Abstract
The stem bark and leaves of Daniellia oliveri were obtained from two sites, Batsari and Zurmi, in Nigeria. Leaves of Leptoderris micrantha were obtained from Agbagi, Nigeria. Essential oils of these plants were obtained by hydrodistillation and analyzed by gas chromatography-mass spectrometry. The major components in the bark essential oil of D. oliveri were δ-cadinene (12.8%), α-muurolene (6.7%), α-calacorene (5.9%), and caryophyllene oxide (5.5%). The major components in the leaf essential oils from Batsari and Zurmi, respectively, were humulene epoxide II (8.0% and 16.3%), caryophyllene oxide (7.4% and 12.4%), pentadecanal (8.9% and 6.0%), phytone (6.5% and 2.2%), δ-cadinene (5.3% and 3.0%), and α-muurolene (5.3% and 2.6%). The major components in the leaf essential oil of L. micrantha were incensole (16.2%), phytone (15.4%), pentadecanal (13.7%), α-pinene (7.7%), and iso-phytol (5.2%). The essential oils were screened for antibacterial activity against Bacillus cereus, Cutibacterium acnes, Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Pseudomonas aeruginosa, and Serratia marcescens, and for antifungal activity against Aspergillus fumigatus, Aspergillus niger, Cryptococcus neoformans, Microsporum canis, Microsporum gypseum, Trichophyton mentagrophytes, Trichophyton rubrum, and Candida albicans, using the microbroth dilution method. The leaf essential oils of D. oliveri and L. micrantha showed only marginal activity against the panel of microorganisms. However, D. oliveri bark essential oil showed notable antifungal activity against Aspergillus niger and Trichophyton rubrum with a minimum inhibitory concentration of 78.1 µg/mL for each. This is the first report on the essential oil compositions of D. oliveri and L. micrantha from Nigeria and their antimicrobial activities.
Daniellia oliveri (Rolfe) Hutch. & Dalziel (Caesalpinioideae, Fabaceae) is a deciduous tree with a flat-topped crown. 1 The tree ranges from Senegal to South Sudan and Uganda, south of Sahal, where it is the most widespread tree species of the Savannah. 2 In Nigeria, the tree is known as “iya” in Yoruba, “maje” in Hausa, and “abwa” in Ibo. 3 In northern Nigeria, leaves of D. oliveri are used to treat diabetes, gastrointestinal problems, diarrhea, as a diuretic and an aphrodisiac, 4 the bark and the resin are used as a mosquito repellent, 5 and extracts of the bark are used in Burkina Faso to treat small ruminant gastrointestinal parasites. 6 The resin of D. oliveri has yielded the labdane diterpenoids daniellic acid 7 and oliveric acid, 8 while triterpenoids and flavonoids have been isolated from the leaves. 9
Leptoderris micrantha Dunn (Papilionoideae, Fabaceae) is a liana with brown bark and light pink or purple flowers. The plant is native to Guinea, Ghana, and Southern Nigeria and is called “atari obuko” or “ewe awo” in southwestern Nigeria. A leaf decoction is taken as an aphrodisiac and treatment for male impotence, 10 and as a treatment for psychosis, dropsy, swellings, edema, gout, and pulmonary troubles. 11 The leaves of L. micrantha have been reported to contain rotenone. 12 As part of our continuing investigation on essential oils from Nigerian medicinal plants and their biological activities, this investigation was focused on the analysis and antimicrobial properties of the leaf and bark essential oils of D. oliveri and the leaf essential oil of L. micrantha.
Results and Discussion
Chemical Compositions
Daniellia oliveri
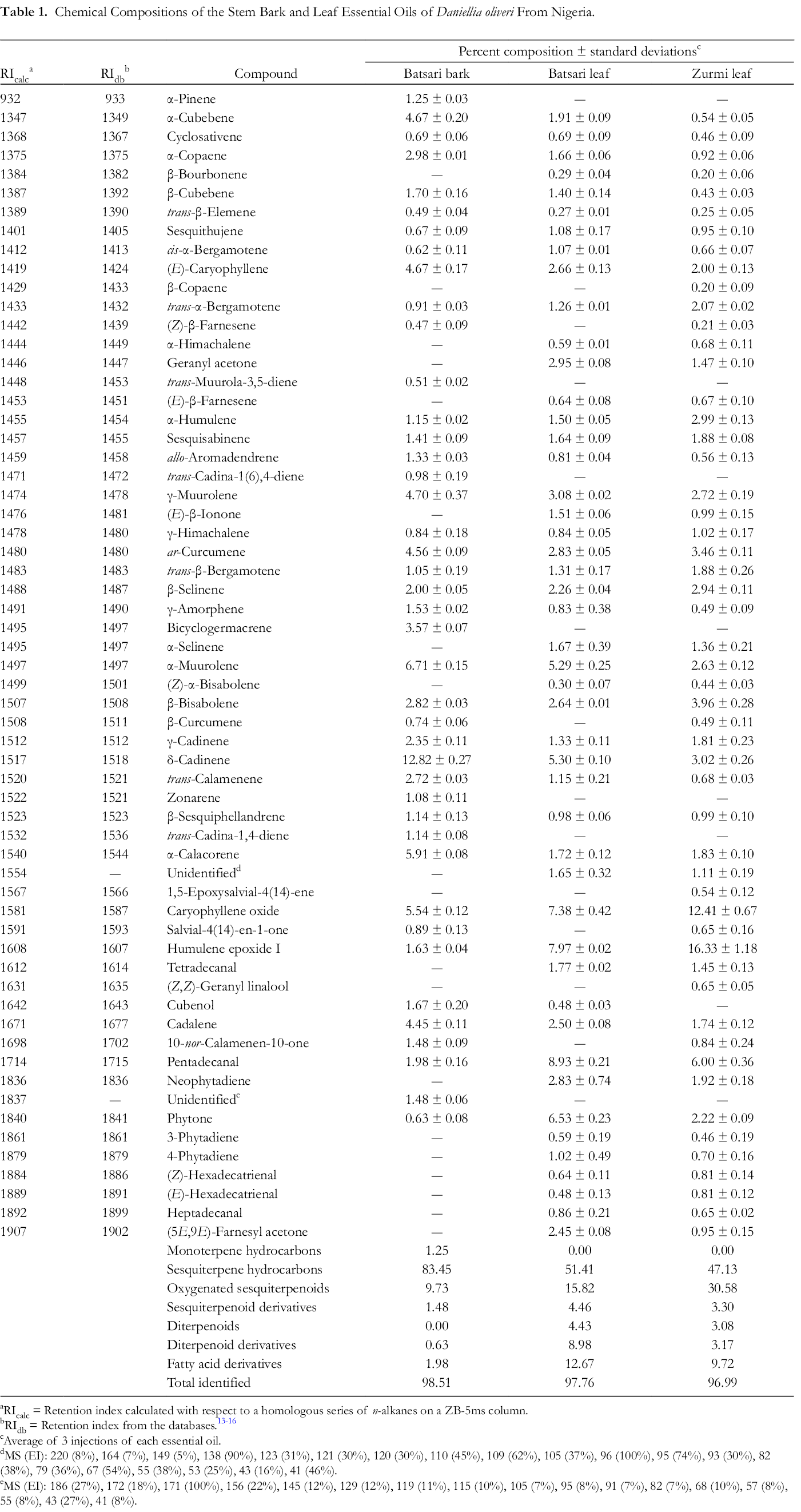
Leaf and stem bark essential oils of D. oliveri were obtained from Batsari (Katsina State) in yields of 1.54% and 1.94%, respectively. The leaf essential oil of D. oliveri from Zurmi (Zamfara State) was obtained in 1.62% yield. The essential oils were pale-yellow in color.
Sesquiterpenoids dominated the essential oils of D. oliveri (Table 1). The bark essential oil from Batsari was composed largely of δ-cadinene (12.8%), α-muurolene (6.7%), α-calacorene (5.9%), and caryophyllene oxide (5.5%). The major components in the leaf essential oils from Batsari and Zurmi, respectively, were humulene epoxide II (8.0% and 16.3%), caryophyllene oxide (7.4% and 12.4%), pentadecanal (8.9% and 6.0%), phytone (6.5% and 2.2%), δ-cadinene (5.3% and 3.0%), and α-muurolene (5.3% and 2.6%).
Chemical Compositions of the Stem Bark and Leaf Essential Oils of Daniellia oliveri From Nigeria.
aRIcalc = Retention index calculated with respect to a homologous series of n-alkanes on a ZB-5ms column.
cAverage of 3 injections of each essential oil.
dMS (EI): 220 (8%), 164 (7%), 149 (5%), 138 (90%), 123 (31%), 121 (30%), 120 (30%), 110 (45%), 109 (62%), 105 (37%), 96 (100%), 95 (74%), 93 (30%), 82 (38%), 79 (36%), 67 (54%), 55 (38%), 53 (25%), 43 (16%), 41 (46%).
eMS (EI): 186 (27%), 172 (18%), 171 (100%), 156 (22%), 145 (12%), 129 (12%), 119 (11%), 115 (10%), 105 (7%), 95 (8%), 91 (7%), 82 (7%), 68 (10%), 57 (8%), 55 (8%), 43 (27%), 41 (8%).
Menut and co-workers had previously obtained and analyzed the bark essential oils of D. oliveri from Benin and Burkina Faso. 17 Although qualitatively similar, there are some notable differences between the bark essential oil from Nigeria and those from Benin and Burkina Faso. Germacrene D was one of the dominant sesquiterpenes in the essential oil from Burkina Faso (29.5%) and a major component in the essential oil from Benin (4.5%), but was not detected in the bark essential oil from Nigeria. Conversely, caryophyllene oxide (5.5%) and cadalene (4.5%) were significant components of the Nigerian bark essential oil, but these components were not reported in the bark essential oils from either Benin or Burkina Faso.
The leaf essential oils, previously reported, from Ivory Coast and from Senegal were dominated by δ-cadinene (24.2% and 31.1%, respectively) 18 Other major components were α-copaene (Ivory Coast, 7.0%; Senegal, 8.3%) and γ-muurolene (Ivory Coast, 4.0%; Senegal, 4.4%), which are consistent with the composition of the leaf essential oils from Nigeria. However, cyperene, found in the leaf essential oils from Ivory Coast and Senegal (3.8% and 4.8%, respectively), was not detected in the leaf essential oils from Nigeria. Conversely, neither pentadecanal nor phytone, major components in the leaf essential oils from Nigeria, were reported in the leaf essential oils from Ivory Coast or Senegal.
Overall, sesquiterpenoids have dominated the leaf and bark essential oils of D. oliveri, but there are quantitative differences in composition, which may be attributed to geographical location as well as seasonal variation.
Leptoderris micrantha
Hydrodistillation of L. micrantha leaves, collected from Agbagi, Ikire (Osun state), Nigeria, yielded a pale-yellow essential oil in 0.53% (w/w) yield. The major components in the leaf essential oil of L. micrantha were incensole (16.2%), phytone (15.4%), pentadecanal (13.7%), α-pinene (7.7%), and iso-phytol (5.2%) (Table 2). As far as we are aware, there have been no previous reports on the essential oil from L. micrantha or any Leptoderris species. Although there are apparently no reports on essential oils from Leptoderris species reported in the literature, there are several essential oils in the Fabaceae worth mentioning. Incensole was a major component (11.1%) in the essential oil of Indigofera aspalathoides from southern India. 19 Phytone and pentadecanal were major components in Lupinus varius essential oil from Jordan (20.5% and 10.2%, respectively) 20 and Vicia caroliniana essential oil from Alabama (21.5% and 5.7%, respectively). 21 Thus, these compounds are not uncommon in essential oils of the Fabaceae.
Chemical Composition of the Leaf Essential Oil of Leptoderris micrantha From Nigeria.

aRIcalc = Retention index calculated with respect to a homologous series of n-alkanes on a ZB-5ms column.
cAverage of 3 injections of the essential oil.
dMS (EI): 196 (5%), 137 (9%), 126 (29%), 111 (28%), 95 (37%), 85 (38%), 81 (38%), 71 (70%), 70 (51%), 69 (55%), 57 (100%), 43 (80%), 41 (52%).
eMS (EI): 272 (3%), 257 (3%), 229 (4%), 189 (4%), 161 (7%), 149 (13%), 135 (17%), 123 (17%), 121 (33%), 111 (23%), 109 (25%), 107 (30%), 97 (35%), 95 (37%), 93 (40%), 84 (100%), 81 (45%), 71 (37%), 69 (48%), 67 (32%), 59 (24%), 57 (35%), 55 (45%), 43 (64%), 41 (49%).
Antimicrobial Activity
The bark and leaf essential oils of D. oliveri (Batsari) and the leaf essential oil of L. micrantha have been screened for activity against a panel of 7 bacteria and 8 fungi (Table 3). The leaf essential oils of D. oliveri and L. micrantha showed only marginal activity against the panel of microorganisms with minimum inhibitory concentration (MIC) values ≥156 µg/mL. However, D. oliveri bark essential oil showed notable antifungal activity against Aspergillus niger and Trichophyton rubrum with an MIC of 78.1 µg/mL for each. It is difficult to speculate as to what components in the bark essential oil of D. oliveri may be responsible for the antifungal activity. The major components were δ-cadinene (12.8%), α-muurolene (6.7%), α-calacorene (5.9%), and caryophyllene oxide (5.5%). Caryophyllene oxide has shown only weak activity against A. niger (MIC = 625 µg/mL). 22 As far as we are aware, neither δ-cadinene, α-muurolene, nor α-calacorene have been individually screened for antifungal activity. However, essential oils with these components have shown antifungal activity. For example, the essential oil of Xenophyllum poposum, with δ-cadinene (16.5%), α-muurolene (3.0%), and α-calacorene (1.1%), showed antifungal activity against A. fumigatus (MIC = 25 µg/mL) and T. rubrum (MIC = 50 µg/mL) 23 ; Teucrium montanum essential oil, with δ-cadinene (17.2%), α-muurolene (1.7%), and α-calacorene (5.0%), was active against Fusarium oxysporum in a zone-of-inhibition assay. 24
Antibacterial and Antifungal Activities (MIC, μg/mL) of Daniellia oliveri (Batsari) and Leptoderris micrantha Essential Oils From Nigeria.

Abbreviation: MIC, minimum inhibitory concentration.
aGentamicin for bacteria, amphotericin B for fungi.
Trichophyton rubrum is a common dermatophytic fungus and is commonly involved in tinea pedis (athlete’s foot), tinea cruris (jock itch), and tinea corporis (ringworm). 25 -27 Microsporum canis and T. mentagrophytes are also dermatophytic fungi responsible for ringworm. 28,29 Essential oils have been examined as potential alternatives to conventional drugs for treatment of tinea infections, 30 -32 and the observed antifungal activity of D. oliveri bark essential oil suggests that it may also serve as a treatment option for athlete’s foot, ringworm, or other tinea infections.
Conclusions
This investigation has revealed the compositions of the essential oils from D. oliveri and L. micrantha, two members of the Fabaceae growing in Nigeria. Antimicrobial screening has shown D. oliveri bark essential oil to have promising antifungal properties and may be useful in treating dermal fungal infections such as athlete’s foot or ringworm.
Materials and Methods
Plant Material
Daniellia oliveri leaf and stem bark samples were taken directly from source trees in 2 sites in northern Nigeria, Batsari (Katsina State, 12°45′19.84″ N, 7°14′53.12″ E, 472 m elevation) and Zurmi (Zamfara State, Zurmi Local Government, 12°45′59.99″ N, 6°47′5.99″ E, 390 m elevation). Both leaves and stem bark of D. oliveri were obtained in the month of April, 2019. The plants were taxonomically identified and authenticated by Namadi Sunusi. A voucher specimen (number 01186) has been deposited in the Department of Biological Sciences, Ahmadu Bello University, Nigeria. The leaves and stem bark were manually removed, air-dried in the laboratory for 7‐10 days, and then pulverized using electric blender.
Samples (450 g each) of both leaves and stem bark of D. oliveri were subjected to hydrodistillation in an all-glass Clevenger-type apparatus. Each of the D. oliveri samples and water were mixed in a ratio of 2:6, and the mixture was stirred constantly during hydrodistillation for 3 hours until no more essential oil was apparent in the distillate. The oils were dried over anhydrous sodium sulfate and stored in sealed amber vials under refrigeration (4 °C) prior to analysis.
The fresh leaves of Leptoderris micrantha (1.5 kg) were collected in the month of July, 2019, from Agbagi, Ikire (7°43′0″ N, 4°14′0″ E, 425 m elevation) in Irewole Local Government Area, Osun state, Nigeria. The plant was taxonomically identified and authenticated by Mr S. A. Odewo at the Forest Research Institute of Nigeria (FRIN) Jericho Ibadan, Oyo state, where a voucher specimen (FHI 112550) was deposited. The leaves of L. micrantha were air-dried in the laboratory for 5 days and then pulverized using a blender before hydrodistillation.
The essential oil from the leaves of L. micrantha was obtained by hydrodistillation. The plant materials (500 g) were introduced into a 5-L flask and distilled water was added until it covered the sample. Hydrodistillation was carried out twice for 3-4 hours in an all-glass modified Clevenger apparatus. The essential oil isolated in the arm of Clevenger apparatus was carefully isolated and transferred to a preweighed amber sample bottle, dried with anhydrous sodium sulfate, and stored under refrigeration (4 °C) until ready for analysis. Hydrodistillation of L. micrantha leaves yielded a pale-yellow essential oil in 0.53% (w/w) yield.
Gas Chromatographic-Mass Spectral Analysis
The essential oils were analyzed by gas chromatography-mass spectrometry as previously reported 33 using a Shimadzu GCMS-QP2010 Ultra, electron impact mode with electron energy =70 eV, scan range =40-400 atomic mass units, scan rate =3.0 scans/s, and Shimadzu GC-MS solution software v. 4.45 (Shimadzu Scientific Instruments, Columbia, MD, USA); ZB-5ms fused silica capillary GC column Phenomenex, Torrance, CA, USA; (5% phenyl)-polymethylsiloxane stationary phase, 0.25 µm film thickness; helium carrier gas, column head pressure = 552 kPa, flow rate = 1.37 mL/min; injector temperature = 260 °C, ion source temperature = 260 °C; GC oven temperature program: initial temperature = 50 °C, temperature increased 2 °C/min to 260 °C. For each sample, a 5% w/v solution in CH2Cl2 was prepared, 0.1 µL was injected using a split ratio of 30:1. Retention indices (RIs) were determined using a homologous series of n-alkanes. 34 Identification of the essential oil components was carried out by comparison of mass spectral fragmentation patterns in addition to RI comparison with those reported in the databases 13 -16 using the LabSolutions GCMS solution software version 4.45 (Shimadzu Scientific Instruments, Columbia, MD, USA) and with matching factors >90%.
Antimicrobial Screening
The essential oils were screened for antibacterial activity against Gram-positive bacteria (Bacillus cereus [ATCC No. 14579], Cutibacterium acnes [ATCC No. 11827], Staphylococcus aureus [ATCC No. 29213], Staphylococcus epidermidis [ATCC No. 12228], and Streptococcus pyogenes [ATCC No. 19615]) and Gram-negative bacteria (Pseudomonas aeruginosa [ATCC No. 27853], and Serratia marcescens [ATCC No. 14756]), and for antifungal activity against the molds (Aspergillus fumigatus [ATCC No. 96918], Aspergillus niger [ATCC No. 16888], Cryptococcus neoformans [ATCC No. 32045], Microsporum canis [ATCC No. 11621], Microsporum gypseum [ATCC No. 24102], Trichophyton mentagrophytes [ATCC No. 18748], and Trichophyton rubrum [ATCC No. 28188]), and 1 yeast (Candida albicans [ATCC No. 18804]) using the microbroth dilution technique 35,36 as previously reported. 37,38
Footnotes
Acknowledgments
NSD and WNS participated in this work as part of the activities of the Aromatic Plant Research Center (APRC, https://aromaticplant.org/).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
