Abstract
Objective
Cognitive dysfunction is common in neurodegenerative diseases. Sea buckthorn oil (SBO) has anti-inflammatory and antioxidant properties. So, we want to investigate the effect of SBO on reducing scopolamine-induced oxidative stress, inflammation, and cholinergic changes in PC12 cells, and its impact on ameliorating cognitive dysfunction in APP/SP1 mice. The purpose of this study is to prove that SBO has a positive effect on cognitive dysfunction.
Method
In vitro, the effect of SBO on scopolamine-induced PC12 cells damage is evaluated using the 3- (4,5-dimethylthiazol-2-yl) −2,5-diphenyltetrazolium bromide (MTT) method. Levels of various biochemical markers were also examined by enzyme-linked immunosorbent assay (ELISA), including acetylcholinesterase (AChE), and acetylcholine (ACh). As well as examining the inflammatory factors: interleukin-1β (IL-1β), tumour necrosis factor-α (TNF-α), and interleukin-6 (IL-6). The oxidative stress of cell was detected using reactive oxygen species (ROS) staining. In addition, we detected catalase (CAT), glutathione peroxidase (GSH-Px), and superoxide dismutase (SOD), which are indicators of oxidative stress (OS) in vitro experiments. Moreover, the detection of Nrf2/HO-1 molecules and inflammatory cytokines released by reverse transcription-polymerase chain reaction (RT-PCR) and western blot. In vivo, the memory behavior is evaluated using the Morris water maze(MWM) and step-down test.
Result
SBO showed a significant reduction of AChE activities and increased the viability of scopolamine-treated PC12 cells. Meanwhile, SBO increases the level of GSH-Px, SOD, CAT activities, and ACh, and decreases the level of TNF-α, IL-1β, and IL-6. Moreover, increasing the expression of the Nrf2/HO-1 pathway. SBO has shortened the swimming time to the platform, and increased the exploration time of the target quadrant and the number of times it crosses the platform. Meanwhile, it reduced the number of errors in the step-down test.
Conclusion
SBO has a protective effect against scopolamine-induced PC12 cell injury and a favorable impact on improving cognitive impairment in APP/PS1 mice. Therefore, SBO may have clinical applications in the treatment of cognitive impairment.
Introduction
Some studies have shown that the main characteristic of various neurodegenerative diseases is cognitive impairment. 1 The histological feature is amyloid β peptide (Aβ) deposition outside the cell in the form of neuroinflammatory plaques. Within neurons, neurogenic fiber tangle (NFT) forms aggregates of hyperphosphorylated Tau proteins. 2 The early clinical syndrome of neurodegenerative diseases is mild cognitive impairment (MIC) with fewer functional deficits. Further progression of MIC leads to aphasia, visuospatial deficits, executive dysfunction, and dyscalculia. The conversion rate of MIC to neurodegenerative diseases is estimated at 15% per year, reaching almost 45% within 5 years. 3 Globally, about 50 million people have cognitive dysfunction, and this number will go up to 152 million by 2050. 4 Global patients spend trillions of dollars annually on diseases, which puts a financial burden on themselves and their families. Some research demonstrates that this symptom is related to a few factors, such as OS, neuroinflammation, and cholinergic abnormalities. 5
Intervention strategies include promoting neurogenesis and protecting neurons and synapses. 6 The four FDA-approved drugs (ie, donepezil, carboplatin, galantamine, and memantine) can temporarily relieve symptoms of cognitive impairment. However, they cannot stop the long-term progression of the disease and have certain side effects. 7 Natural compounds (including phenolic acids and flavonoids) improve disease due to their antioxidant, anti-inflammatory, and modulating cholinergic substances. And they are multi-targeted drugs with fewer side effects than synthetic drugs. 8 Sea buckthorn oil (SBO) can be extracted from seeds by mechanical cold pressing. 9 It contains phenolic acids and flavonoid compounds. These have been shown to have anti-inflammatory, antioxidant, neuroprotective, and anti-aging effects. 10 The main flavonoids in SBO are rutin, isorhamnetin, and quercetin. They have neurotrophic effects, which can induce neuronal cell differentiation and alleviate symptoms of neurodegenerative diseases. 11 Among them, rutin, catechin, and quercetin have been shown to have synergistic effects in antioxidant and neuroprotective aspects.12–14 Based on the above evidence, SBO is selected as a potential therapeutic agent in this study.
Inhalation therapy is the most common method of administration in aromatherapy trials. In recent years, many researchers have attempted to utilize the olfactory/trigeminal pathway to deliver drugs (non-invasively) to the brain. 15 Studies have shown that inhalation therapy can treat brain disorders such as insomnia, Alzheimer's disease (AD), and Parkinson's disease. 16 The advantages of inhaled administration are safety, avoidance of hepatic primary metabolism, non-invasiveness, and ease of administration. 18
Recent evidence suggests that SBO synergizes with neurotrophic factors in inducing neural protrusion growth. 17 However, there are limited studies on improving cognitive impairment by SBO. To evaluate the effect of SBO on improving cognitive impairment in cells and mice. We measured neuroinflammation, cholinergic activity, and oxidative stress levels and explored the antioxidant mechanism by scopolamine-induced PC12 injury model. Then, we treated mice by nebulized inhalation of SBO and evaluated the effect of SBO on the cognitive ability of mice using (MWM) and step-down test to elucidate that SBO has a beneficial role in the treatment of cognitive impairment.
Material and Method
Cells Culture and Treatment
PC12 cell lines are obtained from Procell Life Science&Technology Co. Ltd (Wuhan, China). Cells are cultured using RPMI-1640 medium (Genview) supplemented with 5% fetal bovine serum (Applied Biosystems) and 15% Horse Serum (Genview), penicillin (100 μg/mL), and streptomycin (100 μg/mL) in the humidified incubator at 37°C with 5% CO2. Fresh medium and subculture are replaced according to cell state. For the PC12 cell experiment, cells in the logarithmic growth phase are seeded in the plate at a density of 1 × 105 cells/mL. The cell morphology is observed using a phase contrast microscope (Nikon Ts2R).
Determination of Cells Viability
Cell viability is determined by MTT (bromide M5655 Sigma-Aldrich), human and experimental toxicological analyses are performed as described previously. Put simply, the PC12 cells are set up in a 96-well plate. SBO is extracted from seeds, which are purchased from Shanghai Yuanye Bio-Technology Co., Ltd SBO is mixed with dimethyl sulfoxide(DMSO) in a 1:1 ratio, which is diluted with the culture medium. To explore the neuroprotective effect of SBO polyamines induced in PC12 cells, According to a previous study, 18 we pretreated the PC12 cell with a concentration gradient of SBO (0.125, 0.25, 0.5, 1 μL/mL) for 12 h. The positive control group is treated with donepezil at a concentration of 4 μg/mL. No intervention is performed in the model group within the first 12 h, then adding to the optimum dosage dose of polyamine-(scopolamine-) (4 mg/mL) for the next 4 h, then cells are incubated with 0.5% MTT for 4 h. After that aspirating the medium, 100 μL of DMSO is added in a 96-well plate. The optical density(OD) is measured at 570 nm by a microplate reader (SpectraMax i3x). At last, cell viability is calculated by the ratio of OD.
Cell viability (%) = (sample group OD-blank group OD) /(control group OD-blank group OD) × 100%.
Enzyme-Linked Immunosorbent Assays
The PC12 cells are inoculated and cultured in a 24-well plate, and the SBO is added after 24 h. cell supernatant is collected after treatment with SCOP for 4 h. The IL-6, TNF-ɑ, and IL-1β are determined concentrations by the manufacturer's instructions (1, 110, 602, 579, 409 and 1, 117, 203 Beijing Dakewei Biotechnology Co, Ltd). Ach content and AchE activity are assessed by the instructions (ml093046 Shanghai Mlbio Co, Ltd and CSB-E17001Fh Wuhan Cusabio Co., Ltd) of the Ach assay kit and AchE activity assay kit.
Intracellular Antioxidant Biochemical Indicators Assay
PC12 cells are set up in a 6-well plate and treated with SBO after 24 h. Cell bodies are collected by centrifugation after 12 h and treated with scopolamine for 4 h. The activity levels of CAT, GSH-Px, and SOD are determined by the manufacturer's instructions (BC0205, BC1195, and BC0715 Beijing Solarbio Science & Technology Co., Ltd).
Real-Time PCR
The PC12 cells are set up and cultured in a 6-well plate. Total RNA is extracted from the PC12 cell using TRIzol (Procell Life Science&Technology Co., Ltd, Wuhan, China) in a low temperature environment. Then, cDNA is generated by adding a commercial RT-PCR kit (ABclonal Co, Ltd, Beijing, China). After that, real-time PCR is conducted using the SYBR Green Quanti Tect RT-PCR kit (Novizan Biotechnology Co., LTD, Nanjing, China), and each sample is repeated three times for analysis. β-actin is used as an internal reference for all of the samples. The relative mRNA expression of target genes is calculated using the 2-ΔΔCt method. Specific primers are as follows:
TNF-α, Forward 5′-CAGGCGGTGCCTATGTCTC-3′. Reverse 5′- CGATCACCCCGAAGTTCAGTAG-3′; IL-6 Forward 5′- CTGCAAGAGACTTCCATCCAG-3′. Reverse 5′- AGTGGTATAGACAGGTCTGTTGG-3′; IL-1β Forward 5′-GAAATGCCACCTTTTGACAGTG-3′. Reverse 5′-TGGATGCTCTCATCAGGACAG-3′; β-actin Forward 5′-GCAGTTGGTTGGAGCAA-3′. Reverse 5′-ATGCCGTGGATACTTGGA-3′.
Reactive Oxygen Species Assay
The PC12 cells are set up in Laser Confocal Petri Dish. The level of intracellular Reactive Oxygen Species (ROS) is detected in the cell using the fluorescent probe 2′,7′-dichlorodihydrofluorescein (DCHF, Sigma, United States of America). Within the cell, DCHF can be oxidized to fluorescent 2,7-dichlorofluorescein (DCF) by intracellular reactive oxygen species. The fluorescence detection is monitored using a fluorescence microscope (Nikon G-L, JP). ROS is quantified as the relative fluorescence intensity of DCHF in each cell within the scanned area. The values in the figure indicate the relative fluorescence intensity. Image-J software is used to calculate the average fluorescence intensity of cells.
Immunoblotting Assay
The PC12 cells are inoculated in the 6-well plate. Immunoblotting assay is performed as described previously. Lysis of cell with protein lysate (Beyotime Biotechnology) for 10 min on the ice. Protein concentration is determined using a bicinchoninic acid (BCA) protein assay kit (Beyotime Biotechnology). The protein concentration in the cell is determined using the BCA protein assay kit (Beyotime Biotech). Protein-like flats (20 μg/well) are injected into the wells of a 12% sodium dodecyl sulfate-polyacrylamide gel and electroblotted onto polyvinylidene difluoride (PVDF) membrane. The membranes are closed with 5% PBS-Tween 20 (PBST; 0.05%) skim milk powder for 1 h at 25°C, and respectively incubated with anti-Nrf2 (A21729, AB clone), anti-Ho-1 (A23650, AB clone), or anti-actin (ACTB) antibodies (Sigma) overnight at 4°C. After washing by PBST, PVDF membranes are incubated with appropriate HRP-coupled secondary antibodies (AB clone) for 1 h at normal atmospheric temperature. The immunoreactive bands are formed by color development with 3,30-diameter-aminobenzidine. The relative amounts of proteins are analyzed using Image J software.
Animal Ethics Statement
The learning memory disorder model group used the APP/PS1 transgenic mice, and the normal group used C57BL/6J mice (normal mice) of the same genetic background (6 weeks old, 20.5 ± 0.7 g). All the mice are purchased from Beijing Vital River Laboratory Animal Technology Co. During the 8-month acclimatization period, the animal has free access to food and drinking water and is housed in controlled chambers (temperature 24 ± 2 °C; relative humidity 45-60%; photoperiod 12 h/day; light hours 06:00-18:00). The experimental protocols followed the health guidelines for the Care and Use of Laboratory Animal.
Animal Experiment Design
C57BL/6J mice are used in the normal group; APP/PS1 mice are used in the carrier growth group, model group, SBO group, and positive control group, with 5 mice in each group Vehicle. According to the previous condition, we choose the following dose-response of SBO for the study. 19 The model and donepezil group inhaled a mixture of propanetriol and propylene glycol (4:6) in a volume of 5 μL for 3 s with a pause of 237 s. The solvent used to dilute the SBO is a solution (4:6 v/v) of glycerol and propylene glycol. SBO (1%) is exposed to an electronic vaporizer (RELX) and nebulized. The mice of the SBO group are exposed to the oil vapor. One group sniffed SBO vapor for 1 time/day, 1 h/time, for 30 days (high-dose group); the other group of mice inhaled SBO vapors for 1 time/day, 0.5 h/time, for 30 days (low-dose group); while the mice in the positive control group are treated with donepezil (3 mg/kg/day), which is given by oral gavage 1 time/day for 30 days. Normal group mice are fed normally without any intervention.
Morris Water Maze
Referring to the experimental methods and criteria in the literature, we carried out the subsequent experiments. 20 The Morris water maze (MWM) includes the first 5 days of positioning and navigation experiments, as well as the last day of space exploration experiments. Mice are trained to find hidden underwater platforms during a 5-day location navigation test. The time it took the mice to locate the platform is measured as the evasion latency. A 1-day space exploration test is conducted after platform removal. Swimming trajectories, activity time in the target quadrant, and the number of times the mice crossed the platform are measured over a period of 120 s. All data are recorded by a Morris software visual tracking system (Anhui Zhenghua Bio-Instrumentation Equipment Co. Ltd).
The Step-Down Passive Avoidance
According to the previous condition, 20 The memory retention ability of mice is detected by the step-down passive avoidance (SDA) test. The test chamber consisted of transparent Plexiglas. The bottom of the glass chamber consists of electrically stimulated connecting wires and electrocution devices, and a cylindrical rubber platform is placed in the corner. During acclimatization, mice are subjected to 180 s of acclimatization exploration in a permissive test chamber. After 24 h, the escape latency of mice and the number of errors were recorded within 3 mins.
Characterization of SBO by HPLC Analysis
According to the previous condition, the composition of SBO is analyzed by high performance liquid chromatography (HPLC). 21 Quercetin, lignans, isorhamnetin, catechin, and gallic acid are as standard compounds for HPLC. The HPLC analytical condition is been shown in Table 1.
HPLC Analytical Conditions.

Instrument and working conditions for Gallic acid, Catechin, Quercetin, Rutin, and Isorhamneti analysis by high-performance liquid chromatography (HPLC).
Statistical Analysis
All experiments are independently repeated at least 3 times. The normal distribution of data is analyzed using Graphpad Prism 9.0.0 (121). Normally distributed data are expressed as mean ± standard error and compared using one-way ANOVA in prism 9.0.0. Statistical significance is set at p < 0.05. The average fluorescence intensity is calculated using Image-J software, and images are processed using Adobe Photoshop CS2021 (Adobe Photoshop 2021).
Result
Effect of SBO on Scopolamine-Induced Damage in PC12 Cells
The PC12 cells are generally used as a model for cognitive impairment, which can evaluate the effect on cellular activity and protein expression levels of various substances. 22 In vitro, we determined the cytotoxicity of PC12 cells, and the result is shown in (Figure 1A). All target compounds showed low cytotoxicity to PC12 cells when the concentration was less than 0.1%. Then, we selected the concentration with lower toxicity to PC12 cells and detected the cytoprotective effect of the target compound against scopolamine-induced cell damage by MTT assay. The result of the MTT assay is shown in (Figure 1B). The viability of cells in the model group is lower than that of the normal group. Compared with the cells in the model group, the survival rate of cells treated with SBO concentrations of 0.5‰, 0.25‰, and 0.125‰ is significantly increased, indicating that SBO has a cytoprotective effect on PC12-damaged cells.

(A) The PC12 cell of viability with different concentrations of SBO. (B) Effect of SBO on the scopolamine damage in PC12 cell.

Effects of SBO on the mRNA levels of pro-inflammation cytokines in PC12 cells. The mRNA levels of (A) IL-6, (B) IL-1β, and (C) TNF-α in PC12 cells are detected by RT-PCR assay. Effects of SBO on the protein levels of pro-inflammation cytokines in PC12 cells. The protein levels of (a) IL-6, (b) IL-1β, and (c) TNF-α in PC12 cells are detected by ELISA. Different superscript letters are statistically significant differences (p < 0.05).
The Test of IL-6, TNF-α, and IL-1β Levels in PC12 Cells
We detected the mRNA levels of IL-6, IL-1β, and TNF-α by RT-PCR. The results are as follows: in the model group, the mRNA expression levels of IL-6, IL-1β, and TNF-α are significantly increased compared with the normal group, while the mRNA expression levels are significantly decreased in the positive control group and SBO group, and there is no significant difference between the positive control group and the normal group in IL-6 and IL-1β (p>0.05). After that, we detected the levels of IL-6, IL-1β, and TNF-α by ELISA. In the model group, the expression levels of inflammatory factors of IL-6, IL-1β, and TNF-α are significantly increased compared with the normal group. In the positive control group and SBO group, the expression levels of the inflammatory factors significantly decreased. There is no significant difference between the positive control group and the SBO group (Figures 3a-3c, p>0.05). The results showed that SBO significantly improved the level of inflammatory expression in scopolamine-induced PC12 cells.

Acetylcholine content (A) and acetylcholinesterase activity (B) are measured by assay kit instructions. Statistically significant differences between different superscript letters (p < 0.05).
SBO Amelioration of Scopolamine-Induced Cholinergic Disturbances in PC12 Cells
Since the model is closely related to cholinergic injury, we investigated the function of the cholinergic system. Figure 3 A shows the ACh levels in the hippocampus. Compared with the control group, the model group showed a decrease in ACh levels. Meanwhile the the positive control group and SBO group significantly increased ACh levels. There is no significant difference in ACh between the SBO group (p > 0.05). In the model group, scopolamine significantly increased AChE activity (Figure 3B), which significantly decreased in the SBO group. Compared with the model group, SBO (0.125, 0.25, 0.5 μL/mL) administration decreased AChE activity, with no significant difference between SBO groups (p > 0.05).
Ach is the first neurotransmitter to be identified, which is important in memory function and closely related to the pathological mechanism of AD. AchE is an enzyme that metabolizes Ach in the synaptic gap, leading to cognitive dysfunction. To further evaluate the protective effect of SBO on scopolamine-induced PC12 cell injury, we assessed Ach content and AchE activity. Scopolamine is a muscarinic cholinergic receptor antagonist that can lead to impaired cholinergic transmission, which can result in cognitive impairment.23,24 As expected, SBO ameliorated scopolamine-induced cholinergic dysfunction in PC12 cells. In the model group, the amount of Ach in the cell supernatants is reduced (Figure 3A), and the activity of AchE is higher than in the normal group (Figure 3B); in the SBO group and the positive control group, the cholinergic properties are significantly improved compared with the model group.
Effect of SBO on the Activities of SOD, CAT and GSH-Px
SOD, CAT, and GSH-Px are the main antioxidant enzymes that scavenge free radicals produced during oxidative stress in brain tissue. The specific activities of SOD (Figure 4A), CAT (Figure 4B), and GSH-Px (Figure 4C) are significantly decreased in scopolamine-treated cells compared to the normal group. Treatment of PC12 cells with different concentrations of SBO (0.25‰ and 0.125‰) and donepezil significantly increased the activities of SOD, CAT, and GSH-Px in cells compared to the model group. There is no significant difference between the SBO-treated group and the positive control group (p > 0.05). Thus suggesting that SBO exerts a protective effect against oxidative stress in PC12 cells.

Effect of SBO on the oxidative damage in PC12 cells. (A–C) Effect of SBO on Cell supernatant SOD, CAT, and GSH-Px levels. Statistically significant differences between different superscript letters (p < 0.05).
Effect of SBO on ROS Generation
ROS refers to the intracellular reactive oxygen species, which always participate in redox reactions and metabolism in the cell. However, in the brains of patients, the ROS level is abnormally increased due to oxidative stress, which disrupts the brain environment of patients. High concentrations of ROS in cells can cause mitochondrial dysfunction or damage. In order to investigate whether SBO could affect the production of ROS, we measured the relative level of ROS by DCFH-DA staining. After treating the cell with different scopolamine, fluorescence is observed using a fluorescence microscope to assess intracellular ROS level. The fluorescence intensity is much higher in the model group compared to the normal group (Figure 5A). The relative ROS values for the normal, model, and control groups are 1, 2.54, and 1.53 respectively. The ROS values for the groups treated with no SBO concentration (0.125, 0.25, and 0.5 μL/mL) are 1.82, 1.73, and 2.17 respectively. The ROS value were 1.82, 1.73, and 2.17. respectively in the group treated without a concentration of SBO (0.125, 0.25, 0.5 μL/mL). ROS level increased in the SBO group compared to the control group, but there is no significant difference. The result suggests that treatment of PC12 cells with SBO inhibits ROS production (p < 0.05) (Figure 5B).

Effects of SBO on cell viability and Scopolamine-induced oxidative stress in PC12 cells. Cells are pre-treated with SBO (0.125, 0.25, 0.5 μL/mL) for 24 h, and then stimulated with Scopolamine (4 mg/mL) for 4 h. (A) The ROS level is detected by laser confocal microscopy. (B) The above is the ROS level for each group. The value in the figure represents the relative fluorescence intensity of the cell, which is determined by laser scanning confocal microscopy. The average fluorescence intensity is calculated using the Image-J software. Different superscript letters are statistically significant differences (p < 0.05).

PC12 cells are treated with all kinds of SBO at 0.125, 0.25, 0.5 μL/mL, and donepezil 0.4 μg/mL for 12 h. Then the cell is subjected to an optimal dose of Sopolamine-(scopolamine-) (4 mg/mL) for another 4 h. The protein levels of Nrf2 and HO-1 are detected by Western blot assay. (A) Western blotting of Nrf2, HO-1and β-actin. (B) Analysis of Nrf2 protein levels. (C) Analysis of HO-1 protein levels. Different super-scripts letters are statistically significant differences (p < 0.05).
SBO Regulates the Levels of Antioxidant-Related Proteins
The (Nrf2)/(HO-1) pathway is important in regulating the expression of various antioxidant and anti-inflammatory genes. 25 Under ischemia-reperfusion injury, Nrf2 translocates to the nucleus, activates HO-1, and further represses inflammatory proteins.26,27 In the case of oxidative stress, the activated Nrf2 enters the nucleus, forms a dimer with the Maf protein, and then interacts with antioxidant response elements (ARE) to activate heme oxygenase-1 (HO-1), thereby inhibiting ROS production.27,28 Therefore, we explored the effects of SBO on the Nrf2/HO-1 pathway. In the model group, the expression of Nrf2 and HO-1 proteins is decreased compared to the normal group. However, the expression of Nrf2 and HO-1 is increased in the SBO and positive control group with significant differences compared to the model group. (p < 0.05) And at SBO content of 0.25, 0.5 μL/mL, there is no significant difference in Nrf2 protein expression compared to the normal group (p < 0.05). The results indicated that SBO increased the expression of the antioxidant protein Nrf2 and its downstream protein HO-1. Therefore, SBO might slow down progression through the Nrf2/HO-1 pathway to slow down the development of disease.
SBO Intervention Improved Cognitive Dysfunction in Mice
The trajectories of each group in the place navigation test are shown in Figure 7A. Compared with control mice, the escape latency of mice significantly increased about MWM on the second day (Figure 7C) (p < 0.05). The escape latency in the SBO-L and SBO-H groups decreased compared to mice on the second day. There is no significant difference on the fourth day. In the space exploration test, the trajectory of each group is shown in Figure 7B. Control mice spent more time in the target quadrant (Figure 7D) and crossed the platform more frequently (Figure 7E). In the SDA test, we detected that the treatment of SBO significantly increases step-down latency in mice (Figure 7F) (p < 0.05) and reduced the error rate (Figure 7G). There is no significant difference between the SBO group and the positive control group. It proved that memory acquisition ability is poor in mice. However, this phenomenon is improved by SBO intervention. Meanwhile, there is no significant difference between the SBO group and the control group in the target quadrant and platform crossings. The results indicated that SBO at a dose of (0.6, 1.2 μL/day could significantly improve cognitive dysfunction in mice (p < 0.05).

Effects of SBO on the MWM in mice. (A) Swimming trajectories of mice in the place navigation test. (B) Swimming trajectories of mice in the space exploration test. (C) Escape latency in the place navigation test. (D) Time spent in the target quadrant in the space exploration test. (E) Times of crossing platform in the space exploration test. (F, G) On the last day during MWM, the number of errors and latency in the step-down passive avoidance test. Different superscript letters are statistically significant differences (p < 0.05).
In order to identify the anti-inflammatory and antioxidant components in SBO, we used HPLC analysis of standard flavonoids, polyphenols,29,30 rutin, quercetin, isorhamnetin, gallic acid, and catechins are detected in SBO (Figure 8A-D). According to the content of their standard compounds of rutin and quercetin in the SBO samples are calculated based on the standard area curves of the compounds (Figure 8E, F). The pharmacological activities of these constituents are characterized by anti-inflammatory, antioxidant, and cholinergic effects.
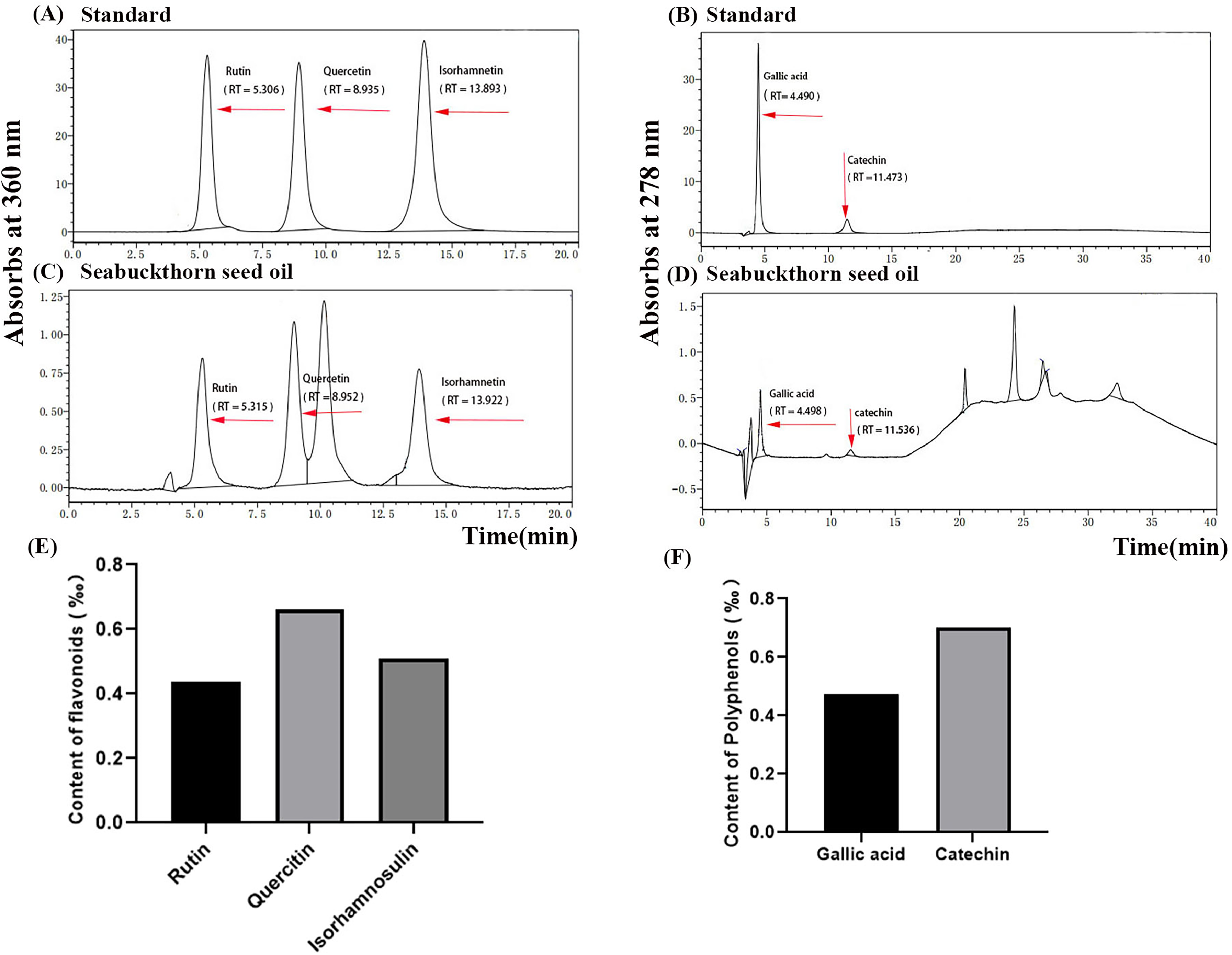
The compound profiles in SBO are analyzed by high performance liquid chromatography (HPLC) with standard compounds (rutin, quercetin, isorhamnetin, gallic acid, and catechin) (Figure 8A–D) in Table 1. The contents of flavonoids and polyphenols are calculated (Figure 8E-F) from the standard curves of the standard compounds.
Discussions
PC12 cells have many characteristics of neural cells and are often used to replace neural cells in research. 26 The cells are widely used to study various aspects of the nervous system in vitro.31,32 Scopolamine is a nonselective muscarinic ACh receptor (mAChR) antagonist. 33 It affects memory in animals and humans, producing a model similar to early symptoms of cognitive dysfunction. 34 Therefore, this study used scopolamine to model cognitive dysfunction in PC12 cells.
Our results showed that SBO decreased TNF-α, IL-6, and IL-1β levels in PC12 cells, indicating that SBO has good anti-inflammatory effects. There is a pronounced and severe inflammation in the brain of cognitive impairment. 35 Inflammation is mediated predominantly by microglial cells and astrocytes.36,37 Continuous activation of microglia in the brain produces many pro-inflammatory cytokines, directly affecting neurons and synapses.36,37 Increased neuroinflammation leads to decreased neuronal growth. These changes can lead to cognitive impairment. 38 Gallic acid and rutin in SBO inhibited the expression of inflammatory factors. 39 At the same time, the expression of the genome of inflammatory genes is attenuated. 40 So as to slow down the process of cognitive impairment.
In our study, the SBO treatment group significantly reduced ROS production. The activity of SOD, CAT, and GSH-Px and the expression of transcription factors between Nrf2/HO-1 signaling pathways were significantly increased. OS is associated with aging and is involved in cognitive impairment pathology. Under high stress conditions, cells produce large amounts of ROS. SOD, CAT, and GSH-Px are antioxidant enzymes that play a key role in inhibiting ROS production. 41 Meanwhile, Nrf2/HO-1 signaling plays an important role in protecting nerve cell from oxidative damage, which inhibits intracellular ROS level through the induction of heme oxygenase-1 (HO-1). 42 Oxidative stress causes neuronal and synaptic damage, and neurons and synapses are associated with cognitive function. 43 Catechins in SBO significantly increased CAT, GSH-Px, and SOD activities, 44 and rutin enhanced the expression of Nrf2 and downstream target HO-1 transcription factors. Thus slowing down the process of cognitive impairment.
In addition, the study's results showed that SBO reduced AchE activity and increased Ach level. The cholinergic system is essential in memory function, and reduced levels of acetylcholine in the brain are considered vital factors for cognitive impairment. 45 AChE is a critical enzyme in hydrolyzing ACh in vivo. Cholinesterase inhibitors block AChE and accordingly inhibit ACh catabolism and increase extracellular levels of ACh in the brain. 46 In the present study, SBO showed good AChE inhibitory activity in vitro. Quercetin contained in SBO inhibits acetylcholinesterase through the hydroxyl group on the benzene ring. 47 Inhibition of AChE promotes communication between nerves and alleviates symptoms of memory loss. Thereby improving the symptoms of cognitive impairment.
Our results showed that SBO improved the cognitive deficits of mice in the MWM test and the SDA test experiment. Oxidative stress, neuroinflammation, and cholinergic alterations are important pathological mechanisms in the pathogenesis of cognitive impairment. Under oxidative stress, excessive accumulation of ROS causes neuronal damage, such as neuroinflammation. 48 ACh exerts an anti-inflammatory effect by inhibiting the translocation of NFκ-β, thus synthesizing proinflammatory cytokines and improving redox status.49,50 In age-related neurodegenerative diseases such as AD, the microglia and astrocytes proliferate around the NFT and senile plaques. 51 In an activated state, these cells enhance the neuroinflammatory processes. 52 Meanwhile, cholinergic atrophy can induce abnormal phosphorylation of tau proteins. 53 Hyperphosphorylated tau proteins readily dissociate from microtubules and aggregate to form NFT. This leads to aggravating neuroinflammation and oxidative stress, 54 which reduces the efficiency of neural signaling and disintegrates neuronal networks, Leading to exacerbation of cognitive impairment symptoms. Mild cognitive impairment is a preclinical symptom of Alzheimer's disease, with 40% of these patients progressing to AD after four years, 55 and the development of severe cognitive impairment is also involved.
This study has some limitations: for example, although SBO shows good efficacy, the specific mechanism of action is unclear, and we need further research. In the follow-up study, we will use nuclear magnetic technology to analyze the structural changes in the brain tissue of mice. We will also analyze neuronal damage by Nysted staining experiments and inflammatory infiltration by HE staining. Finally, we will also analyze possible synergies between the components and their mechanisms of action. In the future, we will make the active ingredients of SBO into a medicinal liquid, and through the nebulizer inhaler device, using high-speed oxygen airflow, the medicinal liquid will be formed into a mist, which will then be inhaled by the respiratory tract, to achieve the purpose of relieving and improving the symptoms of cognitive impairment patients.
Conclusions
In conclusion, SBO can effectively attenuate scopolamine-induced PC12 cell damage and protect neuronal cells. This study significantly improved cognitive impairment in mice by inhalation nebulization of SBO.
Limitations
There are some limitations in this study; for example, although essential oils show good efficacy, the specific mechanism of action is not clear,and we need further research. In this study, although we have analysed the effect of SBO extract on the improvement of mice in behavioural experiments, we have not yet analysed its effect on tissue structure and biochemical indicators.
Footnotes
Acknowledgments
This work was supported by The Key innovation Project of Qilu University of Technology (Shandong Academy of Sciences) (2022JBZ01-06)
Author Contribution
Dong Yang1#: Writing – original draft, Formal analysis, Data curation, Software Song Zhang1#: Investigation, Methodology, Project administration, Resources
Duo Wang1: Software, Supervision, Validation, Visualization
Qiulin Yue1: Methodology, Project administration, Software, Validation
Chen Zhao4: Project administration,Visualization, Investigation, Resources
Xin Sun1: Project administration,Visualization, Investigation, Resources
Kunlun Li4: Project administration,Visualization, Investigation, Resources
Baojun Li4: Project administration, Visualization, Investigation, Resources
Le Su*1,5,: Writing – review & editing, Funding acquisition, Supervision, Resources
Yang Zhang*2,: Writing review & editing, Funding acquisition, Supervision, Resources
Lin Zhao*,1,3: Writing – review & editing, Funding acquisition, Supervision, Resources
Conflict of Interest Statement
BL and KL were employed by the company Shangdong Zhuoran Biotechnology Co., Ltd The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability Statement
All datasets during the current study are publicly available, available upon reasonable reques data sharing is applicable to this article.
Ethics Approval and Consent to Participate
All experimental procedures were performed under the Guidance of the National Research Council's Guide for the Care and Use of Laboratory Animals and approved by the Animal Ethics Committee of Qilu University of Technology (Shandong Academy of Sciences) (No. SWS20220303).
Funding
This work was supported by Yantai Development Zone Science and Technology Leading Talents Project (grant number 2020CXRC4, Lin Zhao, and Orlando Borrás - Hidalgo), Key innovation Project of Qilu University of Technology (Shandong Academy of Sciences) (2022JBZ01-06), the XPCC Financial Science and Technology Project in 2022 (No. 2022AB002).
