experiment progressed, emphasizing a stronger inhibitory effect by AG.

Weekly Analysis of Fructosamine Content [Graph of Fructosamine Content versus Days; AG – Aminoguanidine, BSA – Bovine Serum Albumin, G – Glucose, PC – Phycocyanin].
By day 14, AG achieved a notable reduction of 21.76%, while PC managed only 12.45% (Figure 7). This trend continued on day 21, with AG reaching 23.70%, compared to PC, which is 17.17%. On day 28, AG maintained its superior performance with a reduction of 24.72%, whereas PC showed 16.39%.
Total AGEs Determination
The accumulation of AGEs was monitored by fluorescence spectroscopy over a 28-day period (Figure 8). The fluorescence intensity corresponding to AGEs increased steadily in the BSA + G system, indicating the progressive nature of glycation. The addition of PC significantly reduced AGE formation, achieving 67.81% inhibition by day 28.

Determination of Total Fluorescent AGEs by spectrofluorometry [Graph of Intensity versus Wavelength; AG – Aminoguanidine, BSA – Bovine Serum Albumin, G – Glucose, PC – Phycocyanin].
AG, on the other hand, was more effective, showing 87.32% inhibition under identical experimental conditions. These results suggest that while PC can inhibit AGE formation, AG remains a more potent antiglycation agent in this regard.
Individual Fluorescent AGEs
The individual AGEs formed in the BSA-glucose system were analyzed based on their fluorescence intensity (Figure 9). The most prominent AGE detected was argpyrimidine, followed by pentosidine, andvesperlysine, while crossline exhibited the lowest fluorescence intensity.

Determination of Individual Fluorescent AGEs by spectrofluorometry [Graph of Intensity versus Individual AGEs; AG – Aminoguanidine, BSA – Bovine Serum Albumin, G – Glucose, pc – Phycocyanin].
The presence of PC led to significant inhibition of the formation of these AGEs, with notable reductions in all the fluorescent AGEs measured. This effect highlights ability of PC to inhibit multiple stages of the glycation process, thereby reducing the formation of various glycation products.
Discussions
The dual investigations into the fluorescence quenching and antiglycation properties of PC were undertaken to elucidate its bioactivity at the molecular level. The primary aim was to understand whether ability of PC to interact with proteins, as demonstrated through its binding to BSA, contributes to its observed antiglycation effects.26,30 This approach integrates biophysical and biochemical perspectives, providing a holistic understanding of potential of PC as a therapeutic agent in mitigating protein glycation, a key process implicated in diabetes and aging-related complications.11,12,24
Fluorescence quenching analysis confirmed that PC interacts with BSA through a static quenching mechanism, forming a stable complex³³−³5. The double-log plot indicated a binding site number slightly above 2 at 25 °C, likely due to experimental variability or fitting deviations. However, previous studies suggest that PC primarily binds to a single high-affinity site, likely within subdomain IIA. Stern-Volmer analysis further supported this interaction, showing a strong binding affinity (Ka) and a single dominant binding site³³,³5. Thermodynamic analysis revealed a spontaneous interaction driven by hydrophobic forces and hydrogen bonding³4, suggesting that PC may influence BSA's structural and functional properties³7,³8.
The antiglycation study further validated the ability of PC to protect proteins from glycation-induced damage. Glycation is a non-enzymatic reaction between reducing sugars and protein amino groups, leading to the formation of AGEs.20,23–25 The suppression of browning intensity and AGE-specific fluorescence in the presence of PC demonstrated its efficacy in inhibiting glycation. For instance, PC reduced browning by up to 54.57%, protein carbonyl content by 17.45% and fructosamine content by 16.39% over 28 days (Figure 10). This inhibition was accompanied by reduced AGE fluorescence by 67.81%, highlighting its protective effects against early and late stages of glycation.11,24,27

Comparison of Percent Inhibition by PC and AG [Graph of Percent Inhibition versus Antiglycating Activity of PC and AG; AG – Aminoguanidine, PC – Phycocyanin].
The observed antiglycation effects are likely linked to interaction of PC with BSA.8,15,17 By forming a stable complex, PC may shield critical amino acid residues, such as lysine and arginine, from glycation-induced modifications.15,31–33 This interaction could also limit the conformational changes in BSA that make these residues more susceptible to glycation.13–17,31–36 Furthermore, inherent antioxidant properties of PC may contribute to the suppression of oxidative stress, a key driver of protein glycation.11,24,27
Interestingly, while aminoguanidine (AG), a well-established synthetic antiglycation agent, showed superior efficacy in several parameters, dual role of PC as both a protein-binding agent and an antiglycation inhibitor underscores its therapeutic potential.15,17 Unlike AG, which acts primarily through trapping dicarbonyl intermediates, 17 PC appears to exert a multifaceted protective effect, combining direct protein interaction with antioxidant activity.31–33,36 This makes PC a promising natural alternative or adjunct to existing antiglycation therapies.
The correlation between the quenching and antiglycation studies lies in the central role of the PC-BSA complex.34–36 The binding of PC to BSA not only alters the protein's fluorescence properties but also reduces the availability of reactive sites for glycation. 35 This dual mechanism highlights the potential of PC to modulate protein behavior and protect against glycation, especially in physiological conditions where proteins like albumin are abundant and play critical roles in maintaining homeostasis.31–33,37–39
In conclusion, the combined findings demonstrate that ability of PC to interact with proteins is closely tied to its antiglycation properties.32,33 By forming stable complexes with proteins, PC offers a protective mechanism against glycation, which, coupled with its antioxidant activity, positions it as a versatile bioactive compound with potential therapeutic applications.37–39 Future studies could explore its effects in vivo and expand the scope to include other glycation-prone proteins, thereby enhancing our understanding of its role in combating glycation-related pathologies.
Moreover, these findings have significant clinical implications, particularly in the prevention of glycation-associated complications such as diabetic nephropathy, retinopathy, and neurodegenerative disorders. Given that serum albumin plays a critical role in maintaining blood osmotic pressure and transporting biomolecules, the ability of PC to bind and stabilize albumin under hyperglycemic conditions suggests its potential as an adjunct therapy for diabetes management. To translate these findings into clinical applications, further studies involving human serum albumin (HSA) and in vivo models are necessary to validate its efficacy and safety. Additionally, evaluating its pharmacokinetics and bioavailability will be crucial for determining its suitability for therapeutic use.
Conclusion
The current study examined the interaction between PC and BSA, revealing a strong binding affinity and the spontaneous formation of a PC-BSA complex. The analysis of binding parameters, derived from the Stern-Volmer quenching model, indicated that the quenching mechanism followed a dynamic process, with the quenching efficiency increasing with temperature. Thermodynamic calculations suggested that the interaction between PC and BSA was driven predominantly by hydrophobic forces and occurred spontaneously, as evidenced by negative values for Gibbs free energy (ΔG).
Additionally, the ability of PC to inhibit the formation of early, intermediate, and advanced glycation end-products underscores its potential as a natural inhibitor of glycation. This inhibition suggests that PC may prevent the accumulation of harmful AGEs, which are associated with various degenerative diseases. Overall, the findings highlight multifaceted bioactivity of phycocyanin and indicate its potential as a therapeutic candidate for managing glycation-induced protein modifications, which are implicated in numerous pathologies, including diabetes and neurodegenerative disorders. Further studies could explore the detailed mechanisms by which PC interacts with glycation intermediates and its long-term efficacy in clinical applications.
Footnotes
Acknowledgements
The authors like to thank the funding of Researchers Supporting Project Number (RSPD2025R710) from King Saud University, Riyadh, Saudi Arabia. The authors are also grateful to Dr Vani B. for generously providing axenic culture of S. platensis PCC 7345.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Ethical Considerations
Ethical Approval is not applicable for this article.
Authors Contributions/CRediT
AA, and MSO conceptualized the study. AP collected the data and experimented. AP analyzed the data. AA supervised the work. All authors interpreted the analyzed data, wrote and reviewed the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Researchers Supporting Project Number (RSPD2025R710) from King Saud University, Riyadh, Saudi Arabia.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All the data are analyzed and presented in the article.