Abstract
Significance Statement
Because lung tissue is susceptible to ionizing radiation, excessive radiation doses can cause damaged cells to produce and release a variety of pro-inflammatory or pro-fibrotic cytokines, which can increase cell damage and death. Radiation-induced lung injuries are frequently experienced by patients receiving radiotherapy for thoracic malignancies, and these injuries can progress to radiation-induced lung damage (RILI). Finding safe and effective preventive compounds that protect individuals from harmful effects is essential. Thus, the goal of this study is to assess how Nattokinase helps to lessen the negative effects of high-dose γ-radiation exposure on the lungs. Nattokinase may help maintain pulmonary function by balancing the levels of fibrotic factors such as NOX-4, inhibiting TRX, LDH, and TRAP activation. And through decreasing the expression levels of the inflammatory mediators MCP-1, IL-6, NLRP-3, STAT-3 and SOCS-3
Introduction
Ionizing radiation is emitted into the atmosphere by a variety of sources, including the Sun and man-made ones including nuclear power plants, medical treatments, and military and industrial uses.
1
This may result in the organism's eventual death as well as serious acute impacts. Organ damage, immunosuppression, and endocrine disruption may occur even after exposure
There is a significant risk of lung illnesses from IR exposure. Radiation-induced lung injuries (RILI) are frequently experienced by patients undergoing radiotherapy for thoracic malignancies. 5 These injuries can progress to radiation-induced lung damage. Two complications of radiation-induced fibrosis of the lung and early radiation pneumonitis (RP) arise from the complex pathological process of RILI.6,7 These results limit the therapeutic dose of radiation applied to cancer patients. Due to lung tissue's sensitivity to ionizing radiation, excessive radiation doses can cause damaged cells to produce and release a variety of pro-inflammatory or pro-fibrotic cytokines, which increases cell damage and death. 8
As of right now There is no effective treatment for lung damage brought on by radiation. Finding effective and safe prophylactic drugs that insulate individuals from harmful effects is vital. According to Nuszkiewicz et al these substances have the ability to counteract ROS detrimental consequences in order to avoid diseases linked to ROS. 9 These substances function in various ways, mostly through modifying the antioxidant defense system. The efficacy and compatibility of plant-based products, including herbal medicine, in offering radioprotection have been well investigated. 10
The term “Bacillus-fermented food” refers to natto, a cheese-like dish produced from soybeans fermented with Bacillus subtilis. 11 Nattokinase is an alkaline protease having 275 amino acid residues and a molecular weight of roughly 28 kilodalton (kDa). 12 NK is a natural product with proven safety for human consumption following oral administration. 13 Strong fibrinolytic/antithrombotic properties, lipid-lowering and anti-atherosclerotic properties, antihypertensive properties, antiplatelet/anticoagulant properties, and neuroprotective properties are only a few of the numerous advantages of nattokinase. 14 It has 4-fold greater thrombus lysing activity than plasmin, which is the body's natural defense against clots. The very potent thrombus lysing activity of nattoki-nase comes from its ability to produce plasmin from plas-minogen, and increases the level of t-PA. Apart from this,studies have shown that nattokinase is stable in the intestines and has shown no side effects like allergies, abdominal painor bleeding disorders. 15 Nk intake leads to positive outcomes in the pathophysiology of Alzahimer disease in rat models. 13 In chronic inflammatory disease model, long-term NK supplementation ameliorated dextran sulphate sodium (DSS)-induced chronic colitis, inhibited colonic injury and prevented cell apoptosis in colonic tissue. 16 Different from the mechanism against acute inflammation, NK mitigated chronic colitis through reducing the expression of indoleamine 2,3-dioxygenase 1 (IDO-1), an important contributor of inflammatory bowel disease. In addition, it was reported that NK alleviated neuroinflammation in retinopathy murine model, by switching the proinflammatory microglia into an anti-inflammatory phenotype via nuclear factor erythroid 2-related factor 2/heme oxygenase-1 (Nrf2/HO-1) pathway. 17 In accordance, another study also found that NK attenuated bisphenol A or gamma irradiation-induced neuroinflammatory by activation of Nrf2. 18 Remarkably, disturbances in the proper HO-1 level are associated with the pathogenesis of age-dependent disorders. In light of the crosstalk between inflammation and aging, the multiple anti-inflammatory effects of NK may be partially responsible for its benefits on aging-linked diseases.
Thus the goal of this study is to evaluate how nattokinase mitigates the negative consequences of lung exposure to high doses of gamma radiation (γ-radiation). Specifically, this research aims to: 1- Confirm the effects of nattokinase on oxidative stress and the inflammatory response in lung tissues following γ-radiation exposure. 2- Investigate how nattokinase may help maintain pulmonary function by balancing the levels of fibrotic factors such as NOX-4, while inhibiting TRX (Thioredoxin), LDH (Lactate Dehydrogenase), and TRAP (Total Radical-Trapping Antioxidant Potential) activation. 3-Provide valuable insights into the effects of nattokinase on the gene expression levels of key inflammatory markers, including MCP-1, IL-6, NLRP3, STAT3, and SOCS3.
By achieving these objectives, the study aims to elucidate the therapeutic potential of nattokinase in protecting lung health following radiation exposure and its role in modulating oxidative stress, inflammation, and fibrotic responses.
Materials and Methods
Materials
Nattokinase was purchased from ChemicalBook INC, 2024 (CAS No. #133876-92-3, 418 BROADWAY, STE N AL, USA). LDH rat ELISA kit (Cat# EKN46642-96T, Biomatik USA, LLC). NOX4 ELISA kit (Cat# EKU06295-96T, Biomatik USA, LLC). TRX and TRAP rat ELISA kit (Cat# CSB-E09729r, CSB-E08491r, CUSABIO, CUBIO Innovation Center), Houston, TX 77054, USA.
Radiation Facility
Canadian γ-cell-40 (60Co source) was used to give rats a single-dose, 5 gray (Gy) gamma irradiation. The National Centre for Radiation Research and Technology NCRRT (Cairo, Egypt) used a Canadian to irradiate the rat's entire body. The experiment was carried out with a dosage rate of 0.401 Gy/min.
Animals
Male Swiss albino rats, weighing between 120 and 150 g, were utilized in the study. They were obtained from the National Centre for Radiation Research and Technology's breeding section. After acclimatization, the rats were housed on a commercial pellet diet with an endless supply of water for a week.
Ethics Approval Statement
The National Institute of Health's (NIH No 85:23, revised 1996) guidelines for animal care and the guidelines established by the National Center for Radiation Research and Technology's (NCRRT) Ethics Committee, Atomic Energy Authority, Cairo, Egypt, governed the use and care of laboratory rats in experimental settings. This research protocol was organized and carried out in compliance with the council for international organizations of medical sciences (CIOMS) and International Council for Laboratory Animal Science (ICLAS) (international guiding principles for biomedical research involving animals 2012), and the ARRIVE guidelines 2.0. It was approved by the committee based on the 3Rs (replace, educe, and refine) principles for animal experimentation (Approval No F/59A/23). 19
Design of the ARRIVE Experiment
40 rats were split up into 4 equal groups. (1) Control group (C): Normal rats were did not receive any therapy and didn’t exposed to radiation. (2) Rats exposed to a single-dose, 5Gy gamma irradiation were in the radiation group (R). (3) The rats in the Nattokinase group (N) received an oral dose of 720 Fibrinolytic Units (FU)/kg body weight /day of the enzyme for ten days. 18 (4) Radiation + Nattokinase group (NR): rats first exposed to 5Gy gamma irradiation and then received oral dose of nattokinase treatment
Gathering Tissue Samples for Examination
Rats were decapitated and subsequently slaughtered at the conclusion of the experiment while under a mild ketamine anesthesia (30 mg/kg, intraperitonally i.p.). 20 For the purpose of separating sera and performing biochemical analyses, blood samples were extracted and centrifuged via cardiac perforation. The tissues from the lungs were gathered and ready for histological analysis. After homogenizing the remaining tissue samples at a ratio of 1:10 times weight/volume (w/v) in phosphate buffer (0.1 M, pH 7.4, ice cold), they were kept for additional biochemical analysis.
Biochemical Assay
Yoshioka et al reported that the oxidative status in serum was evaluated using the identification of malondialdehyde (MDA), a marker for lipid peroxidation. Thiobarbituric acid reactive chemicals (TBARS), which have a wavelength of 532 nanometers (nm), are created by the thiobarbituric acid test. 21 Estimating catalase (CAT) activity was done using the methods outlined by Aebi. 22 This method's basic idea is to calculate the rate at which hydrogen peroxide (H2O2) decomposes at 240 nm. The expressed results are in U/mg protein. The total super-oxide dismutase activity (SOD, enzyme commission number EC 1.15.1.11) for cuppor (Cu), zinc (Zn), and manganese (Mn) was measured using the Sun et al techniques. 23 The assay's foundation was blocking the xanthine-xanthine oxidase system's ability to reduce Nitro Blue Tetrazolium (NBT) as a super-oxide producer. U/mg protein is the expression of SOD activity. Total protein content was calculated using the Lowry et al technique. 24 The assay's foundation was blocking the xanthine-xanthine oxidase system's ability to reduce NBT as a super-oxide producer. U/mg protein is the expression of SOD activity. We calculated the total protein content using the procedure outlined by Lowry et al 24
Enzyme-Linked Immunosorbent Assay (ELISA)
Using an ELISA Kit for rats, the concentrations of lactate dehydrogenase (LDH), Thioredoxin 1 (TRX1), Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX4), and the quantity of tartrate resistant acid phosphatase 5 were determined by following the manufacturer's instructions. Using an ELISA reader (Bio-Tek Instruments Inc., Winooski, VT, USA), the optical density (OD) value was measured at 450 nm.
Quantitative Real-Time PCR
RNA isolation and reverse transcription: RNA was extracted from lung tissue using the RNeasy plus mini kit (Qiagen, Venlo, The Netherlands), according to the manufacturer's instructions. Genomic DNA was eliminated by a DNase-on-column treatment supplied with the kit. The RNA concentration was determine spectrophotometrically at 260 nm using the Nano Drop ND-1000 spectrophotometer (Thermo Fisher scientific, Waltham, USA) and RNA purity was checked by means of the absorbance ratio at 260/280 nm. RNA integrity was assessed by electrophoresis on 2% agarose gels. RNA (1 μg) were used in the subsequent cDNA synthesis reaction, which was performed using the Reverse Transcription System (Promega, Leiden, The Netherlands). Total RNA was incubated at 70 °C for 10 min to prevent secondary structures. The RNA was supplemented with MgCl2 (25 mM), RTase buffer (10X), dNTP mixture (10 mM), oligod (t) primers, RNase inhibitor (20 U) and AMV reverse transcriptase (20 U/μl). This mixture was incubated at 42 °C for 1 h.
Quantitative real time PCR: qRT-PCR was performed in an optical 96-well plate with an ABI PRISM 7500 fast sequence detection system (Applied Biosystems, Carlsbad, California) and universal cycling conditions of 40 cycles of 15 s at 95 °C and 60 s at 60 °C after an initial denaturation step at 95 °C for 10 min. Each 10 μl reaction contained 5 μl SYBR Green Master Mix (Applied Biosystems), 0.3 μl gene-specific forward and reverses primers (10 μM), 2.5 μl cDNA and 1.9 μl nuclease-free water. The sequences of PCR primer pairs used for each gene are shown in Table 1. Data were analysed with the ABI Prism sequence detection system software and quantified using the v1·7 Sequence Detection Software from PE Biosystems (Foster City, CA). Relative expression of studied genes was calculated using the comparative threshold cycle method. All values were normalized to the endogenous control β-actin. 25
Primers Used for QRT-PCR.

Histopathological Examination
Rats in various groups were autopsied, and samples from their lung tissues were preserved for 24 h at 10% formalin saline. After that, they were cleaned with tap water and serially diluted (methyl, ethyl, and 100% ethyl) alcohol to dehydrate them. Following that, The samples underwent a 24-h paraffin immersion at 56oC in a hot air oven after being washed in xylene. Tissue blocks wrapped in paraffin wax were created and sectioned at a thickness of 4 mm using a slide microtome. Finally, the tissue sections were collected on glass slides, deparaffinized, and stained with hematoxylin and eosin (H&E) stain for routine examination using a light electric microscope, following the protocol described by Bancroft et al 26
Grading of Histopathological Alterations
According to Arsad et al, 27 histopathological changes in the lungs were evaluated as follows: (0) meant there were no changes, (+), (++), and (+++) meant there were mild, moderate, and severe changes.
Statistical Analysis
ANOVA (one-way analysis of variance) was used for statistics, and Tukey-Kramer post hoc multiple comparisons between treatment means were then performed. Statistical analyses were performed using Prism, version 6 (GraphPad Software, La Jolla, CA). A significance level of P ˂ .05 will be applied.
Results
Effect of Nattokinase on MDA, SOD, and CAT in Lung Tissue of Irradiated Rats
The results showed that the N group for MDA in the lung tissue drastically decreased, but the SOD and CAT activities were substantially higher when compared to the control (C) group. Furthermore, there was an oxidative stress brought on by the γ-radiation exposure, as evidenced by a considerable increase (P < .05) in MDA and an important reduction (P < .05) in SOD and CAT activity in the R group as opposed to the (C) group. As seen in Figure (1), the IR impact was greatly enhanced by concurrent nattokinase treatment (NR), which demonstrated a considerable increase (P < .05) in SOD and Catalase activities and a significant drop (P < .05) in MDA when compared to the R groups, respectively.

Effect of Nattokinase on MDA, SOD, and Catalase Induced by γ-Radiation in Lung Tissue. Mean ± Sstanderd Error (SEM) (n = 10) is Represented by Each Value. Columns Marked with ‘a’, ‘b’, ‘c’, and ‘d’ are Noteworthy from C, N, R and NR, Respectively, at (P < .05). Normal Rats are Represented by C, Nattokinase-Treated Rats by N, and 5Gy-Exposed Rats by R.
Effect of Nattokinase on LDH in Lung Tissue of Irradiated Rats
Figure (2) represents the effect of the treatment of nattokinase on LDH in lung tissue in that the LDH concentration was significantly diminished regarding the C group. Otherwise, the exposure to high-dose radiation elevates LDH concentration (P < .05) compared to the C group. However, the treatment of natokinase (N) after γ-radiation exhibited a pronounced decrease (P < .05) in the NR group regarding the R group.

Nattokinase Impact on LDH in Lung Tissue of Different Animal Groups. Mean ± SEM (n = 10) is Represented by Each Value. Columns Marked with ‘a’, ‘b’, ‘c’, and ‘d’ are Significant from C, N, R and NR, respectively, at (P < .05). Normal Rats are Represented by C, Nattokinase-Treated Rats by N, and 5Gy-Exposed Rats by R.
Effect of Nattokinase on TRX in Lung Tissue of Irradiated Rats
Rats given NK alone (N) showed no appreciable variation in the quantity of TRX in lung tissue as compared to the control group (C). However, following the IR exposure, an enormous increase was noted in the (R) group. In the meantime, the NK therapy greatly enhanced the IR impact in NR rats, who showed a considerable (P < .05) decline in TRX when compared to the R group Figure (3).

Nattokinase Influence on TRX in Lung Tissue of Tested Rats. Each Number is the Mean ± SEM (n = 10). The Significance of Columns Labeled ‘a’, ‘b’, ‘c’, and ‘d’ is Determined by C, N, R and NR, Respectively, at (P < .05). Rats Treated with Nattokinase (N), Rats Subjected to 5Gy (R), and Rats Representing Normalcy (C) are Displayed.
Effect of Nattokinase on NOX4 in Lung Tissue of Irradiated Rats
Figure (4) illustrates the impact of NK as a treatment on NADPH oxidase 4 (NOX4) in lung tissue.The statistics showed that there was no discernible difference between the N and C groups. Concurrently, the irradiation led to a tremendous (P < .05) increase in NOX4 in reaction to the C group. The substantial reduction in the NR group (P < .05) after being exposed to the R group indicated the presence of the N effect.

Effect of Nattokinase on NOX4 in Lung Tissue of all Rat Groups. Mean ± SEM is Represented by Each Value. (10 in total). The Columns that have ‘a̴ are Significantly Different from C, ‘b̴ Significant from Nattokinase Group, ‘c̴ Significant from Radiated Group, and ‘d̴ Significant from NR’ Indicated at (P < .05). Normal Rats are Represented by C, Nattokinase-Treated Rats by N, and 5Gy-Exposed Rats by R. NR Rats Exposed to Gamma Radiation and then Treated by Nattokinase.
Effect of Nattokinase on TRAP in Lung Tissue of Irradiated Rats
Figure 5 shows the tartrate-resistant Acid Phosphatase 5 (TRAP/ acid phosphatase 5 (ACP5)) level that was determined in the lung tissue. In comparison to the C group, the statistics showed an enormous decrease in the N group. As a result of radiation, TRAP in the R group was considerably higher (P < .05) than in the C group. Interestingly, nattokinase treatment resulted in an impressive reduction in TRAP following gamma radiation exposure in the NR group compared to either the N or R groups.

Influence of Nattokinase on TRAP in Lung Tissue. Mean ± SEM (n = 10) is Represented by Each Value. The Columns that have ‘a̴ Significant from C, ‘b̴ Significant from N, ‘c̴ Significant from R, and ‘d̴ Significant from NR’ are Indicated at (P < .05). Normal Rats are Represented by C, Nattokinase-Treated Rats by N, and 5Gy-Exposed Rats by R. NR Rats Exposed to Gamma Radiation and then Treated by Nattokinase.
Influence of Nattokinase on Inflammatory Responses in γ -Irradiated Rats
In Lung
The data represented in Figure (6) showed that, there were no significant changes observed in the gene expression of

Influence of Nattokinase on the Gene Expression of MCP-1, IL-6, NLRP-3, STAT-3 and SOCS-3 in Lung Tissue of γ-Irradiated Rats. Each Value Represents Mean ± SEM (n = 6). The Columns that have ‘a̴ Significant from C, ‘b̴ Significant from N, ‘c̴ Significant from R, and ‘d̴ Significant from NR’ are Indicated at (P < .05). Normal Rats are Represented by C, Nattokinase-Treated Rats by N, and 5Gy-Exposed Rats by R. NR Rats Exposed to Gamma Radiation and then Treated by Nattokinase.
Histopathological Findings of Lung Tissue
Figure 7 presents the histopathological examination of lung tissue from different animal groups. The C-group displayed healthy alveoli, bronchi, and bronchioles along with normal lung tissue (Figure 7a), with a lesion score 0. On the other hand, the lung tissue of the R group showed severe broncho pneumonia, with significant inflammation in the bronchi and interstitial tissue (Figure 7b) and a severe lesion score +++. The lung tissue of the N group (Figure 7c) displayed no histological changes, with a lesion score of 0. Finally, the lung tissue of the NR group showed a marked regression in the inflammatory reaction; it was still mild (Figure 7d) with a mild lesion score of +
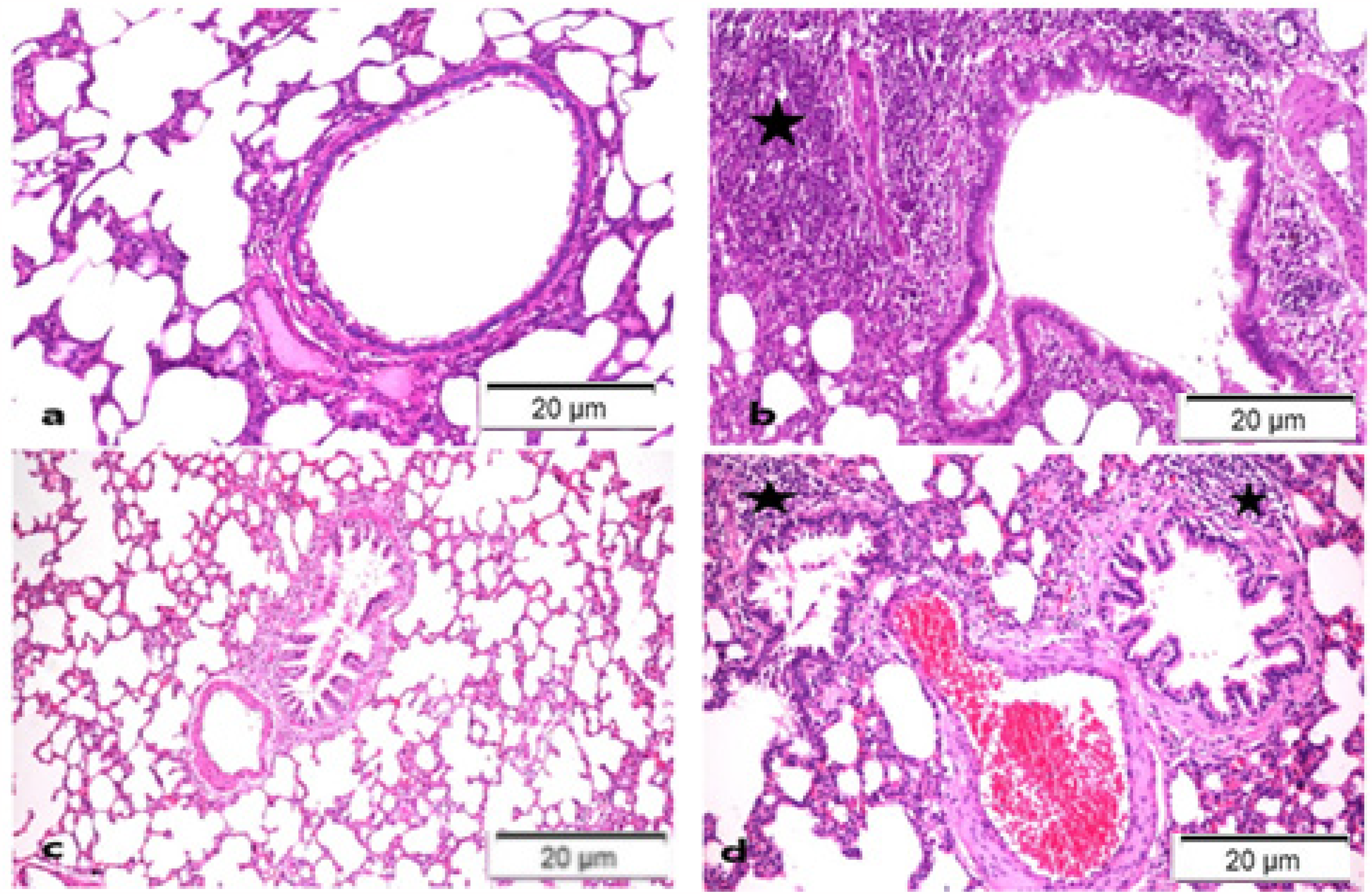
H&E X200-Stained Photomicrographs of the Lungs from Several Experimental Groups. Demonstrate the Following: a. Control Negative Normal Group (0), b. R group with Severe Bronchopneumonia (*) (+++), c. Nattokinase Group with Apparently Healthy Tissue (0), d. R + N Group Mild Bronchopneumonia (+).
Discussion
It is hard to completely avoid being exposed to ionizing radiation in the modern environment. However, there are significant worries over the possibility of contracting cancer or other non-cancerous diseases as a result of this exposure, which can happen as a result of unintentional incidents or medical operations. 28 Research has shown that pro-oxidant enzymes and inflammatory mediators may rise over time in response to high doses of ionizing radiation received during radiotherapy. This results in the constant generation of nitric oxide (NO) and ROS. 29 These substances are linked to extensive DNA damage and mutation, which can cause genomic instability and an elevated risk of developing cancer. 30
According to Rahi et al RILI can result in inflammation, fluid accumulation, breathing difficulties, reduced lung function, and respiratory failure. 6 The present investigation documented the detrimental effects on the pulmonary tissues of rats given rather high doses of whole-body gamma irradiation (5 Gy). The formation of a lipid peroxidation product (MDA) increased while the activity of two important endogenous antioxidant enzymes, SOD and CAT, significantly decreased. These outcomes concur with those of Ahmadvet al., who shown that radiation increases MDA levels while inhibiting SOD and GPx (glutathione peroxidase) activity. 31 Research has shown that increased oxidative stress in irradiated tissues is how IR causes adverse effects. 32 Because cells include 80% water, IR promotes radiolysis of water molecules. This increases the creation of ROS, which combine with macromolecules like proteins, lipids, and nucleic acids to cause cell malfunction and death. 9
Furthermore, Bezerra et al provided evidence that oxidative stress, which results from an imbalance between the generation of oxidants and antioxidant defenses, is a vital part of the altered alveolar environment in lung fibrosis. 33 Furthermore, IR causes irradiated cells’ intracellular signaling pathways to become active, which causes endogenous ROS to accumulate. 34 Radiation pneumonitis in humans and radiation-induced lung damage in rats have both been linked to elevated oxidative stress. 7 On the other hand, TRX, glutathione, SOD, CAT, and peroxiredoxin (Prx) systems are among the network of antioxidant systems that help eliminate ROS. 35
Disulfide oxidoreductase activity is exhibited by the peptide enzyme TRX-1 at a molecular weight of around 12 kDa. 36 According to Oberacker et al, TRX-1 is essential for several cellular processes, such as redox homeostasis, proliferation, DNA synthesis, transcription factor modulation, and control of cell death. 37 TRX-1's effects, however, can differ according on the nature and severity of the ailment. TRX-1, for instance, can defend against damaging insults in ischemia-reperfusion injury when apoptosis is involved. 38 However, by scavenging ROS involved in the mechanism, elevated TRX-1 in many human malignancies can reduce the efficacy of radiation therapy or chemotherapy. 39 In the present work, the level of TRX-1 was revealed to have considerably increased in the R group. TRX-1 is known to scavenge ROS directly, which helps to protect the tissue from oxidative stress. This finding is supported by Wang et al's research from 2022, which also demonstrated that TRX-1 could directly scavenge ROS, and this may be why the level of TRX-1 decreases after treatment with NK. 40
As exposure to IR is inevitably accompanied by production of high levels of reactive oxygen species (ROS), so the management of this action could provide a method to avoid the deleterious effects on normal tissues upon radiation exposure. Therefore, radiation countermeasure agents should be used to reduce the hazardous effects of IR. Radioprotectors are various agents that act via different mechanisms involving: scavenging of free radicals and ROS; improvement of the DNA repair process; synchronizing of cells; enhancing antioxidant and redox-sensitive genes; modulating cytokines and growth factors; inhibiting apoptosis; repurposing of drugs; and tissue regeneration. Scavenging of free radicals is the most common mechanism of radioprotection, whereas the alteration of growth factors, cytokines and redox genes appears to be an effective strategy. 41 Our previous work stated that fucoxanthine have a noticeable radioprotective action mediated through its regulatory effect on the apelin-13/APJ/NF-κB signaling pathway which attributed to its antioxidant and anti-inflammatory activity that was reflected in different physiological processes. It could be recommended to use FX in cases of radiation exposure to protect normal tissues. 42 In addition Azab et al, study the radioprotective effect of Withania somnifera (Ashwagandha) and found that Ashwagandha root extract counteract acute and chronic impact of γ-radiation on liver and spleen of rats. It could exerts radio-protective influences because of its antioxidants and anti-inflammatory capabilities. 43 What is more, Sirtuin (ultra pure trans-Resveratrol; t-Res) had a radio-protective effect via its antioxidant and anti-inflammatory effects on liver and kidney of rats subjected to a harmful whole body single or a fractionated radiation at a dose of 6 Gy. This is attributed to its ability to down-regulate the hypoxia factor (HIF-1α) and increase Nrf-2 gene expression, which is associated with a decline in lipid peroxidation (MDA) and enhancement in antioxidant enzyme (CAT), which supports its ability to curb inflammatory responses in vulnerable tissues. 44 Moreover, Pomegranate peel extract sensitizes hepatocellular carcinoma cells to ionizing radiation, induces apoptosis and inhibits MAPK, JAK/STAT3, β-Catenin/NOTCH and SOCS3 signaling. 30
Nattokinase attenuates bisphenol A or gamma irradiation-mediated hepatic and neural toxicity by activation of Nrf2 and suppression of inflammatory mediators in rats. NK improved liver functions, impaired oxidative status, the cholinergic deficits, and minified the misfolded proteins aggregates. Furthermore, NK alleviated the neuroinflammation via modulating NF-κB/Nrf2/HO-1 pathway and glial cell activation in addition to their antiapoptotic effect. 18
Furthermore, as this work shows, NK improves animal lipid metabolism by inhibiting low-density lipoprotein.45,46 In accordance with Fu et al, who discovered that dietary fermented NK supplementation improved lion-head goslings’ growth performance, serum biochemical parameters, and antioxidant capacity, our results showed that NK increases antioxidant SOD and CAT while decreasing lipid peroxidation through decreasing MDA levels. 47 This could be because the fermented meal contains beneficial metabolites that are produced after fermentation, like vitamins and short peptides. According to Fu et al, these compounds aid in balancing the body's production of pro- and antioxidant-oxidants. 48
A cytosolic enzyme with high activity is LDH. 48 Cell death and damage are tracked by the presence of LDH outside of cells.49 Furthermore, it is frequently regarded as a traditional inflammatory marker. 50 In the present investigation, exposure to IR resulted in an increase in LDH activity. According to Judge et al, the expression of lactate dehydrogenase-A (LDHA), which stimulates myofibroblast development, causes an abnormal rise in lactate generation in lung tissue from both humans and animals when exposed to IR. 51
Radiation also causes lung epithelial cells to undergo apoptosis, which results in the capillary luminal dilatation and desquamation of alveolar walls, which increase vascular permeability and result in interstitial edema. 52 According to Park et al, increased alveolar protein levels signify direct radiation damage by increasing vascular permeability. 52 In lung parenchymal cells, elevated LDH activites are the cause of this damage. 53 Furthermore, it was shown by Judge et al that IR causes dose-dependent LDHA induction, lactate generation, and extracellular acidification in primary human lung fibroblasts. 51 However, as previously noted in other research, the administration of NK reduces LDH activity because it functions as a naturally occurring anti-inflammatory chemical.12,54
Lung damage brought on by radiation frequently does not heal completely and can result in dysregulated processes that compromise the organ. The main generator of ROS during various disease processes is the NADPH oxidase family. Endothelial cells have four different isoforms of NOX: NOX1, NOX2, NOX4, and NOX 5. 55 Compared to other forms, research conducted in 2017 by Chen et al revealed that Human lung microvascular and pulmonary artery endothelial cells express NOX4 primarily. This study also revealed an increase in NOX4 expression after exposure to 5Gy ionizing radiation, which is consistent with the findings of Najafi et al, who showed that high doses of ionizing radiation can result in a chronic upregulation of NOX, including NOX2 and NOX4. 29 Furthermore, Serrander et al, reported that treatment with NK substantially decreased NOX4 expression. NOX4 is known to be constitutively active with the most common measurable ROS, hydrogen peroxide (H2O2). 56
Fibroblasts, myofibroblasts, and inflammatory cells are accumulated in Radiation-Induced Lung Fibrosis (RILF). Hypoxia in the lung tissue causes ongoing macrophage-associated production of reactive oxygen/nitrogen species (ROS/RNS) after either single or fractionated irradiation, damaging the lung tissue.57,58 Furthermore, it was suggested that the pro-fibrotic “alternatively activated” state of macrophages is linked to lung fibrosis. 59 One of the metalloprotein enzymes in the family is TRAP/ACP5. 60 According to Tanner et al, activated alveolar macrophages exhibit elevated expression of TRAP5,61 which is a potentially useful biomarker for chronic inflammation. 62 The present investigation revealed a considerable rise in TRAP5a levels subsequent to irradiation, which is in line with other findings that have demonstrated the activation of alveolar macrophages in inflammatory settings, such as high-dose irradiation, hence elevating TRAP5a levels. 57 But NK injection dramatically decreased TRAP5a activity, presumably because it inhibits NOX2 signaling in macrophages, which is one of NK's anti-inflammatory and anti-oxidative stress properties. 63
The results presented in Figure 6 provide valuable insights into the effects of nattokinase on gene expression related to inflammation and immune response in lung tissues following gamma radiation exposure. Notably, the data indicate that the treatment with nattokinase did not lead to significant changes in the expression levels of MCP-1, IL-6, NLRP3, STAT3, and SOCS3 in the control group, suggesting that nattokinase, at the administered dosage, does not exert pro-inflammatory effects under normal conditions. However, in groups exposed to 5 Gy of gamma radiation, there was a significant elevation (p < .05) in the expression levels of the aforementioned genes. This increase highlights the inflammatory response triggered by radiation, which is consistent with prior studies that demonstrate the activation of pro-inflammatory pathways following such exposure. Radiation-induced lung injury (RILI) is characterized by heightened levels of inflammatory cytokines and mediators, such as MCP-1 and IL-6, which play critical roles in recruiting immune cells and perpetuating inflammation. 64
MCP-1 (monocyte chemoattractant protein-1) is significantly upregulated following radiation exposure and plays a pivotal role in the recruitment of monocytes and macrophages to the site of injury in lung tissues. This recruitment contributes to the inflammatory response observed in RILI. Elevated levels of MCP-1 have been linked to increased inflammation and the development of lung injury post-radiation. 65 While IL-6 is a key cytokine involved in inflammatory processes and tissue repair. 66 IL-6 is a key pro-inflammatory cytokine that induces the STAT3 pathway and is significantly elevated in response to radiation. It plays a critical role in mediating inflammation and is associated with the activation of fibrotic pathways in lung tissues. Elevated IL-6 levels correlate with the severity of RILI. 64 Suppressor of Cytokine Signaling 3 (SOCS3) negatively regulates the IL-6/STAT3 signaling pathway. In the setting of RILI, dysfunctional SOCS3 expression can lead to uncontrolled STAT3 activation, promoting persistent inflammation and fibrosis. Proper regulation of SOCS3 is therefore crucial in preventing chronic lung injury following radiation exposure. 67 NLRP3 (NOD-Like Receptor Pyrin 3) is a component of the inflammasome that can be activated in response to cellular stress induced by radiation. Radiation exposure can lead to the formation of reactive oxygen species (ROS), which further activate NLRP3, resulting in the release of pro-inflammatory cytokines, such as IL-1β. The upregulation of NLRP3, a component of the inflammasome, indicates increased activation of innate immune responses, correlating with findings that link NLRP3 activation to radiation-induced lung damage. 68 Simultaneously, the elevation of STAT3, a transcription factor associated with both inflammation and fibrosis, reflects the potential for long-term tissue remodeling and damage following radiation exposure. 69
The striking finding of nattokinase treatment is its ability to significantly reduce (p < .05) the elevated gene expression levels of MCP-1, IL-6, NLRP3, STAT3, and SOCS3 in the irradiated groups (NR groups) compared to the respective radiation-only groups (R groups). This indicates that nattokinase may possess protective effects against RILI. The fibrinolytic and anti-inflammatory properties of nattokinase can contribute to reducing tissue inflammation and injury post-radiation. Nattokinase has been shown to enhance fibrinolytic activity and potentially improve blood flow and healing processes, which may mitigate the inflammatory cascade initiated by radiation. 67
Furthermore, the significant downregulation of SOCS3, a negative regulator of IL-6 signaling, in the NR group suggests that nattokinase might help rebalance the inflammatory response, thereby preventing the chronic inflammation that characterizes RILI. This aligns with emerging data regarding the use of natural compounds in modulating inflammatory responses and suggests that nattokinase's mechanisms could be further explored as a therapeutic approach to alleviate RILI.
The biochemical data were validated by the histological exams, which showed significant bronchopneumonia in the R group together with a noticeable inflammatory response in the bronchi and interstitial tissue (Figure 6a). These findings are consistent with a prior study of Jiang et al, that showed inflammation following early exposure to ionizing radiation (IR), which was followed by the infiltration of neutrophils or macrophages into lung tissue and the activation of inflammatory cytokines. 70 Furthermore, pulmonary epithelial damage was reported by Groves et al in a number of radiation lung injury models. 71 Early exposure to external radiation causes alterations in the epithelial lung components, especially the existence of Type II pneumocytes, vasodilatation, and direct endothelial damage. 31 On the other hand, administration of NK showed an improvement in lung tissue damage by reducing inflammatory reactions, which could be due to the anti-inflammatory impact of NK (Figure 7).
This study presents some significant findings regarding the role of nattokinase in mitigating lung tissue damage induced by gamma irradiation, but it's clear that there are several limitations that need to be acknowledged.
Suggestions for Future Research
By addressing these limitations and exploring these suggestions, future research can build on our findings and contribute to a deeper understanding of nattokinase's potential as a radioprotector.
Conclusion
In conclusion, this study illustrates the protective effects of nattokinase against the harmful consequences of lung exposure to high doses of gamma radiation (γ-radiation). The findings confirm that nattokinase positively influences oxidative stress and the inflammatory response, significantly aiding in the maintenance of pulmonary function. By balancing the levels of key fibrotic factors such as NOX-4 and inhibiting the activation of TRX, LDH, and TRAP, nattokinase demonstrates a multifaceted approach to mitigating radiation-induced lung injury. Moreover, the ability of nattokinase to reduce the expression levels of inflammatory markers MCP-1, IL-6, NLRP3, STAT3, and SOCS3 highlights its role in attenuating the inflammatory response triggered by γ-radiation exposure. These results suggest that nattokinase may serve as a promising therapeutic agent for preventing or alleviating radiation-induced lung injury.
Overall, this study provides valuable insights into the molecular mechanisms through which nattokinase acts, paving the way for future research aimed at exploring its clinical applications in radioprotection and the management of pulmonary health in patients undergoing radiation therapy. As a natural substance, nattokinase is often viewed as a safer alternative to synthetic drugs, which may carry a higher risk of side effects.
Footnotes
Acknowledgements
Thanks to all the authors.
Ethical Considerations
All experiments complied with ARRIVE standards. The Ethical Committee (REC) of the NCRRT, Atomic Energy Authority, Cairo, Egypt, approved the study (Approval No: F/59A/23).
Author Contributions/CRediT
MKA was the study’s designer. For the practical work, N.M.E., F.M.S., and R.E.M. were responsible. The data analysis and paper draught were completed by N.M.E. A.A.H. rewrote and thoroughly examined the work. The manuscript was approved by all authors Every writer has read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research, writing, and/or publication were all done without financial assistance.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All information provided in this manuscript is contained inside the manuscript.
