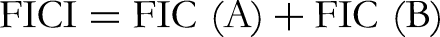Abstract
The combined effects of the essential oil (EO) mixtures can significantly enhance their antimicrobial activity by harnessing the synergistic interaction among their diverse bioactive compounds. Additionally, combining EOs with conventional antimicrobial agents can improve or restore the efficacy of these treatments, providing a promising strategy for combating pathogenic and resistant microorganisms. Various methods have been used to evaluate the EO-based combinations, resulting in different types of interactions. This review highlights that EOs from several medicinal and aromatic herbs exhibit synergistic effects when used in mixtures or in combination with antibiotic and antifungal drugs against pathogenic and resistant bacterial and fungal strains. Additive, indifferent and antagonistic effects have also been reported. Recorded synergistic effects suggest the potential therapeutic effects of EOs as natural alternatives or complements to conventional antimicrobial drugs to combat antimicrobial resistance. Despite the demonstrated synergistic effects of many EOs and conventional antimicrobials, there is a notable lack of studies elucidating the mechanisms underlying these synergistic interactions and their specific targets within microbial cells. In this review, the investigations of synergistic studies of EO with conventional antibiotics and the progress in antimicrobial effect of EO mixtures have been reported.
Keywords
Introduction
The global emergence of multidrug-resistant microorganisms has become a critical public health issue, largely due to the overuse and misuse of antimicrobial drugs. This problem spans a wide range of pathogens, including bacteria, fungi, viruses, and parasites, all of which have developed resistance to conventional antibiotic, antifungal, antiviral and antiparasitic treatments.1,2 As a result, antimicrobial resistance is now recognized as an urgent and pervasive global challenge. The ubiquitous occurrence of multidrug-resistant bacteria has severely compromised the efficacy of current antibiotic therapies, leading to thousands of deaths worldwide annually and the number is expected to rise the millions by 2050.3,4 Therefore, there is an urgent need for the development of new and innovative antimicrobial alternatives to address the growing problem of multidrug resistance. In this context, the use of plant secondary metabolites such as essential oils (EOs) could present a promising alternative strategy. EOs are volatile, natural, fragrant liquids extracted from various parts of medicinal and aromatic plants, especially leaves and flowers. These oils are synthesized through complex metabolic pathways, primarily aimed at protecting plants from a range of pathogenic microbes.5–7 Extensive research in this area have shown that several EOs can act as antibacterial and antifungal agents against a broad spectrum of human pathogens, including antibiotic-resistant strains.5,8–10 The broad-spectrum biological activities of EOs can be attributed to the complexity and variability of their chemical compositions. One of the advantages of EOs is that they are produced naturally as complex mixtures that act on several targets. As a result, the risk of resistance development is lower.5,11,12 In general, the antimicrobial activities of EOs are strongly linked to their hydrophobicity. This characteristic allows them to penetrate microbial cells and cause functional and structural alterations. Thus, the membrane is often altered, inducing an increase in its permeability leading to cytoplasm leakage and cell degeneration.13–15 However, the majority of EOs have been shown to be effective at relatively high concentrations and it has been reported that higher concentrations of individual EOs are required to achieve in vivo antimicrobial potency similar to levels established by in vitro assays. Therefore, mixture of EOs constitute a new approach to increase their antimicrobial efficacy through the synergistic interactions generated between their different components.16–21 Furthermore, there are many reports of enhanced antimicrobial activity when EOs are used in combination with conventional antibiotic and antifungals drugs.22–26 The constituents of EOs can disrupt cell membrane integrity, thereby facilitating the penetration of antimicrobial agents to their target sites. This review aims to provide an overview of studies investigating the combined antimicrobial effect of EOs among themselves, and with conventional antimicrobial agents to enhance their efficacies. It presents current knowledge on the synergy between EOs and antibiotics, report some documented mechanisms underlying these interactions, and the use of innovative technologies in EO mixtures.
Methodology
The articles used for this review were collected from the electronic databases: Web of Science, Pubmed, Scopus, SciFinder, and Google Scholar. The titles used to perform the searches consisted of keywords including: antimicrobial activity, essential oil, mixtures, synergy, antibiotics, antifungals, interaction, mechanism of action; checkerboard, and mixture design, among others related to the subject of the review. To focus on recent research and maintain relevance, only articles published between 2017 and 2022 were considered for the synergy with antibiotics. Articles in different languages were taken into consideration. Then, the articles were rigorously selected according to the focus of the review. Studies that reported the combined antimicrobial effects of EOs and the synergistic interactions of EOs with conventional antimicrobials, whether or not they included information on the mechanisms of action, were considered for review.
EOs with Antimicrobial Activities
Medicinal and aromatic plants have been used for centuries as remedies to treat human diseases, especially those of infectious origin. These plants contain therapeutically valuable compounds called “secondary metabolites”, which are typically derived from secondary metabolism. EOs are a class of secondary metabolites with a complex composition. They are volatile, hydrophobic and liposoluble substances. EOs are being intensively explored for their potential application as antimicrobial agents or in combination therapy with conventional antimicrobials.22,23,27,28 This use makes sense in the current context of increased spread of resistant strains that increasingly limit the efficacy of conventional antimicrobials. Studies in this area have shown that several EOs can act as antibacterial and antifungal agents against a broad spectrum of human pathogens, including antibiotic-resistant strains. EOs exert their antimicrobial effects through multiple mechanisms of action, making them effective against a variety of microorganisms. These mechanisms include the disruption of cell wall synthesis, damage to cytoplasmic membranes, interference with membrane proteins, leakage of cellular contents, and inhibition of DNA and RNA synthesis (Figure 1).12,29,30 For instance, oregano and thyme EOs rich in carvacrol and thymol were reported to disrupt bacterial cell membranes, leading to increased permeability and cell death.12,31,32 Similarly, eugenol from clove and cinnamaldehyde from cinnamon EOs induce apoptosis-like cell death in yeast cells by compromising membrane integrity. 33 Generally, EOs from clove, thyme, cinnamon, lavender have shown significant antimicrobial effects against both Gram-positive and Gram-negative bacteria, yeast and fungi.8,15,22,28,34 EOs of oregano and thyme, for example, have demonstrated strong antibacterial activity due to their high phenolic content, effectively inhibiting the growth of many pathogenic bacteria.28,35–37 These EOs have also shown potent antifungal properties against Candida and fungi species, by disrupting fungal cell wall and membrane integrity, and inhibiting spore germination and enzymatic activity.29,33,34,38,39

Schematic illustration of EO antibacterial mechanisms against the pathogenic bacterial cell.
Methods Used to Evaluate the Synergistic Interactions
Different interaction assessment methods can sometimes produce different conclusions for the same combination of antimicrobial agents. However there is generally a good degree of agreement between the methods, which have been compared and discussed by several authors.40–42 The interaction between antimicrobial agents can generate four different types of effects: synergistic, additive, antagonistic, or indifferent effects. An interaction is said to be synergistic when the combined effect is greater than the sum of the two effects. An additive effect is when the combined effect is equal to the sum of the effects of the individual substances. Additivity is sometimes referred to as indifference because there is no interaction between the antimicrobial agents being tested. Antagonism is defined if the effect of one or both compounds is less when applied together than when applied individually.43,44 The most commonly used method to study antimicrobial interactions is the Checkerboard assay, in which antimicrobial agents are combined in microplates such that one antimicrobial agent varies horizontally along the x-axis and the other vertically along the y-axis in 2-fold serial dilutions.
42
Dilutions range generally from the MIC to several dilutions below the MIC (up to 2056-fold).
41
In some studies, the same checkerboard principle has been referred to as microdilution method, utilizing various antibiotic concentrations but a single subinhibitory concentration of the EO (eg MIC/4 and MIC/32).26,45 Inhibitory combinations are observed after incubation, from which interaction types can be determined.
42
The results of the Checkerboard test are interpreted by plotting an isobole or isobologram (Figure 2) or by calculating the fractional inhibitory concentration (FIC) and the FIC index (FICI). To represent the results as isoboles (or isobologram) the MICs of the individual antimicrobials tested are plotted on the x and y axes, and joined to create a straight line called the additivity line. The concentrations of the inhibiting combinations are also plotted. Synergy results in a concave curve relative to the additivity line, while antagonistic effects take the form of a convex curve. The FIC of the two antimicrobials and the FIC index (FCI) are calculated by the following formulas:
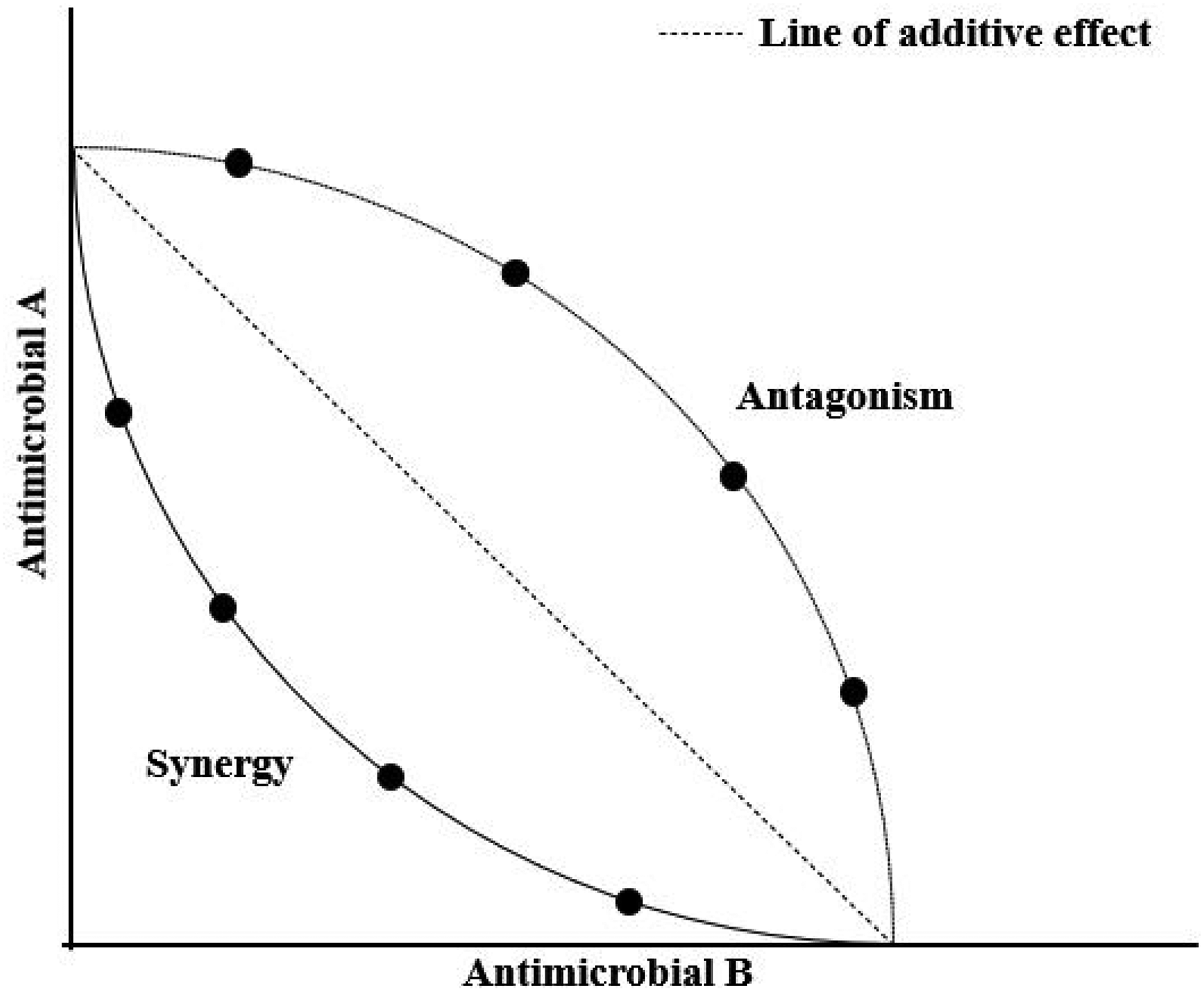
Isobologram for two antimicrobials A and B in a checkerboard titration.
The types of interactions are determined by comparing the calculated FICI to the limits set, which differ between authors (Table 1). Checkerboard method has been used in some studies to evaluate the interaction between two EOs in binary mixtures,20,35,46–49 and often to assess the synergistic effect of EOs with antibiotics.22–24,50–54 In this case, calculating the gain of the antibiotic MIC is essential to determine how many times it has been reduced by the addition of the EO. Another method used in several studies for evaluating antimicrobial interactions of EOs with conventional antibiotics is the time-kill test.24,53,55,56 This test determines the number of viable microbial cells present in the liquid medium in the presence of a particular combination of antimicrobial agents at different times, providing a curve of the dynamics of the combination's effect on cell growth and viability over time. A synergistic interaction is defined when the microbicidal potency of the first agent is enhanced by a sub-inhibitory concentration of the second agent, whereas an antagonistic interaction is generated if the effect of the first component is inhibited by the second. 44 Another method used in some studies to assess the synergy of EO with certain antibiotics is the disc diffusion method, by applying well-defined volumes of both substances together on the same disc. 55 The results are interpreted as synergy if the zone of inhibition for the combination treatment is greater than the sum of the zones for the EO and the corresponding antibiotic; an additive effect is indicated if the zone of inhibition for the combination treatment equaled the sum of the zones for the EO and the corresponding antibiotic; and antagonism is indicated if the zone of inhibition for the combination treatment is less than the sum of the zones for the EO and the corresponding antibiotic. Generally, all of these methods were often used to quantify interactions between antimicrobial agents, especially between EOs and antibiotics or antifungals. However, a new method has been used recently to study the antimicrobial interactions between EOs.17–19,57 This method is based on mixture designs (or experimental designs) in which EOs alone, binary and ternary combinations are included (Figure 3). The experimental data obtained are fitted to different statistical models using data processing software (eg Statistica, SAS JMP, Stat graphic, Expert design…etc) in which the independent variables are the proportions of EOs in the mixture and the dependent variables are the response obtained towards the target organism (MIC, ZI, LD50, IC50…etc). The type of interaction is determined by analyzing the sign and significance of the coefficients of the regression function (Figure 4). The advantage of this method is that the mixture design aims not only to create general designs on the responses and interactions between the EOs, but also to optimize the antimicrobial activity be determining optimized combinations that provide optimal antimicrobial effects.

Examples of mixture designs used to study the interactions between EOs. 58 Points 1, 2, and 3 correspond to the individual EOs, points on the edges of the triangle (4, 5, 6, 4’, 5’, 6’) consist of binary combinations, while points 7, 8, 9, and 10 constitute the ternary combination.

Schematic representation of various methods for studying EO/EO and EO/antibiotic combinations, along with the interpretation and presentation of their results.
The FICI Ranges Used to Determine the Type of Interaction Between Antimicrobial Agents.

Synergistic Interaction of EOs with Antibiotics
A number of studies suggest the synergistic combinations of EOs with conventional antimicrobials as an innovative approach to combat the emergence of antimicrobial resistance and enhance or restore the efficacy of these antimicrobial drugs. The concerted action of these combinations against multiple cellular targets could provide broad-spectrum antimicrobial activity, and thus may be effective in reducing or reversing antimicrobial resistance. 63 Indeed, interactions between EOs and antibiotics have been shown to be effective against many fungal and bacterial cells (both Gram-positive and Gram-negative ones), including multidrug-resistant pathogenic strains. Table 2 reports the studied plant EOs in synergistic effects with conventional antimicrobials. Diverse antimicrobials were used in these synergy studies included antibiotics and antifungals from various chemical families. The families of antibiotics with the greatest presence in synergistic trials include aminoglycosides, penicillins, cephalosporins, aminoglycosides and carbapenems. The antifungal family azoles and polyenes were also well represented in the trials. Regarding EOs, the majority belonging to the genus Thymus, Calamintha, and Mentha from the Lamiaceae family. Different types of interaction have been reported, with many of these combinations showing significant synergistic effects. For instance, Alaoui Jamali et al (2017) investigated the synergistic effects of EOs derived from three Thymus species (Thymus leptobotrys, T. pallidus and T. ciliatus) in combination with the antibiotic cefixime and showed a remarkable synergistic effect against Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Bacillus subtilis, Micrococcus luteus, and Staphylococcus aureus. The study conducted by El Atki et al (2019) demonstrated that Cinnamomum cassia EO exhibited synergistic effects in combination with the conventional antibiotics ampicillin and chloramphenicol and additive effects with streptomycin against E. coli, S. aureus and P. aeruginosa strains. The combination of Satureja montana EO with gentamicin generated additive and synergistic interaction against nine reference strains of S. aureus, E. coli and Listeria monocytogenes. Observation of these treated bacterial cells under a scanning electron microscope showed that synergistic combinations produced alterations in cell morphology through pore formation and cell surface collapse. Synergistic combinations also reduced biofilm formation. 51 In contrast, the combination of Calamintha sylvatica EO with gentamicin showed indifference against S. aureus, antagonism against E. coli, additivity against P. aeruginosa, and synergy against Salmonella abony. Conversely, Calamintha vardarensis EO, when combined with gentamicin, generated an additivity against S. aureus and P. aeruginosa, antagonism against E. coli, and synergy against S. abony. 64 A synergistic effect was also noted when Thymus serpyllum EO was combined with vancomycin and tetracycline against S. aureus, E. coli, K. pneumoniae and with fluconazole and amphotericin B against C. albicans. 65 Melaleuca leucadendra EO exhibited strong synergistic effect against C. albicans in combination with the antifungal agents econazole, ketoconazole and itraconazole. 56 In another study, Pelargonium endlicherianum EO interacted indifferently with gentamicin and synergistically with cefepime by altering the cell membrane permeability of K. pneumoniae. 66 The findings by Tullio et al (2019) highlight synergistic and additive interactions for the combinations of Mentha piperita EO with fluconazole, itraconazole and ketoconazole against some azole sensitive and resistant Candida spp, Cryptococcus neoformans, Trichophyton mentagrophytes, Microsporum canis and Microsporum gypseum. In addition, the synergy with the antibiotic ciprofloxacin and the antifungal fluconazole of the EOs of Cannabis sativa, Periploca laevigata, Lavandula maroccana, Thymus atlanticus, Linaria ventricosa and Argania spinosa was very interesting against Gram-positive bacteria (B. subtilis; M. luteus; S. aureus), Gram-negative (E. coli; P. aeruginosa; K. pneumoniae) and yeast species (C. albicans; C. glabrata; C. krusei; C. parapsilosis).22,23,67–70 Boonyanugomol et al (2017) 55 tested the synergy of Zingiber cassumunar EO with some combinations of antibiotics: β-lactamase inhibitors (piperacillin + tazobactam + amoxicillin-clavulanic acid), Cephems (ceftazidime + cefepime + cefotaxime + ceftriaxone), carbapenems (imipenem + meropenem + ertapenem), aminoglycosides (gentamicin + amikacin), tetracyclines (tetracycline + doxycycline), fluoroquinolones (ciprofloxacin + levofloxacin) and inhibitors of the folate biosynthesis pathway (trimethoprim + sulfamethoxazole), and the results were synergistic against Acinetobacter baumannii. In addition, Origanum vulgare EO acts synergistically with polymyxin B by destabilizing and disrupting the bacterial cell membrane and fragmenting the DNA of A. baumannii cells. 37 In another study, Salvia fruticosa, S. officinalis, and S. sclarea EOs reduce the effective dose of tetracycline, decrease the efflux of the antibiotic, and decrease the expression of the Tet(K) gene encoding the efflux protein, in clinical isolates of tetracycline-resistant Staphylococcus epidermidis. 71 Similarly, the EO obtained from Chenopodium ambrosioides reduced synergistically the MICs of tetracycline and ethidium bromide against a S. aureus strain carrying the Tet(K) gene, indicating inhibition of the efflux pump. 72
Summary of Synergistic Interaction of EOs with Antimicrobial Drugs (Antibiotics and Antifungals) Against Bacteria and Fungi Strains.

Combined Antimicrobial Activity of EO Mixtures
The use of EO mixtures has been developed recently as a new approach to improve their bioactivities as natural antimicrobial, antioxidant, insecticide and acaricide agents, by taking advantage of the synergistic and additive interactions generated between their different classes of constituents. In microbiology, this new strategy has been developed to increase the potential use of EOs in the control of resistant pathogenic microorganisms involved in human infections and/or food spoilage. Different species were studied in EO mixtures (Table 3). The majority of these species belong to the genus Thymus, Origanum, Lavandula, Rosmarinus, Mentha, Cinnamonum, Satureja, myrthus, Artemisia and Melaleuca. These species have generated different types of interaction when combined in a binary, ternary or even sometimes quaternary way. The binary mixtures were evaluated generally by the checkerboard method, volume/volume mixing method, or different percentages of the EOs. On the other hand, the majority of studies on ternary and quaternary mixtures employed the design of experiments method. However, a few studies tested equi-proportional mixtures or varying percentages, accompanied by the microdilution assay for ternary and quaternary mixtures.86–90 Exceptionally, Vavala et al 91 used the checkerboard method with time kill analysis, even for a mixture of three oils. In general, the methods used were categorized in the following order: checkerboard method, volume-to-volume ratios, mixture design, and varying percentage combinations. Based on the results, several binary EO mixtures showed synergistic interactions against pathogenic strains of Trichophyton, Staphylococcus, Salmonella, Bacillus, Pseudomonas, Listeria, Enterobacter and E. coli.16,20,21,92–95 Nevertheless, additive, indifferent or antagonistic interactions have been also recorded by some EO binary and ternary combinations against various microorganisms.35,46,87,96–99 For instance, the combination of Origanum vulgare and Rosmarinus officinalis EOs showed synergy against L. monocytogenes, Yersinia enterocolitica and Aeromonas hydrophilla, and addition against Pseudomonas fluorescens. 100 The combination of Cinnamomum zeylanicum with Eugenia caryophyllata EOs showed addition against Bacillus cereus, B. subtilis, S. aureus, and indifference against E. coli and Salmonella typhimurium, while that with Thymus vulgaris EO exhibited additive effect against all these tested bacteria. 96 In another study, Fahimi et al 98 tested binary combinations of EOs from Thymus vulgaris, Lavandula angustifolia, Rosmarinus officinalis and Mentha piperita against four bacterial strains, and the results showed that the combinations of Mentha piperita with Thymus vulgaris or Lavandula angustifolia possess high synergism interaction against S. aureus. All the other tested combinations exhibited antagonistic effects. The study conducted by Ayari et al 49 showed that the EO of Thymus vulgaris interact synergistically in binary combinations with EOs from Melaleuca alternifolia, Cinnamomum cassia, and Origanum compactum against B. cereus and Paenibacillus amylolyticus. The ternary mixtures of Satureja hortensis/Origanum vulgare subsp. hirtum /Origanum vulgare subsp. vulgare 87 and Mentha suaveolens/Rosmarinus officinalis/Melaleuca alternifolia 91 showed synergistic effects against Helicobacter pylori and Pseudomonas syringae, respectively. On the other hand, the different ternary combinations studied using the mixture design method have shown different types of interaction depending on the tested combinations and the targeted microorganisms, and they allowed the prediction of optimal EO mixtures that represent the highest antimicrobial effect. According to Chraibi et al, 18 the optimal mixture predicted against E. coli, S. aureus and Candida tropicalis corresponded to 54%/46%; 56%/44% and 55%/45% of Mentha piperita and M. pulegium EOs, respectively. In another study, Ouedrhiri et al 101 showed that 17.1% of Myrtus communis, 39.6% of Artemisia herba-alba and 43.1% of Thymus serpyllum represents the greatest antibacterial activity against S. aureus, E. coli and B. subtilis. However, Fadil et al 19 found 55% T. vulgaris and 45% M. communis as an optimal mixture providing the highest effect against Salmonella typhimurium. It is important to note that the only study that investigated a quaternary EO mixture using the mixture design method, was reported by Falleh et al, 102 in which the combined effect of the four EOs was evaluated against E. coli and only one optimal mixture of three EOs (2.4% Syzygium aromaticum, 38.2% Lavandula stoechas, and 59.4% Cinnamomum zeylanicum) was found.
Summary of Antimicrobial Activity of EO Mixtures.

Nanoemulsion and Encapsulation of EO Mixtures
The use of EOs in mixtures has demonstrated enhanced antimicrobial properties, making them a promising solution for various applications. To further harness their potential, some of these mixtures have recently been developed into innovative formulations through advanced technologies such as nanoemulsions and encapsulation. These methods significantly improve the physical stability, bioavailability, and targeted delivery of EOs, addressing inherent challenges such as volatility, hydrophobicity, and susceptibility to environmental degradation.17,121,122 By optimizing these delivery systems, these technologies amplify the antimicrobial, antifungal, and synergistic effects of EO mixtures, providing sustainable and highly effective solutions across diverse fields. In this context, the encapsulation of a synergistic EO mixture of Pimpinella anisum and Coriandrum sativum has been explored using chitosan-based nanoemulsions. The encapsulated mixture showed superior antifungal and antiaflatoxigenic activities against Aspergillus flavus, compared to the unencapsulated formulation. It effectively inhibited fungal proliferation, reduced aflatoxin B1 secretion, and minimized lipid peroxidation in stored rice. Investigation of mechanism of antifungal action revealed that this encapsulated EO mixture disrupted ergosterol biosynthesis and caused irreversible plasma membrane damage in Aspergillus flavus cells. Furthermore, fumigation with the nanoemulsion significantly decreased methylglyoxal levels in fungal cells, elucidating its biochemical mechanism for inhibiting aflatoxin production. 122 In another investigation, the antibacterial efficacy of an EO mixture obtained from Ammodaucus leucotrichus, Thymus vulgaris, and Lavandula maroccana was assessed against multidrug-resistant bacteria S. aureus, E. coli, and P. aeruginosa. Using an augmented simplex-centroid design, an optimized EO mixture was identified and experimentally validated. This optimized mixture was formulated into a nanoemulsion, achieving significantly enhanced antibacterial activity compared to its non-emulsified form. Additionally, the optimized mixture was tested in combination with antibiotics gentamicin and amoxicillin, leading to a reduction in their MICs of up to 64-fold, demonstrating its potential synergistic in combating antibiotic resistance. 17 Furthermore, the nanoencapsulation of an EO mixture composed of Salvia rosmarinus and Cedrus atlantica was optimized using a response surface design, employing gum arabic as the encapsulation material. This optimized formulation significantly enhanced the antifungal activity of the EO mixture against the brown rot fungi Gloeophyllum trabeum and Poria placenta. The nanoencapsulation process improved the physico-chemical stability of the EO mixture and also amplified its inhibitory effects on fungal growth. 121 These findings demonstrate the transformative role of nanoemulsification and encapsulation technologies in optimizing EO mixtures. By enhancing their stability, efficacy, and antimicrobial properties, these innovations pave the way for potential applications in food safety and human medicine.
Conclusions and Future Perspectives
The antimicrobial synergistic effect of EOs, either among themselves or with pharmaceutical antimicrobial drugs, has been well-documented against human pathogenic and resistant microorganisms. Combinations between EOs have been used to enhance their therapeutic properties or their potential as food preservatives, while synergistic activity with antibiotics has been explored to improve or restore the efficacy of clinical antimicrobials that have become ineffective due to the growing issue of antimicrobial resistance. This review highlights that EOs from several medicinal and aromatic herbs exhibit synergistic effects when used in EO mixtures or in combination with antibiotic and antifungal drugs against many bacterial and fungal species. Despite the demonstrated synergistic effects of many EOs and conventional antimicrobials, there is a lack in studies elucidating the mechanisms underlying these synergistic interactions and their targets within microbial cells. Future studies addressing the mechanisms of action of EO combinations require interdisciplinary research efforts and innovative approaches. Enhancing the understanding of these interactions is crucial for unlocking the full therapeutic potential of EO combinations. Another critical area for future research is elucidating the molecular mechanisms underlying EO-antibiotic interactions, providing valuable insights for designing more effective combinations. Moreover, research should focus on developing novel delivery systems for EO-antibiotic formulations particularly through nanoencapsulation and nanoemulsion technologies. These methods can significantly enhance the stability and bioavailability of EOs, enable targeted delivery and ensuring that their therapeutic properties are preserved and more effectively utilized. These advancements will be essential for overcoming the challenges associated with EO-antibiotic combinations and maximizing their efficacy in combating antimicrobial resistance and related microbial infections.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.